Introduction
Carotenoids are natural hydrophobic pigments (Correâ-Filho et al., 2019) found in various fruits and vegetables, but they can also be synthesized by microorganisms (Mahfoudhi and Hamdi, 2015). However, carotenoids cannot be synthesized by humans, making their intake through external sources necessary (Bockuviene and Sereikaite, 2020). Among carotenoids, β-carotene is one of the most widely distributed in plant-based foods, including fruits and vegetables (Correâ-Filho et al., 2019) and is recognized as a provitamin A source. Due to its antioxidant properties, β-carotene offers health benefits, such as reducing the risk of cardiovascular diseases, diabetes, and preventing macular degeneration (Bockuviene and Sereikaite, 2020). However, β-carotene has low chemical stability, poor water solubility, and low bioavailability (Lin et al., 2021). Additionally, it is sensitive to factors like oxygen, light, and heat (Zhu et al., 2021). Maintaining the stability of β-carotene is a challenge for industry.
The microencapsulation emerged as a technique that protects compounds from external factors, providing them with greater stability during storage and allowing for controlled release in the gastrointestinal system (Lin et al., 2021). In this process, microencapsulation involves forming a protective layer around the compounds (Al-Maqtari et al., 2021). Common wall materials used for this purpose include biopolymers, such as proteins and carbohydrates (Mahfoudhi and Hamdi, 2015). Maltodextrin, is often used as a wall material in microencapsulation because it offers good protection against oxidation. However, its main drawback is its low emulsifying capacity, which is why it is typically combined with other polymers (Carneiro et al., 2013). In this regard, gums are widely used due to their solubility, low viscosity, emulsifying properties, and relatively low cost (Mahfoudhi and Hamdi, 2015). Gum Arabic, consists of approximately 2 % proteins and a high proportion of carbohydrates. Because of this composition, gum Arabic has been extensively used in microencapsulation for its emulsifying properties (Correâ-Filho et al., 2019). Other authors have used proteins, such as soy protein, which has been shown to create more resistant and stable coatings than other plant-based proteins, in addition to its emulsifying properties (Al-Maqtari et al., 2021).
There are several techniques for microencapsulation, however, spray drying and freeze-drying are the most commonly used (Mahfoudhi and Hamdi, 2015). The choice of method depends on the properties of the desired final products. Freeze-drying is widely used for drying heat-sensitive compounds, as it relies on the sublimation of water from a frozen product without high temperatures (Al-Maqtari et al., 2021). Although there is extensive research on the use of freeze-drying for the encapsulation of ingredients, only a few studies (Mahfoudhi and Hamdi, 2015; Bockuviene and Sereikaite, 2020; Lin et al., 2021; Zhu et al., 2021) focus on β-carotene encapsulation through freeze-drying using soy protein isolate and soy milk. To our knowledge, none of the published studies report on the influence of different types of wall materials on the encapsulation efficiency, stability, and bioaccessibility of freeze-dried microencapsulated β-carotene during storage. Therefore, the objective of this work was to study the encapsulation of β-carotene by freeze-drying, using maltodextrin and mixtures with other encapsulating agents, evaluating encapsulation efficiency, as well as the stability of the encapsulated β-carotene during storage at 25 °C and 4 °C, antioxidant activity, and bioaccessibility.
Material and methods
Materials
Maltodextrin and gum Arabic were provided by IMSA and MEYER Laboratories ®, Mexico, respectively. The food-grade soy protein was supplied by Mundo del Sabor, Mexico. β-carotene (C9750, synthetic, ≥ 93 %), 1,1-diphenyl-2-picrylhydrazyl (DPPH), pancreatin (porcine pancreas, P1500), and bile salts (bovine bile, B3883) were purchased from Sigma Aldrich, USA. Pepsin was supplied by Matheson Coleman and Bell Manufacturing Chemists, USA. All chemicals used were of analytical grade and stored according to the manufacturer’s instructions.
The soy milk was prepared according to Mendoza-Avendaño et al. (2019), with slight modifications. The soybeans, washed with tap water and soap, were immersed in boiling water at a 1:4 (w/v) ratio for 5 min, then blended for 3 min with water in the same 1:4 (w/v) proportion. The suspension was filtered through a fine cloth and the resulting liquid, known as soy milk, was pasteurized at 12 psi for 12 min. Finally, the soy milk was freeze-dried for 48 h at - 40 °C with a chamber pressure of 0.280 mbar using a Labconco freeze dryer (FreeZone 4.5, USA) and stored at 4 °C.
Preparation of the dispersions
The encapsulating agents were dissolved in distilled water at room temperature and hydrated for 24 h under refrigeration. Afterward, the wall material solutions were autoclaved at 12 psi for 12 min. Subsequently, the encapsulating agent solutions were mixed using an Ultra-Turrax homogenizer (MA-102, USA) to ensure that the total solid concentration in each dispersion was 20 % (w/v). The composition of the dispersions is shown in Table 1. For treatment T1, maltodextrin was stirred at 10,000 rpm for 30 min, after which β-carotene was added at a concentration of 80 mg/100 mL of solution. For subsequent treatments (T2, T3, T4 and T5), maltodextrin was first added and stirred for 3 min at 10,000 rpm, then the other encapsulating agents were incorporated one by one, and the mixture was homogenized for 30 m. Finally, β-carotene was added at a concentration of 80 mg/100 mL. Stirring in all cases was carried out using the Ultra-Turrax (MA-102, USA).
Table 1 The formulations used in this study were based on the following encapsulating agents: Maltodextrin, gum Arabic, soy milk, soy protein.
Tabla 1 Formulaciones utilizadas a base de Maltodextrina, goma Arábiga, leche de soya y proteína de soya.
| Treatments | Concentration (g/100 mL) | |||
|---|---|---|---|---|
| Maltodextrin (MD) | Gum Arabic (GA) | Soy milk (SM) | Soy protein (SP) | |
| T1 | 20 | - | - | - |
| T2 | 10 | 10 | - | - |
| T3 | 10 | - | 10 | - |
| T4 | 10 | - | - | 10 |
| T5 | 10 | 3.33 | 3.33 | 3.33 |
- Not added.
- No adicionado.
Production of microcapsules by freeze-drying
The previously prepared emulsions were frozen using liquid nitrogen and subsequently freeze-dried for 48 h at -40 ºC with a chamber pressure of 0.280 mbar. The dry powders (microcapsules) were collected, crushed in a mortar and placed in vacuum-sealed metallized bags.
The yield process (YP) for freeze-drying was calculated using the methodology of Al-Maqtari et al. (2021) with the following equation (1):
Physical and physicochemical properties of the microcapsules
Encapsulation efficiency (EE)
The encapsulation efficiency (EE) was determined based on the ratio of the surface β-carotene content to the total β-carotene content, following the methodology of Correâ-Filho et al. (2019) with some modifications. To determine the total β-carotene, 10 mg of microcapsules were dissolved in 2 mL of distilled water and stirred for 3 min using a vortex (IKA®, Germany). Subsequently, 25 mL of acetone were added, which was then stirred for 3 min. Afterward, the mixture was centrifuged at 2,650 g for 20 min (centrifuge Z 326 K, Hermle Labortechnik, Germany). For the determination of surface β-carotene, 10 mg of microcapsules were dispersed in 10 mL of acetone and stirred for 3 min using a vortex. The mixture was then centrifuged at 2,650 g for 20 min. The total and surface β-carotene content were quantified using UV-VIS spectrophotometry at 450 nm (spectrophotometer DU® 730, Beckman Coulter®, USA). The EE was calculated using equation (2):
Moisture content and water activity (aw)
The moisture content of the microcapsules was determined using the methodology outlined in NOM-116-SSA1-1994, while the water activity was measured with a portable hygrometer (HygroPalm AW1, Rotronic, Switzerland).
Water solubility index (WSI)
The WSI was determined according to Navarro-Flores et al. (2020) where one gram of microcapsules was dispersed in 12 mL of water and stirred for 3 min. The mixture was incubated in a water bath at 30 °C for 30 min, followed by centrifugation at 2,030 g for 10 min. Finally, the supernatant was dried at 105 °C until a constant weight was achieved. The WSI was calculated according to equation (3):
Fourier transform infrared spectroscopy
Fourier Transform Infrared Spectroscopy (FTIR, Thermo Fisher Nicolet™ iS™ 10, USA) was used to obtain the infrared spectra of the microcapsules. For that, 40 scans were performed with a spectral resolution of 4 cm⁻¹, and each spectrum was recorded in the wavenumber range of 400 to 4,000 cm⁻¹ (Martínez-Molina et al., 2022).
Morphology of the microcapsules
The morphology of the microcapsules was examined using scanning electron microscopy with a JEOL equipment (JCM7000, USA). The microcapsules were placed on a sample holder and vacuum-coated with a thin layer of gold. Finally, the samples were scanned at 10 kV (Martínez-Molina et al., 2022).
Antioxidant activity (AA)
According to Tomovska et al. (2016), the AA of the microcapsules was determined using the DPPH free radical method, with some modifications. A sample of 2 mg was diluted in 6 mL of distilled water, followed by the addition of 3 mL of a methanolic DPPH solution (50 μM). For the blank, a sample of 2 mg was diluted in 6 mL of distilled water, and then 3 mL of methanol were added. For the control, 6 mL of water was mixed with 3 mL of the DPPH solution. The mixtures were stirred and allowed to rest in the dark for 30 min. The absorbance was then measured at 517 nm. The results were expressed as a percentage of DPPH radical scavenging activity and calculated using equation (4).
Storage stability of microcapsules
The microcapsules were stored at 4 ºC and 25 ºC in the dark in vacuum-sealed bags. Refrigerated storage was conducted for 90 days, with samples taken every 30 days, while storage at room temperature lasted for 45 d, with samples taken every 15 days. This was done to determine the stability of the microcapsules by monitoring the β-carotene content, percentage of release, and measuring AA during storage.
Gastrointestinal simulation
The microcapsules were subjected to conditions similar to those of the gastrointestinal tract, following the in vitro methodology of Picot and Lacroix (2004). For gastric simulation, 50 mg of the microcapsules were dispersed in 10 mL of gastric juice. The gastric juice was prepared by dissolving 0.26 g/L of pepsin in sterile distilled water, adjusting the pH to 1.9 with a 0.1 N HCl solution. The microcapsules exposed to gastric juice were incubated with stirring at 150 rpm for 30 min at 37 ºC. Subsequently, 20 mL of hexane were added, the solution was hand shaken, and then centrifuged at 2,650 g for 20 min at 8 ºC.
For the intestinal simulation, the process from the gastric phase was repeated for another sample of 50 mg of microcapsules, and subsequently, the pH was increased to 7.5 using a 1 N NaOH solution. The resulting solution was supplemented with 1.2 mL of concentrated sodium phosphate buffer solution (0.25 M, pH 7.5) and 0.53 mL of bile salt solution (150 g/L). The pH was adjusted to 7.5, and the volume was brought to 24 mL using distilled water. Finally, 2.67 mL of a pancreatin solution (2.44 g/L) in sodium phosphate buffer (0.02 M, pH 7.5) were added, achieving a final volume of 26.67 mL. The mixture was incubated at 150 rpm for 6 h at 37 °C. Then, 20 mL of hexane were added, the solution was vortexed, and centrifuged at 2,650 g for 20 min at 8 °C.
The β-carotene content in the organic fraction of the gastric and intestinal phases was determined using high-performance liquid chromatography (HPLC) under the conditions detailed in the following section.
Quantification of β-carotene by HPLC
The quantification of β-carotene during gastrointestinal simulation was performed following the methodology of Daood et al. (2014), using HPLC coupled to a UV-Vis detector and a C18 column (3 μm, 250 × 4.6 mm). The separation of β-carotene was achieved using a gradient elution of (A) methanol-water (93:7), (B) methanol, and (C) 30 % 1,2-dichloromethane in methanol. The elution started with 100 % A, then shifted to 100% B in 5 min, followed by 100 % C in 20 min, and finally returned to 100 % A within 5 min. The flow rate was set at 0.7 mL/min, and the detection occurred at 450 nm. For quantification, the β-carotene peak area at the maximum absorption wavelength was measured, and its concentration was determined using a standard curve of β-carotene C9750 (Sigma Aldrich, USA).
Experimental design and statistical analysis
The experiments were conducted using a completely randomized experimental design in triplicate. Data were analyzed through an analysis of variance (ANOVA) using the Statgraphics Centurion® statistical software package. Mean comparisons were performed using Tukey’s test (p ≤ 0.05) to determine significant differences among treatments.
Results and discussion
The microcapsules were characterized in terms of their physical and physicochemical properties after freeze-drying and throughout storage at 25 °C and 4 °C.
Physical and physicochemical properties of the microcapsules
Yield process (YP)
The YP values ranged from 82.78 % to 88.95 %. The results showed no statistically significant difference (p ≤ 0.05) in the process yield concerning the different encapsulating agents mixtures (Table 2). These findings are consistent with those reported by Al-Maqtari et al. (2021), who encapsulated a plant extract through freeze-drying using different encapsulating agents, reporting yields between 83.16 % and 87.95 %. Similarly, Sousa de Oliveira et al. (2020) achieved YP between 85.3 % and 88.5 % using various plant proteins to encapsulate oil through freeze-drying.
Table 2 Yield process (YP), encapsulation efficiency (EE), moisture content, water activity, and water solubility index of β-carotene micro-encapsulated with different wall materials.
Tabla 2 Rendimiento de proceso (YP), eficiencia de encapsulación (EE), contenido de humedad, actividad de agua e índice de solubilidad en agua de β-caroteno microencapsulado con diferentes materiales de pared.
| Variables | T1 | T2 | T3 | T4 | T5 | Tukey |
|---|---|---|---|---|---|---|
| YP (%) | 88.95±2.67 a | 83.11±3.41 a | 82.78±2.25 a | 84.43±0.54 a | 86.72±4.44 a | 7.96 |
| EE (%) | 24.50±2.98 b | 50.40±3.96 a | 23.50±0.82 b | 49.18±5.18 a | 15.90±0.54 b | 8.70 |
| Moisture content (%) | 5.70±1.0 a | 5.54±0.18 a | 5.15±0.08 a | 3.60±0.23 b | 5.06±0.1 a | 1.26 |
| aw | 0.23±0.0 a | 0.11±0.0 d | 0.20±0.0 b | 0.09±0.0 e | 0.14±0.0 c | 0.003 |
| WSI (%) | 92.32±0.12 a | 92.33±0.47 a | 67.22±1.86 c | 86.86±1.15 b | 65.57±0.14 c | 2.97 |
Treatments with maltodextrin (T1), maltodextrin-gum Arabic (T2), maltodextrin-soy milk (T3), maltodextrin-soy protein (T4), and a mixture of all agents (T5). Means followed by different lowercase letters in the same column are significantly different (p ≤ 0.05) according to Tukey’s test.
Tratamientos con maltodextrina (T1), maltodextrina-goma Arábiga (T2), maltodextrina-leche de soya (T3), maltodextrina-proteína de soya (T4), y mezcla de todos los agentes (T5). Medias seguidas de diferentes letras minúsculas en una misma columna son significativamente diferentes (p ≤ 0.05) según la prueba de Tukey.
Encapsulation efficiency (EE)
In microencapsulation processes, EE is an important criterion for determining process efficiency. The highest EE (50.40 ± 3.96 %) was achieved with the maltodextrin-gum Arabic mixture (Table 2). This is likely due to gum Arabic’s emulsifying properties (Mahfoudhi and Hamdi, 2015; Carneiro et al., 2013; Al-Maqtari et al., 2021). Correâ-Filho et al. (2019) suggest that carotenoids may hydrophobically interact with the hydrophobic protein regions present in gum Arabic.
Like T2, the T4 treatment (maltodextrin-soy protein) also exhibited high EE (49.18 ± 5.18 %). This can be attributed to the pre-treatment of the agents, which involved pasteurization. The heating of soy protein probably exposed more hydrophobic amino acid residues on the protein’s surface, likely interacting via hydrophobic bonds with β-carotene (Zhang et al., 2021). However, T5 treatment, which contained a mixture of all encapsulating agents, unexpectedly resulted in a lower EE (15.36 ± 0.54 %). This may be due to greater interactions between the encapsulating agents themselves rather than with β-carotene.
Moisture content
The moisture content of a given food is often linked to its stability, quality, and composition, which could influence its storage, packaging, and processing (Yamashita et al., 2017). The moisture content of the microcapsules ranged from 3.6 % to 5.7 % (Table 2). These values are considered safe, as Fredes et al. (2018) reported that the acceptable moisture content for powdered foods is between 3 % and 10 %. These results are similar to those reported by Mahfoudhi and Hamdi (2015), who found moisture values of approximately 2 % in microcapsules using gum Arabic and gum almond in freeze-dried samples.
Water activity (aw)
Water activity plays a crucial role in determining both quality changes and microbial growth or survival (Pan-utai and Iamtham, 2020). Values below 0.6 indicate that the product is safe from microbial growth (Zhu et al., 2021). As observed in Table 2, treatment T1 has the highest aw value (0.23 ± 0.0) compared to the other treatments (p ≤ 0.05). These results are similar to those reported by Zhu et al. (2021), who obtained values of 0.23 - 0.234 using modified starch to coencapsulate β-carotene through freeze-drying. Al-Maqtari et al. (2021) suggest that the differences in aw values among formulations may be due to the chemical composition of the encapsulating agents used and the drying time.
Water solubility index (WSI)
Solubility is the final stage of particle dissolution, which is also a key factor in food quality and is assessed by testing the dissolution rate of microparticles in water (Konaré et al., 2023). The WSI is also related to the reconstitution of the powder (Pan-utai and Iamtham, 2020) in suspensions. The WSI for T1 and T2 were around 92 % (Table 2), similar to those reported by Mahfoudhi and Hamdi (2015). These results may be attributed to the fact that maltodextrin and gum Arabic are carbohydrates with a high capacity for water absorption (Sousa de Oliveira et al., 2020; Al-Maqtari et al., 2021). In contrast, for T3, T4, and T5 treatments, the WSI values were lower (Table 2), probably because soy milk and soy protein influenced the water absorption time (Sousa de Oliveira et al., 2020). Soy protein is partially soluble in water, which leads to a decrease in the WSI of microparticles (Fredes et al., 2018). During the pasteurization of soy milk, soy proteins tend to denature due to heating and interact with each other, causing globulins to become less soluble in water, as reported by Nishinari et al. (2014).
Fourier Transform Infrared Spectroscopy (FTIR)
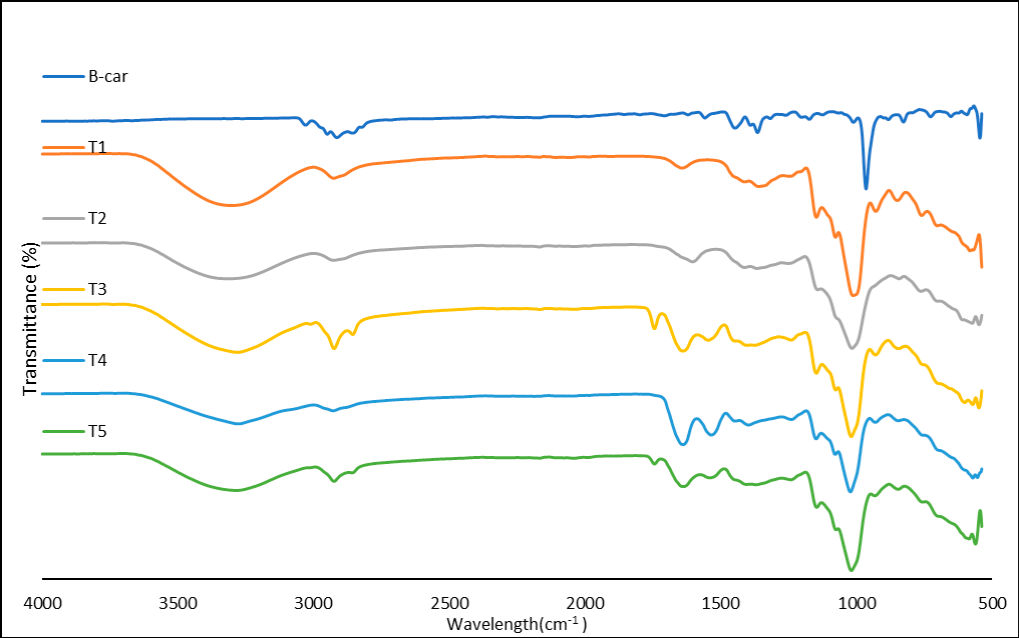
Encapsulation efficiency is probably related to the possible chemical interactions between the compound to be encapsulated and the encapsulating agents. Therefore, it is important to know what chemical interactions were observed in the freeze-dried samples. The intermolecular interactions between β-carotene and the encapsulating agents were studied using FTIR (Figure 1). The spectrum for β-carotene shows signals at 2,948 and 2,913 cm⁻¹, as well as at 1,446 cm⁻¹ corresponding to the C-H bond, at 1,364 cm⁻¹ corresponding to the terminal methyl group (-CH₃), at 3,028 cm⁻¹ indicating the presence of unsaturations, at 1,557 and 1,620 cm⁻¹ demonstrating the presence of polyene, and at 964 cm⁻¹ indicating trans conjugation. These signals are similar to those reported by Zhang et al. (2021) and Lin et al. (2021).
Treatment T1 shows signals at 3,300 cm⁻¹ indicating the presence of O-H groups, as well as at 2,924 and 1,359 cm⁻¹ indicating C-H bonds characteristic of carbohydrates, at 701 cm⁻¹ corresponds to -(CH₂)-n chains, with n ≥ 4, and a signal at 1,012 cm⁻¹. Additionally, treatment T2 presents signals at 3,300 cm⁻¹ indicating O-H groups; 2,925 and 1,365 cm⁻¹ C-H bonds characteristic of carbohydrates, and 761 cm⁻¹ for -(CH₂)-n chains, with n ≥ 4; and a signal at 1,016 cm⁻¹. These results are similar to those reported by Mansour et al. (2020), who used gum Arabic as an encapsulating agent. On the other hand, treatment T3 has signals at 3,281 cm⁻¹ indicating O-H groups, which may also relate to amine signals and nitro groups possibly associated with the lecithin from soy milk, 1,743 and 1,638 cm⁻¹ are characteristic of carbonyl groups, 2,923 and 2,855 cm⁻¹, as well as 1,374 cm⁻¹, indicate C-H bonds, and a signal at 1,019 cm⁻¹.
Conversely, treatment T4 shows signals at 3,277 cm⁻¹ that are characteristic of the stretching vibration of O-H and N-H bonds, similar to those reported by Zhang et al. (2021), who used whey protein. Signals at 1,638 and 1,534 cm⁻¹ are characteristic of C=O and N-H bonds, respectively, as also reported by Mansour et al. (2020), at 1,396 and 1,239 cm⁻¹ are characteristic of the carboxyl group, possibly related to the amino acids of the protein or attributed to C-N stretching and N-H bending vibrations of amide III, as reported by Mansour et al. (2020), and a signal at 2,927 cm⁻¹ relates to C-H bonds, as well as a signal at 1,022 cm⁻¹.
Meanwhile, treatment T5 presents signals at 3,283 cm⁻¹ characteristic of the stretching vibration of O-H and N-H bonds, at 1,636 and 1,539 cm⁻¹ correspond to N-H bonds, due to soy protein and milk, at 1,743 and 1,638 cm⁻¹ are characteristic of carbonyl groups, at 2,924 and 1,369 cm⁻¹ indicate C-H bonds of carbohydrates, and finally, a signal at 1,018 cm⁻¹.
Finally, the signal at 966 cm⁻¹ of β-carotene was likely shifted, with signals appearing between 1,012 and 1,022 cm⁻¹ in all treatments after encapsulation due to possible interactions between β-carotene and the encapsulating agents. Lin et al. (2021) reported that all β-carotene signals were restricted after encapsulation, possibly due to hydrophobic interactions between β-carotene and the encapsulating materials.
Microstructure of the microcapsules
Although freeze-drying allows us to obtain highly porous products and, therefore, enable quick hydration, it was important for us to know this structure. All samples (Figure 2) exhibited fragmented, laminar, and rough structures with a smooth surface, similar to those reported for freeze-dried materials (Lin et al., 2021; Zhu et al., 2021; Stoica et al., 2022). The rough structure of the microparticles is often attributed to their contraction after significant moisture loss and subsequent cooling, as reported by Stoica et al. (2022). Al-Maqtari et al. (2021) noted that products obtained by freeze-drying displayed a fragmented structure with irregular shapes of varying sizes. This is probably due to the crushing process after drying as reported by Sousa de Oliveira et al. (2020). The amorphous and irregular structures can protect the trapped molecules from high temperatures and oxygen exposure as reported by Konaré et al. (2023).
Stability of microcapsules during storage
So far, we have seen that encapsulating agents change the properties of microcapsules. However, it is important to know the capacity of encapsulating agents for preserving β-carotene. Table 3 shows that as storage time increases, degradation also increases. The stability of β-carotene was influenced by the combination of materials used and storage temperature (Table 3). In Table 3, it is also noted that treatment T3 exhibits the highest concentration of β-carotene (3.80 µg/g) at the end of storage (45 d), while treatment T5 shows the lowest concentration of β-carotene (2.47 µg/g). The pores generated during freeze drying may pose a challenge during prolonged storage, as a greater number of pores leads to a larger surface area for oxygen contact and diffusion, which can decrease oxidative stability, as reported by Mahfoudhi and Hamdi (2015). Another possible reason for the reduction in β-carotene concentration in the microcapsules is auto-oxidation. Mordi et al. (2020) mentioned that the auto-oxidation of β-carotene may start with β-carotene in its trans isomer form transitioning to its cis form, a process that can easily occur at 30 ºC. The twisting of the molecule during isomerization can lead to an unpaired electron state, which can readily react with oxygen from the carotenoid itself to form oxidation products.
Table 3 Concentration of β-carotene in microcapsules obtained by freeze-drying stored at room temperature.
Tabla 3 Concentración de β-caroteno en microcápsulas obtenidas por liofilización almacenadas a temperatura ambiente.
| Concentration (mg β-carotene/g powder) | ||||||
|---|---|---|---|---|---|---|
| Time (Days) | T1 | T2 | T3 | T4 | T5 | Tukey |
| 0 | 3.85±0.14 Ca | 4.36±0.02 Ba | 4.94±0.06 Aa | 4.05±0.03 Ca | 4.39±0.06 Ba | 0.20 |
| 15 | 3.40±0.03 Db | 3.90±0.07 Bb | 4.64±0.06 Aa | 2.99±0.03 Eb | 3.73±0.04 Cb | 0.13 |
| 30 | 3.31±0.07 Cb | 3.80±0.14 Bb | 4.18±0.19 Ab | 2.83±0.06 Dc | 2.96±0.11 Dc | 0.34 |
| 45 | 3.21±0.10 Bb | 3.64±0.13 Ab | 3.80±0.09 Ac | 2.68±0.02 Cd | 2.47±0.05 Cd | 0.23 |
| Tukey | 0.25 | 0.26 | 0.30 | 0.10 | 0.19 | |
Treatments with maltodextrin (T1), maltodextrin-gum Arabic (T2), maltodextrin-soy milk (T3), maltodextrin-soy protein (T4), and a mixture of all agents (T5). Means followed by different lowercase letters in the same column are significantly different (p ≤ 0.05) according to Tukey’s test. The means in the same row followed by different uppercase letters are significantly different (p ≤ 0.05).
Tratamientos con maltodextrina (T1), maltodextrina-goma Arábiga (T2), maltodextrina-leche de soya (T3), maltodextrina-proteína de soya (T4), y mezcla de todos los agentes (T5). Las medias en la misma columna seguidas por diferentes letras minúsculas son significativamente diferentes (p ≤ 0.05). Las medias en la misma fila seguidas por diferentes letras mayúsculas son significativamente diferentes (p ≤ 0.05).
At refrigeration temperature, the decrease in carotenoid concentration in the microcapsules was also observed but was slower (Table 4) compared to the microcapsules stored at room temperature. In Table 4, the treatment T3 exhibits the highest concentration of β-carotene (4.14 µg/g) at the end of storage (90 days), while treatment T4 shows the lowest concentration (3.27 µg/g). The freezing temperature is also an important factor in carotenoid degradation. Rezvankhah et al. (2020) mentioned that, the freezing method before freeze drying should be considered as a critical point because it can alter the stability of the microcapsules. Thus, a lower freezing temperature provided greater stability to the freeze-dried β-carotenoid particles, as in our case where we used immersion freezing in liquid nitrogen.
Table 4 Concentration of β-carotene in microcapsules obtained by freeze-drying stored at refrigeration temperature.
Tabla 4 Concentración de β-caroteno en microcápsulas obtenidas por liofilización almacenadas a temperatura de refrigeración.
| Concentration (mg β-carotene/g powder) | ||||||
|---|---|---|---|---|---|---|
| Time (Days) | T1 | T2 | T3 | T4 | T5 | Tukey |
| 0 | 3.85±0.14 Ca | 4.36±0.02 Ba | 4.94±0.06 Aa | 4.05±0.03 Ca | 4.39±0.06 Ba | 0.20 |
| 30 | 3.79±0.27 Ca | 4.1±0.06 BCb | 4.48±0.09 ABb | 4.06±0.02 Ca | 4.26±0.05 ABb | 0.36 |
| 60 | 3.53±0.37 Ca | 4.02±4.4 ABb | 4.4±06 Ab | 3.63±0.0 BCb | 3.85±0.05 BCc | 0.45 |
| 90 | 3.57±0.04 Ca | 4.03±0.05 Ab | 4.14±0.04 Ac | 3.27±0.06 Dc | 3.85±0.02 Bc | 0.12 |
| Tukey | 0.63 | 0.10 | 0.17 | 0.09 | 0.12 | |
Treatments with maltodextrin (T1), maltodextrin-gum Arabic (T2), maltodextrin-soy milk (T3), maltodextrin-soy protein (T4), and a mixture of all agents (T5). Means followed by different lowercase letters in the same column are significantly different (p ≤ 0.05) according to Tukey’s test. The means in the same row followed by different uppercase letters are significantly different (p ≤ 0.05).
Tratamientos con maltodextrina (T1), maltodextrina-goma Arábiga (T2), maltodextrina-leche de soya (T3), maltodextrina-proteína de soya (T4), y mezcla de todos los agentes (T5). Las medias en la misma columna seguidas por diferentes letras minúsculas son significativamente diferentes (p ≤ 0.05). Las medias en la misma fila seguidas por diferentes letras mayúsculas son significativamente diferentes (p ≤ 0.05).
Antioxidant activity (AA)
At the beginning of storage, the highest AA of 28.75 % (Table 5) was observed with treatment T3 (maltodextrin-soy milk). These results are attributed not only to the AA of β-carotene but also to the phenolic and antioxidant compounds, likely present in the soy milk and protein. The antioxidant effect of soy milk is partly due to phospholipids, the main components of lecithins, which can donate a hydrogen atom from their amino group derived from choline and ethanolamine, granting them radical-scavenging properties (Judde et al., 2003). Additionally, the AA of the microcapsules decreased during storage, with a greater reduction in microcapsules stored at room temperature compared to those stored under refrigeration (Tables 5 and 6).
Tabla 5 Actividad antioxidante (AA) de las microcápsulas de β-caroteno almacenado a temperatura ambiente.
| Antioxidant activity at room temperature (%) | ||||||
|---|---|---|---|---|---|---|
| Time (Days) | T1 | T2 | T3 | T4 | T5 | Tukey |
| 0 | 4.66±0.21 Ea | 5.85±0.17 Da | 28.75±0.17 Aa | 25.41±0.48 Ba | 22.47±0.31 Ca | 0.78 |
| 15 | 4.16±0.03 Db | 4.66±0.23 Db | 28.26±0.4 Aa | 24.84±0.94 Ba | 21.38±0.67 Cab | 1.52 |
| 30 | 3.67±0.11 Dc | 3.99±0.41 Dc | 26.30±0.80 Ab | 23.89±0.70 Bab | 21.00±0.67 Cb | 1.59 |
| 45 | 3.48±0.24 Dc | 4.14±0.05 Dbc | 24.99±0.31 Ac | 22.80±0.39 Bb | 20.75±0.38 Cb | 0.81 |
| Tukey | 0.44 | 0.66 | 1.29 | 1.73 | 1.39 | |
Treatments with maltodextrin (T1), maltodextrin-gum Arabic (T2), maltodextrin-soy milk (T3), maltodextrin-soy protein (T4), and a mixture of all agents (T5). Means followed by different lowercase letters in the same column are significantly different (p ≤ 0.05) according to Tukey’s test. The means in the same row followed by different uppercase letters are significantly different (p ≤ 0.05).
Tratamientos con maltodextrina (T1), maltodextrina-goma Arábiga (T2), maltodextrina-leche de soya (T3), maltodextrina-proteína de soya (T4), y mezcla de todos los agentes (T5). Las medias en la misma columna seguidas por diferentes letras minúsculas son significativamente diferentes (p ≤ 0.05). Las medias en la misma fila seguidas por diferentes letras mayúsculas son significativamente diferentes (p ≤ 0.05).
Tabla 6 Actividad antioxidante (AA) de las microcápsulas de β-caroteno almacenado a temperatura de refrigeración.
| Antioxidant activity at refrigeration temperature (%) | ||||||
|---|---|---|---|---|---|---|
| Time (Days) | T1 | T2 | T3 | T4 | T5 | Tukey |
| 0 | 4.66±0.21 Ea | 5.85±0.17 Da | 28.18±0.46 Aa | 25.41±0.48 Ba | 22.47±0.31 Ca | 0.78 |
| 30 | 4.17±0.04 Dab | 5.59±0.12 Da | 27.51±0.84 Aa | 24.94±0.90 Bab | 21.91±0.60 Ca | 1.66 |
| 60 | 3.68±0.31 Ebc | 5.58±0.11 Da | 27.20±0.67 Aab | 24.21±0.79 Bab | 21.75±0.76 Ca | 1.60 |
| 90 | 3.52±0.27 Ec | 5.23±0.03 Db | 25.36±0.94 Ab | 23.38±0.27 Bb | 21.43±0.07 Ca | 1.22 |
| Tukey | 0.60 | 0.31 | 1.88 | 1.73 | 1.33 | |
Treatments with maltodextrin (T1), maltodextrin-gum Arabic (T2), maltodextrin-soy milk (T3), maltodextrin-soy protein (T4), and a mixture of all agents (T5). Means followed by different lowercase letters in the same column are significantly different (p ≤ 0.05) according to Tukey’s test. The means in the same row followed by different uppercase letters are significantly different (p ≤ 0.05).
Tratamientos con maltodextrina (T1), maltodextrina-goma Arábiga (T2), maltodextrina-leche de soya (T3), maltodextrina-proteína de soya (T4), y mezcla de todos los agentes (T5). Las medias en la misma columna seguidas por diferentes letras minúsculas son significativamente diferentes (p ≤ 0.05). Las medias en la misma fila seguidas por diferentes letras mayúsculas son significativamente diferentes (p ≤ 0.05).
Similar to T3, treatments T4 (maltodextrin-soy protein) and T5 also exhibited high AA (25.41 and 22.47 %, respectively) at the beginning of storage (Table 5). These values, in addition to the contribution of β-carotene, are attributed to the soy protein. The degradation of soy protein caused by pasteurization exposed more amino acids, which likely contributed some antioxidant activity (Zhang et al., 2021). On the other hand, the AA of treatments T1 and T2 was the lowest (< 6 %). This effect could be attributed to the β-carotene since the AA of maltodextrin and gum Arabic is negligible.
Bioaccessibility
The bioaccessibility for treatments T1, T4, and T5 showed no statistically significant difference (p ≤ 0.05) between the gastric and intestinal phases (Table 7), indicating that β-carotene was released in the gastric phase, and no further release occurred in the intestinal phase. These results are likely due to the protective effects of the encapsulating agents, particularly maltodextrin and soy protein, which are substances that disperse in the upper gastrointestinal tract, as reported by Rezvankhah et al. (2020). Additionally, soy protein is highly sensitive to pH changes and to gastric pepsin, which hydrolyzes proteins, as well as to enzymes secreted by the pancreas in the small intestine used in the simulation (Biehler and Bohn, 2010; Rezvankhah et al., 2020). These results align with Konaré et al. (2023), who reported that the bioaccessibility of encapsulated compounds is influenced by the encapsulating agents, the nature of their combination, as well as the structure of the capsules and the encapsulation method.
Tabla 7 Bioaccesibilidad de β-caroteno microencapsulado.
| Bioaccessibility (%) | |||
|---|---|---|---|
| Treatment | Gastric phase | Intestinal phase | Tukey |
| T1 | 19.86±2.22 Ab | 18.74±2.60 Ab | 5.48 |
| T2 | 17.33±1.68 Bb | 23.84±1.48 Ab | 3.59 |
| T3 | 31.53±1.90 Ba | 53.63±5.29 Aa | 9.01 |
| T4 | 18.69±0.62 Ab | 21.49±1.85 Ab | 3.18 |
| T5 | 26.74±3.10 Aa | 24.59±0.73 Ab | 5.11 |
| Tukey | 5.56 | 7.70 | |
Treatments with maltodextrin (T1), maltodextrin-gum Arabic (T2), maltodextrin-soy milk (T3), maltodextrin-soy protein (T4), and a mixture of all agents (T5). Means followed by different lowercase letters in the same column are significantly different (p ≤ 0.05) according to Tukey’s test. The means in the same row followed by different uppercase letters are significantly different (p ≤ 0.05).
Tratamientos con maltodextrina (T1), maltodextrina-goma Arábiga (T2), maltodextrina-leche de soya (T3), maltodextrina-proteína de soya (T4), y mezcla de todos los agentes (T5). Las medias en la misma columna seguidas por diferentes letras minúsculas son significativamente diferentes (p ≤ 0.05). Las medias en la misma fila seguidas por diferentes letras mayúsculas son significativamente diferentes (p ≤ 0.05).
On the other hand, treatments T2 and T3 did show a statistically significant difference (p ≤ 0.05) in the release of microencapsulated β-carotene between the gastric and intestinal phases (Table 7). This is likely due to the fact that for treatment T2 (maltodextrin-gum Arabic), some polysaccharides, such as gum Arabic, are dietary fibers and therefore are not degraded in the upper regions of the gastrointestinal tract (Rezvankhah et al., 2020). Gum Arabic has been shown to be effective in increasing the bioaccessibility of carotenoids due to its emulsifying capacity, enhancing carotenoid solubility and the production of lipid micelles in the intestinal phase (Santos et al., 2021). Huang and Zhou (2019) found that the use of gum Arabic helped to delay the release of anthocyanins during in vitro simulated gastrointestinal digestion.
Moreover, T3 enabled approximately 85 % of β-carotene release during the gastric and intestinal phases (Table 7). For treatment T3, soy proteins were hydrolyzed by the enzymes present in intestinal juice, allowing greater release of carotenoids as reported by Rezvankhah et al. (2020). However, unlike treatment T4, the presence of phospholipids helps emulsify carotenoids, enhancing their solubility (Soukoulis and Bohn, 2018). Phosphatidylcholine, likely present in soy milk and one of the main phospholipids in lecithin, could increase the bioaccessibility of β-carotene, as phosphatidylcholine has a high emulsifying capacity, as reported by Mao et al. (2018). Bile is particularly important for emulsifying fats and forming mixed micelles that solubilize and transport lipophilic products (Minekus et al., 2014), such as β-carotene. Bile salts act as surfactants, reducing the size of the gastric emulsion by forming micelles that contain β-carotene in addition to free fatty acids, monoglycerides, and phospholipids (Donhowe and Kong, 2014). Similarly, pancreatin and pH seem to play a predominant role in micelle production (Biehler and Bohn, 2010).
Conclusions
This study investigated the microencapsulation of β-carotene through freeze-drying using various mixtures of encapsulating agents. The drying yields were statistically the same for all treatments. The maltodextrin-gum Arabic and maltodextrin-soy protein mixtures resulted in the highest encapsulation efficiency (about 50 %). Meanwhile, the maltodextrin-soy milk mixture maintained the highest concentration of β-carotene at the end of storage. Storage under refrigeration conditions preserved a higher concentration of β-carotene. Regarding antioxidant activity (AA), the maltodextrin-soy milk treatment demonstrated the highest AA as well as the highest bioaccessibility in the intestinal phase. Scanning electron microscopy revealed that the microcapsules obtained by freeze-drying exhibited a fragmented structure with irregular shapes of various sizes. Fourier transform infrared spectroscopy indicated that the characteristic signals of β-carotene were modified by the signals of the encapsulating agents, suggesting that β-carotene may have been encapsulated through hydrophobic interactions. β-carotene microcapsules could be used as supplements in the food industry for food formulation. In addition, in the pharmaceutical industry, our methodology could be used for the production of stable vitamin compounds for storage and controlled release. Moreover, the maltodextrin-soy protein (MD-SP) mixture could be used for the encapsulation of other fat-soluble antioxidant compounds, with potentially attractive results. Finally, the approach used in this study for the microencapsulation process of β-carotene via freeze-drying can be applied to other sensitive compounds.