INTRODUCTION
For many centuries, the provision of good quality water has been a determining factor for both subsistence and the society economy (Qu et al., 2013). Nevertheless, although the percent of freshwater available is low (0.5%), their unsuitable use and the detriment of the water quality is a common phenomenon around the world (CONAGUA, 2016; Sun et al., 2017). Water pollution depicts one of the most alarming environmental problems in the contemporary world (Pulido-Bosch, 2007; Hernández-Flores et al., 2017). In many countries, particularly those with emerging economies, anthropogenic activities related to population growth, industrial development, mining, the excessive use of fertilizers, pesticides and insecticides makes the water unusable for any human activity (Cifuentes et al., 1994; Literathy, 1996; Gómez-Álvarez et al., 2004; Sjömander Magnusson and van der Merwe, 2005; Gordillo-Martínez et al., 2010; Lu, 2011).
In Mexico, some states with important mining activity such as Zacatecas, San Luis Potosí, Guerrero and Sonora have been affected by the pollution by heavy metals (Hg, Ar, Pb and Cr) attributed to the mining industry and effected by the inadequate management of solid mining wastes (Covarrubias and Peña Cabriales, 2017; Corona-Chávez et al., 2017). On the other hand, INEGI (2013) reported that the surface water or shallow groundwater represents the main source of domestic water supply for a large number of rural populations and many cities in Mexico. Even raw or superficial shallow groundwater without any treatment is still used for human consumption, which may represent a risk to the health of the inhabitants.
Taxco de Alarcón (Tx-A), Guerrero, México (18° 33' 24" N; 99° 36' 17" W), recognized as a city of mining origin founded in the mid-seventeenth century is considered a major tourist destination (CRM, 1999; Des Rivières and Beals, 2011). From the 20th century on, mining in Taxco has been centered on base metals Pb, Cu, Zn and precious metals such as Ag and Au. Unfortunately, linked to their production processes, some elements such as As and heavy metals (potentially toxic elements) were produced and released into the environment in waste. Those pollutants are present in the liquid and solid mining waste disposed in the surrounding environment of Tx-A, where the wind, animals and the rain are some of the possible dispersion factors, altering the properties of the soil and water (Romero et al., 2007; Talavera-Mendoza et al., 2016; Corrales-Pérez and Martín-Romero, 2018).
The city has several different water sources. The main source of supply is the surface water of the Chontacuatlán river captured at 18° 39' 15" N; 99° 34' 27" W. Other supply sources of Tx-A are the springs of Chacuhalco and Tenería, and the San Marcos and El Sombrerito dikes (known as dams). These dikes are located in the recharge zone and, therefore, their capturing capacity is very low. In addition, these dikes are located in areas where the hydraulic load is very high, i.e. where the difference between the level of stored water and the water flow of the Chontalcuatán River is almost 1 km.
In most cities, potable water is treated in treatment plants that aim to eliminate pathogenic contaminants and microbes that may potentially harm the health of users. The methods used as well as the efficiency of the process determine the quality of the water that is sent to different places, and finally determine the use for which it is destined. The evaluation of the chemical and microbiological composition of potable water for human consumption, as well as of the chemical composition of suspended particulate matter is an imperative need, not only for determining the water quality but also to evaluate the efficiency of the treatment methods used.
The city of Tx-A has a water treatment plant (WTP) (Figure 1). The water processed in the WTP comes from the Chontalcuatlán river, the Chacuhalco and Tenería springs, and the San Marcos and El Sombrerito dams. According previous analysis, the water of the Chacuhalco spring shows good quality and presents levels of turbidity below the NOM-127-SSA1-1994 standard (Secretaría de Salud, 2000); therefore, water enters directly to the chlorination stage of the WTP. The water from the other sources is collected in a mixing box before being treated. Tx-A treatment plant uses the pitcher method to determine the amount of flocculant aluminum sulphate Al2(SO4)3 to be added by adjusting, for this, the pH of the water between 6.5 and 7.5 with 20 % CaO and HCl solutions.
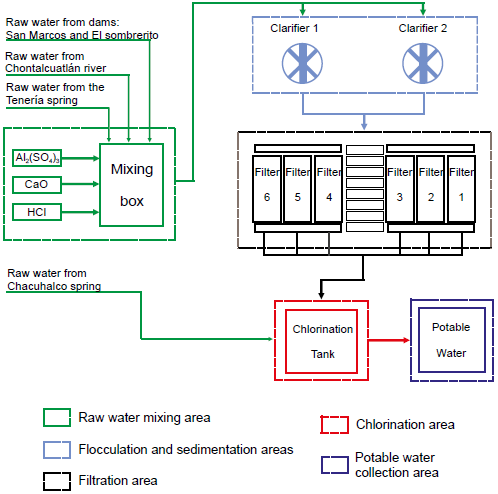
Figure 1 Schematic diagram of the purification process in the Taxco de Alarcón water treatment plant.
The WTP, as many others around the world, is not designed to treat the pollution caused by mining waste and the risk of the presence of this type ofpollutants is high. The pollution of water by As, Cd, Pb, Hg, Tl, among other toxic elements, related to biomagnification of these elements from solid and liquid mining wastes close to urban settlements is an issue of public health concern (Smith and Huyck, 1999; Massoudieh et al., 2010; Talavera-Mendoza et al., 2016).
Thus, due to the importance of this city, its proximity to the mining solid wastes, and the fact that surface water is the main source for supply, we performed this study with the aim of characterize i) the composition and chemical speciation of particulate matter suspended in water to determine the content ofpotentially toxic elements, and ii) the chemical and bacteriological composition of the potable water of Tx-A, according to the Mexican official standard NOM-127-SSA1-1994.
MATERIALS AND METHODS
Thirty-three water samples from the Taxco de Alarcón city were analyzed (Figure 2). One raw water sample comes from the Chacuhalco spring (sample 1, Figure 2), and the second sample is a mixture ofwater of the Chontalcuatlán river, the Teneria spring, and the San Marcos and El Sombrerito dams (sample 2, Figure 2). The third sample was taken from the WTP output (sample 3, Figure 2). Finally, thirty samples come from the water distribution network collected directly from the tap (samples 4-33, Figure 2). The source of the treated water, and the treatment method have remained the same for a decade. Sampling was carried out in July 2007, at the beginning of the rainy season. The selection of sampling points was random; in Figure 2, the sampling points are located on a map of the city of Tx-A.
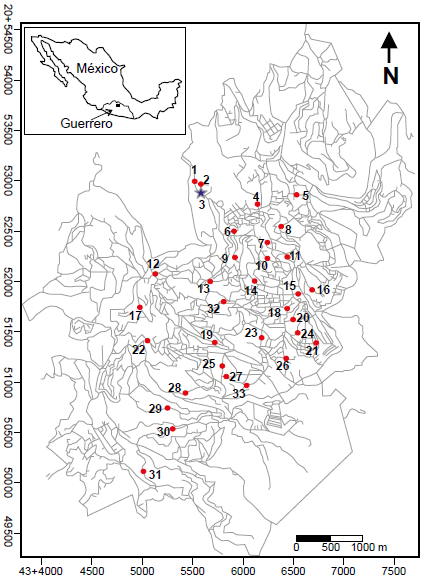
Figure 2 Map of the urban area of Taxco de Alarcón, Guerrero, showing the location of the municipal water treatment plant (blue star), collection sites of samples of raw water (red circles labeled 1 and 2) and household tap water (red circles labeled 3-33).
At each sampling site, the temperature, pH, Eh and electrical conductivity were determined. Temperature and pH were measured by potentiometry with an Oakton portable pH meter. The Eh was measured with a Cole-Parmer model waterproof ORPtestr Double Junction. Conductivity was measured with a Horiba Model ES-14 E conductivity meter with automatic temperature compensation and stabilization.
Three aliquots were taken at each point, two of them in Nalgene™ high density polypropylene bottles prewashed with diluted HNO3, deionized water and tempered with the sample water. For the bacteriological analyzes, a 250 mL aliquot was taken in sterile containers. The collected samples were placed in a cooler at <15 °C and transported to the laboratory for analysis at the end of the day. To determine the anions, the aliquot was 1 L, whereas for the analysis of the metals the aliquot was 500 mL acidified with 2 mL of ultrapure concentrated HNO3 (Ultrex II).
The alkalinity was determined by the titration method with 0.02 M HCl using phenolphthalein, bromocresol green, and methyl red as colorimetric indicators. The chlorides were determined by titration with 0.02 M AgNO3 and 0.04 M K2CrO4. The sulfate (SO42-), nitrate (NO3- as N), fluorine (F-) and turbidity contents were analyzed by colorimetry using a HACH model DR/890. BaCl2 and citric acid were used for the sulfates; for the nitrates a cadmium reduction reagent was used, whereas for the fluoride SPANDS was used. Only absorbence was measured for turbidity, comparing it with a target.
Bacteriological analysis was performed by the 0.45 μm cellulose acetate membrane filtration method using ampoules with Hach's m-ColiBlue24®- culture medium. The bacteria were incubated in a Cole-Parmer 52100-100 convection oven at a temperature of 37 oC for 24 h.
The cations concentration was determined in acidified and filtered samples (0.45 μm cellulose acetate) by the technique of inductively coupled plasma atomic emission spectroscopy (ICP-AES) in a Perkin Elmer, Optima 3200 DV. Sodium was analyzed on a 100 Perkin Elmer AAnalyst 800 Atomic Absorption Spectrometer. During the analysis, four certified High-Purity water standards (CWW-TM-D; CWW-TM-H; CWW-TM-A and CWW-TM-E) were used.
On the other hand, to assess the presence, concentration and bio-availability of toxic metals in the water, the extractable metal content was determined treating 1.0 g of suspended particulate matter obtained from sample 13 (Figure 2) with concentrated HNO3 (assuming that the nature of the particulate is the same in all the samples). Furthermore, to establish the potential release effect of toxic elements of suspended material in potable water with greater precision, a sequential extraction of metals from the particulate was carried out following the methodology proposed by Tessier et al. (1979). It has been shown that the presence of toxic metals (total or partial concentration) in particulate matter does not necessarily mean that they are bioavailable (Tessier et al., 1979) and, therefore, have harmful effects on biota or man. The bioavailability of an element depends largely on the chemical species in which it is found. For example, high concentrations of Cd in a sulfur phase with low solubility will be less bioaccessible than very low concentrations of Cd in a water-soluble phase. For this reason, the understanding of the fraction in which a particular metal is associated and its concentration provides more information on the bioavailability of the metal and, therefore, a better approximation to the true impact on health. According to Tessier et al. (1979), the metals can be separated into five chemical fractions: 1) exchangeable fraction (soluble + adsorbed); 2) fraction bound to carbonates; 3) fraction associated with oxyhydroxides of Fe-Mn; 4) fraction bound to sulfides and organic matter, and 5) residual fraction (insoluble). Total metals and sequential extraction solutions were analyzed in an ICP-AES Perkin-Elmer, optima 3200 DV (Talavera-Mendoza et al., 2016).
RESULTS
Physicochemical and bacteriological characteristics of water samples
Raw water
The real detection limits (DL) reported by Talavera-Mendoza et al. (2006) were used for water samples analyzed under the same analytical conditions, on the same equipment and using the same sample preparation procedure (Table 1, Table 2).
Table 1 Physicochemical characteristics of water samples from Taxco de Alarcón, Guerrero.
| Sample | pH | Conda | Turbidity | Tempb | Ehc | NO3--N | HCO3- | CO32- | Cl- | SO42- | SiO2 | F- | Na+ | K+ | Ca2+ | Mg2+ | TDSd | Hardness |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| mS∙cm-1 | NTUe | °C | mV | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | ||
| DLf | -- | -- | 1.0 | -- | -- | 0.100 | 5.0 | 5.0 | 1.00 | 1.00 | 5.0 | 0.010 | 0.050 | 0.050 | 0.050 | 0.050 | -- | -- |
| 1 | 6.6 | 0.103 | 8 | 19.6 | 551 | 0.2 | 70.8 | <DL | <DL | <DL | 75.9 | 0.11 | 10.1 | 3.0 | 12.6 | 0.2 | 133 | 32.4 |
| 2 | 8.4 | 0.636 | 5 | 18.5 | 426 | 2.4 | 187 | 10 | 9.6 | 70.0 | 38.9 | 0.44 | 63.5 | 7.4 | 54.9 | 11 | 434 | 182.5 |
| 3 | 5.7 | 0.136 | 32 | 22.1 | 885 | 0.8 | 4.9 | <DL | 5.2 | 45.6 | 24.9 | 0.03 | 5.6 | 2.6 | 10.8 | 1.9 | 92 | 34.6 |
| 4 | 7.3 | 0.299 | 21 | 19.7 | 714 | 0.8 | 70.8 | <DL | 30.5 | 49.5 | 37.8 | 0.17 | 19.6 | 4.1 | 24.7 | 6.1 | 227 | 86.6 |
| 5 | 7.9 | 0.431 | 4 | 18.6 | 510 | 1.0 | 85.4 | <DL | 54.8 | 68.5 | 30.3 | 0.25 | 33.8 | 6.5 | 35.5 | 6.7 | 310 | 116.0 |
| 6 | 6.8 | 0.076 | 92 | 20.7 | 453 | 0.8 | 36.6 | <DL | 1.8 | 24.1 | 30.1 | NDg | 2.9 | 2.6 | 9.6 | 0.8 | 96 | 27.4 |
| 7 | 7.8 | 0.407 | 93 | 23.1 | 473 | 0.9 | 75.6 | <DL | 46.1 | 61.7 | 26.3 | 0.19 | 26.0 | 4.6 | 31.4 | 5.9 | 268 | 103.0 |
| 8 | 7.8 | 0.312 | 4 | 20.6 | 532 | 1.0 | 63.4 | <DL | 35.7 | 58.5 | 32.8 | 0.21 | 21.7 | 4.3 | 27.1 | 6.0 | 237 | 92.4 |
| 9 | 7.4 | 0.319 | 10 | 20.8 | 730 | 0.9 | 56.1 | <DL | 33.1 | 70.5 | 35.3 | 0.30 | 19.7 | 4.1 | 25.9 | 6.5 | 237 | 91.4 |
| 10 | 6.3 | 0.161 | 7 | 20.8 | 514 | 0.8 | 14.6 | <DL | 12.2 | 45.7 | 24.8 | 0.08 | 8.4 | 2.6 | 12.6 | 2.3 | 114 | 40.8 |
| 11 | 5.2 | 0.132 | 18 | 22.9 | 964 | 0.6 | 9.8 | <DL | 9.6 | 47.1 | 24.2 | ND | 7.0 | 2.4 | 9.9 | 1.6 | 101 | 31.4 |
| 12 | 8.1 | 0.079 | 34 | 19.8 | 422 | 0.5 | 122.0 | <DL | 2.7 | 14.3 | 32.4 | 0.03 | 3.5 | 2.5 | 10.1 | 0.6 | 173 | 27.9 |
| 13 | 7.1 | 0.246 | 20 | 20.8 | 753 | 2.0 | 68.3 | <DL | 18.3 | 37.9 | 47.1 | 0.05 | 12.5 | 3.1 | 25.9 | 3.9 | 201 | 80.5 |
| 14 | 7.0 | 0.297 | 5 | 231 | 480 | 0.7 | 43.9 | <DL | 28.7 | 63.4 | 33.0 | 0.04 | 15.5 | 2.2 | 23.8 | 4.6 | 201 | 78.4 |
| 15 | 7.1 | 0.330 | 11 | 22.3 | 496 | 0.7 | 63.4 | <DL | 40.0 | 68.0 | 38.7 | 0.09 | 22.7 | 1.9 | 28.7 | 6.7 | 253.0 | 99.1 |
| 16 | 7.7 | 0.251 | 7 | 22.8 | 510 | 1.1 | 31.7 | <DL | 22.7 | 68.7 | 23.6 | ND | 13.5 | 3.5 | 22.9 | 4.5 | 183 | 75.7 |
| 17 | 7.6 | 0.076 | 20 | 19.6 | 425 | 0.6 | 39 | <DL | <DL | 10.3 | 30.7 | ND | 3.1 | 2.6 | 9.1 | 0.8 | 83 | 25.8 |
| 18 | 7.5 | 0.314 | 19 | 22.6 | 486 | 0.7 | 56.1 | <DL | 34.2 | 9.8 | 33.5 | 0.05 | 21.6 | 4.0 | 27.8 | 5.8 | 178 | 93.1 |
| 19 | 7.9 | 0.269 | 3 | 21.1 | 495 | 0.6 | 48.8 | <DL | 27.0 | 66.1 | 27.6 | 0.10 | 16.3 | 3.9 | 25.2 | 3.4 | 206 | 76.9 |
| 20 | 7.1 | 0.237 | 20 | 24.1 | 504 | 0.7 | 51.2 | <DL | 26.1 | 40.0 | 29.5 | 0.07 | 15.3 | 3.3 | 20.1 | 4.3 | 177 | 67.7 |
| 21 | 8.2 | 0.35 | 5 | 22.1 | 504 | 1.1 | 80.5 | <DL | 34.8 | 59.6 | 32.7 | 0.12 | 21.5 | 4.6 | 32.2 | 5.7 | 259 | 104.0 |
| 22 | 8.1 | 0.143 | 43 | 21.7 | 878 | 0.8 | 63.4 | <DL | 17.4 | 17.6 | 31.6 | ND | 11.2 | 3.2 | 11.2 | 0.8 | 143 | 31.1 |
| 23 | 6.2 | 0.141 | 32 | 22.1 | 902 | 06 | 12.2 | <DL | 14.0 | 34.5 | 24.1 | ND | 7.4 | 2.8 | 10.1 | 2.1 | 97 | 33.8 |
| 24 | 7.8 | 0.277 | 9 | 22.9 | 549 | 0.8 | 43.9 | <DL | 24.4 | 69.3 | 29.1 | 0.14 | 13.7 | 4.5 | 24.5 | 5.3 | 203 | 82.8 |
| 25 | 7.8 | 0.223 | 15 | 23.4 | 476 | 0.9 | 43.9 | <DL | 20.1 | 39.1 | 31.2 | 0.09 | 12.7 | 4.4 | 19.3 | 3.7 | 162 | 63.5 |
| 26 | 8.3 | 0.135 | 3 | 22.2 | 596 | 0.7 | 68.3 | <DL | 7.0 | 8.5 | 30.6 | ND | 8.5 | 3.3 | 12.8 | 1.7 | 127 | 39.0 |
| 27 | 8.1 | 0.442 | 5 | 22.9 | 453 | 1.0 | 95.2 | <DL | 63.5 | 56.7 | 29.8 | 0.14 | 36.8 | 6.9 | 30.6 | 6.4 | 314.0 | 103.0 |
| 28 | 7.6 | 0.258 | 18 | 21.7 | 484 | 0.8 | 53.7 | <DL | 23.5 | 40.2 | 32.5 | 0.02 | 15.5 | 3.8 | 18.6 | 4.9 | 179 | 66.6 |
| 29 | 7.4 | 0.235 | 9 | 23.6 | 447 | 0.9 | 39.0 | <DL | 19.0 | 40.5 | 29.7 | 0.13 | 12.1 | 3.6 | 18.2 | 4.1 | 154 | 62.1 |
| 30 | 8.2 | 0.082 | 10 | 20.2 | 373 | 0.6 | 43.9 | <DL | 1.8 | 1.1 | 13.0 | ND | 2.8 | 1.5 | 9.8 | 0.5 | 70 | 26.2 |
| 31 | 7.5 | 0.139 | 9 | 24.4 | 469 | 0.6 | 85.4 | <DL | <DL | 1.1 | 39.2 | ND | 7.1 | 2.2 | 15.5 | 0.6 | 134 | 41.3 |
| 32 | 7.5 | 0.242 | 23 | 20.9 | 544 | 1.0 | 36.6 | <DL | 22.7 | 41.5 | 25.6 | 0.04 | 13.6 | 3.3 | 17.3 | 3.8 | 155 | 59.0 |
| 33 | 8.9 | 0.277 | 6 | 24.4 | 414 | 0.5 | 53.7 | 10 | 26.1 | 35.4 | 25.2 | 0.08 | 15.1 | 4.2 | 23.0 | 4.0 | 185 | 74.0 |
Notes: a Conductivity; b Temperature; c Redox potential; d Total dissolved solids; e Nephelometric transfer units; f Detection limit; g Not detected; Data values have a standard deviation less than 5 %.
Table 2 Concentration of trace elements in water samples from Taxco de Alarcón, Guerrero.
| Sample | Fe | Sr | Zn | Mn | As | Ag | Al | Ba | Cd | Co | Cu | Ni | Pb | V | Se |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | mg∙L-1 | |
| DLa | 0.025 | 0.025 | 0.025 | 0.025 | 0.010 | 0.025 | 0.025 | 0.025 | 0.005 | 0.025 | 0.025 | 0.025 | 0.025 | 0.025 | 0.025 |
| 1 | <DL | 0.038 | 0.402 | <DL | <DL | <DL | 0.100 | 0.609 | <DL | <DL | <DL | <DL | <DL | <DL | <DL |
| 2 | 0.049 | 0.337 | 0.871 | <DL | 0.034 | <DL | 0.135 | 0.935 | <DL | <DL | <DL | <DL | <DL | <DL | <DL |
| 3 | 0.093 | 0.065 | 0.042 | 0.032 | <DL | <DL | 1.820 | 0.076 | <DL | <DL | <DL | <DL | <DL | <DL | <DL |
| 4 | 0.112 | 0.153 | 0.44 | <DL | <DL | <DL | 0.874 | 0.068 | <DL | <DL | <DL | <DL | <DL | <DL | <DL |
| 5 | 0.027 | 0.363 | 0.049 | <DL | <DL | <DL | 0.188 | 0.069 | <DL | <DL | <DL | <DL | <DL | <DL | <DL |
| 6 | 0.692 | 0.051 | 0.108 | 0.088 | <DL | <DL | 1.060 | 0.101 | <DL | <DL | <DL | <DL | <DL | <DL | <DL |
| 7 | 0.183 | 0.180 | 0.236 | <DL | <DL | <DL | 0.367 | 0.088 | <DL | <DL | 0.049 | <DL | <DL | <DL | <DL |
| 8 | 0.051 | 0.154 | 0.073 | <DL | <DL | <DL | 0.680 | 0.060 | <DL | <DL | 0.138 | <DL | <DL | <DL | <DL |
| 9 | 0.036 | 0.159 | 0.197 | <DL | <DL | <DL | 0.347 | 0.067 | <DL | <DL | 0.229 | <DL | <DL | <DL | <DL |
| 10 | 0.099 | 0.075 | 0.076 | <DL | <DL | <DL | 1.130 | 0.049 | <DL | <DL | <DL | <DL | <DL | <DL | <DL |
| 11 | 0.283 | 0.063 | 0.435 | 0.033 | <DL | <DL | 2.680 | 0.107 | <DL | <DL | 0.026 | <DL | <DL | <DL | <DL |
| 12 | 0.495 | 0.048 | 0.028 | <DL | <DL | <DL | 0.125 | 0.086 | <DL | <DL | 0.048 | <DL | <DL | <DL | <DL |
| 13 | 0.046 | 0.124 | 1.064 | <DL | <DL | <DL | 0.424 | 0.053 | <DL | <DL | 0.044 | <DL | <DL | <DL | <DL |
| 14 | 0.100 | 0.137 | 0.098 | <DL | <DL | <DL | 1.000 | 0.072 | <DL | <DL | 0.047 | <DL | <DL | <DL | <DL |
| 15 | 0.106 | 0.169 | 0.034 | <DL | <DL | <DL | 0.897 | 0.069 | <DL | <DL | <DL | <DL | <DL | <DL | <DL |
| 16 | 0.031 | 0.122 | 0.572 | <DL | <DL | <DL | 0.077 | 0.069 | <DL | <DL | 0.070 | <DL | <DL | <DL | <DL |
| 17 | 0.808 | 0.045 | 0.042 | 0.031 | <DL | <DL | 0.156 | 0.079 | <DL | <DL | 0.110 | <DL | <DL | <DL | <DL |
| 18 | 0.041 | 0.154 | 0.176 | <DL | <DL | <DL | 0.320 | 0.063 | <DL | <DL | 0.058 | <DL | <DL | <DL | <DL |
| 19 | <DL | 0.129 | 0.025 | <DL | <DL | <DL | 0.054 | 0.051 | <DL | <DL | <DL | <DL | <DL | <DL | <DL |
| 20 | 0.251 | 0.115 | 0.213 | <DL | <DL | <DL | 0.761 | 0.045 | <DL | <DL | 0.047 | <DL | <DL | <DL | <DL |
| 21 | 0.031 | 0.169 | <DL | <DL | <DL | <DL | 0.419 | 0.050 | <DL | <DL | <DL | <DL | <DL | <DL | <DL |
| 22 | 0.565 | 0.055 | 0.716 | <DL | <DL | <DL | 0.165 | 0.064 | <DL | <DL | 0.129 | <DL | <DL | <DL | <DL |
| 23 | 0.176 | 0.071 | 0.214 | 0.040 | <DL | <DL | 2.070 | 0.041 | <DL | <DL | 0.029 | <DL | <DL | <DL | <DL |
| 24 | <DL | 0.146 | 0.027 | <DL | <DL | <DL | 0.123 | 0.043 | <DL | <DL | 0.052 | <DL | <DL | <DL | <DL |
| 25 | 0.126 | 0.128 | <DL | <DL | <DL | <DL | 0.297 | 0.047 | <DL | <DL | 0.047 | <DL | <DL | <DL | <DL |
| 26 | 0.056 | 0.068 | <DL | <DL | <DL | <DL | 0.179 | 0.026 | <DL | <DL | 0.049 | <DL | <DL | <DL | <DL |
| 27 | 0.027 | 0.219 | 0.045 | <DL | 0.018 | <DL | 0.136 | 0.055 | <DL | <DL | 0.038 | <DL | <DL | <DL | <DL |
| 28 | 0.234 | 0.125 | 0.460 | <DL | 0.016 | <DL | 0.620 | 0.058 | <DL | <DL | 0.063 | <DL | <DL | <DL | <DL |
| 29 | 0.091 | 0.115 | 0.916 | <DL | <DL | <DL | 0.167 | 0.066 | <DL | <DL | 0.170 | <DL | <DL | <DL | <DL |
| 30 | 0.071 | 0.033 | 0.231 | <DL | 0.011 | <DL | 0.039 | 0.021 | <DL | <DL | 0.038 | <DL | <DL | <DL | <DL |
| 31 | 0.196 | 0.043 | 0.129 | 0.060 | <DL | <DL | <DL | 0.034 | <DL | <DL | 0.056 | <DL | <DL | <DL | <DL |
| 32 | 0.133 | 0.111 | 0.046 | <DL | <DL | <DL | 0.693 | 0.076 | <DL | <DL | 0.144 | <DL | <DL | <DL | <DL |
| 33 | 0.061 | 0.155 | 0.051 | <DL | <DL | <DL | 0.286 | 0.054 | <DL | <DL | 0.029 | <DL | <DL | <DL | <DL |
Notes: a Detection limit; Data values have a standard deviation less than 5 %.
The temperature of the water of Chacuhalco spring (sample 1; Figure 2) was 19.6 °C; the pH was almost neutral (6.6) and the Eh was 551 mV indicating high oxygenation. The conductivity of Chacuhalco water was 0.103 mS cm-1 and reflects low dissolved solids content. Water has a low turbidity (8 NTU) and is characterized by contents of SO42- and Cl- < DL and low values of HCO3-, NO3-N, F-, Na+, Ca2+, Mg2+ and K+ (Table 1). The hydrogeochemistry of the Chacuhalco spring shows that the water has values of turbidity just above that established in NOM-127-SSA1-1994. The concentration of the majority of the measured metals is below the DL except for Sr (0.038 mg·L-1), Zn (0.402 mg·L-1), Al (0.100 mg·L-1) and Ba (0.609 mg·L-1) (Table 2). Bacteriological analysis indicates that Chacuhalco spring water presents eight colony-forming units (CFU) of total coliforms (TC) (Table 3). This means that the water is not suitable for human consumption, and yet, this water is not treated in the WTP and is only chlorinated before sending it to the homes.
Table 3 Bacteriological analyzes of water samples from Taxco de Alarcón, Guerrero. Fecal coliforms correspond to E. coli.
| Sample | Total coliforms UFC/100 mL |
Fecal coliforms UFC/100 mL |
|---|---|---|
| 1 | 8 | ND |
| 2 | >100 | >20 |
| 3 | ND | ND |
| 4 | 12 | 3 |
| 5 | 7 | 1 |
| 6 | 16 | 2 |
| 7 | 1 | ND |
| 8 | 13 | 8 |
| 9 | 1 | ND |
| 10 | ND | ND |
| 11 | ND | ND |
| 12 | ND | ND |
| 13 | 7 | 3 |
| 14 | ND | ND |
| 15 | ND | ND |
| 16 | ND | ND |
| 17 | 4 | ND |
| 18 | ND | ND |
| 19 | ND | ND |
| 20 | ND | ND |
| 21 | ND | ND |
| 22 | ND | ND |
| 23 | ND | ND |
| 24 | ND | ND |
| 25 | ND | ND |
| 26 | ND | ND |
| 27 | 3 | 1 |
| 28 | ND | ND |
| 29 | ND | ND |
| 30 | ND | ND |
| 31 | 1 | ND |
| 32 | 1 | ND |
| 33 | ND | ND |
ND: No detected.
On the other hand, the mixed sample of the Chontalcuatlán river, the Teneria spring, and the San Marcos and El Sombrerito dams (sample 2) has a slightly basic pH (8.4) and an Eh of 426 mV, which indicates an oxidizing environment. The conductivity of this water was moderate (0.636 mS·cm-1) and therefore the total dissolved solids (TDS) are also moderate (434 mg·L-1). Turbidity is low (5 NTU). Sample 2 has low anion and cation contents (Table 1). With the exception of Ba (0.935 mg·L-1), all metals are within the NOM-127-SSA1-1994, and even Cu, Ag, Mn, Cd, Co, Ni, Pb, Se, and V are found <DL of the method (Table 2). In the bacteriological analysis, more than 20 CFU/100 mL of E. coli and more than 100 CFU/100 mL of TC were found (Table 3). The results indicate that this water does not meet the specifications of NOM-127-SSA1-1994.
Treated water
The water sample treated at the WTP (sample 3, Figure 2) showed a slightly acid pH (5.7). The Eh was 885 mV which indicates that the water is highly oxygenated. It also has low conductivity (0.136 mS cm-1) and consequently low TDS (92 mg·L-1), whereas the turbidity is high (32 NTU).
Sample 3 is characterized by low content of HCO3-, Cl-, SO42-, NO3-N, F-, Na+, K+, Ca2+, and Mg2+ (Table 1). Moreover, concentrations of As, Ag, Cd, Co, Cu, Ni, Pb, Se and V are < DL of the method, whereas the contents of Fe, Sr, Zn, Mn and Ba were low (Table 2). Furthermore, bacteriological analysis indicates that the treated water does not contain E. coli or TC (Table 3). Therefore in sample 3, the parameters that do not meet the NOM-127-SSA1-1994 standard are pH, turbidity and Al content. The acidity of the water is probably due to the chlorination process. On the other hand, the high values of turbidity and Al content are due to the inefficiency of the flocculation process during the treatment process.
Water from the distribution network
The temperatures of the household samples 4 to 33 (Figure 2) range between 18.6 and 24.4 °C. The pH in the household samples displays significant variations, ranging from 5.2 to 8.8. Half of the samples have slightly basic pH values (7.5-9.0), 40 % have circum-neutral pH values (7.5 - 6.5) and only 10 % have slightly acidic pH values (6.5-5.5). The Eh values are variable and range from 373 to 964 mV (Table 1). Conductivity varies between 0.076 and 0.442 mS·cm-1, which translates into TDS between 70 and 314 mg·L-1. The turbidity of the samples is also variable and ranges from 3 to 93 NTU.
The HCO3- contents were variable and range from 9.7 to 122.0 mg·L-1. The contents of SO42- (1.1 - 70.5 mg·L-1) and of Cl- (0.9 - 63.5 mg·L-1) were low. The contents of NO3-N range from 0.5 to 2.0 mg·L-1. The concentrations of F- were very low between <DL and 0.3 mg·L-1.
The content of Ca2+ varied from 9.1 to 35.5 mg·L-1, whereas the concentration of Mg2+ varied between 0.452 and 6.690 mg·L-1. These low concentrations of Ca2+ and Mg2+ translate into a low hardness (<116 mg·L-1 as CaCO3). According to the Durfor and Becker (1964) classification, the household tap water is classified as soft to moderately hard water. In general, all samples are characterized by low concentrations of Na+ (2.7-36.7 mg·L-1) and potassium K+ (1.4 - 6.9 mg·L-1).
The concentrations of Ag, Cd, Co, Ni, Pb, Se and V in all samples of household water were very low (<DL of the analytical method used). The concentrations of Zn range between 0.027 and 0.916 mg·L-1, only three samples were <DL. In most samples, the Mn content was low or <DL (Table 2), and only in five samples was above the detection limit, but with concentrations that are consistently low (0.031-0.088 mg·L-1). On the other hand, the contents of As were equally low since in most samples were <DL. Samples 27, 28 and 30 had concentrations exceeding this limit which varied between 0.011 and 0.018 mg·L-1. The contents of Sr varied between 0.033 and 0.363 mg·L-1. Concentrations of Fe range between <DL and 0.808 mg·L-1, whereas those of Al varies between <DL and 2.68 mg·L-1. The concentrations of Ba were low (0.021-0.107 mg·L-1). Finally, the Cu contents ranged from 0.026 to 0.230 mg·L-1, except in six samples which had concentrations <DL.
The household water samples contain a number of CFUs of variable TC and fall in the range of 0 to16 CFU/100 mL. The number of E. coli CFUs in the samples ranges from 0 to 8 CFU/100 mL (Table 3).
The chemical composition of water reaching households varies greatly and shows that during water distribution and storage, it undergoes important transformations. Comparing the household samples with sample 3 from the output of the WTP, the household water shows important variations in pH, turbidity, Fe and Al contents as well as in TC and fecal coliforms (FC). Sample 3 does not present FC or TC but up to 8 CFU of E. coli and up to 16 CFU of TC were detected in the household water.
Chemical composition of particulate matter
Taxco potable water shows a notorious turbidity. Turbidity in household samples is generated by suspended particles generally of the size of clays (<2 μm) and colloids (<0.3 μm) resulting from an incomplete and/or inefficient flocculation process in the WTP. The results of partial extraction with HNO3 are presented in Table 4.
Table 4 Concentration of metals extractable with HNO3 in particulate matter of household tap water sample 13.
| Element | K | Ca | Mg | Ba | Sr | Al | Cu | Fe | Mn | Pb | Zn | V | Cr | Co | Ni | As |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Concentration (mg·kg-1) | 927 | 3922 | 1106 | 103 | 32.19 | 53000 | 1069 | 9765 | 301 | 126 | 74.5 | 35.14 | 13.54 | 3.86 | 0.24 | 38.12 |
Sequential extraction
The distribution of the analyzed metals (Cd, Co, Cu, Fe, Mn, Pb, V, Zn and As) in the sequential extraction is quite variable indicating that the metals are associated with different chemical fractions (Figure 3, Table 5). Cd, As and Pb are the metals of major ecotoxicologi-cal interest due to the effects they have on biota, including humans (Fergusson, 1990).
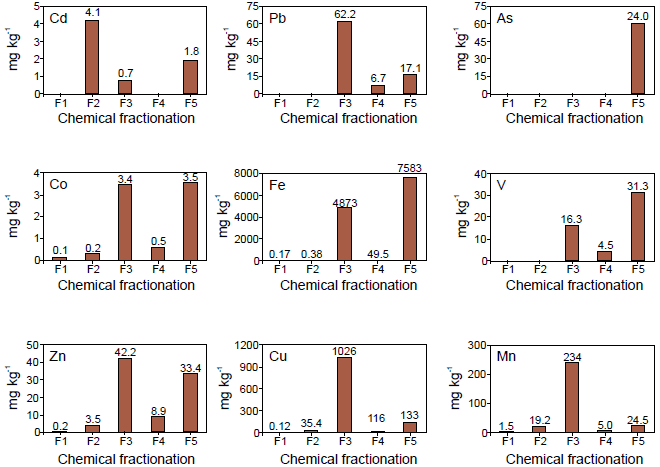
Figure 3 Chemical speciation of the potentially toxic elements detected in the particulate matter of one household tap water sample from Taxco de Alarcón. F1: Exchangeable fraction (soluble + adsorbed); F2: Fraction bound to carbonates; F3: Fraction associated with Fe-Mn oxyhydroxides; F4: Fraction bound to sulfides and organic matter; F5: Residual fraction (insoluble).
Table 5 Chemical fractionation in particulate matter of household tap water sample 13.
| Element | Concentration (mg·kg-1) | Total | TDf | Recovery | ||||
|---|---|---|---|---|---|---|---|---|
| F1a | F2b | F3c | F4d | F5e | (mg∙kg-1) | (mg∙kg-1) | (%) | |
| Cd | <0.5 | 4.17 | 0.744 | <0.5 | 1.88 | 6.79 | 5.62 | 121 |
| Co | 0.161 | 0.278 | 3.44 | 0.576 | 3.56 | 8.01 | 8.03 | 100 |
| Cu | 0.124 | 35.4 | 1026 | 116 | 133 | 1311 | 1087 | 121 |
| Fe | 0.176 | 0.388 | 4873 | 49.5 | 7583 | 12506 | 13811 | 91 |
| Mn | 1.51 | 19.3 | 243 | 5.09 | 24.6 | 293 | 296 | 99 |
| Pb | <0.5 | <0.05 | 62.2 | 6.75 | 17.2 | 86.1 | 141 | 61 |
| V | 0.012 | 0.012 | 16.4 | 4.59 | 31.3 | 52.3 | 48.8 | 107 |
| Zn | 0.285 | 3.58 | 42.3 | 8.96 | 33.5 | 88.6 | 97.3 | 91 |
| As | <0.5 | <0.5 | <0.5 | <0.5 | 24.0 | 24.0 | 18.7 | 128 |
a Exchangeable fraction (soluble + adsorbed); b Fraction bound to carbonates; c Fraction associated with oxyhydroxides of Fe-Mn; d Fraction bound to sulfides and organic matter; e Residual fraction (insoluble); fMetal content in total digestion.
The element Cd is mainly associated with fraction 2 or carbonated fraction (4.17 mg·kg-1), followed by fraction 5 or residual fraction (1.88 mg·kg-1). The fraction of Cd bound to Fe-Mn oxyhydroxides (fraction 3) is lower at 0.744 mg·kg-1, and the content in the exchangeable fraction (fraction 1) and those linked to sulfides and organic matter (fraction 4) is very low or <DL of the method. Although Cd was not detected in fraction 1, which is the most available, the presence of this metal adsorbed to fractions 2 and 3 indicates that there is a moderate potential for releasing them. A change to lower pH (<5.5), as occurs in sample 11, will release Cd from the particulate matter.
The highest concentration of Pb (62.2 mg·kg-1) is associated with the Fe-Mn oxyhydroxides fraction followed by the residual fraction (17.15 mg·kg-1) and, to a lesser extent, in the fraction associated with sulfides and organic matter (6.75 mg·kg-1). The exchangeable fraction and that associated with carbonates are below the DL of the method. Similar to Cd, the fraction associated with Fe-Mn oxyhydroxides will be released if the pH of the water decreases below 5.5. The Pb fraction associated with sulfides and organic matter will be more difficult to release since it requires values of pH <2.0, conditions not registered in the samples studied.
Fortunately, total arsenic is associated with the residual fraction, so the likelihood of its release is extremely low.
Co, Fe, V and Zn have a similar speciation and differ only by the absolute concentrations in each fraction. These metals are mostly bound to the Fe-Mn oxyhydroxides fraction followed by the residual fraction and the fraction associated with sulfides and organic matter. The most available fractions (1 and 2) have the lowest concentrations in these metals (0.161 and 0.278 mg·kg-1, respectively). In particular, the concentration of Co in fraction 3 and 5 (3.44 and 3.56 mg·kg-1, respectively) are similar, whereas the concentration in fraction 4 is much lower (0.576 mg·kg-1). The lowest Fe concentration was found in the exchangeable fraction (0.176 mg·kg-1) and in that associated with carbonates (0.388 mg·kg-1), whereas the highest concentration was found in the residual fraction (7583 mg·kg-1), followed by fraction 3 (4873 mg·kg-1), and finally fraction 4 (49.53 mg·kg-1). The lowest concentrations of V were found in fractions 1 and 2 (0.012 mg·kg-1) and the highest concentration was found in fraction 5 (31.34 mg·kg-1). In fraction 3 the concentration of V is 16.4 mg·kg-1. Finally, in fraction 4 the content of V is 4.59 mg·kg-1. The content of Zn is variable; the fraction with the highest content is 3 followed by fraction 5. The lowest content of Zn was found in the exchangeable fraction. The Zn concentrations in the rest of the fractions are less than 10 mg·kg-1. Cu and Mn have a similar distribution. The highest concentration of Cu and Mn is found in fraction 3, followed by fraction 5 and the lowest concentration is found in fraction 1 (Figure 3).
DISCUSSION
Household water quality with respect to NOM-127-SSA1-1994 standard
Table 6 shows a comparison between the physicochemical and bacteriological parameters analyzed in household water samples and the permissible limits established in the NOM-127-SSA1-1994 standard. Samples 19 and 26 are the only two samples that met the standard. Figure 4 shows the spatial distribution of samples that exceed permissible limits in turbidity, Al, pH, Fe, FC and TC (Figure 4a, 4b, 4c, 4d, 4e, 4f, respectively), established in the NOM-127-SSA1-1994 standard.
Table 6 Range values of physicochemical parameters of Taxco de Alarcón household tap water, compared with the permissible limits in NOM-127-SSA1-1994 standard.
| Parameter | Range of values | Permissible limit in the standard |
Number of samples that exceed the standard |
|---|---|---|---|
| Al (mg·L-1) | 0.039-2.68 | 0.200 | 19 |
| As (mg·L-1) | 0.010-0.018 | 0.05 | 0 |
| Ba (mg·L-1) | 0.021-0.107 | 0.700 | 0 |
| Cd (mg·L-1) | <0.005 | 0.005 | 0 |
| E. Coli (UFC/100 mL) | 0-8 | 0 | 6 |
| Total coliforms (UFC/100 mL) | 0-16 | 0 | 11 |
| Chlorides (mg·L-1) | 0.940-63.5 | 250 | 0 |
| Copper (mg·L-1) | 0.026-0.230 | 2.00 | 0 |
| Total hardness (CaCO3) (mg·L-1) | 25.83-104 | 500 | 0 |
| Fe (mg·L-1) | 0.027-0.808 | 0.300 | 4 |
| Fluoride as F-) (mg·L-1) | 0-0.300 | 1.50 | 0 |
| Mn (mg·L-1) | 0.023-0.088 | 0.15 | 0 |
| Nitrates as N (mg·L-1) | 0.500-2.0 | 10 | 0 |
| pH | 5.2-8.8 | 6.5-8.5 | 5 |
| Pb (mg·L-1) | <0.025 | 0.025 | 0 |
| Na (mg·L-1) | 2.78-36.75 | 200 | 0 |
| Total dissolved solids (mg·L-1) | 69.9-314.5 | 1000 | 0 |
| Sulfates (mg·L-1) | 1.10-70.5 | 400 | 0 |
| Zn (mg·L-1) | 0.027-0.916 | 5.0 | 0 |
| Turbidity (NTU) | 3-93 | 5.0 | 24 |
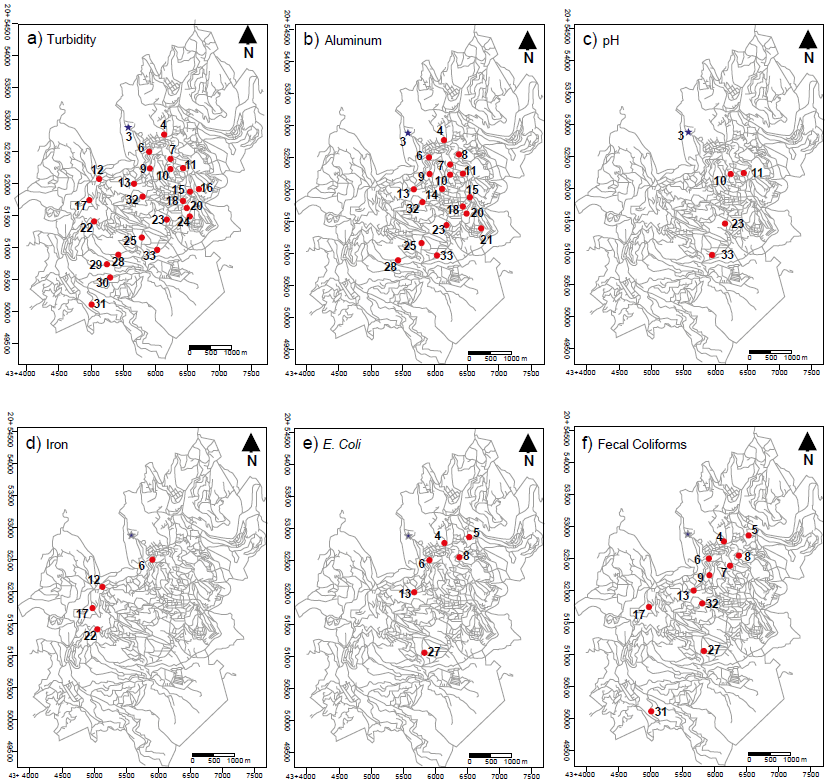
Figure 4 Location of sampling sites of household water samples that do not meet the permissible limits established in the NOM-127-SSA1-1994. Blue star is the location of the water treatment plant.
The pH is one of the physicochemical parameters with the greatest influence on water chemistry. It controls the conductivity, TDS content, alkalinity, hardness and metal content, among other parameters. In this study, the pH plays an important role in the interaction of water with the distribution pipes, an acid pH causes the dissolution of metallic materials of the pipe; in the case of pipes of different materials, e.g. plastic or cement, the effect is less. Also, a high pH causes the accumulation of precipitates.
Turbidity is important due to the fact that suspended particles are mainly composed of clays and organic matter, which have a high retention capacity of potentially toxic elements such as metals and As. Under reducing conditions, the toxic elements retained in the clays become soluble; and therefore, their concentration in water increases (Romero, 2001).
On the other hand, the high Al contents recorded in the potable water samples has its origin in the Al2(SO4)3 used in the WTP for the flocculation process. At least 19 samples exceed the maximum permissible limit in the standard (0.20 mg·L-1) reaching up to 13.4 times the permissible limit, as in sample 11 (Figure 5a). Any excess of this reagent will be solubilized and ionic aluminum will be released.
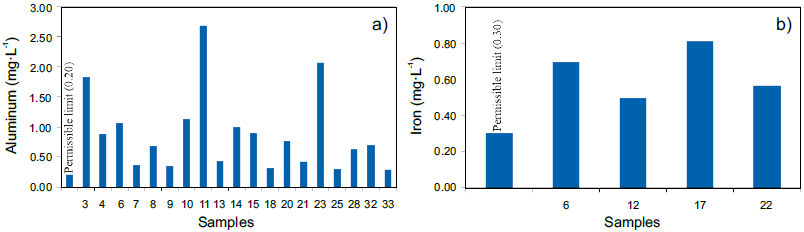
Figure 5 Samples that exceed the permissible limits established in the Official Mexican standard NOM-127-SSA1-1994. a) Aluminum, and b) iron.
The accumulation of aluminum in the brain correlates with the onset of Alzheimer's (Emsley, 1998). On the other hand, the presence of iron (Fe) can be explained by the fact that the water from the WTP has a slightly acidic pH, probably dissolving Fe from the distribution pipelines. At least four samples exceed the permissible limit of 0.30 mg·L-1 and reach up to 2.7 times the permissible limit (Figure 5b). Although Fe is an essential element for humans in small doses (10-30 mg·d-1), high doses can cause health problems (Emsley, 1998).
Finally, fecal coliforms (FC) are subdivided into subgroups and one of them is E. coli which is a bacteria found in human and animal feces, therefore, when the water contains E. coli, it indicates that the water could have been contaminated with human or animal feces, and this represents a health risk (Edberg et al., 2000). The treated water of the WTP did not show the presence of TC or E. coli. There are some factors that can lead to the pollution with coliforms. The water treated in the WTP is distributed to the households using the gravity force. Nevertheless, the difference of height between the WTP and the households results in high pressure in the water network, thus pressure reducing boxes are necessary. Those boxes are found outside throughout the city and are not hermetically sealed and represent a source of contamination for the treated water. On the other hand, most of the households store the water in tanks that not receive maintenance. Thus, the contamination is related to water transportation or storage in tanks at homes.
Potential affectation by the toxic elements contained in the suspended particles
The distribution of the analyzed elements (Cd, Co, Cu, Fe, Mn, Pb, V, Zn and As) in the different chemical fractions of the particulates vary greatly. Although the chemical behavior of these elements is variable and depends on the chemical fraction, their presence in the suspended particles in the household water represents a potential risk to the health of the users if the polluted water is drank.
Under acidic conditions and reduced Eh, the metals are soluble (Tessier et al., 1979; Fergusson, 1990). If the metals are in a basic medium and with an oxidizing Eh, they will oxidize and become insoluble. In the case of Taxco de Alarcón, 90 % of the water that reaches the houses presents a circum-neutral to slightly basic pH (6.5-9.0) and an oxidizing Eh. Therefore, it is very likely that the metals remain insoluble and are retained in the suspended particles.
The elements contained in the exchangeable fraction represent the most soluble elements and, therefore, have greater bioavailability. The amount of metals adsorbed or retained on the surface of a mineral depends on the surface area of each mineral. On the other hand, the type of adsorbed metals depends on the Point Zero Charge (PZC) of each mineral and the pH of the medium in which they are found (Manning and Goldberg, 1996). For example, the PZC of Fe oxides ranges from 5 to 8. When the pH of the water is lower than the PZC of Fe oxides, the surface of this mineral is positively charged, thus attracting all negatively charged compounds and retaining them on its surface. In contrast, when the pH of the water is higher than the PZC, the surface of this mineral is negatively charged releasing the negatively charged elements and retaining positively charged metals. The particulate matter has low but significant concentrations of Co, Cu, Fe, Mn, V and Zn in the exchangeable fraction, which will become soluble with small changes in the physicochemical conditions of the water.
The elements associated with the carbonate fraction are considered to be moderately bioavailable. A pH<6.5 is required to release the elements associated with this fraction. The matter has high concentrations of Cd, Cu and Mn, and smaller but significant in Co, Fe, V and Zn, in the fraction associated with carbonates. Samples 3, 10, 11 and 23 have pH values <6.5 and represent the highest potential for releasing these elements associated with the carbonate fraction.
In the Fe-Mn oxyhydroxide fraction, the associated elements are considered to be moderately bioavailable. In this case, the elements contained could be released into the environment when the pH of the medium reach values <5.5. In the analyzed particulate matter, high concentrations of Cu, Fe, Mn, Pb, V, and Zn, and lower concentrations of Co, were detected in the fraction of oxyhydroxides. Sample 11 is the only one that has a pH value <5.5. However, the elements contained in this fraction will become bioavailable when the water undergoes additional important external transformations (Fergusson, 1990).
The elements bound to the fraction of sulfides and organic matter are considered to have low bioavailability. Extreme pH conditions (<2.0) are necessary for release the elements. This situation is unlikely to occur in both the distribution system and the homes. As an example, an acid pH (~) is reached in gastric juices. In environments like these, the elements associated to sulfides and organic matter fraction represent a potential hazard. Fortunately, tap water is not usually used for drinking. The elements associated to the fraction of sulfides and organic matter in the particulate matter are Cu and Fe and in smaller proportion Co, Mn, Pb, V and Zn.
Finally, the elements associated with the residual fraction are considered non-bioavailable because they are associated with silicate minerals with a very low solubility product, and require extreme physicochemical conditions over a long period of time. The elements associated with this fraction do not represent a potential danger for biota. In the particulate matter, all the analyzed elements are present in this fraction and stand out for their concentration: Fe (7583 mg·L-1) and, due to its toxicological importance, Pb (17.15 mg·L-1) and As (24.0 mg·L-1). The latter was only detected in residual fraction so it does not represent a potential danger to the inhabitants.
CONCLUSIONS
The water supply to the city of Taxco de Alarcón comes from surface water from rivers, dams and springs of the zone. The main supply comes from the Chontalcuatlán river, the springs of Chacuhalco and Tenería within the city, and from the San Marcos and El Sombrerito dams. The obtained and analyzed results indicate that the water quality from these sources is not adequate for potable use because the Chacuhalco spring presents turbidity levels above the NOM-127-SSA1-1994. In addition, eight CFU/100 mL of TC were detected. Mixed water from Chontalcuatlán river water, the Tenería spring, and the San Marcos and El Sombrerito dams is not suitable for human consumption since they do not comply with Mexican regulations; they exceed the limits in barium and have more than 20 FC and more than 100 CFU/100 mL TC. For these reasons, the water from these sources is treated in the WTP before sending the water to the homes. Unfortunately, the chemical analyzes carried out in the plant's output water indicate that the treatment process is inefficient because the water sent to the homes is slightly acidic (pH 5.7), below the range established in the Mexican standard (6.5-8.5). Furthermore, suspended particles were observed, which results in a very high turbidity value (32 NTU) compared to that established in the standard (5 NTU). On the other hand, the output water contains high concentrations of Al (1.82 mg·L-1) that exceed the maximum permissible limit in the standard (0.200mg·L-1) in up to 9 times.
Particulate matter is characterized by high concentrations of total metals (Al, Cu, Fe, Pb, As, Cr, V, Zn, Sr, Ni). The speciation showed that Cu, Mn, Zn, Co, V and Fe have different concentrations in the exchangeable fraction and carbonate-associated fraction. Cd is associated with the carbonate fraction; the Pb is in the fraction associated with Fe-Mn oxyhydroxides, whereas the As is in the residual fraction, which is the most stable chemical fraction.
Finally, samples 19 and 26 from households were the only two samples that meet the Official Mexican Standard (NOM-127-SSA1-1994) in all measured parameters. However, overall the results provide information on the quality of potable water in the city of Taxco de Alarcón and indicate that modifications must be carried out in the water treatment processes, especially in the distribution network and water storage tanks, so that the water meets the standards of quality established by current regulations.











 nueva página del texto (beta)
nueva página del texto (beta)


