1. Introduction
In the last fifty years, numerous studies were focused on the Holocene palaeoenviromental reconstruction of coastal areas, such as estuaries, deltas, bays or lagoons (Allison and Niemi, 2010; Melis et al., 2017; Bianchette et al., 2022; Mayoral et al., 2024). The main results are usually based on the multidisciplinary analysis of continuous sediment cores, including the characterization of the main sedimentary facies, the vertical distribution of selected trace metals, the faunal record and the age models obtained from radiocarbon datings, among other data (Marocco et al., 1996; Ben Khalifa et al., 2019; Wilson and Hayek, 2023). In Mexico, this research yield interesting findings on climate changes and sea level oscillations, marine transgressions, heavy metal enrichment due to anthropic contribution or changes in sediment accumulation rates (Bianchette et al., 2016; Bocanegra-Ramírez et al., 2019; Pitchamani et al., 2022; Jupin et al., 2023; Velandia-Aquino et al., 2023).
The faunal record of coastal cores is a powerful tool for pinpointing the different paleoenvironmental scenarios that have taken place in a given area over the last few thousand years (Peros et al., 2015; Marco-Barba et al., 2019; Korsgaard et al., 2024). In Mexico, molluscs, diatoms, pollen or ostracods have been used for this purpose (Palacios-Fest et al., 2006; Bocanegra Ramírez et al., 2019; Arenas et al., 2022; Suárez-Mozo et al., 2023).
Foraminifera are also one of the most interesting groups in this multidisciplinary research of these cores (e.g.Laprida et al., 2011; Ruiz et al., 2012; Alday et al., 2013; García Artola et al., 2023). In estuarine and lagoonal environments of Mexico, these microorganisms are very useful because changes in their species and associations detect very precisely variations in environmental parameters such as salinity, organic matter content, contamination by heavy metals or agricultural waste, sea level changes, transgressions and regressions, or even earthquakes or tsunamis (Phleger and Lankford, 1978; Coral Hinostroza and Segura-Vernis, 1979; Ramírez-Herrera et al., 2009; Gutiérrez-Ayala et al., 2012; Sánchez et al., 2020; Hardage et al., 2022; Sánchez and Gómez-León, 2024).
In southwestern Spain, the benthic foraminifera of the main estuaries have been previously studied and seven main assemblages have been differentiated in the salt marshes [Trochammina inflata (Montagu, 1808) and Entzia macrescens (Brady, 1870)], tributary channels and channel margins [Ammonia tepida (Cushman, 1926) -as Ammonia inflata (Seguenza) in previous papers-, Astrononion stelligerum (d'Orbigny, 1839), Cribroelphidium vadescens Cushman and Bronniman, 1948 and Ammonia ammoniformis (d'Orbigny, 1826)] and the estuarine mouths [Ammonia beccarii (Linnaeus, 1758) and miliolids] (González-Regalado et al., 2001, 2019a). These previous studies have been applied to the palaeoenvironmental reconstruction of cores and surface sections of this estuary (e.g.González-Regalado et al., 2019b; Arroyo et al., 2021).
In this paper, a multidisciplinary analysis of a continuous sediment core extracted in an old cove located in the middle estuary of the Tinto River (SW Spain) is carried out. Its main objective is the identification of the main paleoenvironmental changes that this area has undergone from the Miocene to the present, based mainly on the sedimentary facies and foraminiferal analyses.
2. Study area
The Tinto River is a small river (100 km length) that runs through southwestern Spain to its mouth in the Atlantic Ocean, where it forms a wide estuary at its confluence with the Odiel River (Figure 1A-B). This estuary can be divided into three zones (Borrego, 1992), depending on the interaction between river currents and tides: i) fluvial estuary, with a dynamic dominated by river action, which gives rise to a set of braided channels; (ii) middle estuary, characterized by a mixture of fresh and marine waters and the presence of several islands consisting of marshes (Figure 1B: e.g., Bacuta Island); and (iii) marine estuary, dominated by tides and protected by two sandy spits (Punta Umbría, Punta Arenillas), with a central barrier island (Figure 1B: Saltés island) formed by cheniers and washover fans that delimits two main channels (Punta Umbría channel, Padre Santo channel).
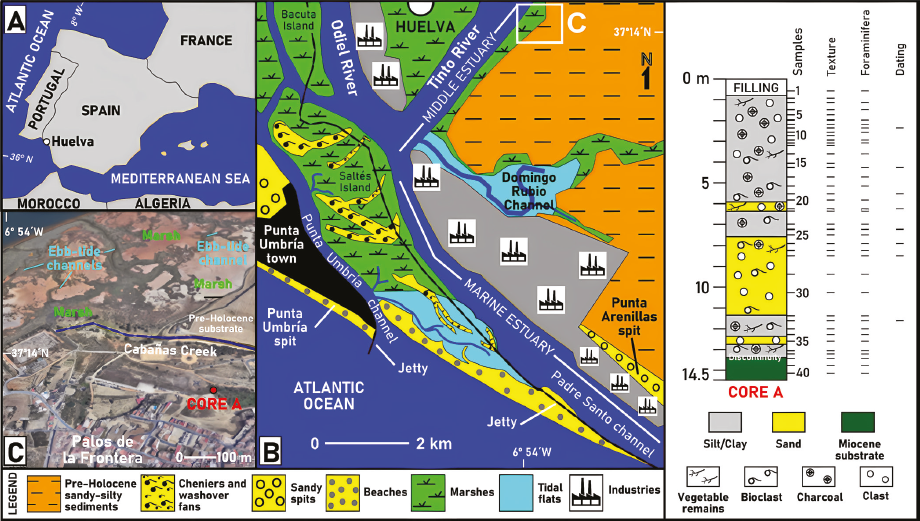
Figure 1 A-B. Location and geomorphology of the Tinto-Odiel estuary. C. Location of La Fontanilla cove and core A.
This estuary was excavated mainly during the Pleistocene by river action and was subsequently flooded during the Holocene transgression, which had its maximum towards 7000-6500 cal. kyr BP (Zazo et al.,1994) and whose influence lasted until 5.2 cal. kyr BP (Highstand Phase; Boski et al., 2002). These fluviomarine actions caused the formation of some coves (Figure 1B-C: e.g., La Fontanilla cove) and the erosion of the Neogene formations that make up the estuary substrate, such as the upper part of the Gibraleon Clay Formation (Tortonian-Messinian; Civis et al., 1987) and the Huelva Sand Formation (Pliocene; Civis et al., 1987). These geological formations contain an abundant record of benthic foraminifera, composed mainly of Ammonia beccarii (Linnaeus, 1758), Ammonia inflata (Seguenza, 1862), Nonion faba (Fichtel and Moll, 1798) (identified as Florilus boueanum in some previous papers), Cibicidoides pseudoungerianus (Cushman, 1922), Pullenia bulloides (d'Orbigny, 1846), Bolivina spp., Bulimina spp. and Globobulimina spp. (Sierro et al., 1982; González-Regalado and Civis, 1987; González-Regalado, 1989).
3. Material and methods
Core A (Figure 1C: 14.5 m length; 3.5 amsl) was extracted by usual rotary drilling techniques in the inner part of La Fontanilla, an old cove located in the middle estuary of the Tinto River near the town of Palos de la Frontera (SW Spain). Forty samples were collected for a multidisciplinary analysis, according to the vertical arrangement of the sedimentary facies and their boundaries. Forty samples (20 g) were wet-sieved with pressurized water and floated using sieves of 63, 125, 250, 500, 1000 and 2000 pm diameter for textural analysis. Forty additional samples were selected for foraminiferal analysis. In each subsample, twenty grams of sediment were separated and levigated through a 125 pm mesh sieve, a sieve frequently used in the analysis of benthic foraminifera and adequate for ecological analysis (e.g.Milker and Schmiedl, 2012; Klootwijk and Alve, 2022).
The residue was dried in an oven at a constant temperature of 40°C for a period of no less than one day. Foraminifera were classified according to Loeblich and Tappan (1987), Milker and Schmiedl (2012) and the World Register of Marine Species (WoRMS). Results were compared with the foraminiferal assemblages determined in the Neogene formations found in the vicinity of core A (see previous section).
Eight dates were produced at the National Center of Accelerators (CNA, Seville, Spain), with a subsequent calibration using the program CALIB version 8.2 (SHCal20 curve for organic matter and wood).
The final results correspond to calibrated ages using 2σ intervals and the mean calibrated ages were used to produce a chronological framework of core A. Sedimentation rates were extrapolated using the linear interpolation of CLAM 2.3.2 software (Blaauw, 2010).
4. Results
4.1. SEDIMENTARY FACIES
The textural analysis allows to delimitate six sedimentary facies (Figure 2A-B). Facies 1 (F1; 14.51-3.5 m depth; samples A-40 and A-39) consists of massive greenish clayey silts (silt + clay: 87-88%; sand: 11-13%) with scarce macrofaunal remains. These fine sediments belong to the Gibraleon Clay Formation (Civis et al., 1987) and are separated from facies 2 (F2; 13.5-11.6 m depth; samples A-38 to A-33) by an erosional discontinuity. F2 is characterized by an increasing grain size (sand: 23-56%), abundant plant debris, small charcoal grains and numerous brackish gastropods [mainly Peringia ulvae (Pennant, 1777)].
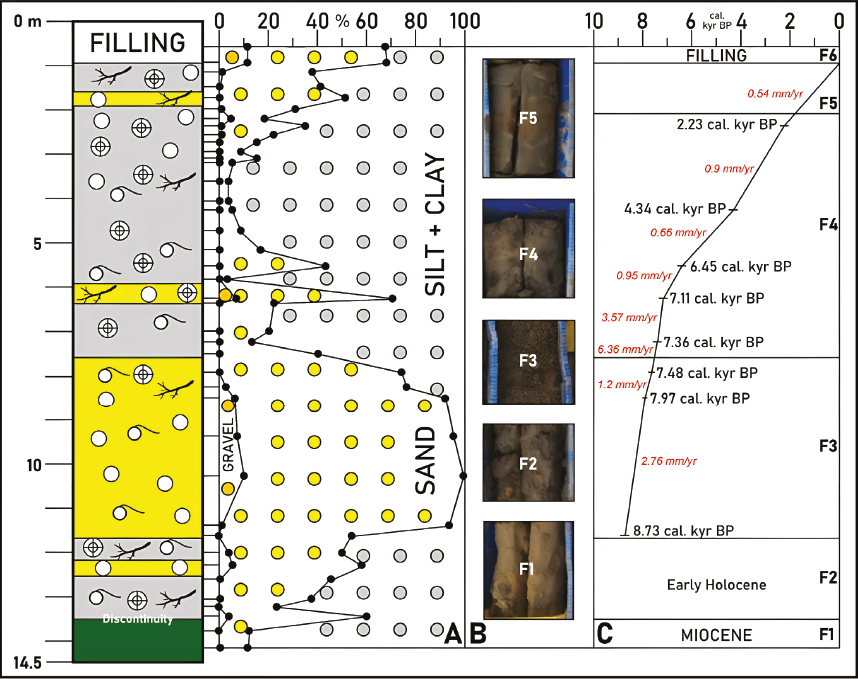
Figure 2 A. Textural analysis of core A. B. Photographs of the different sedimentary facies. C. Age model of core A.
Facies 3 (F3; 11.6-7.6 m depth; samples A-32 to A-26) is composed of fine and very fine sands (sand: 53.6-91%) with scarce percentages of gravels (<10%) and silts (< 8.5%) in most samples. This sandy facies includes numerous quartz clasts and frequent shells of brackish gastropods. Near the top of these facies, silt contents increase (24-26%) between 8.2 m depth and 7.6 m depth, coinciding with frequent plant debris, charcoal remains and a few shells of marine bivalves. This decrease in grain size is even more evident in facies 4 (F4; 7.6-1.8 m depth; samples A-25 to A-6), dominated mainly by muddy sediments (silt + clay: 30.3-97.8%; mean: 80.8%) with very variable percentages of sand (2.2-62.2%; mean: 18.5%). Charcoal and plant remains are very abundant, while macrofauna is very scarce or even absent, with a few shells of gastropods restricted to samples A-21 and A-22. Numerous ceramic fragments have been collected in the uppermost 1.2 m of this facies (3-1.8 m depth).
Facies 5 (F5; 1.8-0.9 m depth; samples A-5 to A-3) is characterized by silty sands and sandy silts (sand: 37.9-51.6%; silt + clay: 48.4-61.8%) with abundant millimetre-sized traces of charcoal. Ceramic fragments are frequent in the upper part of these facies, close to the contact with the recent filling (facies F6; 0.9-0 m depth; samples A-2 and A-1). This recent filling took place during the late 20th and early 21st century and consists of muddy sands (sand: 53-56.5%; silt + clay: 32.4-36.1%) with significant gravel content (11%). These sediments include some shells of pulmonate gastropods.
4.2. DATING AND SEDIMENTATION RATES
The silty-clayey sediments of F1 belong to the upper part of the Gibraleon Clay Formation (Civis et al., 1987). Its age is Upper Miocene, according to previous biostratigraphic and magnetostratigraphic studies (Sierro, 1987; Salazar et al., 2016). These sediments are separated from an upper erosional unconformity, which evidences a later erosive period that caused the disappearance of the Pliocene sandy-silty sediments of the Huelva Sand Formation. In this area, a strong erosion of the Neogene formations developed during the Pleistocene, due to the action of fluvial dynamics (Cáceres, 1995). According to these data and the age of the upper sediments (Table 1; limit F2-F3: ~8.73 cal. kyr BP), F2 was probably deposited during the early Holocene.
Table 1 Radiocarbon database of core A.
| Sample | Depth (m) | Laboratory code | Sample material | δ13C% |
14C age (yr BP) |
Calibrated yr BP (2σ) |
Probability distribution |
Mean probability (cal. Kyr BP) |
|---|---|---|---|---|---|---|---|---|
| A-8 | 2.3 | CNA-4919 | Organic matter | -32.7 | 2188 ± 32 | 2321 2103 | 1 | 2.23 |
| A-16 | 4.2 | CNA-4921 | Organic matter | -29.8 | 3904 ± 33 | 4420 4236 | 0.997 | 4.34 |
| A-19 | 5.4 | CNA-4922 | Organic matter | -24.4 | 5668 ± 51 | 6564 6311 | 0.981 | 6.45 |
| A-21 | 6.15 | CNA-4924 | Organic matter | -26.5 | 6196 ± 36 | 7169 6984 | 0.938 | 7.08 |
| A-24 | 7.2 | CNA-4925 | Organic matter | -26.2 | 6443 ± 36 | 7424 7279 | 1 | 7.36 |
| A-26 | 7.8 | CNA-4926 | Organic matter | -24.2 | 6566 ± 35 | 7513 7425 | 0.87 | 7.47 |
| A-28 | 8.3 | CNA-4927 | Wood | -20.7 | 7143 ± 37 | 8020-7928 | 0.908 | 7.97 |
| A-32 | 11.6 | CNA-4931 | Organic matter | -5.3 | 7911 ± 34 | 8790-8598 8980-8913 |
0.739 0.129 |
8.73 |
The age of F3 ranges from ~8.73 cal. kyr BP to ~7.42 cal. kyr BP (Figure 2C). This interval can be divided into two periods of low mean sedimentation rates (MSR) between ~8.73-7.97 cal kyr BP (MSR~2.76 mm/yr) and ~7.97-7.48 cal. kyr BP (MSR~1.2 mm/yr) compared to a final period (~7.48-7.42 cal. kyr BP) with an increasing MSR (~6.36 mm/yr), which continued in the lower part of F4 until ~7.36 cal. kyr BE In other cores collected in several estuaries in southwestern Spain, MSR~5 mm/yr have been found between 10000 cal. kyr BP and 6500 cal. kyr BP, coinciding with the maximum of the MIS-1 transgression in this area (Lario et al., 2002).
F4 was deposited between ~7.42 cal kyr BP and around ~2 cal kyr BP (Figure 2C). The initial moderate MSR of this interval was followed by a progressive decline between 7.36-7.11 cal. kyr BP (MSR~3.57 mm/yr), 7.11-6.45 cal. kyr BP (MSR~0.95 mm/yr), 6.45-4.34 cal. kyr BP (MSR~0.66 mm/yr) and 4.34-2 cal. kyr BP (MSR~0.9 mm/yr). This decrease in MSR has been also observed in the marine zone of the Tinto-Odiel estuary and other adjacent estuaries during this period (MSR~1.5-2 mm/yr; Lario et al., 2002). This decreasing trend persists during the deposition of F2 (~2 cal. kyr BP-Present; MSR~0.54 mm/yr). Recent sedimentation rates deduced in several short cores obtained in the middle part of the Tinto-Odiel estuary are very variable, ranging from 3.9 mm/yr to 0.33 mm/ yr in some tidal flats and other estuarine environments (Davis et al., 2000).
4.3.BENTHIC FORAMINIFERA
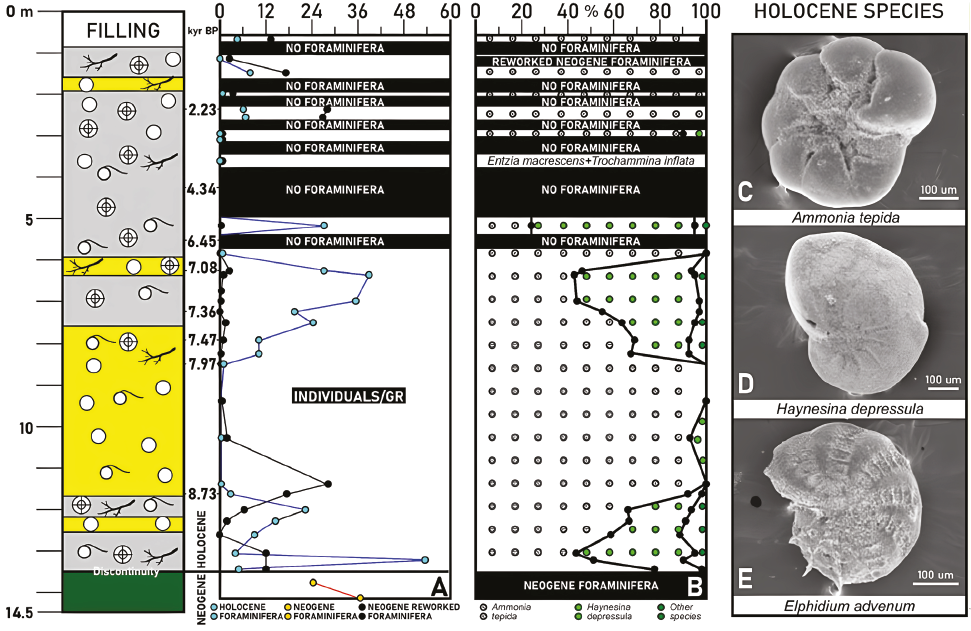
A total of 11616 benthic foraminifera belonging to 69 species have been identified (Table 2). These microorganisms are frequent and diversified especially between 14.5-10.8 m depth (94-1304 individuals/20 grams; 9-33 species/sample), 8.2-5.3 m depth (227-810 individuals/20 grams; 9-16 species/sample), as well as in some samples collected in the uppermost 2.5 m of core A (50-690 individuals/20 grams; 18-26 species/sample).
Table 1 Distribution of benthic foraminifera in core A
| SPECIES/SAMPLES | A-40 | A-39 | A-38 | A-37 | A-36 | A-35 | A-34 | A-33 | A-32 | A-31 | A-30 | A-29 | A-28 | A-27 | A-26 | A-25 | A-24 | A-23 | A-22 | A-21 | A-20 | A-18 | A-14 | A-12 | A-11 | A-9 | A-8 | A-6 | A-4 | A-3 | A-1 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ammonia tepida (Cushman, 1926) | 79 | 559 | 37 | 105 | 193 | 293 | 51 | 7 | 14 | 10 | 11 | 135 | 140 | 309 | 215 | 311 | 327 | 267 | 2 | 131 | 137 | 116 | 17 | 160 | 90 | |||||||
| Elphidium aculeatum (D'Orbigny, 1 846) | 1 | 3 | ||||||||||||||||||||||||||||||
| Elphidium advenum (Cushman, 1922) | 7 | 2 | 5 | 3 | 5 | 2 | II | 12 | 6 | 2 | 2 | 2 | ||||||||||||||||||||
| Elphidium crispum (Linnaeus, 1758) | 1 | 1 | ||||||||||||||||||||||||||||||
| Elphidium macellum (Fitchel & Moll, 1798) | 1 | 2 | ||||||||||||||||||||||||||||||
| HOLOCENE | Elphidium sp. | 2 | ||||||||||||||||||||||||||||||
| Entzia macrescens (Brady, 1870) | 1 | |||||||||||||||||||||||||||||||
| Haynesina depressula (Walker and jacob, 1798) | 21 | 401 | 44 | 53 | 73 | 120 | 3 | 1 | 51 | 45 | 150 | 168 | 368 | 413 | 270 | 382 | 1 | 3 | 3 | 2 | ||||||||||||
| Haynesina germánica (Ehrenberg, 1840) | 2 | 91 | 2 | 14 | 20 | 24 | 10 | 5 | 12 | 3 | 21 | 35 | 57 | 23 | ||||||||||||||||||
| Trochammina inflata (Montagu, 1808) | 1 | 1 | 1 | |||||||||||||||||||||||||||||
| Ammonia beccarii (Linnaeus, 1758) | 17 | 36 | 37 | 42 | 2 | 12 | 29 | 69 | 105 | 10 | 4 | 4 | 10 | 10 | 3 | 4 | 12 | 1 | 7 | 1 | 5 | 8 | 5 | |||||||||
| Ammonia inflata (Seguenza, 1862) | 104 | 51 | ||||||||||||||||||||||||||||||
| Amphicorina scalaris (Bastch, 1791) | 7 | 5 | 9 | 1 | 3 | 13 | 15 | 2 | 1 | 1 | 4 | 1 | 1 | 3 | 28 | 23 | 2 | 10 | 1 | 9 | ||||||||||||
| Amphicorina semicostada (Costa, 1853) | 1 | |||||||||||||||||||||||||||||||
| Amphicorina tenuicostada (Costa, 1853) | 15 | |||||||||||||||||||||||||||||||
| Anomalonoides granosus (Hantken, 1875) | 1 | 1 | ||||||||||||||||||||||||||||||
| Banerella gibbosa (D'Orbigny, 1826) | 1 | 1 | ||||||||||||||||||||||||||||||
| Biloculina sp. | 1 | |||||||||||||||||||||||||||||||
| Bolivina alata (Seguenza, 1862) | 1 | |||||||||||||||||||||||||||||||
| Bolivina catanensis (Seguenza, 1862) | 2 | |||||||||||||||||||||||||||||||
| Bolivina fastigia Cushman, 1936 | 1 | |||||||||||||||||||||||||||||||
| Bolivina puctata D'Orbigny, 1839 | 1 | 1 | 1 | 1 | 1 | 1 | 4 | 1 | 1 | |||||||||||||||||||||||
| Bolivina spatulala (Williamson, 1858) | 22 | 12 | 9 | 13 | 4 | 17 | 19 | 1 | 1 | 3 | 2 | 29 | 31 | 1 | 22 | 3 | 25 | |||||||||||||||
| Bulimina aculeata D'Orbigny, 1826 | 1 | |||||||||||||||||||||||||||||||
| Bulimina costata D'Orbigny, 1852 | 1 | 2 | 1 | 1 | 6 | 1 | 1 | 5 | 6 | |||||||||||||||||||||||
| Bolivina elongata D'Orbigny, 1846 | 229 | 64 | 26 | 38 | 3 | 9 | 10 | 56 | 78 | 1 | 9 | 14 | 2 | 3 | 7 | 8 | 3 | 4 | 102 | 117 | 7 | 47 | 4 | 36 | ||||||||
| Cancris auricula (Fitchel and Moll, 1798) | 6 | 2 | 1 | 1 | 1 | 14 | II | 3 | ||||||||||||||||||||||||
| Cancris oblougus (Williamson, 1858) | 12 | |||||||||||||||||||||||||||||||
| Cassidulina carinata Silvestri, 1896 | 2 | 15 | 1 | 6 | 13 | 25 | 2 | 1 | 1 | 2 | 3 | 54 | 22 | 4 | 27 | 3 | 16 | |||||||||||||||
| Cassidulina levigata D'Orbigny, 1826 | 1 | |||||||||||||||||||||||||||||||
| Cibicides floridanus (Cushman, 1918) | 2 | 1 | 1 | |||||||||||||||||||||||||||||
| Criboelphidium williamsoni (Haynes, 1973) | 1 | |||||||||||||||||||||||||||||||
| Favulina hexagona (Williamson, 1848) | 1 | 1 | ||||||||||||||||||||||||||||||
| Fisuruina piriformis (Buchner, 1940) | 1 | 1 | ||||||||||||||||||||||||||||||
| Fureskoina subacuta (D'Orbigny, 1846) | 16 | 13 | 10 | 17 | 8 | 27 | 59 | 2 | 1 | 3 | 46 | 62 | 2 | II | 1 | 8 | ||||||||||||||||
| Globobulimina affinis (D'Orbigny, 1839) | 45 | 1 | 4 | 3 | 15 | 18 | 2 | 6 | 1 | 2 | 9 | 3 | 4 | |||||||||||||||||||
| Globobulimina auriculata (Baylei, 1894) | 2 | 3 | 3 | 1 | ||||||||||||||||||||||||||||
| Gyroidina umbonata (Silvestri, 1898) | 2 | |||||||||||||||||||||||||||||||
| Hansenisca soldanii (D'Orbigny, 1826) | 1 | 1 | 3 | 4 | 2 | 6 | 15 | 3 | 7 | |||||||||||||||||||||||
| NEOGENE | Heterolepa bellincionii (Giannini and Tavani, I960) | 4 | 7 | 1 | 5 | 7 | 18 | 2 | 1 | 2 | 1 | 14 | 4 | 12 | 9 | |||||||||||||||||
| Homaloedra williamsoni (Alcock, 1865) | 1 | |||||||||||||||||||||||||||||||
| Lagena semistriata Williamson, 1848 | 1 | |||||||||||||||||||||||||||||||
| Lagena striata (D'Orbigny, 1839) | 2 | 6 | 3 | 1 | 2 | 3 | 1 | 3 | 5 | 1 | 1 | 4 | ||||||||||||||||||||
| Lenticulina calcar (Linnaeus, 1758) | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 2 | 2 | 1 | 2 | |||||||||||||||||||||
| Lenticulina inornata (D'Orbigny, 1846) | 3 | 1 | 1 | 2 | 1 | |||||||||||||||||||||||||||
| Lenticulina rotulata (Lamarck, 1804) | 5 | 3 | 3 | 2 | 1 | 2 | 1 | 1 | 3 | 2 | 2 | 3 | 1 | |||||||||||||||||||
| Lenticulina sp. | 2 | 1 | 4 | 5 | 2 | 8 | 2 | 5 | 5 | 3 | 1 | 3 | 1 | 3 | 9 | 8 | 2 | |||||||||||||||
| Lobaluta lobatula (Walker and Jacob, 1798) | 1 | 2 | ||||||||||||||||||||||||||||||
| Marginulinopsis costata (Batsch,, 1791) | 2 | 1 | 1 | 1 | 1 | 1 | ||||||||||||||||||||||||||
| Martinottiella communis (D'Ortbingy, 1846) | 1 | 2 | ||||||||||||||||||||||||||||||
| Melonis pompilioides (Fitchel and Moll, 1798) | 3 | 1 | 1 | 5 | 7 | 1 | 1 | 10 | 5 | 1 | 4 | 1 | ||||||||||||||||||||
| Nonion faba (Fitchel and Moll, 1798) | 257 | 297 | 114 | 71 | 12 | 53 | 75 | 178 | 9 | 3 | 2 | 7 | 17 | II | 4 | 8 | 15 | 5 | 2 | 4 | 6 | 185 | 240 | 33 | 159 | 23 | 126 | |||||
| Nonionoides turgidus (Williamson, 1858) | 3 | 1 | ||||||||||||||||||||||||||||||
| Oolina borealis Loeblich and Tappan, 1954 | 2 | 1 | ||||||||||||||||||||||||||||||
| Pandaglandulina dinapolii Loeblich and Tappan, 1955 | 1 | 1 | 2 | 1 | 3 | |||||||||||||||||||||||||||
| Planorbulina medditerranensis D'Orbigny, 1826 | 1 | 2 | ||||||||||||||||||||||||||||||
| Pullenia bulloides (D'Orbigny, 1846) | 2 | 1 | 3 | 1 | ||||||||||||||||||||||||||||
| Pullenia quinqueloba (Reuss, 1851) | 1 | 1 | ||||||||||||||||||||||||||||||
| Pyramidulina raphanus (Linnaeus, 1758) | 1 | 1 | ||||||||||||||||||||||||||||||
| Quinqueloculina seminulum (Linneus, 1758) | 5 | 1 | 2 | |||||||||||||||||||||||||||||
| Rectuvigerina multicostata (Cushman and Jarvis, 1929) | 1 | |||||||||||||||||||||||||||||||
| Saracenaria ita/ica Defrance, 1824 | 1 | |||||||||||||||||||||||||||||||
| Sigmolopsis celata (Costa, 1855) | 1 | |||||||||||||||||||||||||||||||
| Textularia agglutinans (D'Orbigny, 1839) | 7 | 25 | 28 | 16 | 6 | 9 | 9 | |||||||||||||||||||||||||
| Textularia sagittula Defrance, 1824 | 2 | |||||||||||||||||||||||||||||||
| Trifarina fornasinii (Selli, 1948) | 3 | |||||||||||||||||||||||||||||||
| Uvigerina mediterranea Hofker, 1932 | 3 | 2 | 1 | 1 | 1 | |||||||||||||||||||||||||||
| Uvigerina tenuistriata Reuss, 1870 | 14 | 4 | 1 | 8 | 1 | 1 | ||||||||||||||||||||||||||
| Valvulineria bradyana Fornasini, 1900 | 3 | |||||||||||||||||||||||||||||||
| TOTAL NUMBER OF BENTHIC FORAMINIFERASTUDIED | 735 | 491 | 343 | 1304 | 94 | 222 | 425 | 785 | 620 | 43 | 23 | 22 | 21 | 227 | 254 | 518 | 398 | 715 | 810 | 620 | 2 | 546 | 6 | 15 | 16 | 690 | 688 | 77 | 500 | 50 | 356 | |
| HOLOCENE SPECIES (%) | 0 | 0 | 29.74 | 81.37 | 90.43 | 80.63 | 68 | 56.43 | 8.87 | 16.28 | 65.22 | 45.45 | 52.38 | 88.55 | 79.13 | 93.63 | 98.49 | 98.18 | 95.93 | 92.9 | 100 | 98.35 | 33.33 | 0 | 6.25 | 20.29 | 17.3 | 22.08 | 52 | 0 | 25.84 | |
| NEOGENE SPECIES (%) | 100 | 100 | 70.26 | 18.63 | 9.57 | 19.37 | 32 | 43.57 | 91.13 | 83.72 | 34.78 | 54.55 | 47.62 | 11.45 | 20.87 | 6.37 | 1.51 | 1.82 | 4.07 | 7.1 | 0 | 1.65 | 66.67 | 100 | 93.75 | 79.71 | 82.7 | 77.92 | 68 | 100 | 74.16 | |
The most abundant species are Ammonia tepida (Cushman, 1926) (3716 individuals; 31.95%), Haynesina depressula (Walker and Jacob, 1798) (2572 individuals; 22.11%), Nonion faba (Fichtell and Moll, 1798) (1914 individuals; 16.46%), Bulimina elongata (d'Orbigny), 1846 (877 individuals; 7.54%), Ammonia beccarii (Linnaeus, 1758) (433 individuals; 3.72%) and Haynesina germanica (Ehrenberg, 1840) (299 individuals; 2.57%), which make up almost 85% of the benthic foraminifera extracted. Most of the remaining species do not exceed 1% of the total number of benthic foraminifera.
5. Discussion
5.1. BIOSTRATIGRAPHICAL REMARKS
The distribution of these microorganisms in Neo-gene and Holocene (paleo)environments in southwestern Spain (Sierro et al., 1982; González-Regalado and Civis, 1987; González-Regalado, 1989; González-Regalado et al., 2001; Abad et al., 2022) allows a differentiation between Neogene and Holocene benthic foraminifera (Table 2). Fifty-nine reworked Neogene species have been determined, which come from the erosion of the Miocene-Pliocene formations surrounding La Fontanilla cove. The main Neogene species are N. faba, B. elongata, A. beccarii, Fursenkoina subacuta (d'Orbigny, 1846), Bolivina spathulata (Williamson, 1858) and Cassidulina carinata (Silvestri, 1896).
These species are abundant in the three basal samples of core A, the basal samples of F2 and the uppermost 2.2 m of this core (Figure 3A-B; up to 28 individuals/gram).
5.2. AUTECOLOGY OF THE MAIN SPECIES
In a general overview, Neogene reworked species are typical of shallow marine environments (< 100 m depth). N. faba and B. elongata are common in infralittoral areas (< 40 m depth), although both species have been reported up to 100 m depth (Mendes et al., 2004; Pipper and Reichenbaker, 2009; García Sanz and Usera, 2015; Tulburé et al., 2017). Ammonia inflata is another species that is also commonly found within this depth range (Colom, 1974; Mateu-Vicens et al., 2014).
A. tepida and H. depressula, the two main Holocene species, are very frequent in estuaries, coastal lagoons, marshes and even bays. They are euryhaline species that can withstand large variations in salinity and environmental stress conditions (see review in Murray, 2006). In these environments, A. tepida is more abundant in more open, deeper areas with higher tidal turnover, such as tidal channels, intertidal mudflats, areas closer to the mouth of the estuaries and lagoons or even shallow infralittoral areas (Jorissen, 1988; Debenay and Guillou, 2002; Blázquez, 2005; Frontalini et al., 2011, 2013). This endopelic species feeds on detritus, cyanobacteria or refractory organic matter (Goldstein and Corliss, 1994; Moodley et al., 2000; De Nooijer, 2007). It is a primary pioneer in paralic environments (Debenay et al., 2009).
H. depressula is well represented in marshes, intertidal flats or bays with freshwater inputs (Cearreta, 1989; Pascual et al., 2002; Debenay and Fernández, 2009; Calvo and Langer, 2010). It is a less tolerant species to stress conditions compared with Ammonia tepida and tends to disappear when organic supplies increase or under low oxygen levels (Jorissen et al., 2018; Melis et al., 2019; Rozic et al., 2022). Although both species are common in sediments with highly variable mud contents (Murray, 2006), H. depressula have negative correlations with sand contents and correlates positively with silt percentages while A. tepida is more abundant in sandy sediments of some Mediterranean coastal environments (Melis et al., 2019).
5.3. PALEOENVIRONMENTAL INTERPRETATION OF CORE
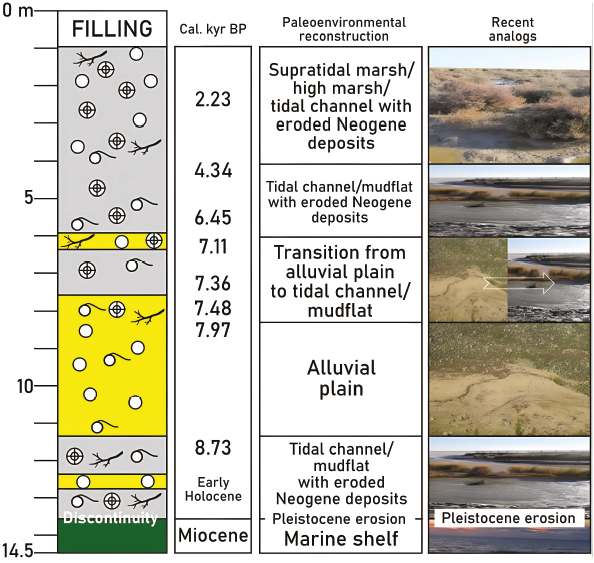
The textural analysis, the paleontological record and the eight dates permit the reconstruction of the paleoenvironmental evolution of core A (Figure 4). The foraminiferal record of the basal clays and silts (F1) is composed exclusively of Neogene species, with an abundant population of N. faba, B. elongata and A. inflata. According to these features, the base of this core is composed of Messinian fine sediments that form the upper part of the Gibraleon Clay Formation, which were deposited on a marine shelf (Sierro et al., 1982; González-Regalado and Civis, 1987; Abad et al., 2022).
F2 (13.5-13.2 m depth) is separated from F1 by an erosional discontinuity. It is characterized by an increasing grain size and the mixing of both Neogene and Holocene foraminiferal species. The topographic height of core A (3.5 m amsl), the contact between F1 and F2 (13.5 m depth), the lithological features cited, the autecology of the Holocene species (see previous section), the age of the upper sediments (Table 2; limit F3-F4: ~8.73 cal kyr BP) and the geological and topographic arrangement of the aforementioned Neogene formations in the surroundings of La Fontanilla cove (Arroyo et al., 2021) would indicate that:
1. A strong erosion caused the dismantling of ~21 m of the Gibraleon Clay Formation and ~4-5 m of the Huelva Sand Formation during the Pleistocene in this area. This erosion has been inferred in other cores collected in this estuary (e.g.Borrego et al., 1999); and
2. In F2, the abundance of A. tepida, H. depressula and H. germanica is characteristic of shallow, restricted brackish intertidal channels or low marshes with muddy substrates (Redois and Debenay, 1996; Debenay and Guillou, 2002; Guerra et al., 2020) and consequently this area was flooded during the early Holocene, coinciding with the progressive advance of the MIS-1 transgression. This paleoenvironment is also inferred by the presence of the gastropod Peringia ulvae, which lives in the inner, silty-sandy areas of estuaries and marshes with low hidrodynamics and important fluvial inputs (Gómez, 2017). The frequent shells of reworked species would be indicative of the transport of alluvial sediments from the nearby Neogene formations.
Near the boundary between F2 and F3 (~8.73 cal kyr BP), the alluvial inputs increase is reflected in an increasing grain size (Figure 2A), the increase of the reworked Neogene foraminifera (Figure 3A-B) and the disappearance of Haynesina spp. (Figure 3C), a species less tolerant than Ammonia tepida to episodes of environmental stress (Jorissen et al., 2018; Melis et al., 2019; Rozic et al., 2022). Most of F3 (13.2-8.2 m depth; ~8.73-7.97 cal. kyr BP) is characterized by sandy sediments with numerous quartzite clasts, frequent specimens of P. ulvae and a scarce foraminiferal population. These features denote the implantation of an alluvial plain, also observed in other cores of this area (e.g.Abad et al., 2022).
The predominance of Ammonia tepida and the scarce abundance of H. depressula confirms the higher affinity of the former species for sandy substrates compared with the latter (Melis et al., 2019). The frequent shells of the gastropod P. ulvae are compatible with this paleoenvironmental reconstruction, because this species is specially adapted to these changing conditions and it crawls about on the surface of the bottom then the burrow for several hours during low tide (Newell, 2009).
The upper part of F3 (8.2-7.6 m; ~7.97-7.42 cal kyr BP) and the lower part of F4 (7.6-5.9 m depth; ~7.42-7.11 cal kyr BP) are characterized by the transition from sandy to silty-clayey substrates, a remarkable predominance of Holocene species and a moderate abundance of Neogene specimens. These features would point to a transition from an alluvial plain to an intertidal mudflat or a shallow tidal channel, because A. tepida and H. depressula are very abundant in these (paleo-) environments (Wukovits et al., 2017, 2018). The presence of marine bivalves, as well as the highest sedimentation rates of core A (~6.36-3.57 mm/yr) points to an increase in tidal action, coinciding with the progress of the MIS-1 transgression, which peaked around 7000-6500 cal. kyr BP in this area (Zazo et al., 1994).
The upper part of F4 (5.9-1.8 m depth; ~7.11-2 cal. kyr BP) is characterized by muddy sediments and the alternation of samples without foraminifera, others dominated by A. tepida and isolated samples with only agglutinated species [Table 1: sample A-14; Entzia macrescens (Brady, 1870) + Trochammina inflata (Montagu, 1808)], very common in high and low marshes all over the world (e.g., Pascual et al., 2019; Kaminski et al., 2020).
This conjunction points to a stressing alternation between periods of waterlogging in this area and others of subaerial exposure, with a periodic transition from intertidal environments to high marshes with abundant vegetation and a continuous transport of Neogene sediments from nearby outcrops (Figure 3A). This period coincides with a marked decrease in sedimentation rates (Fig. 2C; MSR: 0.95-0.66 mm/yr), also observed in other cores from this estuary and other adjacent estuaries during the last 5000 years (Lario et al., 2002).
This paleoenvironmental scenario continued over the last 2000 years (F5), with this alternation of: i) tidal channels, with frequent shells of A. tepida (González-Regalado et al., 2001); and b) marshes without connection with marine fluxes, characterized by the absence of Holocene foraminifera, the presence of only Neogene species and very abundant plant debris. The abundance of Miocene-Pliocene species would indicate a periodic strong erosion from nearby Neogene formations during this interval.
6. Conclusions
The multidisciplinary study of core A collected in the middle estuary of the Tinto River has made it possible to infer the palaeoenvironmental evolution of this sector since the Upper Miocene to the present day.
The substrate of this core consists of Miocene clays deposited on a marine platform, part of which were eroded during the Pleistocene by fluvial dynamics. Subsequently, this area was flooded during the Holocene transgression, with the development of tidal channels or mudflats and an alluvial plain during the early-middle Holocene.
The late Holocene is characterized by tidal channels and marshes with scarce foraminifera or even absence of them. During this palaeoenvironmental evolution, the constant erosion of the adjacent Neogene formations is noted, with the presence of Miocene-Pliocene species in the sediments. Finally, a recent filling makes up the upper part of the core.
Contributions of authors
All authors have contributed to conceptualisation of the paper, review of previous information, field work, interpretation and discussion of results, and in the writing of this article.
Financing
This paper has been mainly financed by the Palos de la Frontera Council. It is also carried out through the following projects: a) DGYCIT project CTM2006-06722/MAR; b) DGYCIT project CGL2006-01412; c) 'Roman cities of the Baetica. CORPVS VRBIVM BAETICARVM (I) (CUB)' (Andalusian Gouvernement); d) 'From the Atlantic to the Tyrrhenian. The Hispanic ports and their commercial relations with Ostia Antica. DEATLANTIR II - HAR2017-89154-P (Plan Nacional de I+D+i); and e) FEDER 2014-2020 project UHU-1260298. Other funds have come from Andalusian Government (groups HUM-132, RNM-238, and RNM-293). It is a contribution to the Research Center in Historical, Cultural and Natural Heritage (CIPHCN) of the University of Huelva.
Conflict of interest
The authors declare that there are no conflicts of interest.
Handling editor
Julieta Masaferro.











 nueva página del texto (beta)
nueva página del texto (beta)