1. Introduction
In the clinical superficial brachytherapy method, it is aimed to give the optimum radiation dose to the target tumor volume using skin patches made of materials such as glass, paper, silicone, and fabric, which are labeled with radionuclides [1]. Jeong et al., used 188Re-labeled paper patches for treating skin cancers [2]. Lee et al. used an adhesive tape patch covered with 165Ho-impregnated polyethylene microfilm in skin cancer treatment [3]. Salgueiro et al. used silicone or natural rubber patches labeled with 32P [4] and Pashazadeh et al. designed patches for superficial skin tumor treatment using microspheres labeled with 90Y [5].
In the above references, average range of beta particles used in the skin patches could be greater in the tissue with respect to the thickness of the superficial skin tumor. As a consequence, beta particles damage not only the target tumor volume but also deeper healthy tissues below the tumor, causing side effects. It is aimed in the treatment to protect the healthy tissue below the tumor volume by selecting short-range radionuclides. Using an external magnetic field is another option to reduce side effects.
In his article published in 1950, Bostick suggested that electrons can be directed by using an external magnetic field in electron beam therapy, hence potential benefits of using magnetic fields in radiation treatments motivated several research studies [6]. The effects of transverse and longitudinal magnetic fields on dose distributions in external photon and electron beam treatments were investigated by different groups [7-9]. Moreno-Barbosa et al. investigated the effects of magnetic fields on dose distributions using the TOPAS Monte Carlo (MC) code, in cases that underwent brachytherapy for lung tumors under clinical conditions [10]. Çavuşoğlu et al. investigated the behavior of beta particles in the magnetic field via GEANT4 MC simulation on the assumption that it would be possible to reduce the damage to normal tissues by using a magnetic field in cancer treatments using beta-emitting radioisotopes [11]. Sucu et al. investigated the dose distribution of Yttrium 90 (90Y), a pure beta emitter radioisotope used in therapy, under the influence of magnetic field [12]. To the best of our knowledge, there is no study in the literature investigating the effect of an external magnetic field on the dose characteristics of skin patch sources used in clinical superficial brachytherapy, which is the focus of this study.
Hindering beta particles from going beyond tumor volume and reducing their side effects may be possible, considering the changes in the orbits and average paths of the charged particles in the magnetic field [13]. A charged particle moving in the magnetic field is under the influence of the Lorentz force. In the vacuum, the charged particle moves in a linear orbit (Fig. 1a) when it moves parallel to the magnetic field vector, in a circular orbit of radius
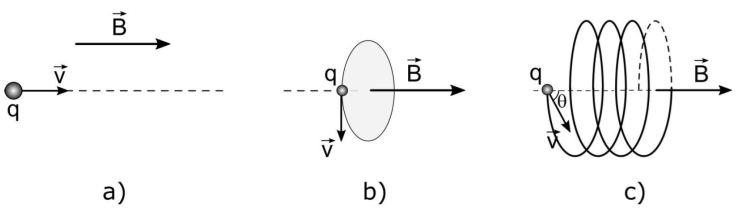
Figure 1 The paths of the charged particle within the magnetic field, depending on the angle between the magnetic field vector and the momentum vector of the particle a) linear, θ = 0 b) circular, θ = 90 c) helical, for any other angle.
Considering that the dose imposed by a 90 Y skin patch source on a tumor tissue by the effect of an external magnetic field can be limited within the tumor volume, and the radiation dose imposed on the healthy tissue underneath the target tumor can be minimized, PDD and TDP dose distributions of 90 Y skin patch source, in transverse magnetic fields were analyzed by using GEANT4 bases GAMOS MC code, in this study.
2. Material and method
In addition to experimental and analytical calculation methods, MC simulation method based on the theory of probability is widely used while calculating dosage in medical applications. It is difficult to obtain and measure precise dose distribution in the neighborhood of beta emitting sources, due to high dose gradient. It is possible to make these calculations with high precision by means of MC simulations. In this study, “GEANT4 based Architecture for Medicine-Oriented Simulations” (GAMOS) [15] code was utilized. It can be used in many applications within the energy interval of 250 eV-100 TeV, depending on the particle type. GAMOS use in medical physics applications has become wide scale in nuclear medicine and radiotherapy [16, 17]. It is possible to make simulations in various fields such as “Positron Emission Tomography” (PET), “Single Photon Emission Computed Tomography” (SPECT) gamma camera, linear accelerator, treatment planning in radiotherapy, by GAMOS code. In regard to these features, GAMOS is comprised of a kernel software involving a number of fields of application.
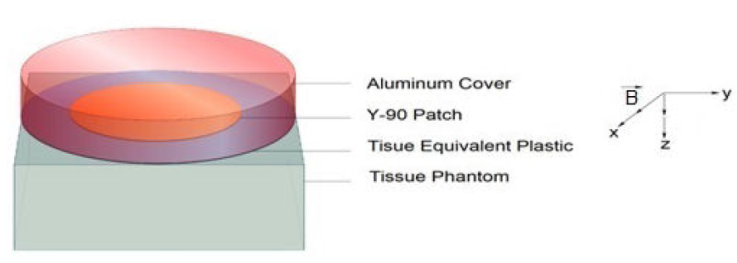
A simulation geometry is formed in ASCII format (Fig. 2). Its radius was chosen as 12.5 mm, and its height as 0.5 mm, in order to be able to make a comparison with the dimensions of the skin patch source of Ref. [20]. The radius of the aluminum protective shield is 20 mm, and its height is 10 mm. The skin patch source is placed within an aluminum protective shield.
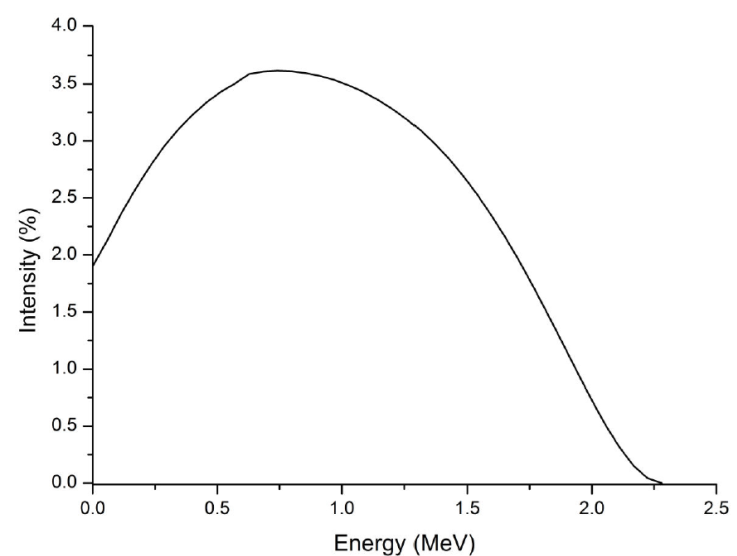
The tissue equivalent plastic material takes place between the tissue phantom and the skin patch source with a thickness of 0.01 mm, for the protection of the skin. The tissue phantom has a volume of 40×40×20 mm3, and it is filled in with voxels with a volume of 0.5×0.5×0.25 mm3. There is no air gap between the layers. 90 Y radionuclide is homogeneously distributed to the whole volume of the skin patch source. The energy spectrum published in the report having the number ICRU 72 was used for 90Y skin patch source (Fig. 3) [18].
Beta particles are isotropically emitted from the skin patch source, with the angle of 𝜋 rad. Tissue equivalent plastic and the elements forming the tissue phantom and their mass fractions are displayed in Table I. The density of the tissue equivalent plastic was 1.127 g/cm3, while the density of the tissue phantom was 1.0 g/cm3. All Monte Carlo calculations in this study were performed using the GmEMPhysics physics pack which also includes low energy models [15]. In order to keep the statistical error below 1%, 109 particles were used. The default production cut value in GAMOS was 100 μm for all operations in all materials. The energy threshold value inside the tissue phantom was 1.06 keV for gamma rays and 84.66 keV for electrons. “Variance Reduction” technique was not used in the calculations. The direction of transverse magnetic field (B), was taken in the perpendicular (x-axis) direction to the central axis of the skin patch source (Fig. 2).
Table I The elements of tissue phantom and tissue equivalent plastic and their mass fractions.
| Elements | Tissue Phantom | Tissue Equivalent Plastic |
|---|---|---|
| C | 0.23219 | 0.77550 |
| Ca | 0.00023 | 0.01838 |
| H | 0.10447 | 0.10133 |
| N | 0.02488 | 0.03506 |
| O | 0.63024 | 0.05232 |
| F | - | 0.01742 |
| Cl | 0.00134 | |
| Fe | 0.00005 | |
| K | 0.00199 | |
| Mg | 0.00013 | |
| Na | 0.00113 | |
| P | 0.00133 | |
| S | 0.00199 | |
| Zn | 0.00003 |
The calculations were implemented for 0 T, 0.6 T, 1.0 T ve 1.5 T magnitudes of transverse magnetic field. Dosimetric data in the tissue phantom were taken from the source up to 10 mm depth with 0.25 mm increments. Simulation outputs were taken as 3D dose files in DOSXYZnrc format. These outputs were evaluated with the code that we wrote in the Matlab R2020a program. The specifications of the PC used in the calculations were as, 2 × Intel Xeon(R) CPU 4 × 2 × (HT) 2,53 GHz, 24 GiB RAM, 64-bit.
3. Results
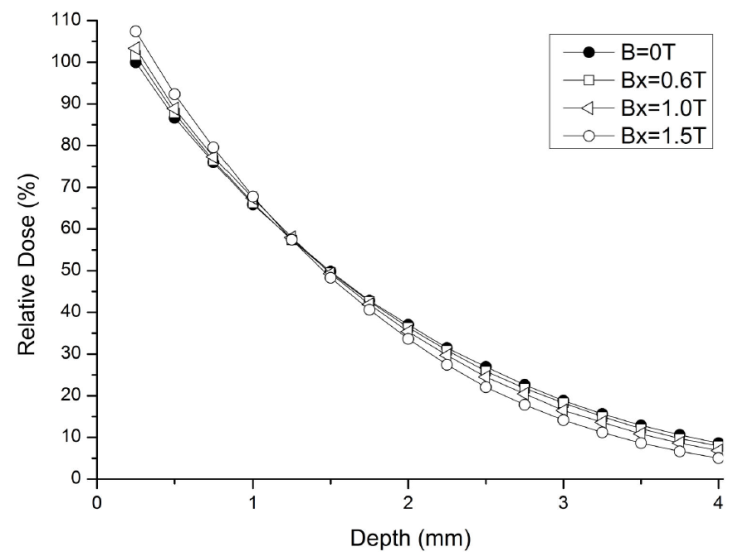
The effect of transverse magnetic field on the PDD dose distribution of 90 Y skin patch source was displayed in Fig. 4. The calculations were implemented for the magnetic field values of 0 T, 0.6 T, 1.0 T and 1.5 T. The values were then normalized with respect to the result of 0 T magnetic field. 0 T magnetic field results of our study were normalized according to the dose at 1 mm depth on the central axis, as suggested in the Ref. [19]. In Fig. 5, dose distributions on the central axis with respect to the transverse magnetic field value were given at 0.25-5.0 mm depth from the tissue phantom surface.
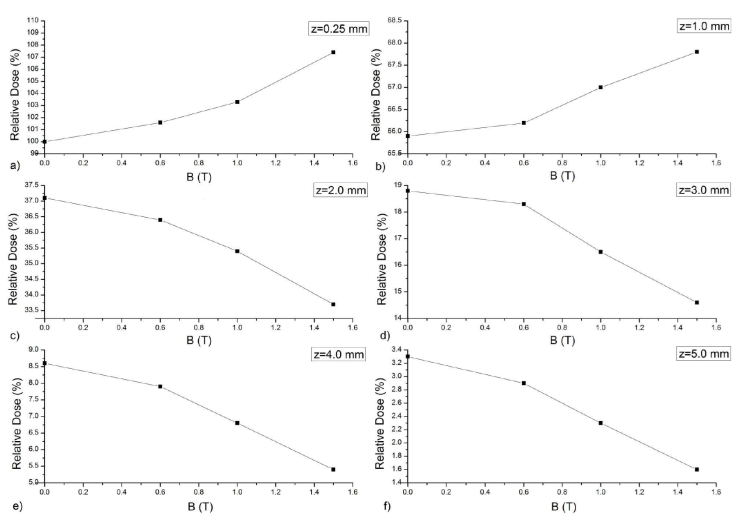
Figure 5 The changes in dose for a) 0.25 mm, b) 1.0 mm, c) 2.0 mm, d) 3.0 mm, e) 4.0 mm and f) 5.0 mm depths from the tissue phantom surface with respect to the change in transverse magnetic field.
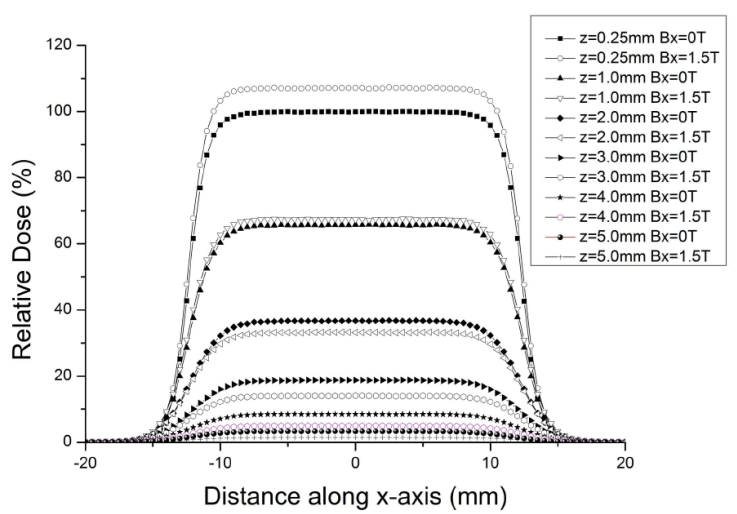
In Fig. 6, the comparison of TDP profiles of 90Y skin patch source was given for 0 T and 1.5 T values of the transverse magnetic field. The calculations were implemented for 0 T, 0.6 T, 1.0 T and 1.5 T values. To avoid too much data intensity, TDP graph was displayed only for 0 T and 1.5 T magnetic field value.
4. Discussion
Depending on the type, stage and location of the tumor, treatment methods such as surgical methods, chemotherapy, curettage, immunotherapy and radiotherapy are used in the treatment of skin cancer. Every treatment method has itsown advantages, disadvantages and limitations. Superficial brachytherapy method has been developed in recent years, enabling the treatment of superficial skin tumors and, hence, avoiding the disadvantages and limitations of the conventional treatment methods. This method is safe for the patient and physician in regard to radiation safety. However, like all other methods, it has side effects although it is easy-to-execute, noninvasive, economical and effective. The ranges of the beta particles used in the skin patch sources of superficial brachytherapy can exceed the target tissue. For this reason, beta particles have a damaging effect not only on the target tissue but also on the healthy tissue and bone underneath the target tumor volume, and cause side effects. The change in the trajectory of the beta particles moving in the magnetic field can be utilized for minimizing the side effects of the skin patch sources used in superficial brachytherapy. Thus, it would be possible to protect the healthy tissue and bone under the tumor by limiting the beta particles within the tumor volume. In accordance with this goal, PDD and TDP dose distributions of 90Y skin patch source, in the presence and in the absence of transverse magnetic field were investigated in this study. Primarily, PDD dose distribution of 90Y skin patch source obtained for 0 T value of the magnetic field was compared with the study of Pashazadeh et al. where they used the Loevinger function, as one of the analytical methods, and EGSnrc MC simulation with a view to determine the dose distribution of 90Y skin patch source [20, 21]. The result of the comparison is displayed in Fig. 7.
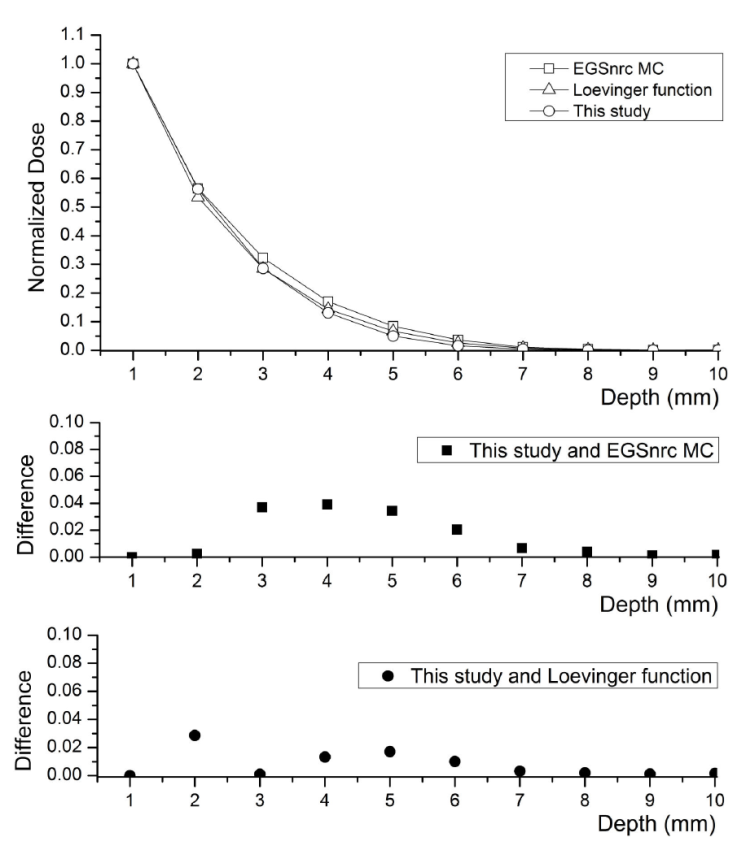
Figure 7 The comparison of the PDD dose distribution of 90Y skin patch source obtained for 0 T value of the transverse magnetic field of this study, with that of Ref. [21]. This study (circle), Loevinger function calculations (triangle) and EGSnrc MC simulation results (square).
There is a maximum difference of 3% between GAMOS MC simulation results in this study and the dose values calculated by the Loevinger function, and a maximum difference of 4% with EGSnrc MC simulation results. It can be seen that dose values obtained by GAMOS MC simulation are lower than the values obtained by EGSnrc MC simulation, and closer to the values obtained by the Loevinger function. These differences are within plausible limits. As for the reasons of this difference, the source is surrounded by a protective aluminum shield for radiation safety in this study, and there is a tissue equivalent plastic material with 1.127 g/cm3 density between the skin patch source and the skin surface to protect the skin. The tissue phantom was modeled and its density was 1.0 g/cm3. Besides, its voxel size was bigger, the number of particles used for calculation was higher, and the energy threshold value was lower.
When the transverse magnetic field was applied to 90Y skin patch source, it was observed that the transverse magnetic field was effective on PDD and TDP dose distributions. When the PDD dose distribution of 90Y skin patch source was examined with respect to the increasing values of the transverse magnetic field, it was observed that there was an increase in the doses from the surface of the tissue phantom up to depths of 1.25 mm as compared to 0 T value of the transverse magnetic field, while a decrease beyond 1.25 mm depth (Fig. 4 and Fig. 5). The entrance skin dose at 0.25 mm depth and percentage differences are given in Table II, with respect to the values of the transverse magnetic field.
Table II The change in the entrance skin dose at 0.25 mm depth and percentage differences with respect to the magnitude of transverse magnetic field.
| B (T) | Entrance Skin Dose (%) | Difference (%) |
|---|---|---|
| 0 | 100.0 | 0 |
| 0.6 | 101.6 | 1.6 |
| 1.0 | 103.3 | 3.3 |
| 1.5 | 107.4 | 7.4 |
The TDP distribution of 90Y skin patch source for 0 T, 0.6 T, 1.0 T and 1.5 T values of the transverse magnetic field was analyzed. The TDP distribution obtained for 1.5 T value of the transverse magnetic field was given, with a view to avoid excessive data intensity (Fig. 6). As seen from Fig. 6, dose values of 90Y skin patch source on the central axis increase at 0.25 mm and 1.0 mm depths, but decrease at 2.0-5.0 mm depths. The changes in TDP dose distribution and percentage differences are given in Table III.
Table III The effect of transverse magnetic field of 1.5 T magnitude on the TDP distribution.
| Depth (mm) | B=0 T | B=1.5 T | Difference (%) |
|---|---|---|---|
| 0.25 | 100.0 | 107.4 | 7.4 |
| 1.0 | 65.8 | 67.7 | 2.8 |
| 1.25 | 57.5 | 57.4 | -0.17 |
| 2.0 | 37.1 | 33.6 | -9.4 |
| 3.0 | 18.8 | 14.1 | -25.0 |
| 4.0 | 8.6 | 5.0 | -41.8 |
| 5.0 | 3.2 | 1.4 | -56.2 |
Zhengue et al. investigated the effects of a magnetic field on the dose distribution of BEBIG type 60Co HDR brachytherapy source, by GEANT4 MC method, and showed that a magnetic field of 3.0 T magnitude caused 40% increase in the dose value at a distance of 5.4 mm from the source center [22]. In their experimental study, with a view to investigate the dose distribution of 90Y source under an external magnetic field, Sucu et al. took measurements at two different depths, namely at 0 mm ve 2.0 mm distance from the source. The results of the measurement showed that, dose increased in the volume close to the source and decreased at 2.0 mm at the rate of 26 %. In their study conducted by using GEANT4 code, Çavuşoğlu et al. showed that, the number of interactions increased within the volume close to the source, depending on the magnitude of transverse magnetic field, and the interaction number decreased with increasing depth. The fact that the interaction number increased in the volume close to the source shows that beta particles were confined within this volume and the local dose increased. In this study, it is seen that the changes in the dose distribution of the 90Y skin patch source under the transverse magnetic field are consistent with the results of the above-mentioned theoretical and experimental studies. In case of a skin tumor of 3.0 mm thickness from the skin surface, total amount of dose accumulating in the target tumor volume is 89%, for the magnetic field magnitude of 0 T. By applying a magnetic field of 1.5 T intensity, it is now seen that 98% of the total dose is confined inside the target tumor volume. By using transverse magnetic field, the dose received by the healthy tissue under the target tumor has decreased 10.1% with respect to 0 T value.
5. Conclusions
The simulation results showed that the transverse magnetic field had a bolus-like effect on the dose distributions of 90 Y skin patch source, and it drew the dose near the tissue level. When the results were compared with the PDD and TDP values corresponding to 0 T value of the magnetic field, the dose values along the central axis increased by 7.4% and 3.1% at the depths of 0.25 mm and 1.0 mm respectively while it decreased by 9.4%, 25.0%, 41.8% and 56.2% at the depths of 2.0 mm, 3.0 mm, 4.0 mm ve 5.0 mm, respectively. In the presence of a superficial skin tumor of 3.0 mm thickness, whereas the dose accumulating in the tumor volume for 0 T value of transverse magnetic field was 89% of the total dose, for 1.5 T value, it increased to 98%, and, hence, the total dose received by the healthy tissue under the tumor decreased by 10.1%. The obtained results showed that it was possible to confine the dose administered to superficial skin tumor within the target tumor volume, and that it was possible to protect the healthy tissue and bone underneath the tumor. In addition, the confinement of the dose inside the target tumor volume by the help of an external magnetic field would enable us to give optimum dose to the tumor volume. Hence, it increases local control and helps the therapy period become shorter.











 nova página do texto(beta)
nova página do texto(beta)