Introduction
Deforestation affects the ecological processes that govern recruitment, as it regulates dispersal, fertile seed production, pollination, germination, and growth. The effects of deforestation also include the deterioration of soil and environmental quality (e.g., humidity, temperature), which together provide the microenvironmental requirements necessary for tree species establishment (Ramírez-Marcial et al., 2001; Garibay-Orijel et al., 2013). In highly deforested sites, the microhabitats of late- successional species are lost. As a result, only pioneer species thrive, leading to the over-representation of some trees and affecting long-term species composition and diversity (Gutiérrez-Granados & Dirzo, 2009; Rebottaro & Cabrelli, 2011; Monárrez-González et al., 2018).
Several pine species are considered invasive in severely deforested areas because these conifers successfully germinate and establish in open areas (González‐Espinosa et al., 1991; Richardson & Bond, 1991; Galindo-Jaimes et al., 2002). Figueroa-Rangel et al. (2008) reported an increase in pine dominance in conifer forests due to logging and deforestation for agricultural areas because shade and humidity are reduced in these environments. Once pine species dominate the forest, the soil becomes more compact, drier, hotter, less acidic, and less fertile, all of which restricts the establishment of non-pine species (Romero-Nájera, 2000; Galindo-Jaimes et al., 2002).
The conifer forest is the second most deforested ecosystem in Mexico, covering only 16.6 % of the national territory (Guerra-de la Cruz & Galicia, 2017; Secretaría de Medio Ambiente y Recursos Naturales [Semarnat, by its acronym in Spanish], 2016). La Malinche National Park (LMNP) harbors part of the conifer forest of the country, and owing to its high diversity and endemisms (Domínguez-Domínguez & Pérez-Ponce de León, 2009; Arriola et al., 2014; Villaseñor & Ortiz, 2014), is one of the ten Priority Inland Regions (PIR) of the central and the Trans-Mexican Volcanic Belt (TVB) regions (Comisión Nacional de Áreas Naturales Protegidas [Conanp, by its acronym in Spanish] & Cooperación Alemana al Desarrollo Sustentable [GIZ, by its acronym in German], 2018). However, forest cover loss due to logging and agriculture continues in the LMNP despite being decreed as a Protected Natural Area in 1938 (Fernández & López- Domínguez, 2005). Today, the LMNP is considered the most significantly isolated mountain in Mexico (Semarnat & Conanp, 2013), with a large portion of the area (~54 %) converted to cropland, human settlements, and induced pastures (Rojas-García & Villers-Ruíz, 2008).
Although the LMNP is a major contributor to biodiversity conservation and connectivity in the Central and TVB regions, only the vegetation composition and structure (Fernández & López-Domínguez, 2005; Rojas- García & Villers-Ruíz, 2008) and the physiological aspects of some tree species have been investigated (Franquiz, 2016; Portillo, 2017; Franquiz, 2018; Montero, 2020; George-Miranda et al., 2022). This study analyzed the impact of deforestation on seedling recruitment and diversity in the LMNP conifer forest. We expected a negative effect of deforestation on tree seedling diversity and recruitment, and a higher abundance of pine seedlings in deforested sites. This research reports valuable information to understand the consequences of deforestation on natural regeneration dynamics, which may be useful in the design of conservation strategies for the LMNP and other conifer forests in Mexico.
Objectives
The specific objectives of the present study were (1) to describe the diversity and recruitment of tree seedlings in the LMNP conifer forest; (2) to evaluate the effect of deforestation on these variables; and (3) to identify the relationship between deforestation and the abundance of pine and non-pine seedlings.
Materials and methods
Study Area
The LMNP stretches across 46093 hectares, covering the states of Tlaxcala (33161 ha) and Puebla (12932 ha) between coordinates 19º06’30”-19º20’19” N and 97º55’32”-98º09’55” W. The climate in the LMNP is temperate subhumid, with a mean annual temperature of 6 °C to 8 °C and a mean annual precipitation of 800 mm to 1000 mm, with heavier rains from June to September (Fernández & López-Domínguez, 2005). The prevailing vegetation types are pine forest, fir forest, and pine-oak forest (Rzedowski, 2006). The dominant tree species include Pinus montezumae Lamb., P. hartwegii Lindl., P. leiophylla Schiede Ex Schltdl. & Cham., Abies religiosa (Kunth) Cham. & Schlecht, and Alnus jorullensis Kunth (Rojas-García & Villers-Ruíz, 2008). Sampling in the LMNP was carried out between 2800 m a.s.l. and 3000 m a.s.l. because this area harbors the highest species diversity. Above this altitude, Abies religiosa and Pinus hartwegii dominate with monospecific forests, while deforestation is greater below this range (Rojas-García & Villers-Ruiz, 2008).
Three 1-km2 forest stands were located on each of the four slopes of the LMNP (North, N; South, S; East, E; and Southeast, SE). Within each forest stand, five circular plots measuring 1000 m2 were located at random (Fig. 1). In each, the current logging was estimated as an approximation of forest use that has partly caused deforestation (Adekunle et al., 2013). Although the current logging measurement does not reflect the historical use, we consider it a useful variable that influences the establishment of tree seedlings since it causes canopy opening and changes in the microenvironment (Pessoa et al., 2017; Monárrez- González et al., 2018).
E, East Slope; W, West Slope; N, North Slope; S, Southeast Slope.
Figure 1 Location of forest stands for sampling seedlings to estimate recruitment and diversity in La Malinche National Park.
For estimating the current logging, the relative density and basal area of damaged individuals (i.e. logged individuals > 20 cm in diameter, taken at 30 cm from the ground, with mechanical damage caused by an ax, chainsaw or/and bowie knife, known in Mexico as machete) within plots were calculated using the following equations:
where:
Ddind = relative density of damaged individuals
ΣDdind = sum of the relative densities of damaged individuals
TD = total density
where:
RGdind =relative basal area (see de Pinto de Lope et al., 2018) of damaged individuals
ΣGdind =sum of the basal areas of damaged individuals
TG = total basal area
Subsequently, to determine the relative logging, the values for the plot of interest were divided by the lowest density and basal area values for the entire LMNP. The overall logging for each forest stand was calculated as the arithmetic mean of the values of the respective plots, which represented the sum of density and basal area values (Sapkota et al., 2010).
As a second indicator of deforestation, the percent forest coverage (%FC) was measured with a convex spherical densitometer (model-A) at four points within each plot (i.e. North, South, East, and West) at a distance of 11 m from the plot center. Forest coverage by stand was calculated as the average value for the plots in each stand.
Recruitment Sampling
Four 10-m2 circular subplots were established within each 1000-m2 plot; these subplots were oriented according to the four cardinal points (North, South, East, West), with 10 m from the center to the edge (Ramírez-Marcial et al., 2001) (Fig. 1). A total of 20 subplots were established in each forest stand. In all subplots, each tree seedling (<0.5 m in height) was recorded, marked (Ramírez-Marcial et al., 2001), and identified with botanical guides based on the vegetative characteristics of adult trees (Perry, 1991).
One year after the sampling plots were established (November 2020), a second sampling was carried out to record the number of new individuals established and thus estimate seedling recruitment per plot, excluding those seedlings counted in the first sampling. Additionally, seedling abundance per plot was recorded as the number of individuals of each species observed in the first sampling (Giacomotti & Reynel, 2018; Ocampo-Zuleta & Bravo, 2019).
Analysis
Seedling diversity was determined from the records of the first sampling and using the additive partitioning method of species diversity introduced by Whittaker (1960): ƴ = α + β, using species richness estimates (Lande, 1996). Alpha diversity (α) is the number of species within-community (forest stand), beta diversity (β) is the exchange of species between communities (forest stands), and gamma diversity (ƴ) is the sum of species of alpha and beta diversity in each forest stand (regional diversity) (Pereyra & Moreno, 2013). These three components (α, β, ƴ) are considered a set of additive elements expressed in the same units (species), which allow for comparing diversity values between sites of interest (Calderón-Patrón et al., 2012). Furthermore, the dominant species were identified from the rank-abundance curves of each species by forest stands.
Relationships
Potential effects of deforestation (i.e., current logging and percent forest coverage) on seedling diversity, abundance and recruitment (dependent variables) were determined by performing Generalized Linear Mixed Models (GLMM) using the Poisson family, whose random variable was the slope; the statistically significance of the models was evaluated using an analysis of variance (ANOVA). Additionally, GLMMs were performed for each species to assess the relationship between deforestation and seedling abundance for pine and non-pine species. All analyses were performed with the program R v. 3 (adipart, AICcmodavg, betapart, Biodiversity, car, lme4, lmerTest, MASS, MuMIn, and vegan packages; R Core Team, 2017).
Results
Recruitment
The largest numbers of recruits (R) were recorded under 3- E (R = 383 individuals) and 3-S (R = 381 individuals). In contrast, the lowest recruitment was observed under 1-W (R = 1 individual) and 2-S (R = 0 individuals) (Table 1). Seedling abundance (Ab) was highest under 2-E (Ab = 32 individuals), while the lowest abundances were recorded under 1-E (Ab = 0 individuals) and 1-S (Ab = 2 individuals; Table 1, Appendix 1).
Table 1 Deforestation, recruitment, and additive diversity of tree seedlings in forest stands (FS) on the four slopes of La Malinche National Park.
| Deforestation | Recruitment | Diversity | |||||
| FS | %Cov | Logging | Recruits | Abundance | α | β | ƴ |
| 1-E | 74.6 | 10.62 | 5 | 0 | 0 | 0 | 0 |
| 2-E | 75 | 18.17 | 11 | 32 | 1 | 3 | 4 |
| 3-E | 51.6 | 5.01 | 383 | 21 | 2 | 2 | 4 |
| 1-W | 73 | 11.69 | 1 | 4 | 1 | 1 | 2 |
| 2-W | 54.8 | 11.86 | 37 | 18 | 2 | 2 | 4 |
| 3-W | 49.6 | 0 | 8 | 16 | 1 | 0 | 1 |
| 1-N | 78.6 | 16.04 | 2 | 8 | 1 | 1 | 2 |
| 2-N | 61.8 | 7.38 | 71 | 10 | 2 | 1 | 3 |
| 3-N | 70.8 | 5.01 | 25 | 15 | 1 | 1 | 2 |
| 1-S | 55 | 1.08 | 136 | 2 | 1 | 0 | 1 |
| 2-S | 64.2 | 0 | 0 | 16 | 2 | 1 | 3 |
| 3-S | 36 | 29.19 | 381 | 17 | 1 | 1 | 2 |
%Cov, Forest coverage percentage; E, East Slope; W, West Slope; N, North Slope; S, Southeast Slope; α, alpha diversity; β, beta diversity; ƴ, gamma diversity (Whittaker, 1960).
Seedling Diversity
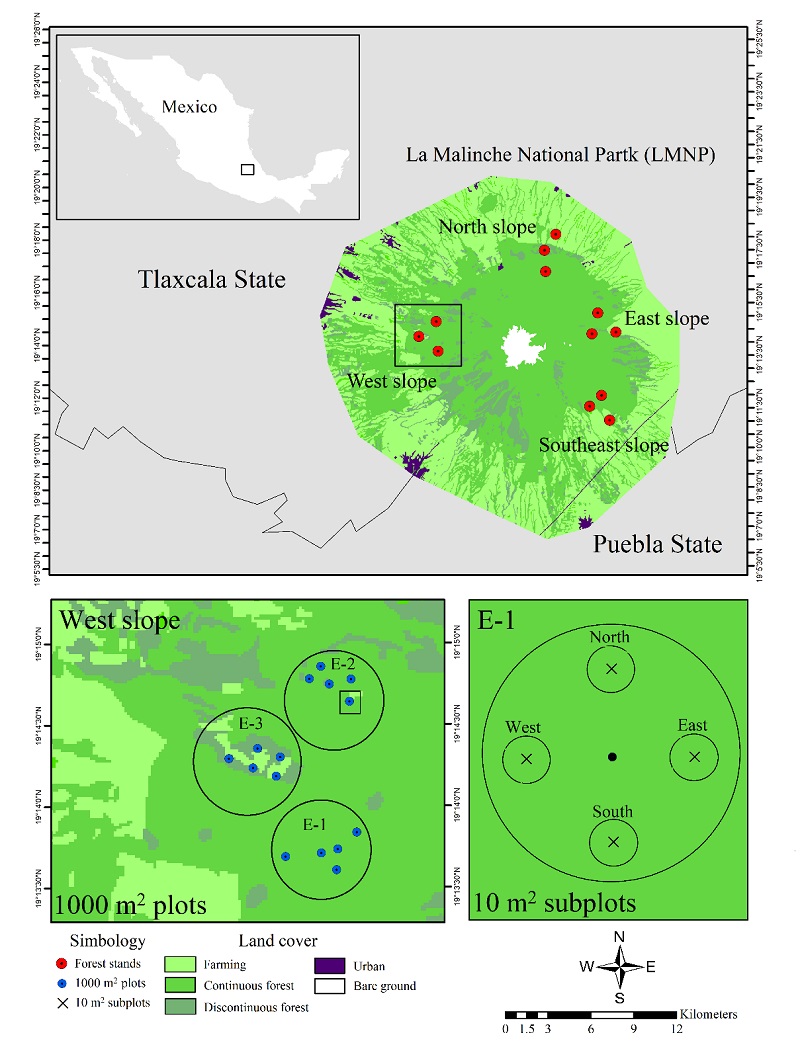
We identified seedlings of seven tree species: Prunus serotina Ehrh., Quercus crassipes, Pinus montezumae, Salix oxylepis Schneid., Abies religiosa, Pinus pseudostrobus, and Arbutus xalapensis (Appendix 1). In general, Pinus montezumae was the dominant species, except in the North slope (2-N and 3-N) and the Southeast slope (1-S and 2-S), where the most abundant species were Salix oxylepis, Quercus crassipes, Prunus serotina, and Arbutus xalapensis, in that order (Fig. 2). Pinus pseudostrobus was the least abundant species over the entire LMNP, with only one seedling recorded (Appendix 1).
E, East slope; W, West slope; N, North slope; S, Southeast slope.
Figure 2 Rank-abundance curves of tree seedlings by regional forest stands of La Malinche National Park.
Alpha diversity (α) was higher under 3-E, 2-W, 2-N, and 2-S (α = 2) and was zero under 1-E. Beta diversity (β) was heterogeneous, being highest in 2-E (β = 3 species) and nil under 1-E, 3-W, and 1-S (β = 0 species). The lowest gamma diversity (ƴ) was observed under 1-E (ƴ = 0 species), but the highest total diversity was found in 2-E, 3- E and 2-W (ƴ = 4 species) (Table 1).
Relationships
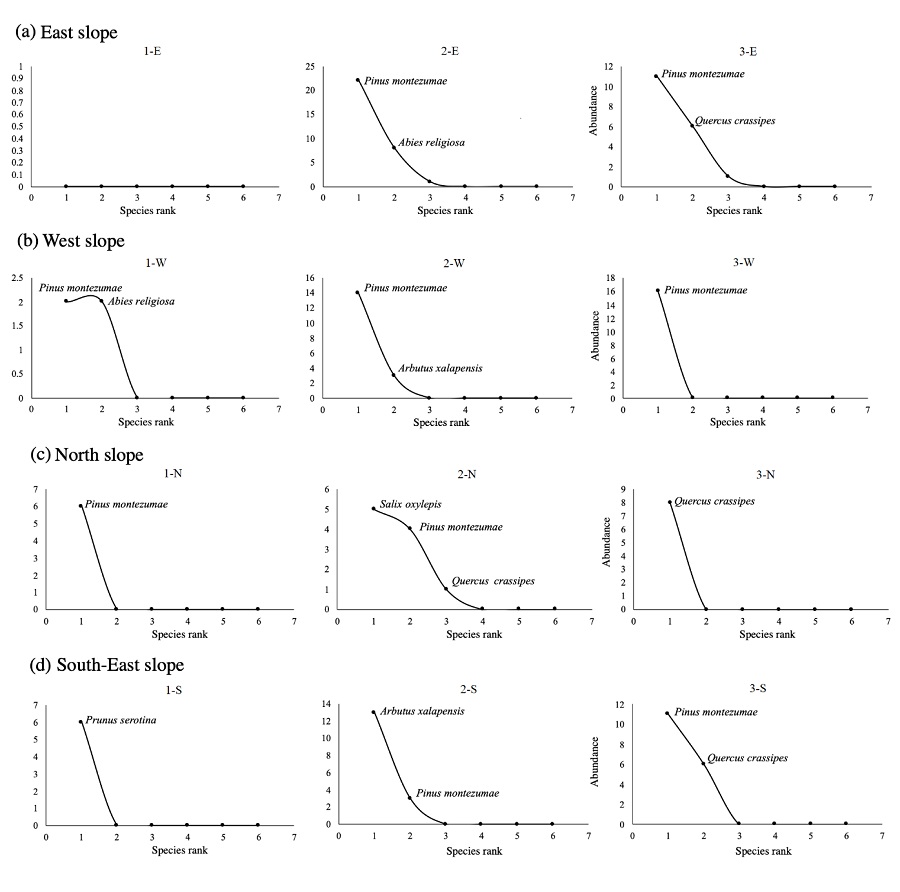
This study found no association between deforestation and tree seedling diversity. Generalized Linear Mixed Models (GLMMs) identified eight statistically significant models that showed relationships of seedling recruitment and abundance with deforestation. Of these, the best models, according to the AIC (Akaike, 1974), were those that explained seedling recruitment (R) with the interaction between logging and percent forest coverage (%FC) (R ~ Logging : %FC + (1|Slope), AIC = 3975, P < 0.001), and the abundance (Ab) with percent forest coverage (Ab ~%FC + (1|Slope), AIC = 390, P < 0.001) (Table 2a and 2b). Recruitment was higher as forest coverage and logging decreased (Fig. 3a), and seedlings abundance increased at lower forest coverage (Fig. 3b).
Table 2 Statistically significant generalized linear mixed models of tree seedling abundance (a), recruitment (b) and species (c) in La Malinche National Park related to logging and percent forest coverage (%FC).
| Model | AIC | BIC | logLik | Deviance | P-value | |
| (a)Abundance | ||||||
| Abundance ~ Logging + (1|Slope) | 394 | 401 | -194 | 280 | <0.001 | |
| Abundance ~ %FC + (1|Slope) | 390 | 396 | -192 | 273 | <0.001 | |
| Abundance ~ Logging + %FC + (1|Slope) | 390 | 398 | -191 | 271 | <0.001 | |
| Abundance ~ Logging : %FC + (1|Slope) | 394 | 401 | -194 | 282 | <0.001 | |
| (b)Recruitment | ||||||
| Recruitment ~ Logging + (1|Slope) | 4091 | 4098 | -2043 | 4185 | <0.001 | |
| Recruitment ~ %FC + (1|Slope) | 4311 | 4317 | -2152 | 3940 | 0.67 | |
| Recruitment ~ Logging + %FC + (1|Slope) | 4067 | 4076 | -2030 | 3832 | <0.001 | |
| Recruitment ~ Logging : %FC + (1|Slope) | 3975 | 3963 | -1975 | 67.5 | <0.001 | |
| (c)Tree species | ||||||
| Quercus crassipes ~ Logging : %FC + (1|Slope) | 103 | 110 | -48.7 | 226 | 0.01 | |
| Pinus montezumae ~ %FC + (1|Slope) | 307 | 313 | -150 | 0.15 | <0.001 | |
| Pinus montezumae ~ Logging + %FC + (1|Slope) | 307 | 315 | -149 | 0.13 | <0.001 | |
| Salix oxylepis ~ %FC + (1|Slope) | 30 | 36 | -12 | 8.62 | 0.04 | |
| Abies religiosa ~ %FC + (1|Slope) | 75 | 81 | -34 | 52.5 | <0.001 | |
| Abies religiosa ~ Logging + %FC + (1|Slope) | 40 | 49 | -16 | 16.2 | <0.001 | |
| Abies religiosa ~ Logging : %FC + (1|Slope) | 37 | 43 | -16 | 23.0 | <0.001 | |
The best models according to the corrected Akaike Information Criterion (AIC; Akaike, 1974) are highlighted in bold. Slope, random variable.
The dots in figure 3a depict each record, and the vertical lines the distance relative to the model’s slope.
Figure 3 Relationships best evaluated by the Akaike Information Criterion (AIC; Akaike, 1974) obtained with generalized linear mixed models between recruitment (a) and abundance (b) of tree seedlings from La Malinche National Park (LMNP) and logging and forest coverage as explanatory variables, using slope as random variable.
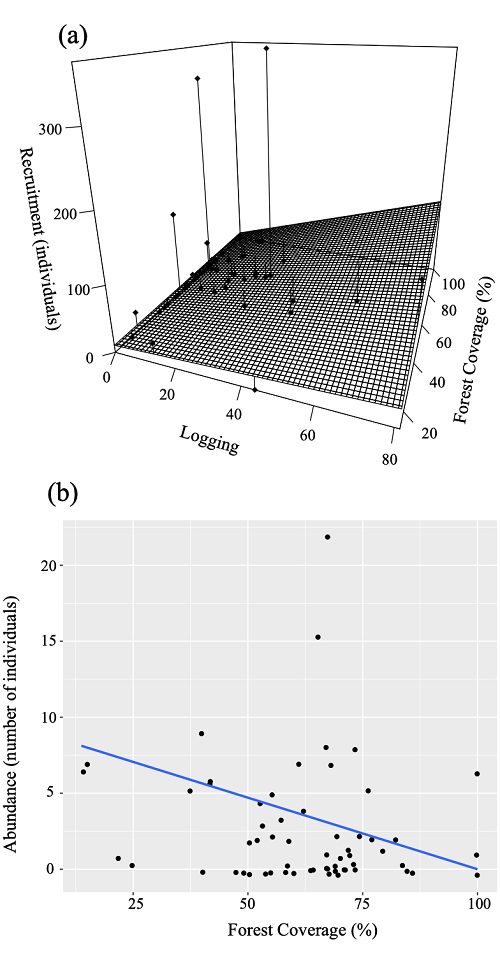
In addition to detecting the interactive effect of logging and forest coverage loss on the abundance of Q. crassipes and Abies religiosa as the optimal models, we also observed the influence of deforestation on Pinus montezumae and S. oxylepis (Table 2c). According to these models, Q. crassipes abundance was higher with the interaction relationship between lower forest coverage and logging, whereas Abies religiosa abundance increased in areas with higher forest coverage and lower logging (Fig. 4a and 4d, respectively). Moreover, Pinus montezumae and S. oxylepis abundances were lower as forest coverage increased (Fig. 4b and 4c).
The dots in Figure 4a and 4d depict each record, and the vertical lines the distance relative to the model’s slope.
Figure 4 Relationships that are best evaluated by the Akaike information criterion (AIC; Akaike, 1974) obtained with generalized linear mixed models between seedlings, by species, from La Malinche National Park (LMNP) and logging and forest coverage as explanatory variables, using slope as random variable.
Discussion
Deforestation gradually modifies ecosystems by influencing species succession and forest dynamics (Trigueros- Bañuelos et al., 2014). The effect on diversity depends on connectivity between forest patches, the requirements of each species, and the environmental conditions that remain after the event (Leibold et al., 2004; Pérez et al., 2013). This study reports an interactive effect of measured deforestation variables on tree seedling recruitment in the LMNP conifer forest. In addition, seedling abundance increased with forest cover loss, as did that of Pinus montezumae, S. oxylepis and Q. crassipes, but the latter was also negatively affected by logging. The case of Abies religiosa is of concern because its abundance decreased with the interactive relationship of logging and forest coverage loss. Likewise, Pinus pseudostrobus is at risk of extirpation (local extinction) due to extremely low recruitment. The results did not fully match our expectations since we detected no effect of deforestation on tree seedling diversity.
Habitat destruction by deforestation modifies microenvironmental conditions and affects seed production and dispersal, thus reducing the recruitment of all species (Sherman et al., 2001; Bullied et al., 2012). These alterations can hamper recolonization and cause floristic homogenization, loss of diversity, and local extinctions (Niemelä, 1999; Sherman et al., 2001; Bullied et al., 2012).
In the LMNP, seedling richness was not associated with deforestation, but the interaction of logging and forest coverage loss influenced seedling recruitment. These results indicate that although forest coverage loss benefits seedling establishment by promoting niche opening, mainly for pioneer species (Pessoa et al., 2017; Monárrez-González et al., 2018), logging showed an adverse influence, probably due to seedling crushing during forest use.
In mature forests, shade-intolerant (pioneer) species, commonly adapted to colonize open areas, are poorly represented. The opposite is observed in highly fragmented forests, where shade-tolerant (late-successional) species are lost due to the disruption of the microenvironment required for them to thrive (Connell, 1978; Sheil & Burslem, 2003). In conifer forests, some pine species are known to persist - and even thrive - in heavily disturbed areas (McKinney & Lockwood, 1999). Unfortunately, pine- dominated areas are less diverse because the soil loses nutrients for the germination of all species; additionally, low humidity and shade further restrict suitable conditions for the establishment of late-successional species (Romero- Nájera, 2000; Galindo-Jaimes et al., 2002).
The present study recorded the dominance of Pinus montezumae in most forest stands. Although a high abundance of Pinus montezumae was expected due to the elevation range covered during field sampling, which corresponds to the natural altitudinal range of the species in the park (2800-3200 m asl), deforestation likely facilitates its colonization, as occurs with other pines (Richardson et al., 2007). Ern (1976) indicated that the former climax forest in the LMNP between 2700 and 2300 m asl was formed by Abies religiosa and Cupressus lindleyi. Abies religiosa was even present in large populations outside the ravines of the wettest slopes - the only sites currently inhabited by this species (S. George-Miranda, pers. obs.). Also, Acuña (1984) reported the abundant presence of firs and cypresses in 1577, indicating the major change in the floristic composition of the LMNP favoring the dominance of Pinus montezumae. These reports support the displacement of species by Pinus montezumae favored by forest coverage loss; nevertheless, deforestation also exerted a beneficial effect on S. oxylepis, a species more abundant in intermediate successional stages (Muñoz-Jiménez et al., 2012; Arriola et al., 2014), and whose abundance was lower than that of Pinus montezumae, being dominant in only a single forest stand (2-N).
Of the seven species of tree seedlings recorded (Salix oxylepis, Quercus crassipes, Pinus montezumae, Pinus pseudostrobus, Prunus serotina, Abies religiosa, and Arbutus xalapensis), the low representation of Pinus pseudostrobus indicates very limited recruitment that threatens the permanence of this species in the LMNP. Likewise, the strong adverse effect of logging and forest coverage loss on Abies religiosa abundance jeopardizes the regeneration of this conifer. According to Ern (1976), Pinus pseudostrobus formed tree associations with Abies religiosa on the LMNP west slope at 2400 m asl. This author also proposed that before the intensive clearing of the natural vegetation in the LMNP, this area was likely covered by mixed forests of Abies religiosa and Pinus pseudostrobus. Unfortunately, all these conditions have been lost.
The conservation of Abies religiosa and Pinus pseudostrobus in the LMNP is uncertain because deforestation has severely affected both species. Abies religiosa requires a relatively specialized microenvironment since its germination is determined by high humidity, while its establishment depends on a relatively low light intensity, requiring intermediate tree coverage levels (Franquiz, 2016; Gallardo-Salazar et al., 2019). For its part, Pinus pseudostrobus is highly susceptible to drought stress and soil degradation associated with severe disturbance (Gómez-Romero et al., 2013). Therefore, future studies should monitor the populations of these species and analyze the relationship between their abundance and soil conditions that may be limiting their establishment (e.g., nutrients, humidity).
Although the low abundance of some species may result from the sampling design, the high loss of forest cover in the LMNP (Valdez et al., 2016) has changed the microhabitat conditions needed for the permanence and establishment of some species (even pioneer species in ecological succession). Therefore, controlling deforestation and forest use in the LMNP is of the utmost importance to mitigate their impact on the survival of poorly represented tree species that require particular habitats. According to our results, specific conservation measures should be implemented in the LMNP to ensure the recruitment and conservation of Abies religiosa and Pinus pseudostrobus. Even though this study does not include environmental factors (e.g. soil characteristics) that could influence the results and limit our conclusions, it is a fundamental basis for understanding the effects of human activities on the natural regeneration of temperate forests.
Conclusions
The information reported herein reveals the interactive effect of deforestation indicators (logging and percent forest coverage) on the recruitment of tree seedlings; however, none of these factors was associated with tree diversity. The dominance of Pinus montezumae, S. oxylepis and Q. crassipes increased with the low forest coverage, and of the latter also with low logging. At the same time, the abundance of Abies religiosa decreased with increasing logging and low forest coverage. The poor representation of Pinus pseudostrobus and the risk of losing optimal habitats for Abies religiosa underline the need to study the effect of microclimatic conditions derived from deforestation to identify the causes of low seedling recruitment and define immediate conservation actions to preserve the diversity levels safeguarded by the LMNP. Future studies should explore the dynamics of forest extraction and other factors that could potentially have an effect on natural regeneration (e.g. soil nutrients, temperature and humidity).
The information reported herein is key for understanding the effects of deforestation on the interactions governing the composition and structure of conifer forests and raises new questions (e.g., recruitment dynamics) that will help define effective actions to conserve biodiversity in the LMNP and other conifer forests of Mexico.











 nueva página del texto (beta)
nueva página del texto (beta)


