Introduction
Marine ecosystems are very complex, and organisms are known to possess bioactive compounds for defense and for the protection of eggs and embryos. Some have chemical mechanisms to synthesize compounds de novo to protect themselves from pathogens (Lauritano & Ianora, 2020).
Currently, about 16,000 natural products have been discovered from marine organisms, for example, in 1949 the first antibacterial product of marine origin, Cephalosporin C, was obtained from a strain of marine fungus Cephalosporium (Pandey, 2019). Among marine organisms, marine invertebrates offer a good source of potential antibacterial drugs (Di Costanzo et al., 2019; Lauritano et al., 2020).
Cephalopods are found in all marine habitats of the world, such as benthic-cryptic; epibenthic, and pelagic in bays and the open sea. However, cephalopods do not have a shell to protect themselves, so they use chemical means, coloration change, and toxins (Monolisha et al., 2013). Cephalopod skin consists of two layers, the epidermis, and the dermis. These layers cover the external surface of the body showing different regional specializations, such as suckers and arms. In addition, the skin is continued with the internal surfaces of the siphon and paleal cavity (Anadon, 2019).
To evaluate the condition of the animals, it is essential to carry out various tests on their organs, including the skin. The characteristics that should be noted on the skin are the presence of desquamation, hematomas, ulcers, and mucosal hypersecretion (FAO, 2011). The skin of most organisms contains mucosal epithelia with important antiparasitic, antifungal, antibacterial, and antiviral properties (Fast et al., 2002). Currently, marine and terrestrial farms present problems arising from diseases transmitted by pathogenic bacteria (OIE, 2019; Prachumwat et al., 2020; Deng et al., 2020; de Lorgeril et al., 2018; Palomares-Reséndiz et al., 2021; Rivera-Benitez et al., 2014; Burniston et al., 2015; Giraldo-Cardona et al., 2019, Pulido-Villamarín et al., 2021). For this reason, this research focuses on the skin of octopuses mainly in the study of mucus (González-Costa et al., 2020; Villanueva et al., 2021).
Recently, there have been many studies on the antibacterial properties of skin mucus from many species of marine organisms against various pathogenic microorganisms for humans and fish (Wei et al., 2010; Bragadeeswaran & Thangaraj, 2011; Vennila et al., 2011; Fuochi et al., 2017; Reverter et al., 2018), of freshwater fishes (Nwabueze, 2014; Pethkar & Lokhande, 2017), marine fishes and catadromous fishes (Fuochi et al., 2017; Pethkar & Lokhande, 2017), elasmobranchs (Coelho et al., 2019; Ritchie et al., 2017), anemones (Subramanian et al., 2011; Stabili et al., 2015), mollusks (Pales Espinosa et al., 2013; Kamiya et al., 2006; Suárez et al., 2021).
The main components of the mucus are glycoproteins and proteoglycans, and to a lesser extent, proteolytic enzymes, lysozymes, immunoglobulins, etc. Unlike other mollusks (Smith, 2002), the chemical nature of mucus in the genus Octopus is limited (Smith & Morin, 2002).
Each species has its own behavior and habitat, lives in different types of aquatic environments, and consumes different types of food, which may influence the amount of mucus secretion and its components within the same species or between different species and may be useful to provide a variety of immune responses. Therefore, the aim of the present study was: To analyze the antibacterial effect of the skin mucus of the common octopus Octopus bimaculatus, against a collection of pathogenic strains for terrestrial and aquatic farm animal collection.
Material and Methods
Specimen Collection
Five octopuses of the genus O. bimaculatus weighing approximately 500 to 700 grams were captured in the community of El Sauzoso, B.C.S. Mexico (24°18'44.5 "N 110°38'26.0 "W) with traps. They were placed in tanks on board of the boat with clean, sterile water. They were washed with a 4% solution of potassium permanganate (KMnO4) before being placed in the tanks and before mucus collection. No chemicals or anesthesia were administered to the octopuses for skin mucus collection. Mucus was collected from five octopuses. Mucus was carefully scraped from the body surface by moving a sterile plastic spatula in an anteroposterior direction, from the head toward the tentacles, and mucus was collected at regular intervals (10 attempts in 3 hrs). Accumulation of mucus from the ventral area was avoided to eliminate intestinal and urogenital contamination (Chong et al., 2005). The mucus samples were brought on ice to the laboratory where they were centrifuged at 13,200 rpm for 10 min at 4°C. The supernatant was placed in a sterile tube and centrifuged again on ice at 11,000 rpm for 30 min. The precipitate was discarded, and the raw mucus extract was stored at -70°C before analysis.
Preparation of raw octopus mucus
Raw octopus mucus was treated with SIGMA® lysozyme enzyme at a final concentration of 10 mg mL-1 in phosphate buffer pH 7.0. The mixture was incubated at optimum pH and temperature for the enzyme to break the peptidoglycan layer of the bacterial cell wall for 2 hrs. The antibacterial activity was performed by a diffusion method in Petri dishes with Müller-Hinton agar using wells (Guerra & Pastrana, 2002).
Reactivation of pathogenic strains
The strains used for the in vitro antibacterial evaluation of the prepared octopus mucus were: Escherichia coli (Amador, 2018), Staphylococcus aureus (Unpublished data), Vibrio harveyi (Parera-Valadez, 2012), Staphylococcus pasteuri (Amador, 2018), Vibrio parahaemolyticus (Amador, 2021) (Table 1). All bacterial strains were obtained from the collection of the Food Science and Technology Laboratory of at Autonomous University of Baja California Sur.
Bacterial strains were cultured according to the protocols and microbiological safety conditions of Eder et al. (2009). Each strain was incubated at 37 °C in nutrient broth (0.5% peptone, 0.5% NaCl, 0.3% meat extract, distilled water, pH 6.8 at 28 °C) for 18-24 h. Subsequently, a viable count of each culture was made and colony-forming units per mL (CFU/mL) were calculated (Barbosa et al., 1995).
Protein estimation
The amount of protein in octopus mucus was determined using the method of Bradford Kruger (2009).
Table 1 Strains from the collection of the Food Science and Technology Laboratory of at Autonomous University of Baja California Sur.
| Isolated | Genus | Species | Place |
|---|---|---|---|
| Oyster | Escherichia | coli | BCS, México. |
| Bovine | Staphylococcus | aureus | BCS, México. |
| Shrimp | Vibrio | harveyi | BCS, México. |
| Oyster | Staphylococcus | pasteuri | BCS, México. |
| Shrimp | Vibrio | parahaemolyticus | BCS, México. |
Petri dish diffusion assay with wells in agar
The antibacterial effect of octopus mucus (OM) extracts on the selected bacterial strains was analyzed by the agar well diffusion method (Valgas et al., 2007). Petri dishes were prepared with 15 mL of Müller-Hinton agar and then the bacterial strains (Table 1) were seeded by surface spreading with 1x107 CFU mL-1. Subsequently, wells with a diameter of 6 mm were aseptically punched with a tip and extracts of the prepared octopus mucus were added (10, 50, and 100 µL mL-1 of mucus extract per well), as positive controls the antibiotics amikacin and chloramphenicol were used at a concentration of 40 µg mL-1 and 20 µg mL-1, respectively, and physiological saline solution (0.85% NaCl) was used as a negative control. The Petri dishes already inoculated and with the extracts were incubated at 37 °C for 24 h. The evaluation of the bactericidal effect was performed by measuring the diameter of the IH formed around the well (Jorgensen & Turnidge, 2015). The diameter of the IH was measured in millimeters (mm) with a vernier, the measurement was made by taking the total diameter of the halo minus the 6 mm of the well. The results of antibacterial activity were compared with positive controls.
Statistical analysis
Results were subjected to Barlett's homoscedasticity test and D'Agostino-Pearson normality test with an α = 0.05 and then a one-way analysis of variance (ANOVA) was performed to compare significant variation in octopus mucus and antibiotics. Determination of factors contributing to significant differences was performed using the LSD multiple comparison test (Sokal & Rohlf, 1980).
Results and Discussion
Octopus mucus may represent a good source of biologically active compounds to be used for different biotechnological purposes. The biological interface between the octopus and its marine environment consists of a mucus layer composed of biochemically diverse secretions of epidermal and epithelial cells (Accogli et al., 2017). It has been suggested that mucus secretion serves an antibacterial purpose, and also helps to collect bacteria from the surrounding waters, and promotes the accumulation of microbial cells, including Vibrio and other marine microorganisms (Troll et al., 2010).
The effects of a new antibacterial peptide (OctoPartenopin), extracted from the suckers of O. vulgaris, against Pseudomonas aeruginosa, Staphylococcus aureus, and Candida albicans were also demonstrated (Maselli et al., 2020). In this research, octopus mucus was used to inhibit the growth of some strains of pathogenic bacteria, where the higher the concentration, the greater the inhibition.
In another study, in vitro and in vivo assays were conducted using octopromycin, a peptide derived from Octopus minor against Acinetobacter baumannii, and revealed that infected fish exhibited a significantly higher relative survival percentage of 37.5 % than infected fish treated with (placebo)PBS 16.6 %, (Rajapaksha et al., 2021).
Soluble proteins
The protein content in the mucus of the selected octopuses varied very little. The general trend of protein content in the epidermal mucus of the species was 3.684 mg mL, while in another research a protein content of 1.0716 mg mL-1 for O. dolfusii and 1.3620 mg mL-1 for O. aegina was found (Monolisha et al.,2013). However, no further information on protein quantification in octopus mucus was found, but in fish, it was shown that raw and partially purified epidermal mucus from Tachysurus dussumieri presented a protein content of 0.48 ± 0.02 mg mL-1 and 0.82 ± 0.05 mg mL-1 (Arulvasu et al., 2012). Similarly, protein was found to be the major component in different skin mucus extracts of Channa micropeltes, Cytusis striatus, Oreochromis niloticus, and Mystus nemurus, which ranged from 4.32 ± 0.28 to 5.79 ± 0.32 mg mL-1 (Rao et al., 2015). Possibly due to the high protein content in the mucus extract of O. bimaculatus had a higher IH against the pathogens selected in this study, as mucus secretion has also been suggested to have an antibacterial purpose (Troll et al., 2010), and in addition to lectins, uncharacterized glycoproteins have also been associated with binding to bacteria in the hemolymph of Octopus vulgaris (Rögener et al., 1987).
Bacterial antagonism assay
The extract of raw octopus mucus and the two antibacterials, chloramphenicol and amikacin, exhibited a strong bactericidal effect against all selected pathogenic strains except for the pathogen V. parahaemolyticus.
In the bioassay, the inhibition of bacterial strains was evaluated using different concentrations of mucus and specific antibiotics. The results showed inhibition halos (IH) (Table 2), where it was observed that at the mucus concentration of 10 µL mL-1, there was a higher IH for the E. coli strain with 19.168 ± 3 mm, the lowest halo for S. aureus with 8.0 ± 0.4 mm and no IH for the V. parahaemolyticus strain. At the concentration of 50 µL mL-1, the highest IH was for E. coli with 20.21 ± 0.4 mm, and the lowest was for V. parahaemolyticus 1.56 ± 0.02. At the concentration of 100 µL mL-1 inhibition was shown in all strains, the highest halo was presented by the S. aureus strain with 14.2 mm ± 0.5 mm and the lowest halo was for V. parahaemolyticus with 3.9 ± 0.2 mm. The antibiotic chloramphenicol 20 lg mL-1 showed the highest inhibition halo for E. coli at 14.6 ± 0.5 mm and the lowest for S. aureus with 2.12 ± 0.1 mm and the antibiotic amikacin 40 lg mL-1 showed the highest IH for S. aureus strain with 8.4 ± 0.6 mm and the lowest for V. parahaemolyticus with 4.0 ± 0.2 mm.
Table 2 Halos of inhibition were shown by selected bacteria exposed to the three different concentrations of octopus mucus and two commercial antibiotics.
| Octopus mucus concentrations HI (mm) | HI antibiotics (mm) | ||||
|---|---|---|---|---|---|
| 10 µL mL-1 | 50 µL mL-1 | 100 µL mL-1 | Chloramphenicol 20 lg mL-1 | Amikacin 40 lg mL-1 | |
| Strains | |||||
| E. coli | 19.168 ± 0.3 | 20.21± 0.4 | 5.52 ± 0.4 | 14.6 ± 0.5 | 5.52 ± 0.4 |
| V. parahaemolyticus | 0 | 3.9 ± 0.2 | 3.9 ± 0.2 | 2.4 ± 0.2 | 4.0 ± 0.2 |
| S. pasteuri | 10.32 ± 0.9 | 10.5 ± 0.45 | 12.7 ± 0.7 | 4.4 ± 0.43 | 5.52 ± 0.4 |
| V. harveyi | 8.82 ± 0.3 | 10.2 ± 0.5 | 12 ± 0.9 | 4.4 ± 0.3 | 6.04 ± 0.5 |
| S. aureus | 8.0 ± 0.4 | 10 ± 0.5 | 14.2 ± 0.5 | 2.12 ± 0.1 | 8.4 ± 0.6 |
HI= Inhibition halos. Halo Diameter of the inhibition halo minus the diameter of the well with an antibacterial.
The five pathogenic strains presented IH in the presence of the two antibiotics. With chloramphenicol 20 µg mL1, the highest IH was of the following strains: E. coli with 14.6 ± 0.5 mm, V. harveyi with 4.4 ± 0.4 mm, S. pasteuri with 4.4 ± 0.43 mm and the lowest of S. aureus with 2.12 ± 0.1 mm. With amikacin 40 lg mL-1 the highest IH was presented by the S. aureus strain with 8.4 ± 0.6 mm, followed by V. harveyi with 6.04 ± 0.5 mm, E. coli and S. pasteuri with 5.52 ± 0.4 mm and the lowest was V. parahaemolyticus with 4 ± 0.2 mm.
The results of the inhibition halos of the selected bacterial strains due to mucus at a concentration of 10 µL mL-1 showed significant differences (p > 0.05) among all strains, however, E. coli was the most inhibited. At the concentration of 50 µL mL-1 there were no differences between the strains S. pasteuri, V. harveyi, S. aureus, but there were differences against V. parahaemolyticus and E. coli (p > 0.05) with the latter having the highest IH of 20.28 mm. With the 100 µL mL-1 mucus significant differences (p > 0.05) were shown between almost all strains: S. aureus and V. harveyi and the E. coli strain had the highest IH of 21.44 mm, except for S. pasteuri. The antibiotic chloramphenicol 20 lg mL-1 did not cause significant differences between the IH of the strains S. pasteuri and V. harveyi, between V. parahaemolyticus and S. aureus, but it did cause significant differences between these groups and against E. coli, the latter presenting the highest IH of 14.8 mm. The antibiotic amikacin 40 lg mL-1 did not cause significant differences between the IH of E. coli and V. harveyi between S. pasteuri and S. aureus, but these two groups did against V. parahaemolyticus (p > 0.05), here the strain with the highest IH was S. aureus with 8.4 mm. Importantly, the V. parahaemolyticus strain was the least inhibited at all three concentrations and antibiotics.
Other reports have shown the antimicrobial activity of aqueous and ethanolic extracts of mucus from different marine organisms. Antimicrobial activity assays carried out with ethanolic extracts of tissues from Octopus dolfusii and Octopus aegina organisms against V. parahaemolyticus showed IH of 34 and 28 mm on average respectively (Monolisha et al., 2013).
Likewise, in methanolic extracts of O. dollfusi tissue, the highest IH of 17 mm was observed against E. coli, another extract of O. aegina showed an IH of 15 mm against V. parahaemolyticus and another of O. aerolatus against S. aureus presented IH of 13 and 14 mm and the lowest IH of 8 mm was observed against Streptococcus sp. with extracts of O. dollfusi, O. aegina and O. aerolatus (Pasiyappazham et al., 2011). In this study, the surface mucus of O. bimaculatus against different pathogens showed the highest IH against E. coli of 21.44 mm, against S. aureus of 14.2 mm, against S. pasteuri of 12.75 mm and against V. harveyi of 12.36 mm. The lowest IH was 4.6 mm in V. parahaemolyticus (Figure 1), similarly, Monolisha et al. (2013) using ethanolic extracts of O. dolfusii tissue against V. anguillarum, V. alginolyticus, V. sclintis, and V. harveyi reported IH of about 27, 22, 21 and 15 mm respectively, also used extracts of O. aegina extracts against the same pathogens reported IH of 25, 21, 21 and 18 mm respectively, which suggests that the genus Octopus presents antibacterial activity in the superficial mucus and could also be in all the internal mucosa of these organisms. Similar studies with extracts of carp mucus showed that the maximum inhibitory effect of carp mucus Hypophthalmichthys nobilis against E. coli reached an IH of 31.00 ± 0.47 mm, that of Ctenopharyngodon idella against P. aeruginosa was 34.33 ± 0.13 m, significantly higher than amikacin 33.33 ± 0.13 mm and that of O. bimaculatus against S. aureus ranged from 10.32 to 12.75 mm (Kumari et al., 2019). It was also reported that S. aureus was more susceptible than other Gram-negative bacteria to growth inhibition by gastropod extracts (Benkendorff et al., 2001), however, no antibacterial activity against E. coli was detected in Mytilus galloprovincialide hemolymph (Hubert et al., 1999). It has been suggested that the resistance of E. coli to antibacterials was due to the complex structure of the Gram-negative cell wall, particularly the lipopolysaccharides of the outer membrane, which excludes most of the active compounds (Im & Khalid, 2020).
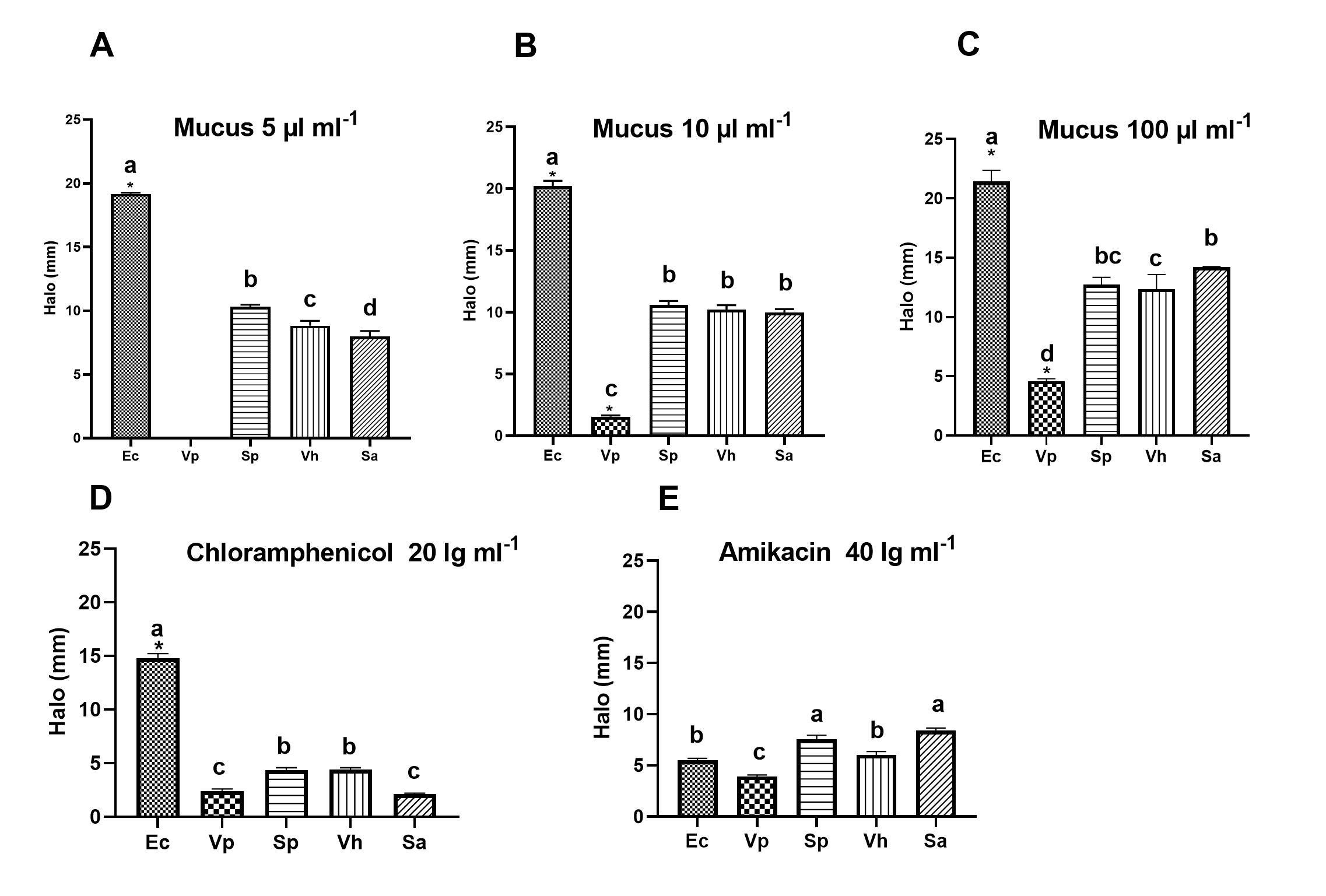
Identical letters denote no significant differences (p > 0.05). In Figure A, there are significant differences between all strains (a) Ec, (b) Sp, (c) Vh, and (d) Sa. Figure B, letter (b) Sp, Vh, Sa there are no differences between them; (a) Ec and (c) Vp means significant differences against a. In figure C, Sp (bc), Sa (b), and Vh (c) there are no differences between them, but there are significant differences (a) Ec and (d) Vp. Figure D no differences between (b) Sp and Vh, (c) Vp and Sa, and significant differences compared to (a) Ec. Figure E, no differences between (a) Sp and Sa, and significant differences in comparison with (c) Vp (p > 0.05) (Tukey HSD).
Figure 1 Halo of inhibition (HI) caused by the octopus mucus extract and two antibiotics on the growth of different selected pathogenic microbial strains. Ec (E. coli), Sa (S. aureus), Vh (V. harveyi), Sp (S. pasteuri), Vp (V. parahaemolyticus).
Conclusions
In the present study, it has been demonstrated that the skin mucus extract of the cephalopod O. bimaculatus has antimicrobial activity against bacteria pathogenic to fish, crustaceans, mollusks, and bovine, and it has been determined that the extract is rich in protein. This suggests that the mucus extract or subsequently purified antimicrobial compounds can be used as an alternative to antibiotics and could perhaps be used in aquaculture and terrestrial farms for chickens, bovine, etc. Since this extract is a natural product, it would help to reduce the problems of resistance to antibiotics that we currently have.
















