Introduction
Bleeding is an important issue in cardiac surgery, about 20% of all blood products are transfused in this scenario around the world1. Bleeding as a complication is associated with poorer results in cardiac surgery. In a retrospective analysis that included 1188 cardiac surgery patients, the presence of excessive post-operative bleeding was associated with an increase incidence of surgical re-exploration (OR = 103.7, 95% CI = 45.6-235.4, p < 0.0001), post-operative stroke (OR = 3.3, 95% CI = 1.6-7-0, p = 0.003), mechanical ventilation for more than 24 h (OR = 3.4, 95% CI = 1.8-6.4, p = 0.0002), stay in the intensive care unit (ICU) for more than 72 h (OR = 1.4, 95% CI = 1.2-3.2, p < 0.0001), and increase mortality (OR = 2.9, 95% CI = 2.9-3.0, p < 0.001)2. Therefore, excessive post-operative bleeding is associated with an increase in hospital costs3.
In addition, there is growing evidence that the transfusion of blood products is an independent factor of increase morbidity, mortality, and hospital costs4,5.
Post-operative bleeding has been established as an important outcome measure in clinical practice and clinical trials. Despite having precise definitions for other complications in cardiac surgery, there is no standardized definition for post-operative bleeding, making difficult to interpret clinical trials and evaluate the management of blood products. We propose the implementation of the universal definition of perioperative bleeding (UDPB) in adult cardiac surgery to standardize nomenclature, improve outcome definitions, and be useful in future clinical trials.
Materials and methods
We conducted a retrospective descriptive and analytical study of one hospital. The data were obtained from a database of the cardiovascular ICU of the National Institute of Cardiology Ignacio Chavez in Mexico City. All adult patients ≥ 18 years old who underwent cardiac surgery from January 1, 2016, to December 31, 2017, were included in the study. Congenital cardiac surgery patients were excluded from the study. The information included in the database was supplemented with information obtained from the files if necessary. The primary objective of the study was to look at mortality associated with the degree of bleeding according to the classification of the UDPB (Table 1). Secondary objectives were to determine whether a greater degree of bleeding was related to an increase stay in the ICU, an increase in hospital stay, and increase events of stroke and acute kidney injury (AKI). The study was approved by the local ethics committee with a waiver of written informed consent.
Table 1 Universal definition for perioperative bleeding in adult cardiac surgery
| Bleeding categories according to the UDPB in adult cardiac surgery | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Bleeding definition | Sternal closure delayed | Post-operative chest tube blood los within 12 h (mL) | PRBC (units) | FFC (units) | PLT (units) | Cryoprecipitate | PCCs | rFVIIa | Re-exploration/tamponade |
| Class 0 (Insignificant) | No | < 600 | 0 | 0 | 0 | No | No | No | No |
| Class 1(Mild) | No | 601-800 | 1 | 0 | 0 | No | No | No | No |
| Class 2(Moderate) | No | 801-1000 | 2-4 | 2-4 | Yes | Yes | Yes | Yes | Yes |
| Class 3(Severe) | Yes | 1001-2000 | 5-10 | 5-10 | N/A | N/A | N/A | No | Yes |
| Class 4(Massive) | N/A | > 2000 | > 10 | > 10 | N/A | N/A | N/A | Yes | N/A |
UDPB: universal definition for perioperative bleeding; PRBC: packed red blood cells; FFP: fresh frozen plasma; PLT: platelet concentrates; PCCs: prothrombin complex concentrates; rFVIIa: recombinant activated factor VII; N/A: not applicable. The number of PRBCs used should only be considered in the UDPB when accompanied by other signs of perioperative bleeding. If different categories indicate mixed definitions of bleeding, the worst definition applies.
Statistical analysis
Continuous variables were represented with medians (interquartile ranges) and for categorical variables number (n) and percentages (%) were shown. Subsequently, patients were divided into five different groups according to the degree of bleeding using the UDPB. For comparisons of continuous variables MannWhitney U-test was used, and Fisher exact test for categorical variables. For those variables with statistical significance (a value ≤ 0.1) a multivariate logistic regression analysis was made, to determine if the degree of bleeding is independently associated with 30-day mortality. Crude mortality by class of bleeding was adjusted to cardiopulmonary bypass (CPB) duration and EuroScore II, which were the variables that were found with significance in the multimodal regression analysis.
Results
A total of 918 patients were obtained. The median age was 59 years, with a proportion of women of 42%. About 21.4% of patients previously used platelet antiaggregant. The highest percentage of surgeries performed in our hospital was valvular type (69.9%). About 32% of the surgeries were an emergency intervention. The overall mortality was 8.1%. Complications for stroke were found in 1% and AKI in 7.8% of the patients. Most of the population was classified as insignificant bleeding class (n = 666, 72.9%), and for massive bleeding the lowest proportion (n = 25, 2.7%) (Table 2).
Table 2 Patient characteristics
| Patient characteristics | |
|---|---|
| Total patients | 918 |
| Demographics | |
| Age (years) | 59 [50-67] |
| Sex female | 384 (42) |
| BMI | 26.7 [23.6-28.8] |
| Comorbidity | |
| High Blood Pressure | 387 (42) |
| Diabetes mellitus type 2 | 205 (22) |
| Chronic renal failure | 23 (2.5) |
| Left ventricular ejection fraction < 50% | 156 (17) |
| Preoperative therapy | |
| Aspirin | 155 (16.9) |
| Clopidogrel | 41 (4.5) |
| Warfarin | 39 (4.2) |
| Pre-operative labs | |
| Hemoglobin, mg/dL | 11.7 [10.2-13.1] |
| Creatinine, mg/dL | 0.9 [0.79-1.10] |
| Type of surgery | |
| Coronary artery bypass graft | 162 (17.6) |
| Valvular | 642 (69.9) |
| Coronary artery bypass graft + Valvular | 78 (8.5) |
| Aorta | 36 (3.9) |
| Operation details | |
| Elective surgery | 624 (68) |
| Emergency surgery | 294 (32) |
| EuroScore II, % | 2 [1.2-4.6] |
| CPB time, min | 120 [97-161] |
| Class of bleeding | |
| Class 0 (Insignificant) | 666 (72.9) |
| Class 1 (Mild) | 75 (8.2) |
| Class 2 (Moderate) | 70 (7.6) |
| Class 3 (Severe) | 82 (8.9) |
| Class 4 (Massive) | 25 (2.7) |
| Outcomes | |
| 30-day mortality | 74 (8.1) |
| Stroke | 9 (1) |
| AKI | 72 (7.8) |
| ICU length of stay, days | 4 [2-5] |
| Hospital length of stay, days | 23 [13-35] |
BMI: body mass index; LVEF: left ventricular ejection fraction; CABG: coronary artery bypass graft; CPB: cardiopulmonary bypass; AKI: acute kidney injury; ICU: intensive care unit. Data presented as median [interquartile range] for continuous variables and counts (percentages) for categorical variables.
In the comparison by groups defined by the class of bleeding, significant differences were found between the CBP and EuroScore II, both being higher in Class 4 compared to the rest. A significant difference was observed in the number of events of stroke and AKI, being more frequent in Class 4 (Table 3).
Table 3 Outcome in patients for bleeding class
| Outcome in patients for bleeding class | ||||||
|---|---|---|---|---|---|---|
| Class 0 | Class 1 | Class 2 | Class 3 | Class 4 | p value | |
| CRF | 17 (2.6) | 1 (1.3) | 3 (4.3) | 2 (2.4) | 0 | 0.74 |
| LVEF < 50% | 113 (17) | 9 (12) | 14 (20) | 17 (20.7) | 3 (12) | 0.55 |
| Aspirin | 116 (17.4) | 11 (14.7) | 11 (15.7) | 14 (17.1) | 3 (12) | 0.92 |
| Clopidogrel | 29 (4.4) | 3 (4) | 4 (5.7) | 5 (6.1) | 0 | 0.73 |
| Warfarin | 28 (4.2) | 1 (1.3) | 4 (5.7) | 4 (4.9) | 2 (8) | 0.57 |
| Creatinine, mg/dL | 0.8 [0.7-1.2] | 0.9 [0.8-1.1] | 0.9 [0.7-1.3] | 1 [0.8-1.1] | 0.9 [0.7-1.2] | 0.92 |
| Hemoglobin, mg/dL | 10.2 [9.7-11.9] | 10.1 [8.6-12.1] | 10.3 [9-13.6] | 11.8 [10-13.6] | 12.1 [9-14.6] | 0.45 |
| EuroScore II, % | 2.3 [1.5-6-8] | 1.8 [1.1-3.3] | 2 [1.2-3.4] | 2 [1.3-6.9] | 4 [1.8-8.3] | 0.003 |
| CPB time, min | 120 [100-140] | 114 [103-150] | 117 [93-163] | 124 [97-152] | 165 [105-223] | 0.0001 |
| Emergency surgery | 205 (30.7) | 21 (28) | 24 (34) | 34 (41.4) | 10 (40) | 0.26 |
| Stroke | 5 (0.8) | 0 | 0 | 3 (3.7) | 1 (4) | 0.03 |
| AKI | 44 (6.6) | 6 (8) | 9 (12.9) | 8 (9.8) | 5 (20) | 0.05 |
| UCI LOS, days | 6 [3-8] | 4 [2-6] | 5 [3-6] | 5 [3-11] | 4 [2-11] | 0.02 |
| HLOS, days | 30 [23-35] | 23 [15-29] | 29 [16-36] | 31 [15-44] | 28 [18-36] | 0.006 |
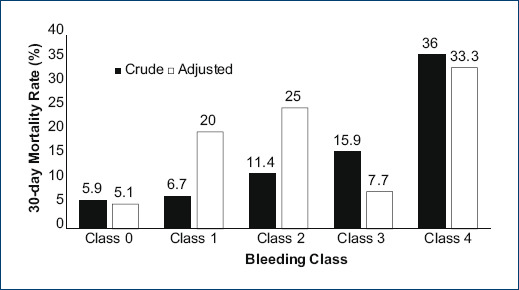
| 30-day Mortality | 39(5.9) | 5(6.7) | 8(11.4) | 13(15.9) | 9(36) | 0.0001 |
CRF: chronic renal failure; LVEF: left ventricular ejection fraction; CPB: cardiopulmonary bypass; AKI: acute kidney injury; ICU LOS: intensive care unit length of stay; HLOS: hospital length of stay. Data presented as median [interquartile range] for continuous variables and counts (percentages) for categorical variables.
In the primary outcome of 30-day mortality, a significant difference was found between the groups, observing that it increased to a higher class of bleeding. This was corroborated by a multivariate logistic regression analysis that was adjusted to EuroScore II and CPB duration, finding an independent association of the bleeding with 30-day mortality (OR, 95%, 5.82 [2.22-15.26], p = 0.0001) (Table 4). Subsequently, an adjustment of the mortality by groups was made, which is shown in figure 1.
Table 4 Multivariate regression analysis for 30-day mortality
| OR (95% IC) | p value | |
|---|---|---|
| Bleeding class* | 5.82 (2.22-15.26) | 0.0001 |
*Adjusted for EuroScore II and cardiopulmonary bypass duration.
Discussion
Excessive bleeding after cardiac surgery is a complex clinical problem despite significant advances in surgical techniques, anesthetic management, and critical care. It is generally accepted is better to avoid the use of blood products as much as possible; however, all cardiac surgery patients are at risk of bleeding and it remains unclear the threshold when the bleeding becomes clinically significant. The way to answer these questions is through the consensus and application of a more precise definition of bleeding.
Mehran et al. published a consensus to define bleeding and be used in cardiovascular clinical trials, but applicable in the context of acute coronary syndromes6. In cardiac surgery, the first studies measure bleeding through post-operative chest tube output, mainly in decision-making to emergency reoperation7-9. On the other hand, other studies quantified the degree of bleeding by the number of globular packages transfused10,11. However, the transfusion strategy of each center may vary, including the hemoglobin threshold, which a globular package is indicated12. Ranucci et al. in a retrospective study of a single center with 16,154 patients defined major bleeding by quantifying the chest tube output and it was associated with higher mortality with a 12% increase of relative risk of death for each 100 ml of increment in the first 12 h of postoperative bleeding13. Taking this as a basis, Dyke et al. established the concept of UDPB, ranking 5 degrees of bleeding considering the chest tube output, delayed sternal closure, the need for surgical re-exploration, and the use of blood products. This classification proposal is more precise in defining mortality in relation to the degree of bleeding14. More recently Colson et al. in a prospective multicenter study define active bleeding as blood loss > 1.5 ml/kg/h for 6 consecutive h in the first 24 h postoperatively, determining that this definition may be more appropriate for clinical decision-making15.
We used the classification proposed by Dyke in our hospital and observed an increase in mortality depending on the degree of bleeding. In the adjustment of mortality with other confounding variables (EuroScore II and CPB duration), the same result was also observed; however, for Classes 1 and 2 the adjusted mortality increases significantly, which can be related to the fact that those two groups have a lower EuroScore II and a lower CPB duration compared to the other groups. The results of our study are like the work of Dyke.
The limitations of this classification and our study are that it does not predict or identify risk factors for bleeding; it does not suggest any treatment or intervention, only attempts to define the degree of perioperative bleeding. We do not compare the UDPB with other classifications suggested in the literature to compare which one is superior to predict worse outcomes or mortality. Another limitation is the retrospective character of the study and only concludes association.
We strongly think the UDPB can be used in cardiac surgery to standardize nomenclature, improve outcome definitions, be useful in future clinical trials, and as a quality measure of care to manage and minimize transfusion.
More multicenter studies are needed to validate this classification and even establish it as a quality parameter in patient care in cardiac surgery.
Conclusions
Most cardiac surgical patients have post-operative bleeding without a clinical consequence. But for those with severe to massive bleeding, it can be catastrophic. We found that the higher the degree in UDPB was associated with higher mortality independently in our hospital population. We propose to use the UDPB for improving bleeding management and minimize transfusion in cardiac surgery patients; it can also serve as a measure for institutional quality improvement.











 nova página do texto(beta)
nova página do texto(beta)