INTRODUCTION
The study of hermatypic corals has focused on tropical coral reef ecosystems, where the optimal environmental conditions are oligotrophic, non-turbid, and thermally stable (Hoegh-Guldberg 2011, Soares 2020). Nevertheless, coral communities inhabiting suboptimal environmental conditions (i.e., high turbidity and sedimentation rates, eutrophication, wide temperature fluctuations, and low pH) have been documented worldwide, suggesting that these communities have a greater capacity to tolerate suboptimal conditions (Camp et al. 2018, Burt et al. 2020, Soares 2020).
Marginal coral communities are ecologically distinct from tropical coral reefs; they show lower biodiversity, a lack of reef accretion, and coral coverage that is represented by species with wide tolerance thresholds (Perry and Larcombe 2003, Browne and Bauman 2023, Schoepf et al. 2023). Based on these and other characteristics, it has been hypothesized that these coral communities have the potential to face stressful local and regional conditions and, therefore, to cope with the effects of climate change (Perry and Larcombe 2003, Schoepf et al. 2023).
Climate change is predicted to be multifactorial, negatively affecting coral reef ecosystems in various ways (Veron et al. 2009), potentially increasing bleaching events (i.e., the loss of photosynthetic endosymbionts) and, eventually, degrading reef ecosystems. This could provoke a loss in the ecological and economic services that coral reefs provide (Veron et al. 2009, Oliver et al. 2018, Sully et al. 2019). Currently, some marginal coral communities inhabit environments resembling those predicted under climate change, allowing for an a priori evaluation of how tropical corals may respond to future environmental changes, as they may face suboptimal conditions (Perry and Larcombe 2003, Camp et al. 2018, Schoepf et al. 2023).
The Gulf of California (GC) represents the northeastern limit of the distribution of hermatypic corals in the Eastern Tropical Pacific (Glynn and Ault 2000). Bahía de los Ángeles (BLA) is located in the central region of the GC, near the Midriff Islands Region, and is considered a high-latitude environment for coral communities (29° N). Thirteen coral species have been reported in BLA, of which 2 are hermatypic (Porites panamensis and Porites sverdrupi), and 11 are ahermatypic (Reyes-Bonilla et al. 2007). The coral community of BLA is not capable of building a reef due to the extremely low temperatures in cold seasons, high turbidity, and high productivity (Reyes-Bonilla et al. 2007). A previous study in BLA reported that P. panamensis colonized rocky areas with a mean coverage of 2.5% (Halfar et al. 2005).
The wide latitudinal distribution of P. panamensis extends from the equator (-0.26° S) to the northern GC (31° N) (Glynn et al. 1994, Reyes-Bonilla et al. 2007), with this coral species being present even in suboptimal environments, such as CO2 seep vents, estuaries, and high latitudes (Norzagaray-López et al. 2015, Zapata and Lozano-Cortés 2015, Oporto-Guerrero et al. 2018). Porites panamensis shows less susceptibility to thermal anomalies than other coral species distributed in the Eastern Tropical Pacific. For example, the 1997-1998 El Niño event triggered massive coral bleaching, leading to 90% mortality in Pocillopora spp. but only 50% mortality in P. panamensis (Carriquiry et al. 2001, Reyes-Bonilla 2001). Furthermore, Reyes-Bonilla et al. (2002) reported widespread bleaching during the 1998-1999 La Niña event, with higher mortality of Pocillopora spp. than other coral taxa, including Porites spp.
Physiological markers, such as endosymbiont density, pigment concentration, and total lipid content, are commonly used to assess coral health. Quantifying the energetic resources and reservoirs of corals allows us to understand acclimation at local and temporal scales, as well as the stress responses of different coral taxa (Kemp et al. 2014, Nielsen et al. 2018, Martínez-Castillo et al. 2020, Chapron et al. 2022). Porites panamensis has developed different physiological strategies that allow it to successfully acclimatize to the local and regional environmental conditions of the Eastern Tropical Pacific (Martínez-Castillo et al. 2020, Santiago-Valentín et al. 2023). However, the physiological response of P. panamensis to the suboptimal conditions of BLA remains unknown, although lower calcification (Cabral-Tena et al. 2013, Norzagaray-López et al. 2015) and high recruitment rates have been reported in this area, which contrasts with what has been reported for their tropical conspecifics (Tejada-Begazo et al. 2022), demonstrating the ability of P. panamensis to cope with suboptimal conditions.
The main objective of this study was to describe the physiological changes involved in the acclimation of P. panamensis to suboptimal environmental conditions in BLA. We hypothesized that P. panamensis would present seasonal acclimation with high lipid content, high endosymbiont density, and low chlorophyll a concentrations during the cold season compared to the warm season. This study contributes to the ongoing efforts to understand the physiological plasticity of marginal coral communities and the physiological mechanisms responsible for the wide tolerance of P. panamensis.
MATERIALS AND METHODS
Study area
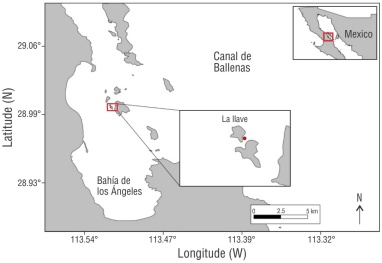
Our study area was located southeast of La Llave Island, within BLA, in the central-western region of the GC (Fig. 1). This area is characterized by large seasonal fluctuations in sea surface temperature (SST), ranging from ~14 °C to ~34 °C, and nutrient enrichment due to vertical mixing in Canal de Ballenas caused by tidal changes and the complex bathymetry (Álvarez-Borrego 2007, Torres-Delgado et al. 2013). Moreover, northwesterly winds in winter and southeasterly winds in summer generate upwelling conditions along the eastern coast of the bay (Martínez-Fuentes et al. 2022) that result in high productivity and eutrophic and turbid water conditions (Halfar et al. 2005, Ledesma-Vázquez et al. 2009, Torres-Delgado et al. 2013, Martínez-Fuentes et al. 2022). The oceanographic conditions of Canal de Ballenas strongly affect the physicochemical conditions of BLA, as the channels connecting the bay facilitate the transport of eutrophic and acidified waters (Amador-Buenrostro et al. 1991, Martínez-Fuentes et al. 2022). Bahía de los Ángeles has 2 well-defined seasons: the cold season (December to May), characterized by low SST and higher nitrate concentrations, and the warm season (June to November), characterized by high SST and lower nitrate concentrations (Martínez-Fuentes et al. 2022).
Field sampling
In total, 2 sampling campaigns were conducted during the cold season (March 2022 and March 2023), and 2 sampling campaigns were conducted in the warm season (October 2022 and August 2023). Healthy colonies were randomly selected in the study area, and coral nubbins (2-cm long [March 2022: n = 15; October 2022: n = 30; March 2023: n = 30; August 2023: n = 30]) were collected (5-10 m depth) during each campaign. Sampling was conducted in triplicate. All samples were individually fixed with a 10% solution of formaldehyde in filtered seawater, transported to the laboratory, and stored at room temperature (~20 °C) until further processing.
Environmental characterization
To characterize the environmental conditions of the waters surrounding our study area during the sampling campaigns, we used SST; the diffuse attenuation coefficient (Kd490), as an indicator of turbidity; the chlorophyll a (Chl a) concentration, as a proxy of the nutrient concentration and primary production; and particulate organic carbon (POC), as a proxy of productivity. The data points represent measurements averaged over 8 days (4 km2 resolution). In addition, we obtained historical SST data (2002-2024) to evaluate the effect of El Niño in BLA. All data were downloaded from the AquaMODIS Giovanni online data system (GIOVANNI 2024). This data was used to correlate the physiological responses of corals with environmental conditions.
Physiological markers
Endosymbiont density and chlorophyll a concentration
Samples were washed with filtered seawater (FSW) and decalcified with 10% acetic acid for ~16 h. The exact area of decalcified tissue was calculated with ImageJ software (Abrámoff et al. 2004). Tissue was homogenized in 1 mL of FSW through 2 sonication cycles (15 s each), with an intermediate dwell time of 15 s, and an amplitude of 70% (Q500, QSONICA, Newtown, USA). Samples were diluted (1:10) with FSW, stained with Lugol, and counted with a hemocytometer (8 counts per sample) using a Motic BA310E microscope (Motic, Hong Kong, China). Counts were normalized to 1 cm2 and are reported as cell·cm-2.
Endosymbiont pigments were extracted from 200 µL of tissue previously homogenized with FSW. Prior to pigment extraction, samples were centrifugated at 1,500 × g for 5 min to eliminate the supernatant. Salt residues were washed with 1 mL of MiliQ and centrifuged under the same conditions. Once the tissue had settled, the supernatant was decanted, and the tissue was immediately resuspended in 1 mL of 100% methanol with a sterile plastic pestle and left to incubate for 24 h at -20 °C in the dark. Chlorophyll a was quantified by spectrophotometry and estimated with the equation of Parsons et al. (1984) with a turbidity blank correction:
where E is the value of wavelength absorbance corrected with the turbidity blank. The total Chl a concentration was normalized per cell. The estimations are expressed as picograms (pg) of Chl a·cell-1.
Total lipid content
Lipids were extracted following the method of Folch et al. (1956) modified by Rodríguez-Troncoso et al. (2010). Briefly, 0.15 g of coral tissue was washed with distilled water and dried at 60 °C for 24 h. Lipids were extracted with a 2:1 chloroform:methanol solution. Precipitation was carried out with KCl (8%), followed by washing with a 1:1 methanol:water solution, vortex mixing, and decanting of the supernatant. The chloroform was evaporated in an oven at 60 °C for 12 h. Finally, the lipids were weighed and expressed as grams of lipids per gram of dry tissue (g of lipid·g-1 DW). All physiological markers are expressed as the mean ± standard deviation (SD).
Statistical analysis
Neither physiological markers nor environmental data exhibited normality or variance homogeneity. Therefore, non-parametric Kruskall-Wallis (α = 0.05), Dunn pairwise tests (α = 0.05), and Bonferroni P-value adjustments were used to assess statistical differences in physiological markers and environmental variables between seasons. Due to the high variability in Kd490, Chl a, and POC, as well as the lack of a clear seasonal pattern (Fig. S1), only the relationships between SST and physiological markers were analyzed using Spearman correlation tests. All analyses were performed using R Statistical Software v. 4.1.3 (R Core Team 2022). Dunn’s pairwise comparisons were conducted with the ‘FSA’ (v. 0.9.5) package. All plots were constructed in GraphPad Prism v. 5.00 (GraphPad Software, La Jolla, USA).
RESULTS
Environmental characterization
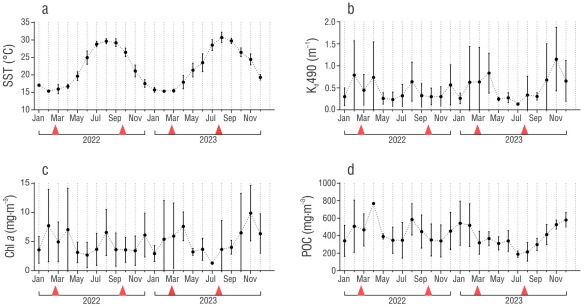
Sea surface temperature varied ~15 °C between the warm and cold seasons (Fig. 2a). The warmest temperatures were observed from July to September (29.42 ± 1.24 °C), and the coldest temperatures were observed from December to March (16.1 ± 1.04 °C). The results of the Kruskal-Wallis test indicated that significant differences in SST were present between seasons (H = 64.65, P = 0.003) (Fig. S1a). Dunn’s pairwise comparisons revealed significant differences between cold and warm seasons (P < 0.001) (Table S1). The Kd490 values varied substantially throughout the year, with peaks in February, April, and August of 2022 and in February, March, April, and November of 2023 (Fig. 2b). Nevertheless, there were no significant differences among seasons (H = 6.3436, P = 0.09) (Fig. S1b). Chlorophyll a exhibited high variation over time (Fig. 2c), with peaks in February and April of 2022 and a larger peak in November of 2023. Mean values of Chl a ranged from 3.7 to 6.6 mg·m-3, but no significant differences were found (H = 5.4722, P = 0.14) (Fig. S1c). Particulate organic carbon also showed high fluctuations over the year (Fig. 2d), with higher concentrations in February, April, and August of 2022 and in January, February, November, and December of 2023. However, the POC concentrations remained above 200 mg·m-3 during all months. The results of the Kruskall-Wallis test revealed significant differences between 2 seasons, and those of Dunn′s test revealed a signficant difference between the warm season of 2023 and cold season of 2023 (H = 11.29, P = 0.01) (Fig. S1d), as well as a significant difference between the warm season of 2022 and the cold season of 2023 (Z = -2.74, P = 0.03) (Table S1).
Endosymbiont density and chlorophyll a
Endosymbiont density was ~2-fold higher in the cold seasons (3.6 × 106 ± 1.04 × 106 cell·cm-2) compared to the warm seasons (1.7 × 106 ± 0.7 × 106 cell·cm-2) in both years (Fig. 3a). The Kruskal-Wallis test showed significant differences (H = 55.3, P < 0.001) in endosymbiont density between seasons, and the pairwise comparison revealed significant differences (P < 0.001) (Table S2) between the warm and cold seasons. The endosymbiont Chl a concentration (Fig. 3b) was found to range from 1.3 pg·cell-1 (warm season) to 2.6 pg·cell-1 (cold season). We observed significant differences in Chl a between seasons (H = 24.60, P < 0.001); a significant increase was identified in the warm season of 2023 compared to that of the warm season of 2022 (Z = -3.99, P < 0.001) (Table S2) and the cold season of 2023 (Z = 4.61, P < 0.001).
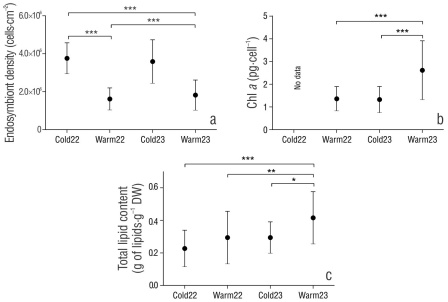
Figure 3 Physiological markers in Porites panamensis used in the present study. Endosymbiont density (a), endosymbiont chlorophyll a (Chl a) concentration (b), and total lipid content (c). Values are means ± SD. Asterisks denote the level of significance of pairwise Dunn tests: *** (P < 0.001), ** (P < 0.01), and * (P < 0.05).
Total lipid content
The mean total lipid content in P. panamensis ranged from 0.22 ± 0.11 g of lipid·g-1 DW to 0.41 ± 0.16 g of lipid·g-1 DW in the warm and cold seasons, respectively. The Kruskal-Wallis test showed significant differences among seasons (H = 17.66, P < 0.001), and the pairwise comparison indicated a significant increase (Table S2) in total lipid content in the warm season of 2023 (Fig. 3c) compared to that of the warm season of 2022 (Z = -2.92, P = 0.01), the cold season of 2022 (Z = 3.83, P < 0.001), and the cold season of 2023 (Z = 2.69, P = 0.02). We did not observe changes in total lipid content between cold seasons.
Spearman correlations
Sea surface temperature and endosymbiont density were significantly and negatively correlated (r = -0.60, P < 0.001). On the other hand, endosymbiont Chl a and total lipid content were significantly and positively correlated with SST (r = 0.37, P < 0.001; r = 0.51, P < 0.001, respectively).
DISCUSSION
Endosymbiont regulation by the host in response to temperature changes as an acclimation mechanism has been widely studied in branching corals, such as Pocillopora damicornis, Acropora humilis, and Acropora formosa (Stimson 1997, Fagoonee et al. 1999, Jandang et al. 2022), and the tropical P. panamensis conspecific (Martínez-Castillo et al. 2020, Santiago-Valentín et al. 2023). Sea surface temperature plays a crucial role in regulating coral physiology (Saxby et al. 2003, Al-Sofyani 2013). Periods of warm temperature increase respiration and photosynthetic rates (Coles and Jokiel 1977, Borell et al. 2008, Al-Sofyani 2013), while cold temperatures decrease metabolism, photosynthetic efficiency, and the translocation of photosynthates to the host (Saxby et al. 2003, Rodríguez-Troncoso et al. 2014). In both cases, if the temperature exceeds the thermo-tolerance limit of the coral, then oxidative stress is induced, resulting in the loss of endosymbionts and, consequently, in bleaching (Brown 1997, Saxby et al. 2003, Lesser 2011). In this sense, despite collecting samples under 3 different temperature regimes, we did not observe bleaching, confirming the wide thermotolerance threshold of P. panamensis.
We sampled in August of 2023, during an El Niño Southern Oscillation (ENSO) event, which caused the fourth global bleaching event (Reimer et al. 2024). In the southern Mexican Pacific, this ENSO event increased SST by ~2 °C above historical records for at least 4 months, causing widespread bleaching and mortality rates of 50-93% in coral cover, affecting the 3 most important reef-building corals: Porites sp., Pocillopora sp., and Pavona sp. (López-Pérez et al. 2024). In our study, we observed a positive thermal anomaly of ~1.48 °C in August 2023 (Fig. 4); however, these conditions did not trigger bleaching in P. panamensis. Previous studies have shown that upwelling due to tidal mixing processes could diminish the effect of El Niño events in the Midriff Islands Region (Santamaría-del-Angel et al. 1994, Álvarez-Borrego 2007), which could explain the observed SST patterns in BLA during 2023. Our results could be interpreted in 2 ways. First, the thermal anomaly during August could be an El Niño effect, which highlighted the thermotolerance of P. panamensis. Second, the effect of the thermal anomaly was masked by upwelling processes, suggesting that BLA is a potential thermal refuge for corals amid rising SST.
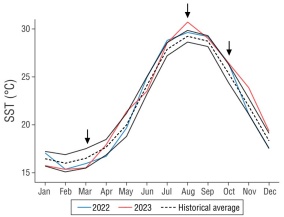
Figure 4 Historical average ± SD of satellite sea surface temperature (SST) data (2002-2024) for Bahía de los Ángeles, Gulf of California. Red and orange color lines refer to the sampling years of this study. Black arrows denote the sampling months.
Seasonal variation in endosymbiont density in natural coral populations has been widely studied, with higher endosymbiont density observed in winter than in summer (Stimson 1997, Fagoonee et al. 1999, Martínez-Castillo et al. 2020, Jandang et al. 2022). The reduction in endosymbiont density during the warmer season has been associated with a host antioxidant response, a strategy to cope with rising temperatures (Liñán-Cabello et al. 2010, Madeira et al. 2015), when dinoflagellate proliferation and photosynthesis accelerate (Coles and Jokiel 1977, Patthanasiri et al. 2022). This effect was observed with a 50% reduction in endosymbiont cells with warm temperatures (Fig. 5a), as a regulatory response of the host to endosymbiont activity. Cold temperatures reduce photosynthesis and photosynthate translocation due to a reduction in photosystem II development (Saxby et al. 2003), with a subsequent reduction in the Chl a concentration (Roth et al. 2012, Rodríguez-Troncoso et al. 2014). We did not observe reductions in the Chl a concentration during periods of either the coldest or highest temperatures, indicating that dinoflagellates, as hosts, also demonstrate high thermotolerance (Fig. 5b). Nevertheless, the high density of endosymbionts during the cold season suggests that the host allowed dinoflagellate proliferation to compensate for the low translocation of photosynthates needed to supply its energetic reservoirs. This is consistent with what has been reported for Pocillopora verrucosa when exposed to cold stress in the eastern Pacific (Rodríguez-Troncoso et al. 2014). In field studies, this pattern has also been observed in Acropora humilis and Pocillopora damicornis in Thailand (Jandang et al. 2022) and in the tropical P. panamensis conspecific (Martínez-Castillo et al. 2020), where Chl a in dinoflagellates decreased with cold temperatures despite an increase in the number of endosymbiont cells in the host.
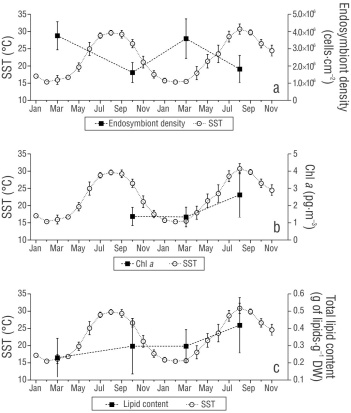
Figure 5 Effect of sea surface temperature (SST) on physiological markers Endosymbiont density (a), endosymbiont chlorophyll a (Chl a) concentration (b), and and total lipid content (c). Values are means ± SD.
Several studies have documented a positive correlation between endosymbiont density and nutrients, especially with inorganic nitrogen (Marubini et al. 1996, Nalley et al. 2023, Zhang et al. 2023). Martínez-Fuentes et al. (2022), following a 2-year monitoring campaign in BLA near La Llave Island, reported an increase in the nitrate concentration during the cold season (13 ± 5 µM) compared to that of the warm season (2 ± 1.5 µM). Considering the positive correlation between endosymbiont density and nutrients in several corals, another explanation of the observed temporal variation in endosymbiont density could be related to the natural fluctuations in nutrient concentrations in BLA. However, we did not measure the nutrient concentration in the water column during the sampling campaigns to evaluate this. Nonetheless, satellite chlorophyll data (Fig. 2c) showed high variability and eutrophic conditions in both years. A continuous supply of nutrients could enhance the proliferation of endosymbiont density within the host.
At the local scale, we observed 5 times more endosymbionts (cell·cm-2) compared to that reported for the tropical P. panamensis conspecific (Martínez-Castillo et al. 2020, Santiago-Valentín et al. 2023), but with lower Chl a concentration (Table 1). Moreover, the Kd490 values indicated that BLA waters were 5 times more turbid than the waters of the tropical P. panamensis conspecific (Rodríguez-Troncoso et al. 2023) due to a high concentration of particulate organic matter, which rapidly attenuated light (Devlin et al. 2008). Corals respond in different ways to dim-light conditions; for example, the host can induce the proliferation of endosymbiont cells, which could increase the amount of photosynthetic pigments (Hoegh-Guldberg and Smith 1989, Fagoonee et al. 1999, Titlyanov et al. 2001, Titlyanov et al. 2002). Dinoflagellate endosymbionts can absorb solar energy at different wavelengths of the light spectrum. While Chl a absorbs light at ~680 nm, other pigments, such as peridinin, absorb a broader spectrum of light (~450-550 nm) (Roth 2014). Therefore, the persistent turbidity in the study area and low penetration of light with short wavelengths could explain the slight variation observed in Chl a, highlighting the role of accessory pigments in dinoflagellate photoacclimation. Furthermore, our results revealed that endosymbiont regulation by the host plays an important role as a photoacclimation mechanism in P. panamensis under turbid conditions.
Table 1 Comparison of environmental conditions in 3 sites in the Mexican Pacific containing Porites panamensis and the physiological markers evaluated in this study. SST: sea surface temperature; Kd490: diffuse attenuation coefficient; chlorophyll a (Chl a); POC: particulate organic carbon; DW: dry tissue.
| Variable | Bahía de los Ángeles (29° N) | Islas Marietas (20° N) | Oaxaca (15° N) | |
| Environmental conditions | SST | 22.2 ± 5.6* | 28.7 ± 2.2 | 28.3 ± 2.1¶ |
| Kd490 | 0.4 ± 0.2* | 0.08 ± 8.8 × 10-3§ | 0.13 ± 1.0¶ | |
| Chl a | 4.5 ± 5.2* | - | - | |
| Nitrate | 4 ± 4.1† | 0.36 ± 0.6‡ | - | |
| POC | 403.3 ± 132.2* | - | - | |
| Physiological marker | Endosymbiont density (cell·cm-2) | 2.6 ± 1.3* | 0.4 ± 0.04‡ | 0.2 ± 0.1¶ |
| Chl a | 1.7 ± 1.0 (pg·cell-1)* | 33.4 ± 5.6 (ng·cell-1)‡ | 26.5 ± 12.5 (pg·cell-1)¶ | |
| Total lipid content (g of lipid·g-1 DW) | 0.32 ± 0.15* | 0.2 ± 0.03‡ | 0.22 ± 0.1¶ |
*Present study, †Martínez-Fuentes et al. (2022), ‡Martínez-Castillo et al. (2022), §Rodríguez-Troncoso et al. (2023), ¶Santiago-Valentín et al. (2022).
The primary energy source for corals in oligotrophic areas is sunlight; photosynthesis is conducted by endosymbionts, which translocate up to 90% of photosynthates to the host (Muscatine and Hand 1958, Trench 1971, Falkowski et al. 1984, Oku et al. 2003). Seasonal changes affect the energetic budget in natural populations of Goniastrea aspera, Acropora millepora, and the tropical P. panamensis conspecific (Table 1), showing high and low lipid content in colder and warmer months, respectively (Yamashiro et al. 2005, Conlan et al. 2020, Martínez-Castillo et al. 2020, Santiago-Valentín et al. 2023), due to the endosymbiont contributing more than 50% of total lipid content within the holobiont, as has been shown for Acropora hyacinthus (Zhang et al. 2023). This contribution of lipids from the endosymbiont to its coral host might be a general pattern in corals, given that a positive correlation between endosymbiotic cell density and lipid content has been reported in at least 8 coral species (Yamashiro et al. 2005). However, we did not observe lipid loss during the warm season despite the ~50% loss in endosymbiotic cells, suggesting that another energy source, such as heterotrophy, which is mainly employed by temperate corals (Ferrier-Pagés et al. 2011), contributed to lipid biosynthesis.
On the other hand, reproductive activity can also reduce lipid content in corals by 10-40% due to the maturation of lipid-enriched eggs (Oku et al. 2003, Viladrich et al. 2016, Viladrich et al. 2017, Conlan et al. 2020). Porites panamensis is a gonochoric breeder that is reproductively active throughout the year, with reproductive peaks during periods of warm temperatures (Glynn et al. 1994, Carpizo-Ituarte et al. 2011, Santiago-Valentín et al. 2023); however, local conditions can modify its reproductive patterns (Santiago‐Valentín et al. 2018). Although there is no data on gamete maturation or the presence of P. panamensis larvae in BLA, high recruitment has been recorded during months of high temperatures, mainly between August and November (Tejada-Begazo et al. 2022), suggesting increased reproductive activity. Despite collecting samples in the warm seasons of both years, when lipids are presumably invested in gamete production, we did not observe a reduction in total lipid content (Fig. 5c).
Previous stable isotope analyses have revealed that corals (Porites spp., Platygyra spp., and Pocillopora spp.) increase heterotrophy in turbid environments (Fox et al. 2018, Travaglione et al. 2023), being able to feed on plankton, ranging in size from picoplankton to mesoplankton (particle sizes of 0.2-1,000 µm), with their consumption contributing ~15-35% to the daily metabolic requirements of healthy colonies (Houlbrèque and Ferrier-Pagès 2009). In addition, Anthony (2006) observed that coral colonies inhabiting turbid waters tend to increase their total lipid content 4-fold compared to their counterparts in clear waters. In this sense, we observed high fluctuations in Chl a and POC over time (Fig. 2c, d), which constitute a potential food source for P. panamensis that could contribute to lipid biosynthesis. If P. panamensis increases heterotrophy, then this increase could compensate for possible lipidic losses due to the reduction in endosymbiont density or the investment of lipids into reproductive activity during the warm season.
Corals can be autotrophic, heterotrophic, or mixotrophic, and their trophic strategy is related to their thermal tolerance. Mixotrophy, the acquisition of nutrients from photosynthesis and heterotrophy, is well-documented in corals and has been suggested to be the most successful nutritional strategy (Houlbrèque and Ferrier-Pagès 2009, Viladrich et al. 2017, Sturaro et al. 2021). Conti-Jerpe et al. (2020) observed that the nutritional strategy is highly related to coral tolerance; autotrophic corals (e.g., Acropora samoensis, A. pruinuosa, and Goniopora lobata) were less tolerant to thermal stress, while mixotrophic and heterotrophic corals (e.g., Porites spp., Pavona spp., Turbinaria spp., and Platygyra spp.) were notably more thermotolerant (Conti-Jerpe et al. 2020). Although we did not measure the nutritional pathways of P. panamensis in BLA, our observations of its thermal tolerance allow us to hypothesize that this species is a mixotrophic organism, which could explain its success under suboptimal environmental conditions due to its ability to acclimatize.
CONCLUSIONS
This study demonstrates the high phenotypic plasticity of P. panamensis through local adaptation to low-light and eutrophic conditions with wide fluctuations in SST. Our results highlight the role of the host through a possible induction of endosymbiont cell proliferation, a photoacclimation mechanism that improves energy uptake. Our results also suggest that P. panamensis exhibits a seasonal response to low-light conditions in BLA, resulting in a 5-fold increase in overall endosymbiont density. On the other hand, the prevalent conditions of high turbidity and productivity could promote a mixotrophic strategy in P. panamensis, which may compensate for lipidic losses in warmer months due to decreased endosymbiont density and increased reproductive activity. Furthermore, upwelling areas in the region could provide refuge from warm SST anomalies, although further studies are needed to evaluate this possibility. Moreover, future studies must also characterize other dinoflagellate pigments that could be beneficial in turbid environments. Isotopic and molecular analysis, such as RNA-seq, should be explored to confirm the mixotrophy in P. panamensis and unravel the fundamental cellular mechanisms involved in the acclimation and resistance of this species. The present study furthers our understanding of P. panamensis physiology under suboptimal conditions, and our results suggest that mixotrophy is a fundamental strategy responsible for the broad tolerance of P. panamensis, highlighting the importance of studying the effects of nutritional strategies in other coral species in the face of local, regional, and global climate impacts.











 texto en
texto en