Introduction
Eosinophilic esophagitis (EoE) is an immuno-allergic disease with symptoms of esophageal dysfunction characterized by eosinophilic infiltration in the esophagus. Its prevalence has increased in recent years, now being the main cause of dysphagia and food impaction in children and adolescents1. Recent studies estimate a prevalence of 25/10,000 individuals in the United States and Europe2. Despite the existence of diagnostic and therapeutic guidelines, its management remains heterogeneous. The symptomatology is non-specific, varying according to age. In young children, it manifests as feeding difficulties, while in older children, dysphagia, retrosternal pain, and food impaction are the main symptoms. At present, a clinical-histological evaluation is necessary to confirm the diagnosis. There is a greater prevalence in males, with the ratio in both children and adults being approximately 3:1. The main objectives of treatment are the resolution of clinical symptoms, maintenance of histological remission, and prevention of long-term complications. Effective treatment has been shown to reverse subepithelial fibrosis and, therefore, can prevent complications such as remodeling and stenosis3.
This work aims to understand the current diagnostic and therapeutic approach to pediatric EoE by healthcare professionals. For this purpose, the young researchers group of the Latin American Society of Pediatric Gastroenterology, Hepatology, and Nutrition (LASPGHAN) conducted a survey directed at pediatric gastroenterologists from Latin America and Spain. Through this survey, the intent is to identify experience and adherence to international guidelines during daily practice.
Method
This is a descriptive, observational, and multicenter study in which pediatric gastroenterologists, both members and non-members of LASPGHAN from Latin America and Spain were invited to respond to an online survey between October and November 2022 that evaluates the management of patients with EoE in terms of diagnosis, treatment, and follow-up. Their responses were to be based on their experience in daily practice.
Participants were invited through an email link to access a Google Forms online questionnaire, available in Spanish and Portuguese, with single-choice questions, multiple-choice questions, and others with free text options. The survey was self-administered and anonymous.
Statistical analysis
The collected data were analyzed using descriptive statistics with Microsoft Excel and Google Forms. In this study, the STROBE-Checklist, specifically designed for cross-sectional studies, was applied.
Ethical considerations
This work complies with current bioethical research regulations. As this was a survey conducted among healthcare professionals without identifying them, it did not require review by the Ethics Committee.
The approach of this work does not involve the manipulation of sensitive data that could compromise the privacy or integrity of individuals.
Within the framework of this research, it is clarified that obtaining informed consent from participants was not necessary, given that the methodology used consisted of a voluntary self-administered survey. Participation in the survey was completely optional, and individuals freely decided to provide their responses without external pressure.
The nature of the self-administered survey ensures that participants have full control over the information they share, and no data are collected that would directly or indirectly identify the respondents or the patients they reference. The questionnaire has been designed so that the privacy and confidentiality of participants are protected at all times.
Likewise, it is guaranteed that the data collected is handled anonymously and in aggregate during analysis, preserving the confidentiality of participants, respecting the ethical principles of research, and aligning with relevant regulations regarding privacy and confidentiality.
Results
A total of 118 pediatric gastroenterologists responded to the survey, with the countries having the greatest participation being Argentina (20%), Brazil (20%), Mexico (13%), Venezuela (11%), Colombia (11%), and Spain (8%) (Fig. 1).
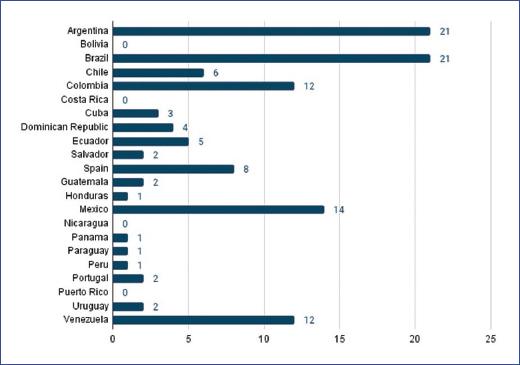
Figure 1 Distribution of participants by country (n). Number of professionals who participated in the survey, distributed by country of origin.
About 28.8% of respondents had more than 20 years in the specialty and 33% had < 5 years. Professionals with 6-10 years and 11-19 years of work in pediatric gastroenterology correspond to 23.7% and 14%, respectively.
More than half of the respondents work in both public and private settings (61% and 60%, respectively). About 40% also work in private practice, and 31% belong to teaching or university hospitals.
About 53% diagnose up to two patients/year with EoE. 31.3% diagnose 3-5 patients. Only 6.7% diagnose 6-10 and 8.5% diagnose more than 10 patients/year.
Diagnostic, therapeutic, and evolutionary management of patients with EoE
DIAGNOSTIC METHODOLOGY AND ENDOSCOPIC FINDINGS
Respondents were asked about how many biopsies and from which portions of the esophagus they perform when EoE is suspected; 55.9% perform 3-4 biopsies in each of the upper and lower segments of the esophagus and 17% take 1-2 biopsies in each of these locations (Fig. 2A). 15.2% of respondents perform 3-4 biopsies randomly and a percentage < 1% (0.85%) perform them in a targeted manner to lesions or abnormalities found in the esophageal mucosa (Fig. 2B). About 8.4% of the participating professionals do not perform endoscopy in their daily practice and therefore refer their patients for this procedure.
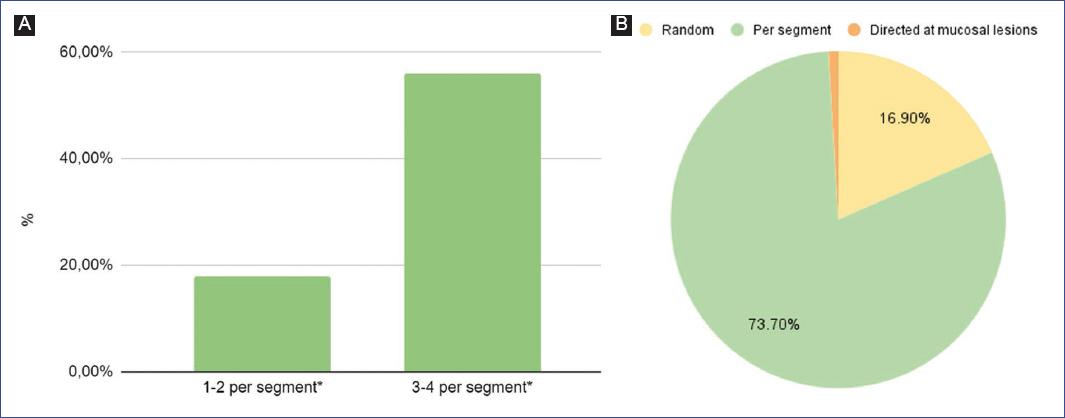
Figure 2 A: number of biopsies performed per esophageal segment for diagnosis of eosinophilic esophagitis. B: approaches to esophageal biopsy sampling for the diagnosis of eosinophilic esophagitis. *Upper and lower segments of the esophagus.
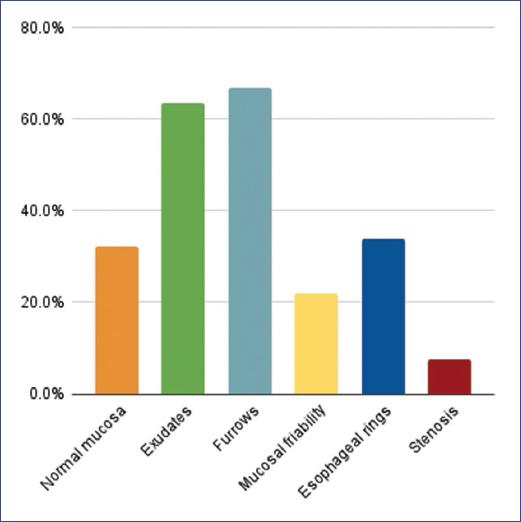
The most frequently observed endoscopic findings are longitudinal furrows (67%) and exudates (64%), followed by concentric rings (34%), normal mucosa (32%), and friability/crepe paper mucosa (22%). Esophageal stenosis was reported in 7.6% (Fig. 3).
The endoscopic reference score for EoE (EREFS) score is a scoring system used to evaluate the severity of EoE based on the presence and severity of five endoscopic characteristics of EoE: edema, concentric rings, exudates, furrows, and stenosis1. About 36.4% of specialists use it during the diagnostic procedure, 34.7% report not using it, and 18.6% are unaware of its existence. The phenotypic characteristics of the disease, based on endoscopic and histopathological findings, are useful for the therapeutic approach. The most frequently observed disease phenotype in this survey is the inflammatory type (79.6%).
THERAPEUTIC MANAGEMENT
The survey inquired about the most commonly used therapeutic strategies at disease diagnosis or during acute flares, and in the maintenance phase, both in patients with inflammatory and stenosing involvement.
The most used first-line treatments in patients with inflammatory involvement are: proton pump inhibitor (PPIs) (33.9%), followed by triple therapy (TT) (PPI + exclusion diet [ED] + topical corticosteroid [TCS]) (26.2%) and combination therapy (ED + PPI) (21%) (Fig. 4). On the other hand, the approach for those with stenosing involvement is: TT (52.5%), combination treatment (TCS + PPI) (13.4%), and topical glucocorticoids (12.37%). 9% use oral corticosteroids and 3% use intravenous steroids.
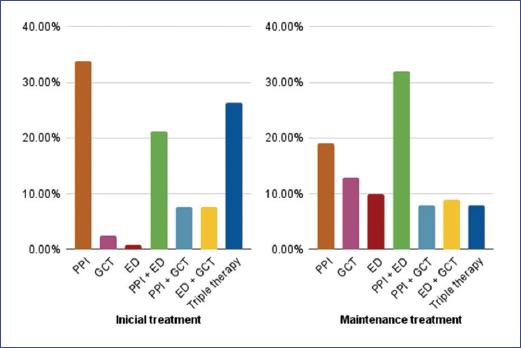
Figure 4 Treatment in non-stricturing eosinophilic esophagitis. Therapeutic approach at the beginning (left) and maintenance (right). DE: elimination diet; GCT: topical steroids; PPI: proton pump inhibitor; Triple therapy: PPI + GCT + ED
For maintenance treatment of patients with inflammatory phenotype, the most commonly used strategies are PPI associated with ED (32.2%), PPI (19.4%), TCS (12.7%), and ED (10%) (Fig. 4). In patients with esophageal stenosis: TT (32.2%), followed to a lesser extent by TCS in association with PPI, oral corticosteroids associated with PPI or diet, and TCS (16% each).
Of the available TCS, 51% of professionals report using budesonide in home preparation, 38.9% use Fluticasone aerosol (swallowed), 16% use commercial budesonide preparation, and 2.5% use oral dispersible budesonide.
Regarding ED, 26% of respondents indicate a diet directed by allergy testing; 25.4% empirically exclude 6-8 foods, 23.7% exclude 2-4 foods, and 10% exclude only 1 food. The remaining 14.4% adapt the ED according to the patient’s clinical history.
Extensively hydrolyzed formulas or elemental formulas as first-line treatment in children under 12 months are used by 52.5% of professionals and in children under 24 months by 23.7%. 21% of gastroenterologists use them in patients with multiple food allergies, 15% in children with severe nutritional compromise at any age, and 6.7% in cases of stenosing phenotype. About 3% of professionals do not have access to this resource and 5.9% report not using them.
At the time of the survey, 3.3% of respondents have patients with EoE under treatment with biological agents.
FOLLOW-UP OF PATIENTS WITH EOE
Post-treatment endoscopic evaluation
The majority of respondents (84.6%) perform the first upper gastrointestinal endoscopy (UGE) follow-up between 8 and 12 weeks after initiating treatment (46.6% and 38%, respectively). 9% of professionals wait more than 12 weeks to request it, and 3.4% do not perform the first endoscopic follow-up if the patient is asymptomatic after establishing treatment.
Regarding the endoscopic schedule for the follow-up of asymptomatic patients with EoE, 35.5% perform control UGE every 6-12 months and 32.2% every 12-24 months; 26.2% of professionals report indicating control endoscopy only if the patient presents symptoms during their evolution, and approximately 5% perform it only in case of therapeutic modifications
Since the stenotic phenotype is not predominant in pediatrics, esophageal dilation is not a routine procedure performed by all pediatric endoscopists. In our survey, only 27.2% of professionals have experience with this therapeutic approach.
Multidisciplinary approach
It was recorded that 52% always request an evaluation of these patients by a pediatric allergist. 27.2% make this referral only if the patient has an allergic history, 12.7% only if they prescribe an ED, and 14% of professionals do not refer their patients to be evaluated by this specialty.
About 47.4% of professionals report always referring their patients with EoE to a pediatric nutritionist. 28.8% only in case of nutritional compromise, 17.5% in case of prescribing targeted diet therapy, 8.2% when prescribing a 4-food ED, and 5% if it is a 6-food ED. 11.3% of respondents do not request an evaluation of the patient by a pediatric nutritionist.
Respondents were asked if they consulted with pediatric endocrinology for patients on corticosteroid treatment. About 16.9% responded that they always make the referral. 60.7% do so only when adverse effects of corticosteroids occur, and 22.8% never indicate evaluation by this specialty.
Prolonged use of corticosteroids
EoE is a pathology that may require the use of steroids for a prolonged period (< 3 months) in many patients.
The most observed adverse effect from the use of TCSs was oropharyngeal candidiasis (38.8%). About 22.8% report having had patients with esophageal candidiasis, 6.7% growth delay, 5% osteopenia or altered basal cortisol, and 4.2% asthenia. About 41.5% have not observed adverse effects from their use.
Despite the prolonged prescription of TCSs, 48.3% do not request basal cortisol measurement. About 22.8% of professionals perform this laboratory test only if the patient presents adverse effects from steroids, and 22% report always requesting a basal cortisol measurement.
Discussion
EoE has been a focus of research since the early nineties when it was recognized as an independent clinical entity. At present, we have guidelines and consensus that guide the medical practice of specialists. It is a disease of immuno-allergic character whose incidence and prevalence are increasing in the pediatric population.
In a study conducted by Shah et al. using the criterion of 15 eosinophils per high-power field for diagnosis, a significant increase in diagnostic sensitivity was demonstrated when increasing the number of esophageal mucosal biopsies obtained, going from 73% with a single sample to 100% with six4. Today, it is recognized that eosinophilic infiltration in the esophagus can be irregular, making it necessary to obtain biopsies from proximal and distal segments, at least six in total, to achieve an accurate diagnosis. In our survey, most professionals perform between three and four biopsies per esophageal segment, complying with international standards.
The endoscopic approach is the first diagnostic test when this pathology is suspected, presenting characteristic but not pathognomonic findings.
The European Society for Pediatric Gastroenterology, Hepatology, and Nutrition (ESPGHAN) Eosinophilic Gastrointestinal Disorders Working Group reports in their multinational longitudinal registry that among 582 patients, whitish exudates and longitudinal furrows were the most frequently reported characteristics. They also observed an endoscopically normal esophagus in 17% of patients, without reporting stenosis5. Similarly, our survey respondents mention exudates and longitudinal furrows as the most frequently observed findings in their practice. Likewise, endoscopists do not usually find stenotic behavior, with the inflammatory phenotype being the most frequent. As we have advanced in our knowledge of this pathology over the years, we currently approach esophageal involvement using an endoscopic score. It is interesting to highlight that in the 2014 ESPGHAN guideline, this type of macroscopic assessment was carried out subjectively by the specialist6. However, we now have a score that allows us to standardize and classify these types of lesions more precisely. The EREFS score was proposed as a tool to classify and rate the presence and severity of the five main endoscopic characteristics of EoE7. In the present survey, almost 19% of professionals are unfamiliar with this score, and more than a third do not use it in their daily practice. Recent studies on the concordance with histological data have obtained contradictory results. Some authors indicate that endoscopists should not base the diagnosis of EoE, nor make presumptions about the activity or remission of this disease based exclusively on endoscopic findings8. On the other hand, Wechsler et al. report in their prospective work a sensitivity and specificity of 89.6% and 87.9%, respectively with the use of this score at the time of diagnostic suspicion7. We understand that it should be reported at the time of the endoscopic procedure as it is a tool that helps us identify with some precision those patients with EoE. Similarly, we emphasize the importance of taking esophageal biopsies in all patients with suspected diagnosis, as it is reported that up to 30% of patients may present with normal-appearing mucosa9.
In relation to treatment objectives, these include the resolution of symptoms and remission of histological activity to avoid fibrostenotic complications. In the 2017 recommendation guideline, PPIs are proposed as effective treatment for symptomatic improvement and endoscopic remission in slightly more than half of patients8. Our survey shows that this therapeutic approach is used in non-stenotic EoE as first-line treatment by the majority of respondents, a third of whom use it as monotherapy. For the fibrostenotic phenotype, PPIs are indicated by more than half of professionals, mainly associated with diet and corticosteroids. Systemic steroids are not currently recommended in the management of patients with EoE8,10. About 3% of respondents have indicated them.
EDs are effective in achieving clinical-histological results in pediatric patients with EoE10. The elimination of six foods results in a higher rate of histological remission when compared to the exclusion of 2-4, but is associated with lower compliance and a greater number of endoscopies. At present, less restrictive empirical diets are the ones of choice, recommending the elimination of 2 foods and increasing the level of restriction to 4 in those patients who do not respond to treatment9. Among the respondents, the number of excluded foods was highly variable in relation to what is reported in the literature. Less than a quarter of the surveyed gastroenterologists restrict between 2 and 4 foods. The empirical exclusion of a single food is the approach least practiced by professionals. This discrepancy between international guidelines and common practice somehow reflects the difficulty we have in implementing an empirical therapy of one, two, or four foods. It seems that a more cautious and restrictive strategy is safer for us, despite the nutritional disorders that this entails.
According to the ESPGHAN guideline for the management of EoE in pediatric patients, the failure of allergy test-directed dietary therapy to induce remission has been attributed to the inability of these tests to accurately detect causal food antigens6,11. In the present survey, however, we observe a high percentage of professionals who rely on these tools to guide dietary treatment.
The British Society for Pediatric Gastroenterology, Hepatology and Nutrition guideline for diagnosis and treatment of EoE states that exclusive elemental formulas have a limited role in the treatment of EoE since, despite their high efficacy, they have very low compliance rates and suggests reserving this tool for patients refractory to other treatments10. In this survey, elemental formulas are used in infants under 1 year of age by half of the respondents. Other reported indications were multiple food allergies, severe nutritional compromise, and stenotic phenotype.
Successful results depend on treatment adherence, which in turn is determined by palatability, cost, third-party coverage, and the effect of treatment on quality of life11,12. Henderson et al. reported a satisfactory clinical response and disease remission (defined as a decrease in eosinophil count < 15 eosinophils/high-powered field) in 96% of patients on an elemental diet, 81% of patients with classic six-food elimination diet (SFED), 80% of patients with modified SFED, and 65% of patients with directed diet13.
In EoE, the different strategies aimed at inducing and maintaining remission are best achieved through a multidisciplinary team. From our survey, we observe that practically half always use this methodology. A smaller percentage reserves this approach for those with some history of atopy or particular dietary treatment.
When reviewing dietary treatment, we observe that many gastroenterologists exclude a large number of foods. Almost 50% decide to initiate this approach by removing at least two or more foods, without opting for a step-by-step ED. This suggests that in practice, we tend to follow a “top-down” treatment approach rather than a “step-up” one. While we could affirm that more than 75% of respondents observe good adherence to the diet, the dietary therapeutic strategies we use highlight the fundamental role of the nutritionist when approaching the patient and their family. This survey reveals that nearly a third of professionals only refer to nutrition specialists when faced with evident nutritional compromise. Similarly, an even smaller percentage refers patients in cases of more restrictive diet therapies. This indicates that a significant portion of professionals only consider nutritional support in situations of high dietary complexity, and not as a routine practice for this pathology.
Regarding the pharmacological approach, monitoring the prolonged use of corticosteroids is not a common practice. Hsu E. and colleagues, in their work on the impact of adrenal insufficiency in pediatric patients under corticosteroid treatment, report that 5 out of 106 patients presented it when morning basal cortisol was measured14. Although the article presents a low percentage, it is important to recognize it and make a timely consultation with the specialist, as cortisol monitoring might be advisable for children with EoE who receive high doses of topical steroids for long periods or if they concomitantly receive corticosteroids through other routes (oral, inhaled, or nasal)8.
Conclusion
This study offers a global view of the management of EoE in Latin America and Spain. However, the heterogeneity in the participation of each country limits the ability to draw conclusions at the local level regarding the management of this pathology in each of them.
The diagnosis of EoE is challenging due to the diversity of its symptoms. Despite the challenges it presents in its practical management, the majority of the surveyed specialists follow the diagnostic guidelines proposed in the latest international guides; however, a not insignificant percentage continues to perform biopsies randomly, which implies a reduction in this yield. While endoscopic control at the beginning of treatment tends to be performed in a timely manner, it becomes very arbitrary at the time of maintenance.
There are discrepancies when it comes to the therapeutic approach. We observe that combination therapy is frequently used both at the initiation and in the follow-up of these patients. It seems difficult to homogeneously follow the guidelines in daily practice. We must be careful when indicating diet therapy in these patients, taking into account that the exclusion of multiple foods from the beginning of treatment can imply a nutritional risk, in addition to difficulties in the social sphere and in relation to the acquisition of skills associated with feeding. Likewise, a more limited exclusion of foods from the beginning would imply a reduction in the number of endoscopic controls.
Finally, we believe that interdisciplinary work is another fundamental pillar to optimize the follow-up and treatment of these patients, facilitating adherence and minimizing long-term adverse effects.











 nueva página del texto (beta)
nueva página del texto (beta)