Introduction
Constant exposure to solar radiation entails negative skin effects induced by reactive oxygen species (ROS) formation as singlet oxygen (1O2). 1O2 is the first excited state of molecular oxygen (3O2) and can be endogenously generated in biological systems through photochemical reactions type II, where UVA/UVB radiation as well as visible light can convert photosensitizer molecules into excited states that transfers absorbed energy to 3O2 to generate 1O2 [1,2]. Of the total solar energy able to reach the earth´s surface, 6.8 % corresponds to UV light, 38.9 % to visible light and 54.3 % to near infrared light [3]. Likewise, from UV light, more than 50 % of UVA can penetrate the dermis, whereas only 14 % of UVB light reaches the epidermis, thereby photochemical generation of 1O2 in the skin is possible due to the presence of endogenous photosensitizer molecules such as porphyrins, bilirubin, B6 vitamers and vitamin K [2,4]. 1O2 is the predominant ROS from type II reactions that is able to react with nucleic acids, unsaturated lipids and aminoacids to yield endoperoxides from [2 + 4] cycloadditions, dioxetanes from [2 + 2] cycloadditions and hydroperoxides from “ene” reactions [5,6].
In recent years due to multiple benefits attributed to natural antioxidants, the cosmetic and dermatology industry has focused on the development of skincare products such as anti-aging creams or sunscreens enriched with antioxidants in order to prevent oxidative damage caused by ROS [7-9]. In this sense, the term antioxidant has been defined as any substance that delays, prevents or removes oxidative damage to a target molecule [10]. Antioxidants can react by depleting molecular oxygen or decreasing its local concentration, removing pro-oxidative metal ions, trapping aggressive reactive oxygen species such as superoxide anion radical or hydrogen peroxide, scavenging chain-initiating radicals like hydroxyl (HO•), alkoxyl (RO•) or peroxyl (ROO•), breaking the chain of a radical sequence or quenching 1O2 [11]. However, increasingly researches have reported pro-oxidant activities of antioxidants such as resveratrol and quercetin [12-14]. Likewise, we previously reported the photosensitizing ability to generate 1O2 of curcumin, resveratrol and quercetin identified through the photo-oxidation of ergosterol method [15]. Also, through this method, the photosensitizing ability of cosmetic colorants to generate 1O2 and the membrane cell damage caused by two of the nine cosmetic colourants evaluated has been reported [16,17]. Furthermore, we recently reported on the pro-oxidant effect of five synthetic hydroxycoumarin-based antioxidants by acting as photosensitizers in 1O2 generation [18]. Therefore, the present study was aimed to determine whether natural antioxidants commonly used in the development of skincare products are able to generate 1O2 by acting as photosensitizing molecules in the photo-oxidation of ergosterol reactions.
Experimental
Reagents
Fisetin, retinol, cyanidin, hesperetin, luteolin, rutin, L-ascorbic acid, apigenin, epigallocatechin gallate, myricetin, kaempferol, ergosterol, eosin yellowish, sodium azide (NaN3) were purchased from Sigma-Aldrich (Corp., St. Louis, Mo., U.S.A.). Caffeic acid, vanillic acid and rosmarinic acid were kindly provided by Prof. Zaira Domínguez from the Universidad Veracruzana. Distilled ethanol, analytical grade was employed as a solvent in photo-oxidation reactions.
Photo-oxidation of ergosterol
Ergosterol was used as 1O2 chemical trap in the determination of the photosensitizing ability through reactions of the photo-oxidation of ergosterol. 14 antioxidants were tested: Fisetin (1), retinol (2), cyanidin (3), hesperetin (4), caffeic acid (5), luteolin (6), rutin (7), vanillic acid (8), ascorbic acid (9), apigenin (10), epigallocatechin gallate (11), rosmarinic acid (12), myricetin (13) and kaempferol (14) (Fig. 1). For each reaction, 1 mM ergosterol and 144 (M antioxidant (initial concentration) was prepared in ethanol [15]. The solution was placed inside a photo-oxidation camera and irradiated (four compact fluorescent lamps) during 2 h under continuous oxygen flux (medicinal grade oxygen, flux rate: 75 mL/s), bubbled using a stainless-steel filter (10 (m HPLC filter). The light intensity was 19623 ±129 lux (YK-10LX light meter). The temperature inside the photo-oxidation camera was 32 °C ± 2. In order to establish reference controls, the photo-oxidation reaction by adding eosin yellowish 144 (M was established as positive control (+) and the reaction without a photosensitizer dye as negative control (−). Sodium azide (1 mM) was used to confirm 1O2 generation in photo-oxidation reactions.
Determination of singlet oxygen by nuclear magnetic resonance
Indirect detection of 1O2 generation was made by proton Nuclear Magnetic Resonance (1H-NMR) recorded on a Bruker Avance HD III spectrometer (500 MHz) and Agilent DD2 500 MHz spectrometer for NaN3 reactions, using CDCl3 as solvent and TMS as an internal reference. Ergosterol traps 1O2 to form ergosterol peroxide. Both sterols were detected by 1H-NMR mixtures analysis based on the comparison of integrals of vinyl signals of H-6 and H-7 protons of ring B of ergosterol (δH-6= 5.57 ppm, dd, 1H; δH-7= 5.38 ppm, dd) and ergosterol peroxide (δH-6= 6.50 ppm, d, 1H; δH-7= 6.25 ppm, d). Once these signals were identified, integration values were obtained from which the conversion ratio of ergosterol into ergosterol peroxide (E:EP) was calculated. This ratio was also calculated for the positive and negative controls.
Antioxidants classification was established taking into reference the E:EP conversion ratio from the negative control. Therefore, antioxidants used in photo-oxidation reactions where the ratio conversion E:EP was higher than the negative control were considered as photosensitizer antioxidants in the generation of 1O2. Conversely, antioxidants used in photo-oxidation reactions where the ratio conversion E:EP was lower than the negative control were considered as antioxidant quenchers of 1O2. MestReNova software (v6.0.2-5475) was used in 1H-NMR analysis and data processing.
Results and discussion
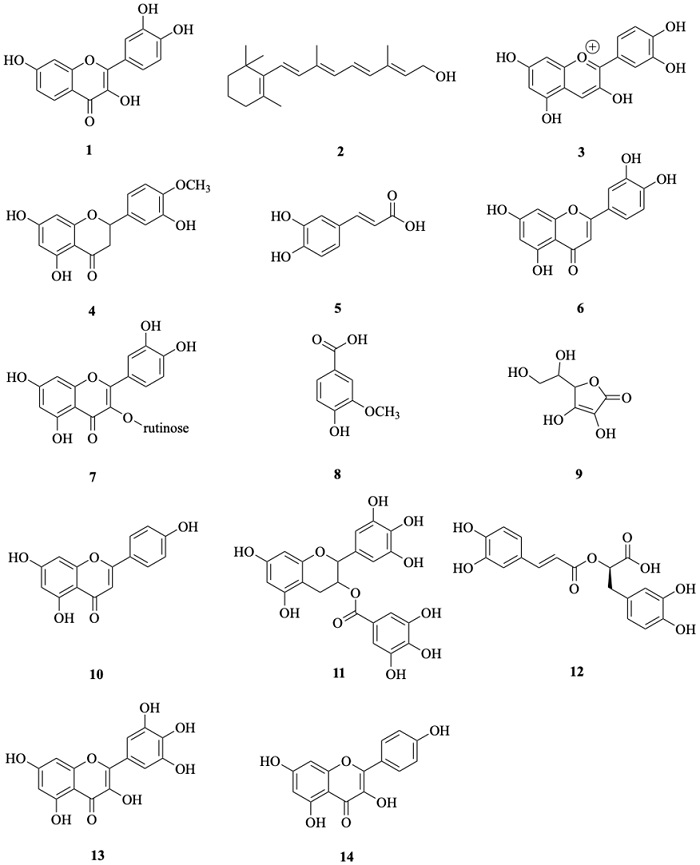
1O2 generation was quantified by 1H-NMR mixtures analysis through the identification of ergosterol peroxide (EP), an oxidation product of ergosterol (E) formed through a Diels-Alder reaction between 1O2 and a conjugate diene system of B ring of ergosterol. Indirect detection of 1O2 through photo-oxidation of ergosterol reactions allowed us to establish the photosensitizing ability of four out of fourteen screened antioxidants: fisetin (1), retinol (2), cyanidin (3), hesperetin (4). Thereby, two double signals at 6.50 and 6.25 ppm attributed to H-6 and H-7 protons from EP were identified in the 1H-NMR spectra, also two double-double signals attributed to H-6 and H-7 protons from E were observed at 5.57 and 5.38 ppm, respectively (Fig. 2). The E:EP conversion ratio was calculated from integration data of this signals and was also expressed as a percentage. Thus, the results obtained from photo-oxidation reactions carried out with 1 and 2 converted 20 % of E into EP and 3 and 4 allowed a 9 % of EP formation (Table 1). This suggests that fisetin, retinol, cyanidin and hesperetin were able to generate 1O2 through a photosensitized mechanism because the EP quantity detected was higher than negative control (5 %).
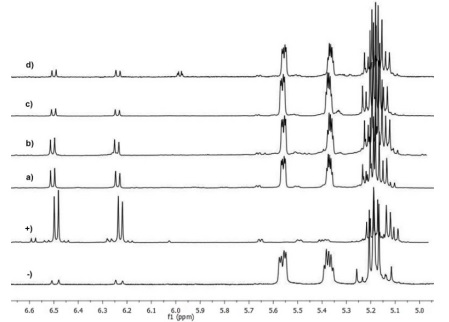
Fig. 2 1H-NMR spectra of mixtures reaction obtained from photo-oxidation reactions carried out with (a) fisetin, (b) retinol, (c) cyanidin and (d) hesperetin. Control (−): reaction without photosensitizer, Control (+): reaction with eosin yellowish as photosensitizer.
Table 1 Conversion ratio of Ergosterol into Ergosterol peroxide (E:PE) calculated from 1H-NMR-signals assignment of H-6 and H-7.
| Antioxidant | Ergosterol | Ergosterol peroxide | Conversion E:EP | |||
| ʃ H-6 | ʃ H-7 | ʃ H-6 | ʃ H-7 | Ratio | EP Percentage | |
| Control (−) | 18H | 18H | 1H | 1H | 18:1 | 5 |
| Control (+) | NS | NS | 1H | 1H | 0:1 | 100 |
| Fisetin | 4H | 4H | 1H | 1H | 4:1 | 20 |
| Retinol | 4H | 4H | 1H | 1H | 4:1 | 20 |
| Cyanidin | 10H | 10H | 1H | 1H | 10:1 | 9 |
| Hesperetin | 10H | 10H | 1H | 1H | 10:1 | 9 |
| Caffeic acid | 177H | 181H | 1H | 1H | 179:1 | 1 |
| Luteolin | 146H | 151H | 1H | 1H | 148:1 | 1 |
| Rutin | 129H | 138H | 1H | 1H | 133:1 | 1 |
| Vanillic acid | 191H | 203H | 1H | 1H | 133:1 | 1 |
| Ascorbic acid | 76H | 77H | 1H | 1H | 76:1 | 1 |
| Apigenin | 62H | 64H | 1H | 1H | 63:1 | 2 |
| Epigallocatequin gallato | 51H | 52H | 1H | 1H | 52:1 | 2 |
| Rosmarinic acid | 42H | 44H | 1H | 1H | 43:1 | 2 |
| Myricetin | 26H | 26H | 1H | 1H | 26:1 | 4 |
| Kaempferol | 20H | 20H | 1H | 1H | 20:1 | 5 |
ʃ: integration values; NS: no signal detected.
Additionally, in order to confirm the presence of 1O2, photo-oxidation of ergosterol reactions with fisetin, retinol, cyanidin and hesperetin were carried out adding sodium azide (1 mM) as specific quencher of 1O2. After the reaction time, a substantial reduction in the quantity of EP generated during photo-oxidation reactions was observed (Fig. 3).
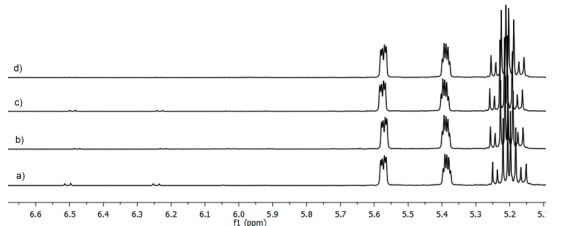
Fig. 3 1H-NMR spectra of mixtures reaction carried out with (a) fisetin, (b) retinol, (c) cyanidin, (d) hesperetin plus NaN3 (1 mM).
On the other hand, signals attributed to EP were barely detected in the 1H-NMR spectra of the remaining antioxidants tested, while signals corresponding to E were clearly visible (Fig. 4). Thus, the E:EP conversion ratios obtained were lower than negative control in photo-oxidation reactions carried out with caffeic acid (5), luteolin (6), rutin (7), vanillic acid (8) and ascorbic acid (9), in which only a 1 % of EP was formed. In a similar way, the E:EP conversion ratios in photo-oxidation reactions carried out with apigenin (10), epigallocatechin gallate (11) and rosmarinic acid (12) allowed the formation of 2 % of EP. Finally E:EP conversion ratios similar to the 5 % of EP detected in the negative control were obtained in photo-oxidation reactions carried out with myricetin (13) and kaempferol (14)(Table 1). Therefore, because of the EP quantity detected in photo-oxidation reactions was ( 5 % (negative control), we can assume that they were not able to generate 1O2 through a photosensitized mechanism thereby they were considered as antioxidants able to quench 1O2.
Fig. 4 1H-NMR spectra of mixtures reaction obtained from photo-oxidation reactions carried out with (a) caffeic acid, (b) luteolin, (c) rutin, (d) vanillic acid, (e) ascorbic acid, (f) apigenin, (g) epigallocatechin gallate, (h) rosmarinic acid, (i) myricetin, (j) kaempferol. Control (−): reaction without photosensitizer, Control (+): reaction with eosin yellowish as photosensitizer.
Identification of compounds with the ability to photosensitize the generation of 1O2 should be considered an important issue owing to the fact that the presence of 1O2 in cells is related to skin photoaging. Cholesterol peroxidation can be caused through ene-reaction between 1O2 and the cholesterol double bond in carbons 5 and 6 to form cholesterol 5(-hydroperoxide, as the major product and cholesterol 6(/(-hydroperoxide as the minor products [19]. The significance of cholesterol peroxidation products in photoaging has been clearly established because a mixture of cholesterol 5-hydroperoxide and cholesterol 7-hydroperoxide induces the activation of matrix metalloproteinase-9 (MMP-9), a protein implicated in collagen degradation. Loss of collagen in the skin results in wrinkles appearing and sagging skin, a hallmark associated with skin photoaging [20]. Several researches have shown the efficient antioxidant, anti-inflammatory and anti-aging activity of fisetin [21,22], retinol [23], cyanidin [24] and hesperetin [25], however our results show that under specific conditions they are able to act as photosensitizer compounds in photochemical reactions and stimulate the generation of 1O2. Hence, the evaluation of the photosensitizing ability of compounds used in skincare formulations should be considered an important issue by the dermatology and cosmetic industry.
Concerning antioxidants that show ability to quench 1O2, several researches have reported, not only on their anti-inflammatory, anti-aging and free radical scavenging properties but also their ability to provide a protector effect against UVA/UVB-induced skin damage [26-30]. Likewise, no phototoxic effect has been reported on caffeic acid and rutin [31]. Therefore, the ability to quench 1O2 added to all the beneficial properties reported for caffeic acid, luteolin, rutin, vanillic acid, ascorbic acid, apigenin, epigallocatechin gallate, rosmarinic acid, myricetin and kaempferol, allow us to suggest that they could be considered as promising compounds to diminish, prevent or avoid skin photoaging caused by 1O2.
Through the photo-oxidation of ergosterol, the photosensitizing ability of antioxidant compounds was evidenced. Thus, fisetin, retinol, hesperetin and cyanidin aside from their antioxidant activity could show a pro-oxidant effect caused by 1O2. Moreover, the identification of antioxidant compounds with the ability to quench 1O2, was seen to be possible because they provided protection to ergosterol against oxidation caused by 1O2. Consequently, the results obtained allow us to increase the antioxidant classification based on their activity against 1O2 as we previously proposed [15]. Therefore, we suggest that caffeic acid, luteolin, rutin, vanillic acid, ascorbic acid, apigenin, epigallocatechin gallate, rosmarinic acid, myricetin and kaempferol can be classified as type 1 antioxidants: antioxidant quenchers of 1O2; and fisetin, retinol, hesperetin and cyanidin as type 2 antioxidants: photosensitizer antioxidants in generation of 1O2.
Conclusions
Antioxidants provide several health benefits. However, the endogenous generation of 1O2 through photosensitized mechanisms joined to non-photosensitized mechanisms could increase the presence of 1O2 in an organism, which can cause damage to cell membrane components and induce skin photoaging. Hence the incorporation of antioxidants in skincare products as anti-aging treatments or sunscreens should be supported by a previous evaluation of their photosensitizing ability in order to increase their safety.