Revisión de literatura
Lippia graveolens and its activity against bacteria associated with bovine mastitis: Literature review
Vargas-Monter, Jorge12
 http://orcid.org/0000-0001-9845-2598
http://orcid.org/0000-0001-9845-2598
Rivero-Pérez, Nallely1*
 http://orcid.org/0000-0002-6154-9983
http://orcid.org/0000-0002-6154-9983
Valladares-Carranza, Benjamín3
 http://orcid.org/0000-0003-0306-3560
http://orcid.org/0000-0003-0306-3560
Ojeda-Ramírez, Deyanira1
 http://orcid.org/0000-0003-4649-2699
http://orcid.org/0000-0003-4649-2699
Noguez-Estrada, Juan12
 http://orcid.org/0000-0002-0493-8843
http://orcid.org/0000-0002-0493-8843
Zaragoza-Bastida, Adrian**1
 http://orcid.org/0000-0002-8537-5025
http://orcid.org/0000-0002-8537-5025
 http://orcid.org/0000-0001-9845-2598
http://orcid.org/0000-0001-9845-2598Rivero-Pérez, Nallely1*
 http://orcid.org/0000-0002-6154-9983
http://orcid.org/0000-0002-6154-9983Valladares-Carranza, Benjamín3
 http://orcid.org/0000-0003-0306-3560
http://orcid.org/0000-0003-0306-3560Ojeda-Ramírez, Deyanira1
 http://orcid.org/0000-0003-4649-2699
http://orcid.org/0000-0003-4649-2699Noguez-Estrada, Juan12
 http://orcid.org/0000-0002-0493-8843
http://orcid.org/0000-0002-0493-8843Zaragoza-Bastida, Adrian**1
 http://orcid.org/0000-0002-8537-5025
http://orcid.org/0000-0002-8537-5025-
Publication dates-
July 04, 2025
Jan-Dec , 2025
- Article in PDF
- Article in XML
- Automatic translation
- Send this article by e-mail
- Share this article +
ABSTRACT
Bovine mastitis is an infectious disease of the mammary gland caused by the invasion of pathogens, among them the bacterial etiology is one of the most important and the treatment of these infections has currently been complicated by the resistance generated by Gram positive bacteria and Gram negative to conventional antimicrobials. The objective of this research was to carry out a bibliographic review of Lippia graveolens and its activity against bacteria associated with bovine mastitis. Scientific reports on the phytochemical composition of wild oregano (L. graveolens) and the antibacterial activity against bacteria associated with bovine mastitis were consulted. The metabolites identified in L. graveolens with the highest reported antibacterial activity were naringenin, quercetin, luteolin as well as the terpenes thymol and carvacrol. L. graveolens contains secondary metabolites with reports of antibacterial activity, so it could be an alternative treatment against bacteria associated with bovine mastitis.
Keywords:
:
Lippia graveolens, secondary metabolites, antibacterial activity, mastitis
INTRODUCTION
Bovine mastitis is inflammation of the mammary gland caused by the invasion of pathogenic microorganisms that destroy milk-secreting tissues. More than 100 species associated with bovine mastitis have been reported in bacterial etiology (Sharun et al., 2021; Morales et al., 2023). The most common bacteria in cases of mastitis are: Staphylococcus aureus, Streptococcus agalactiae, Streptococcus uberis, Streptococcus dysgalactiae, Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa, and coagulase-negative Staphylococcus (Pascu et al., 2022; Morales et al., 2023).
-
Sharun et al., 2021Advances in therapeutic and managemental approaches of bovine mastitis: a comprehensive reviewVeterinary Quarterly, 2021
-
Morales et al., 2023Bovine mastitis, a worldwide impact disease: prevalence, antimicrobial resistance, and viable alternative approachesVeterinary and Animal Science, 2023
-
Pascu et al., 2022Etiology of mastitis and antimicrobial resistance in dairy cattle farms in the western part of RomaniaAntibiotics, 2022
-
Morales et al., 2023Bovine mastitis, a worldwide impact disease: prevalence, antimicrobial resistance, and viable alternative approachesVeterinary and Animal Science, 2023
Mastitis is treated with chemical antimicrobials; however, their continuous and excessive use has led to the development of antimicrobial resistance, creating a global problem in animal and human health due to the interaction of bacteria from these two populations and the transfer of intergenic resistance (Galarza et al., 2021; Wang et al., 2021; Li et al., 2023). Bacterial resistance leads to increased treatment costs and premature culling of animals, which has prompted the scientific community to search for new alternatives for the treatment of bovine mastitis (Kovačević et al., 2022; Morales et al., 2023).
-
Galarza et al., 2021Staphylococcus aureus resistentes à meticilina em animais de fazenda em américa do Sul: uma revisão sistemáticaVive Revista de Salud, 2021
-
Wang et al., 2021Phage vB_PaeS-PAJD-1 rescues murine mastitis infected with multidrug-resistant Pseudomonas aeruginosaFrontiers in Cellular and Infection Microbiology, 2021
-
Li et al., 2023Alternatives to antibiotics for treatment of mastitis in dairy cowsFrontiers in Veterinary Science, 2023
-
Kovačević et al., 2022Is there a relationship between antimicrobial use and antibiotic resistance of the most common mastitis pathogens in dairy cows?Antibiotics, 2022
-
Morales et al., 2023Bovine mastitis, a worldwide impact disease: prevalence, antimicrobial resistance, and viable alternative approachesVeterinary and Animal Science, 2023
Within the flora diversity, Mexican oregano (Lippia graveolens) is a plant of interest due to its phytochemical composition. This wild shrub has been used for culinary purposes and in traditional medicine to treat respiratory and digestive diseases, inflammation, headaches, and rheumatism (Bautista et al., 2021). There are reports that demonstrate the antibacterial activity of L. graveolens using different methods of secondary metabolite extraction and concentrations at which antibacterial activity has been determined against various bacterial genera, including those associated with bovine mastitis (Bautista et al., 2021;Cortés et al., 2021; Kovačević et al., 2022; Garcia et al., 2022). The objective of this research was to conduct a literature review of L. graveolens and its activity against bacteria associated with bovine mastitis.
-
Bautista et al., 2021Mexican Oregano (Lippia graveolens Kunth) as Source of Bioactive Compounds: A ReviewMolecules, 2021
-
Bautista et al., 2021Mexican Oregano (Lippia graveolens Kunth) as Source of Bioactive Compounds: A ReviewMolecules, 2021
-
Cortés et al., 2021Identification and quantification of phenolic compounds from Mexican oregano (Lippia graveolens HBK) hydroethanolic extracts and evaluation of its antioxidant capacityMolecules, 2021
-
Kovačević et al., 2022Is there a relationship between antimicrobial use and antibiotic resistance of the most common mastitis pathogens in dairy cows?Antibiotics, 2022
-
Garcia et al., 2022Loading and release of phenolic compounds present in Mexican oregano (Lippia graveolens) in different chitosan bio-polymeric cationic matrixesPolymers, 2022
METHODOLOGY
To conduct this review, an exhaustive search was carried out in the following databases: PubMed, ScienceDirect, and Google Scholar, for studies published up to 2024. The following headings and keywords were used: L. graveolens, plant extracts, bovine mastitis, and antibacterial activity. Full-text documents were reviewed, and duplicate documents were removed. The exclusion criteria were inadequate methods and lack of access to the full text.
PLANT BOTANY
L. graveolens is a perennial shrub that grows to a height of two meters. It has exfoliating bark branches with opposite petiolate leaves that are oval-lanceolate in shape, with a rough, scabrous upper surface, glandular striae, densely hairy underside, obtuse apex, and variously crenate margin (Ocampo et al., 2009). Flowering occurs during the rainy season (Bueno, 2014). Its inflorescence is indeterminate, subglobose spike-like, with white, zygomorphic corollas and small, 4 mm hermaphroditic flowers, in quantities of 2 to 20 flowers. It has small indehiscent capsule fruits with seeds without endosperm (Figure 1) (Ocampo et al., 2009).
-
Ocampo et al., 2009Biología reproductiva del orégano mexicano (Lippia graveolens Kunth) en tres condiciones de aprovechamientoAgrociencia, 2009
-
Bueno, 2014Composition of essential oil from Lippia graveolens. Relationship between spectral light quality and thymol and carvacrol contentJournal of essential oil research, 2014
-
Ocampo et al., 2009Biología reproductiva del orégano mexicano (Lippia graveolens Kunth) en tres condiciones de aprovechamientoAgrociencia, 2009
Thumbnail
Figure 1
Lippia graveolens in Orizabita, Ixmiquilpan, Hidalgo, Mexico
Lippia graveolens in Orizabita, Ixmiquilpan, Hidalgo, Mexico
The plant is wild and is found in the hills of temperate, arid, and semi-arid areas of Mexico. It adapts to altitudes between 1 400 and 2 300 meters, in stony soils with a sandy loam texture. It prefers a dry and semi-dry climate, with temperatures ranging from 20 to 24 °C and precipitation between 182 and 267 mm (Figure 1) (Martínez et al., 2014). It is an aromatic plant used in cooking and herbal medicine for the treatment of digestive disorders. In Chihuahua, Durango, Tamaulipas, Coahuila, Jalisco, Zacatecas, Querétaro, Hidalgo, and Baja California, its foliage is collected for sale in local markets (Bueno, 2014).
-
Martínez et al., 2014Genetic diversity and genetic structure in wild populations of Mexican oregano (Lippia graveolens HBK) and its relationship with the chemical composition of the essential oilPlant systematics and evolution, 2014
-
Bueno, 2014Composition of essential oil from Lippia graveolens. Relationship between spectral light quality and thymol and carvacrol contentJournal of essential oil research, 2014
COMPOSICIÓN FITOQUÍMICA PHYTOCHEMICAL COMPOSITION
The phytochemical characterization of L. graveolens from aqueous extracts, hydroalcoholic extracts, and essential oils shows differences in its phytochemical composition (González et al., 2017; Bernal et al., 2022). Extraction has been carried out using conventional techniques such as maceration with alcohols and water in different proportions, and by current technologies such as ultrasound-assisted extraction using eutectic solvents and supercritical fluid extraction using carbon dioxide as a solvent (Bernal et al., 2023). The presence of metabolites differs depending on the extraction methods, as shown in Table 1, with phenolic compounds and monoterpenes standing out (González et al., 2017).
-
González et al., 2017Pharmacological evaluation of the anxiolytic-like effects of Lippia graveolens and bioactive compoundsPharmaceutical biology, 2017
-
Bernal et al., 2022Spray-dried microencapsulation of oregano (Lippia graveolens) polyphenols with maltodextrin enhances their stability during in vitro digestionJournal of Chemistry, 2022
-
Bernal et al., 2023Composition of essential oil from Lippia graveolens. Relationship between spectral light quality and thymol and carvacrol contentJournal of essential oil research, 2014
-
González et al., 2017Pharmacological evaluation of the anxiolytic-like effects of Lippia graveolens and bioactive compoundsPharmaceutical biology, 2017
Table 1
Phytochemical compounds identified in plant extracts of Lippia graveolens
Phytochemical compounds identified in plant extracts of Lippia graveolens
| Extract | Extraction method | Solvent | Compounds | Reference |
|---|---|---|---|---|
| Aqueous | Ultrasound and supercritical fluid using CO2 | Choline chlorideethylene glycol, choline chlorideglycerol, and choline chloridelactic acid. | Caffeic, protocatechuic, and rosmarinic acids. Quercetin, luteolin, naringenin, eriodictyol, carvacrol | Garcia et al. (2022)) González et al. (2017) Soto et al. (2019) Bernal et al.(2023) |
| Hexane | Maceration | Hexane | Thymol, m-cymene-8-ol, methyl salicylate, carvacrol, and linalool. | González et al.(2017) |
| Ethyl acetate | Maceration | Ethyl acetate | p-cymene, thymol, cirsimaritin, naringenin | |
| Ethanolic | Maceration | Ethanol Supercritical CO2 modified with ethanol after steam distillation | Naringenin, taxifolin, eriodictyol, caffeic acid, luteolin, coumaric acid, quercetin 3 O-glucoside, 2-hydroxybenzoic acid, apigenin, quercetin, phloridzin, acacetin, sakuranetin, cirsimaritin, chrysoeriol. | Arias et al. (2023) Bernal et al. (2023) González et al. (2017) |
| Hydroalcoholic | Maceration | Methanol and Chloroform | Loganin, secologanin, secoxiloganin, dimethylsecologanoside, loganic acid, 8-epi-loganic acid, carioptosidic acid and its 6-O-p-coumaroyl and 6-O-caffeoyl derivatives, naringenin, pinocembrin, lapachenol and icterogenin, luteolin-7-O-glucoside, apigenin 7-O-glucoside, phloridzin, taxifolin, eriodictyol, scutellarein, luteolin, quercetin, and galangin. | Cortes et al. (2021) Picos et al. (2021) Leyva et al. (2016) Rastrelli et al. (1998) Lin et al. (2007) |
| Essential oil | Hydro- distillation | Carvacrol, α-terpinyl acetate, m-cymene, thymol, β-pinene, and α-thujene, linalool, humulene Sesquiterpene: isocaryophyllene, γ-terpinene. | Hernández et al.(2009) Martínez et al. (2014) Nonato et al. (2022) Castillo et al. (2023) |
-
Garcia et al. (2022)Loading and release of phenolic compounds present in Mexican oregano (Lippia graveolens) in different chitosan bio-polymeric cationic matrixesPolymers, 2022
-
González et al. (2017)Pharmacological evaluation of the anxiolytic-like effects of Lippia graveolens and bioactive compoundsPharmaceutical biology, 2017
-
Soto et al. (2019)Extraction yield and kinetic study of Lippia graveolens with supercritical CO2The Journal of Supercritical Fluids, 2019
-
Bernal et al.(2023)Green extracts and UPLC-TQS-MS/MS profiling of flavonoids from Mexican Oregano (Lippia graveolens) using natural deep eutectic solvents/ultrasoundassisted and supercritical fluidsPlants, 2023
-
González et al.(2017)Pharmacological evaluation of the anxiolytic-like effects of Lippia graveolens and bioactive compoundsPharmaceutical biology, 2017
-
Arias et al. (2023)Simultaneous extraction with two phases (modified supercritical CO2 and CO2-expanded liquid) to enhance sustainable extraction/isolation of pinocembrin from Lippia origanoides (Verbenaceae)Advances in Sample Preparation, 2023
-
Bernal et al. (2023)Green extracts and UPLC-TQS-MS/MS profiling of flavonoids from Mexican Oregano (Lippia graveolens) using natural deep eutectic solvents/ultrasoundassisted and supercritical fluidsPlants, 2023
-
González et al. (2017)Pharmacological evaluation of the anxiolytic-like effects of Lippia graveolens and bioactive compoundsPharmaceutical biology, 2017
-
Cortes et al. (2021)Identification and quantification of phenolic compounds from Mexican oregano (Lippia graveolens HBK) hydroethanolic extracts and evaluation of its antioxidant capacityMolecules, 2021
-
Picos et al. (2021)Supercritical CO2 extraction of oregano (Lippia graveolens) phenolic compounds with antioxidant, α-amylase and α-glucosidase inhibitory capacityJournal of Food Measurement and Characterization, 2021
-
Leyva et al. (2016)Protective role of terpenes and polyphenols from three species of Oregano (Lippia graveolens, Lippia palmeri and Hedeoma patens) on the suppression of lipopolysaccharide-induced inflammation in RAW 264.7 macrophage cellsJournal of Ethnopharmacology, 2016
-
Rastrelli et al. (1998)Iridoids from Lippia graveolensPhytochemistry, 1998
-
Lin et al. (2007)Identification and quantification of flavonoids of Mexican oregano (Lippia graveolens) by LC-DAD-ESI/MS analysisJournal of food composition and analysis, 2007
-
Martínez et al. (2014)Genetic diversity and genetic structure in wild populations of Mexican oregano (Lippia graveolens HBK) and its relationship with the chemical composition of the essential oilPlant systematics and evolution, 2014
-
Nonato et al. (2022)Comparative analysis of chemical profiles and antioxidant activities of essential oils obtained from species of Lippia L. by chemometricsFood Chemistry, 2022
-
Castillo et al. (2023)Impact of fractional distillation on physicochemical and biological properties of oregano essential oil of Lippia graveolens HBK grown wild in MexicoJournal of Essential Oil Bearing Plants, 2023
In L. graveolens, the most abundant flavonoids are: quercetin, luteolin, naringenin, eriodictyol, luteolin, hesperidin, and phloridzin (Bernal et al., 2023). The metabolites naringenin, quercetin, phloridzin, and cirsimaritin are chemical markers of the Lippia genus (Bernal et al., 2022). The best flavonoid profile is obtained from methanolic leaf extract, in which three major iridoids have been found: carioptosidic acid with two derivatives: 6'-O-p-coumaroyl and 6'-O-caffeoyl, and seven minor iridoids: loganin, secologanin, secoxiloganin, dimethyl, secologanoside, loganic acid, 8-epi-loganic acid, and carioptoside (Rastrelli et al., 1998; Lin et al., 2007). Monoterpenes are the main components of essential oils of the Lippia genus (Cortés et al., 2021; Bernal et al., 2023). In water-, hexane-, and methyl acetate-based extracts and in hydrodistillation processes for obtaining essential oil, the presence of the following monoterpenes has been reported: thymol, carvacrol, limonene, b-caryophyllene, r-cymene, camphor, linalool, and a-pinene, which may vary according to the chemotype and extraction method (Calvo et al., 2014; Garcia et al., 2022). In this regard, Vernin (2001) reported that the essential oil of L. graveolens from Mexico and Central America has concentrations of 35 to 71% carvacrol and 5 to 7% thymol. Calvo et al. (2014) reported the presence of more than 70 compounds in its essential oils and identified three chemotypes of the plant: two phenolic (carvacrol and thymol) and one non-phenolic chemotype of oxygenated sesquiterpenes (βcaryophyllene, α-humulene, and caryophyllene oxide). Plant's habitat determines the oil composition, with the highest concentration of carvacrol obtained from plants growing in a semi-arid climate with thin, rocky soils (Torres et al., 2022). However, there are reports of young plants growing in less arid conditions with deep soils, which provide a higher presence of thymol, and those growing in sub-humid climates have a higher amount of oxygenated sesquiterpenes (Llamas et al., 2022).
-
Bernal et al., 2023Green extracts and UPLC-TQS-MS/MS profiling of flavonoids from Mexican Oregano (Lippia graveolens) using natural deep eutectic solvents/ultrasoundassisted and supercritical fluidsPlants, 2023
-
Bernal et al., 2022Spray-dried microencapsulation of oregano (Lippia graveolens) polyphenols with maltodextrin enhances their stability during in vitro digestionJournal of Chemistry, 2022
-
Rastrelli et al., 1998Iridoids from Lippia graveolensPhytochemistry, 1998
-
Lin et al., 2007Identification and quantification of flavonoids of Mexican oregano (Lippia graveolens) by LC-DAD-ESI/MS analysisJournal of food composition and analysis, 2007
-
Cortés et al., 2021Identification and quantification of phenolic compounds from Mexican oregano (Lippia graveolens HBK) hydroethanolic extracts and evaluation of its antioxidant capacityMolecules, 2021
-
Bernal et al., 2023Green extracts and UPLC-TQS-MS/MS profiling of flavonoids from Mexican Oregano (Lippia graveolens) using natural deep eutectic solvents/ultrasoundassisted and supercritical fluidsPlants, 2023
-
Calvo et al., 2014Phytochemical Diversity of the Essential Oils of Mexican Oregano (Lippia graveolens Kunth) Populations along an Edapho‐Climatic GradientChemistry & biodiversity, 2014
-
Garcia et al., 2022Loading and release of phenolic compounds present in Mexican oregano (Lippia graveolens) in different chitosan bio-polymeric cationic matrixesPolymers, 2022
-
Vernin (2001)Analysis of the essential oil of Lippia graveolens HBK from El SalvadorFlavour and fragrance journal, 2001
-
Calvo et al. (2014) Phytochemical Diversity of the Essential Oils of Mexican Oregano (Lippia graveolens Kunth) Populations along an Edapho‐Climatic GradientChemistry & biodiversity, 2014
-
Torres et al., 2022Chemical comparison of the essential oils of Lippia Origanoides in two agroclimatic zones of the Colombian Caribbean coastDyna, 2022
-
Llamas et al., 2022Impact of the in situ-ex situ management of Mexican oregano Lippia origanoides Kunth in northwestern YucatanBotanical Sciences, 2022
METABOLITES AND ANTIBACTERIAL ACTIVITY
Terpenes
Carvacrol and thymol are most prevalent in L. graveolens (Calamaco et al., 2023), and their concentration is affected by edaphic and climatic factors in the plant's habitat (Cortés et al., 2021). Carvacrol (2-methyl-5-1-methylethylphenol) provides the characteristic aroma of oregano (Ultee et al., 2000). It is synthesized from cymene via the mevalonate pathway and is a monoterpene that is insoluble in water and soluble in ethanol, carbon tetrachloride, and diethyl ether (Lee et al., 2017). Its stereochemistry (Figure 2) of a single phenolic ring with three substituents of functional groups (Memar et al., 2017) gives it antibacterial, antioxidant, anticancer, and anti-inflammatory properties (Tapia et al., 2017).
-
Calamaco et al., 2023Revisión sobre el orégano mexicano Lippia graveolens HBK.(Sinonimia Lippia berlandieri Schauer) y su aceite esencialInvestigación y Desarrollo en Ciencia y Tecnología de Alimentos, 2023
-
Cortés et al., 2021Identification and quantification of phenolic compounds from Mexican oregano (Lippia graveolens HBK) hydroethanolic extracts and evaluation of its antioxidant capacityMolecules, 2021
-
Ultee et al., 2000Adaptation of the food-borne pathogen Bacillus cereus to carvacrolArchives of microbiology, 2000
-
Lee et al., 2017Carvacrol‐rich oregano oil and thymol‐rich thyme red oil inhibit biofilm formation and the virulence of uropathogenic Escherichia coliJournal of applied microbiology, 2017
-
Memar et al., 2017Carvacrol and thymol: strong antimicrobial agents against resistant isolatesReviews and Research in Medical Microbiology, 2017
-
Tapia et al., 2017Carvacrol as potential quorum sensing inhibitor of Pseudomonas aeruginosa and biofilm production on stainless steel surfacesFood Control, 2017
Thumbnail
Figure 2
Terpenes reported in Lippia graveolens
Terpenes reported in Lippia graveolens
In bacteria, carvacrol induces cell lysis by altering lipophilic compounds and hydrophobic parts of the cytoplasmic membrane, increasing cation permeability (H+ and K+), generating lipopolysaccharide efflux and reactive oxygen species production, inhibiting ATPase activity, microbial DNA replication, and energy synthesis, causing cell death (Gallegos et al, 2022). However, Ultee et al. (2000) reported that bacteria can adapt to carvacrol and modify the fatty acid composition of the membrane and reduce its permeability.
-
Gallegos et al, 2022Inhibition of bacterial mobility by terpenoid compounds and plant essential oilsTropical and Subtropical Agroecosystems, 2022
-
Ultee et al. (2000)Adaptation of the food-borne pathogen Bacillus cereus to carvacrolArchives of microbiology, 2000
Thymol is an isomer of carvacrol (Figure 2). It is an aromatic substance with a white crystalline color, low solubility in water, and high solubility in organic solvents. It has a neutral pH but can have alkaline characteristics in aqueous solutions due to the deprotonation of phenol (Chizzola, 2013). It has bactericidal, fungicidal, insecticidal, nematicidal, and varroacidal activity (Gallegos et al., 2022). Its antibacterial effect in vitro against Escherichia coli, Salmonella spp. and S. aureus has been reported at a concentration of 0.75 mg/mL (Shapira-Mimran 2007; Gallegos et al., 2019). At concentrations of 1 and 2 % in oregano essential oil, it has greater antimicrobial activity against Gram-positive bacteria and less against Gram-negative bacteria (Du et al., 2015; Erazo et al., 2017). In vitro studies on Gram-negative enterobacteria found greater antibacterial activity of thymol from Lippia berlandieri compared to other antimicrobials (Garcia et al., 2022; Garcia et al., 2022). The antibacterial mechanism of action is similar to that of carvacrol, binding to bacterial membranes in a hydrophobic manner via hydrogen bonds, affecting the outer and inner membranes, increasing permeability and the loss of potassium ions and intracellular ATP, causing cell death (Di Pasqua et al., 2010).
-
Chizzola, 2013Regular monoterpenes and sesquiterpenes (essential oils)Natural products, 2013
-
Gallegos et al., 2022Inhibition of bacterial mobility by terpenoid compounds and plant essential oilsTropical and Subtropical Agroecosystems, 2022
-
Shapira-Mimran 2007Isolation and characterization of Escherichia coli mutants exhibiting altered response to thymolMicrobial Drug Resistance, 2007
-
Gallegos et al., 2019Actividad antibacteriana de cinco compuestos terpenoides: carvacrol, limoneno, linalool, αterpineno y timolTropical and Subtropical Agroecosystems, 2019
-
Du et al., 2015In vitro antibacterial activity of thymol and carvacrol and their effects on broiler chickens challenged with Clostridium perfringensJournal of animal science and biotechnology, 2015
-
Erazo et al., 2017Efecto antimicrobiano del cinamaldehído, timol, eugenol y quitosano sobre cepas de Streptococcus mutansRevista Cubana de Estomatología, 2017
-
Garcia et al., 2022Loading and release of phenolic compounds present in Mexican oregano (Lippia graveolens) in different chitosan bio-polymeric cationic matrixesPolymers, 2022
-
Garcia et al., 2022Loading and release of phenolic compounds present in Mexican oregano (Lippia graveolens) in different chitosan bio-polymeric cationic matrixesPolymers, 2022
-
Di Pasqua et al., 2010Changes in the proteome of Salmonella enterica serovar Thompson as stress adaptation to sublethal concentrations of thymolProteomics, 2010
Flavonoids
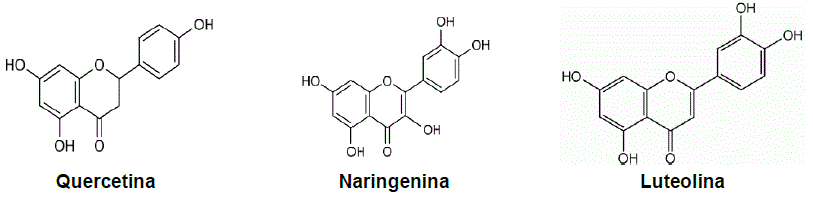
In L. graveolens, the flavonoids (Figure 3) with the highest reported biological activity are naringenin, quercetin, and luteolin (Lin et al., 2007). Naringenin is a bioactive compound with hepatoprotective, antiatherogenic, anti-inflammatory, antimutagenic, anticarcinogenic, and antimicrobial activity (Ke et al., 2017). In reaction with alkyl iodides, it forms O-alkyl compounds of naringenin with antibacterial potential against Escherichia coli, Staphylococcus aureus, and Bacillus subtilis (Kozłowska et al., 2019). A derivative of O-alkyl naringenin is sakuranetin (7-O-methylnaringenin), which has significant antimicrobial activity against Gram-negative and Gram-positive bacteria. Naringenin inhibits the growth of Staphylococcus aureus by affecting the cell membrane and fatty acid composition. In Escherichia coli, it acts on the genes associated with the biosynthesis of membrane fatty acids (Wang et al., 2018).
-
Lin et al., 2007Identification and quantification of flavonoids of Mexican oregano (Lippia graveolens) by LC-DAD-ESI/MS analysisJournal of food composition and analysis, 2007
-
Ke et al., 2017Citrus flavonoid naringenin reduces mammary tumor cell viability, adipose mass, and adipose inflammation in obese ovariectomized miceMolecular nutrition & food research, 2017
-
Kozłowska et al., 2019Novel O-alkyl derivatives of naringenin and their oximes with antimicrobial and anticancer activityMolecules, 2019
-
Wang et al., 2018Modification of membrane properties and fatty acids biosynthesis-related genes in Escherichia coli and Staphylococcus aureus: Implications for the antibacterial mechanism of naringeninBiochimica et Biophysica. Acta (BBA)-Biomembranes, 2018
Thumbnail
Figure 3
Flavonoids reported in Lippia graveolens
Flavonoids reported in Lippia graveolens
Quercetin is a flavonol based on the flavone structure nC6 (ring A)-C3 (ring C)-C6 (ring B). It has broad-spectrum antibacterial activity, breaks down the cell wall of bacteria, inhibits nucleic acid synthesis, and reduces enzyme activity (Wang et al.,2018). Specifically, in Escherichia coli, it alters the activity of adenosine triphosphate (Plaper et al., 2003). According to Hooda et al. (2020), impregnating quercetin with silver nanoparticles inhibits the growth of bacteria: Klebsiella pneumoniae (ATCC790603), Enterococcus faecalis (ATCC51299), Proteus vulgaris (ATCC426), Escherichia coli (ATCC25922), Staphylococcus aureus (ATCC4330), and Pseudomonas aeruginosa (ATCC27853).
-
Wang et al.,2018Modification of membrane properties and fatty acids biosynthesis-related genes in Escherichia coli and Staphylococcus aureus: Implications for the antibacterial mechanism of naringeninBiochimica et Biophysica. Acta (BBA)-Biomembranes, 2018
-
Plaper et al., 2003Characterization of quercetin binding site on DNA gyraseBiochemical and biophysical research communications, 2003
-
Hooda et al. (2020Effect of quercitin impregnated silver nanoparticle on growth of some clinical pathogensMaterials Today: Proceedings, 2020
Luteolin (3',4',5,7-tetrahydroxyflavone) is a polyphenol of the flavone family, with a molecular structure (C6-C3-C6) of two benzene rings and a third ring containing oxygen, and a double bond between carbons C2 and C3 (Figure 3). Its structure favors its biochemical and biological activity (Wu et al., 2019). This flavonoid has various biological activities, such as antioxidant, anti-inflammatory, antimicrobial, anticarcinogenic, and hypoglycemic, hypolipidemic, hypotensive, and immunomodulatory effects (Wu et al., 2019). In bacteria, luteolin affects the integrity of the cell wall and membrane, inhibiting nucleic acid synthesis and protein expression and interfering with energy metabolism (Guo et al., 2022). In a study, Qian et al. (2021) found that luteolin impairs cell membrane morphology and affects biofilm formation in Staphylococcus aureus and Escherichia coli. In studies against Trueperella pyogenes, a minimum inhibitory concentration (MIC) of luteolin of 78 μg/mL was reported, and at half the MIC, susceptibility to methicillin- and macrolide-resistant Staphylococcus increases, providing an alternative treatment for resistant pathogens (Guo et al., 2022).
-
Wu et al., 2019Delivery luteolin with folacin-modified nanoparticle for glioma therapyInternational Journal of Nanomedicine, 2019
-
Wu et al., 2019Delivery luteolin with folacin-modified nanoparticle for glioma therapyInternational Journal of Nanomedicine, 2019
-
Guo et al., 2022Luteolin increases susceptibility to macrolides by inhibiting MsrA efflux pump in Trueperella pyogenesVeterinary Research, 2022
-
Qian et al. (2021)Mechanisms of Action of Luteolin Against Single-and Dual-Species of Escherichia coli and Enterobacter cloacae and Its Antibiofilm ActivitiesApplied Biochemistry and Biotechnology, 2020
-
Guo et al., 2022Luteolin increases susceptibility to macrolides by inhibiting MsrA efflux pump in Trueperella pyogenesVeterinary Research, 2022
ACTIVITY OF Lippia spp. AGAINST RESISTANT BACTERIA ASSOCIATED WITH BOVINE MASTITIS
There are reports of the antibacterial activity of Lippia spp. extracts on resistant bacteria such as Streptococcus spp., Staphylococcus aureus, Pseudomonas aeruginosa, Klebsiella pneumoniae, and Escherichia coli (Gupta et al., 2020; Pinheiro et al., 2022; Nonato et al., 2022). The minimum inhibitory concentration in the evaluation of Lippia spp. extracts and metabolites on bacteria varies depending on the species evaluated, extraction methodology, and evaluation (Rani et al., 2022; Nonato et al., 2022; Suarez et al., 2024).
-
Gupta et al., 2020Essential oils and mastitis in dairy animals: a reviewHaryana Veterinarian, 2020
-
Pinheiro et al., 2022Inhibitory and bactericidal activities of Lippia origanoides essential oil against Escherichia coli, Klebsiella pneumoniae and Pseudomonas aeruginosa multidrug resistantResearch, Society and Development, 2022
-
Nonato et al., 2022Comparative analysis of chemical profiles and antioxidant activities of essential oils obtained from species of Lippia L. by chemometricsFood Chemistry, 2022
-
Rani et al., 2022Antibacterial activity and mechanism of essential oils in combination with medium‐chain fatty acids against predominant bovine mastitis pathogensLetters in Applied Microbiology, 2022
-
Nonato et al., 2022Comparative analysis of chemical profiles and antioxidant activities of essential oils obtained from species of Lippia L. by chemometricsFood Chemistry, 2022
-
Suarez et al., 2024Screening of essential oils against oxacillin-resistant Staphylococcus aureus strains isolated from bovine mastitisBoletín Latinoamericano y del Caribe de Plantas Medicinales y Aromáticas, 2024
In Streptococcus spp., an MIC of 67 mg/mL is reported for supercritical fluid from L. graveolens, while for metabolites: thymol, MICs of 0.31 to 8.0 mg/mL are reported, and for carvacrol, 0.31 to 9 mg/mL (Table 2). In Staphylococcus spp., the MIC for ethanolic extracts of Lippia spp. is reported to be 128 to 512 µg/mL, for essential oil 53.3 to 512 µg/mL, while for thymol it was 0.15 to 0. 75 µg/mL and carvacrol from 0.38 to 1.3 µg/mL (Dal Pozzo et al., 2011; Rani et al., 2022; Gallegos et al., 2019; Nonato et al., 2022).
-
Dal Pozzo et al., 2011Atividade de óleos essenciais de plantas condimentares frente Staphylococcus spp. isolados de mastite bovinaArquivo Brasileiro de Medicina Veterinária e Zootecnia, 2011
-
Rani et al., 2022Antibacterial activity and mechanism of essential oils in combination with medium‐chain fatty acids against predominant bovine mastitis pathogensLetters in Applied Microbiology, 2022
-
Gallegos et al., 2019Actividad antibacteriana de cinco compuestos terpenoides: carvacrol, limoneno, linalool, αterpineno y timolTropical and Subtropical Agroecosystems, 2019
-
Nonato et al., 2022Comparative analysis of chemical profiles and antioxidant activities of essential oils obtained from species of Lippia L. by chemometricsFood Chemistry, 2022
Tabla 2
Actividad antibacteriana de Lippia spp y sus metabolitos contra patógenos resistentes asociados a mastitis bovina
Actividad antibacteriana de Lippia spp y sus metabolitos contra patógenos resistentes asociados a mastitis bovina
| Bacteria | Extracts and compounds | Minimum Inhibitory Concentration (MIC) | Reference |
|---|---|---|---|
| Streptococcus spp. | Thymol Carvacrol |
0.31-0.63 µL/mL 0.16-0.63 µL/mL |
Gupta et al. (2020) |
| Streptococcus agalactiae | Supercritical fluid from L. graveolens | 67 mg/mL | García et al. (2019) |
| Thymol Carvacrol | 8.0 mg/mL 9.0 mg/mL | Gupta et al. (2020) | |
| Staphylococcus aureus | Ethanolic extract of L. alba
Ethanolic extract of L. sidoides Ethanolic extract of L. gracilis Essential oil of L. alba Essential oil of L. sidoides Essential oil of L. gracilis |
853 µg/mL 128 µg/mL 512 µg/mL 256 µg/mL 53.3 µg/mL 512 µg/mL |
Nonato et al. (2022) |
| Essential oil of L. graveolens | 12 µL/mL | Suarez et al. (2024) | |
| Thymol | 0.15-0.75 mg/mL | Gallegos et al. (2019) | |
| Carvacrol | 0.38-0.45 mg /mL | Rani et al. (2022) | |
| Staphylococcus spp. | L. graveolens essential oil
Thymol Carvacrol |
1.6 mg /mL 0.4 a 0.5 mg/mL 0.8 a 1.3 mg/mL |
Dal Pozzo et al. (2011) |
| Pseudomonas aeruginosa | Ethanolic extract of L. alba
Ethanolic extract of L. sidoides Ethanolic extract of L. gracilis Essential oil of L. alba Essential oil of L. sidoides Essential oil of L. gracilis |
213.3 µg/mL 128 µg/mL 512 µg/mL 1024 µg/mL 298.6 µg/mL |
Nonato et al. (2022) |
| Essential oil of L. berlandieri | 682 µg/mL | ||
| Essential oil of L. origanoides | 80 µg/mL | Reyes et al. (2020) | |
| Essential oil of L. origanoides | 2500 μg/mL | Pinheiro et al. (2022) | |
| Mixture of essential oils of L. salvifolia: L. sidoides (9:1) | 0.37 µg/mL | >Gupta et al. (2020) | |
| Escherichia coli | Extracto etanólico de L. origanoides | 5.0 μL | Castellanos et al. (2020) |
| Ethanolic extract of L. alba
Ethanolic extract of L. sidoides Ethanolic extract of L. gracilis Essential oil of L. alba 1Essential oil of L. sidoides Essential oil of L. gracilis |
768 µg/mL 74.6 µg/mL 256 µg/mL 106.6 µg/mL 106.6 µg/mL 426.6 µg/mL |
Nonato et al. (2022) | |
| L. origanoides essential oil | 312 μg/mL | Pinheiro et al. (2022) | |
| L. berlandieri essential o | 4 μg/mL | Bautista et al. (2021) | |
| Mixture of L. salvifolia essential oils: L. sidoides (9:1) | 0.37 µg/mL | Gupta et al. (2020) | |
| Thymol | 0.15-0.38 mg/mL | Gallegos et al. (2019) | |
| Carvacrol | 0.15-0.75 mg/mL | Rani et al. (2022) | |
| Klebsiella pneumoniae | L. origanoides essential oil
Thymol Carvacrol |
312 μg/mL 0.75 mg/mL 0.75 mg/mL |
Pinheiro et al. (2022) Rani et al. (2022) |
-
Gupta et al. (2020)Essential oils and mastitis in dairy animals: a reviewHaryana Veterinarian, 2020
-
García et al. (2019)Inclusión de extracto de Lippia graveolens (Kunth) en la alimentación de Oreochromis niloticus (Linnaeus, 1758) para la prevención de estreptococosis por Streptococcus agalactiaeAquaTIC, 2019
-
Gupta et al. (2020)Essential oils and mastitis in dairy animals: a reviewHaryana Veterinarian, 2020
-
Nonato et al. (2022)Comparative analysis of chemical profiles and antioxidant activities of essential oils obtained from species of Lippia L. by chemometricsFood Chemistry, 2022
-
Suarez et al. (2024)Screening of essential oils against oxacillin-resistant Staphylococcus aureus strains isolated from bovine mastitisBoletín Latinoamericano y del Caribe de Plantas Medicinales y Aromáticas, 2024
-
Gallegos et al. (2019)Actividad antibacteriana de cinco compuestos terpenoides: carvacrol, limoneno, linalool, αterpineno y timolTropical and Subtropical Agroecosystems, 2019
-
Rani et al. (2022)Antibacterial activity and mechanism of essential oils in combination with medium‐chain fatty acids against predominant bovine mastitis pathogensLetters in Applied Microbiology, 2022
-
Dal Pozzo et al. (2011)Atividade de óleos essenciais de plantas condimentares frente Staphylococcus spp. isolados de mastite bovinaArquivo Brasileiro de Medicina Veterinária e Zootecnia, 2011
-
Nonato et al. (2022)Comparative analysis of chemical profiles and antioxidant activities of essential oils obtained from species of Lippia L. by chemometricsFood Chemistry, 2022
-
Reyes et al. (2020)Inhibitory Effect of Mexican Oregano (Lippia berlandieri Schauer) Essential Oil on Pseudomonas aeruginosa and Salmonella Thyphimurium Biofilm FormationFrontiers in Sustainable Food Systems, 2020
-
Pinheiro et al. (2022)Inhibitory and bactericidal activities of Lippia origanoides essential oil against Escherichia coli, Klebsiella pneumoniae and Pseudomonas aeruginosa multidrug resistantResearch, Society and Development, 2022
-
>Gupta et al. (2020)Essential oils and mastitis in dairy animals: a reviewHaryana Veterinarian, 2020
-
Castellanos et al. (2020)Evaluación antimicrobiana del aceite esencial de Lippia graveolens como inhibidor de crecimiento de Salmonella sp, E. coli y Enterococcus spe-CUCBA, 2020
-
Nonato et al. (2022)Comparative analysis of chemical profiles and antioxidant activities of essential oils obtained from species of Lippia L. by chemometricsFood Chemistry, 2022
-
Pinheiro et al. (2022)Inhibitory and bactericidal activities of Lippia origanoides essential oil against Escherichia coli, Klebsiella pneumoniae and Pseudomonas aeruginosa multidrug resistantResearch, Society and Development, 2022
-
Bautista et al. (2021)Mexican Oregano (Lippia graveolens Kunth) as Source of Bioactive Compounds: A ReviewMolecules, 2021
-
Gupta et al. (2020)Essential oils and mastitis in dairy animals: a reviewHaryana Veterinarian, 2020
-
Gallegos et al. (2019)Actividad antibacteriana de cinco compuestos terpenoides: carvacrol, limoneno, linalool, αterpineno y timolTropical and Subtropical Agroecosystems, 2019
-
Rani et al. (2022)Antibacterial activity and mechanism of essential oils in combination with medium‐chain fatty acids against predominant bovine mastitis pathogensLetters in Applied Microbiology, 2022
-
Pinheiro et al. (2022)Inhibitory and bactericidal activities of Lippia origanoides essential oil against Escherichia coli, Klebsiella pneumoniae and Pseudomonas aeruginosa multidrug resistantResearch, Society and Development, 2022
-
Rani et al. (2022)Antibacterial activity and mechanism of essential oils in combination with medium‐chain fatty acids against predominant bovine mastitis pathogensLetters in Applied Microbiology, 2022
In Pseudomonas aeruginosa, an MIC for ethanolic extracts of 128 to 512 µg/mL is reported, while for essential oils it is 0.37 to 80 µg/mL. In the case of Escherichia coli, the following MIC reports were found: ethanolic extracts (74.6 to 256 µg/mL), essential oils (0.37 to 426 µg/mL), thymol (0.15 to 0.38 µg/mL), and carvacrol (0.15 to 0.75 µg/mL).
Essential oils and their compounds (carvacrol and thymol) have the highest antibacterial activity in resistant bacteria isolated from mastitis (Rani et al., 2022; Nonato et al., 2022; Suarez et al., 2024). Pinheiro et al. (2022) mention that the essential oil has inhibitory and bactericidal action against strains of Escherichia coli and Klebsiella pneumoniae, but not against Pseudomonas aeruginosa, where its effectiveness is reduced by the formation of biofilm and the action of efflux pumps, intrinsic characteristics of this species. The combination of ethanolic extracts of Lippia spp. with antimicrobials reduces the MICs of amikacin, gentamicin, and cephalothin, but has an antagonistic effect with benzylpenicillin and other natural extracts (Nonato et al., 2022). Currently, synergies between extracts and antimicrobials are being investigated to improve efficacy, reduce toxicity, and bacterial resistance (Garcia et al., 2019; Pinheiro et al., 2022; Nonato et al., 2023; Suarez et al., 2024).
-
Rani et al., 2022Antibacterial activity and mechanism of essential oils in combination with medium‐chain fatty acids against predominant bovine mastitis pathogensLetters in Applied Microbiology, 2022
-
Nonato et al., 2022Comparative analysis of chemical profiles and antioxidant activities of essential oils obtained from species of Lippia L. by chemometricsFood Chemistry, 2022
-
Suarez et al., 2024Screening of essential oils against oxacillin-resistant Staphylococcus aureus strains isolated from bovine mastitisBoletín Latinoamericano y del Caribe de Plantas Medicinales y Aromáticas, 2024
-
Pinheiro et al. (2022)Inhibitory and bactericidal activities of Lippia origanoides essential oil against Escherichia coli, Klebsiella pneumoniae and Pseudomonas aeruginosa multidrug resistantResearch, Society and Development, 2022
-
Nonato et al., 2022Comparative analysis of chemical profiles and antioxidant activities of essential oils obtained from species of Lippia L. by chemometricsFood Chemistry, 2022
-
Garcia et al., 2019Inclusión de extracto de Lippia graveolens (Kunth) en la alimentación de Oreochromis niloticus (Linnaeus, 1758) para la prevención de estreptococosis por Streptococcus agalactiaeAquaTIC, 2019
-
Pinheiro et al., 2022Inhibitory and bactericidal activities of Lippia origanoides essential oil against Escherichia coli, Klebsiella pneumoniae and Pseudomonas aeruginosa multidrug resistantResearch, Society and Development, 2022
-
Nonato et al., 2023Comparative analysis of chemical profiles and antioxidant activities of essential oils obtained from species of Lippia L. by chemometricsFood Chemistry, 2022
-
Suarez et al., 2024Screening of essential oils against oxacillin-resistant Staphylococcus aureus strains isolated from bovine mastitisBoletín Latinoamericano y del Caribe de Plantas Medicinales y Aromáticas, 2024
CONCLUSIONS
The main secondary metabolites of L. graveolens are flavonoids and monoterpenes, and their concentration varies according to the soil and climate conditions of the plant's habitat. Its antibacterial activity has been demonstrated against various bacterial genera of importance in health, including those associated with bovine mastitis. The greatest antibacterial activity of L. graveolens has been associated with thymol and carvacrol; however, activity has also been reported due to the presence of naringenin, quercetin, and luteolin. In the search for alternatives to combat resistant or multidrug-resistant bacteria associated with bovine mastitis, the secondary metabolites of Lippia graveolens represent an option for studying new treatments.
LITERATURA CITADA
- Arias J, Muñoz F, Mejía J, Kumar A, Villa AL, Martínez JR, Stashenko EE. 2023. Simultaneous extraction with two phases (modified supercritical CO2 and CO2-expanded liquid) to enhance sustainable extraction/isolation of pinocembrin from Lippia origanoides (Verbenaceae). Advances in Sample Preparation. 100059(6):1-12. ISSN: 2772-5820. https://doi.org/10.1016/j.sampre.2023.100059 Links
- Bautista HI, Aguilar CN, Martínez AGC, Torres LC, Ilina A, Flores GAC, Chávez GML. 2021. Mexican Oregano (Lippia graveolens Kunth) as Source of Bioactive Compounds: A Review. Molecules. 26(17):1-12. ISSN: 1420-3049. https://doi.org/10.3390/molecules26175156 Links
- Bernal MMDJ, Carrasco PMDC, Heredia JB, Bastidas BPDJ, Gutiérrez GEP, León FJ, Angulo EMÁ. 2023. Green extracts and UPLC-TQS-MS/MS profiling of flavonoids from Mexican Oregano (Lippia graveolens) using natural deep eutectic solvents/ultrasoundassisted and supercritical fluids. Plants.12(8):1-12. ISSN: 2223-7747. https://doi.org/10.3390/plants12081692 Links
- Bernal MMDJ, Gutiérrez GEP, Contreras AL, Muy RMD, López MLX, Heredia JB. 2022. Spray-dried microencapsulation of oregano (Lippia graveolens) polyphenols with maltodextrin enhances their stability during in vitro digestion. Journal of Chemistry. 2022 (141):1-10. ISSN:1916-9698. https://doi.org/10.1155/2022/8740141 Links
- Bueno DAY, Cervantes MJ, Obledo VEN. 2014. Composition of essential oil from Lippia graveolens. Relationship between spectral light quality and thymol and carvacrol content. Journal of essential oil research. 26(3):153-160. ISSN: 2163-8152 https://doi.org/10.1080/10412905.2013.840808 Links
- Calamaco ZG, Montfort GRC, Marszalek JE, González GV. 2023. Revisión sobre el orégano mexicano Lippia graveolens HBK.(Sinonimia Lippia berlandieri Schauer) y su aceite esencial. Investigación y Desarrollo en Ciencia y Tecnología de Alimentos. 8(1):861-871. ISSN: 2448-7503. https://doi.org/10.29105/idcyta.v8i1.109 Links
- Calvo ILM, Parra TV, Acosta AV, Escalante EF, Díaz VL, Dzib GR, Peña RLM. 2014. Phytochemical Diversity of the Essential Oils of Mexican Oregano (Lippia graveolens Kunth) Populations along an Edapho‐Climatic Gradient. Chemistry & biodiversity. 11(7):1010-1021. ISSN: 1612-1872. https://doi.org/10.1002/cbdv.201300389 Links
- Castellanos HOA, Rodríguez SMD, Acevedo HGJ, Rayn, CA, Rodríguez SA. 2020. Evaluación antimicrobiana del aceite esencial de Lippia graveolens como inhibidor de crecimiento de Salmonella sp, E. coli y Enterococcus sp. e-CUCBA. 2020 (14):1-6. ISSN: 2448-5225. https://doi.org/10.32870/e-cucba.v0i14.155 Links
- Castillo HGA, Espinosa ME, Haro GJN, García FJA, Andrews HE, Velázquez MM. 2023. Impact of fractional distillation on physicochemical and biological properties of oregano essential oil of Lippia graveolens HBK grown wild in Mexico. Journal of Essential Oil Bearing Plants. 26(6):1515-1525. ISSN: 0972-060X. https://doi.org/10.1080/0972060X.2023.2295418 Links
- Chizzola R. 2013. Regular monoterpenes and sesquiterpenes (essential oils). Natural products. 2023(10):973-978. ISSN: 1520-6025. https://doi.org/10.1007/978-3-642-22144-6130 Links
- Cortés CMDC, Flores MH, Orozco AI, León CC, Suárez JA, Estarrón EM, López MI. 2021. Identification and quantification of phenolic compounds from Mexican oregano (Lippia graveolens HBK) hydroethanolic extracts and evaluation of its antioxidant capacity. Molecules. 26(3-702):1-18. ISSN: 1420-3049. https://doi.org/10.3390/molecules26030702 Links
- Cui H, Zhang C, Li C, Lin L. 2019. Antibacterial mechanism of oregano essential oil. Industrial Crops and Products. 139(111498):1-17. ISSN: 0926-6690. https://doi.org/10.1016/j.indcrop.2019.111498 Links
- Dal Pozzo M, Santurio DF, Rossatto L, Vargas AC, Alves SH, Loreto ES, Viegas J. 2011. Atividade de óleos essenciais de plantas condimentares frente Staphylococcus spp. isolados de mastite bovina. Arquivo Brasileiro de Medicina Veterinária e Zootecnia. 63(5):1229-1232. ISSN: 1678-4162. https://doi.org/10.1590/S0102-09352011000500026 Links
- Di Pasqua R, Mamone G, Ferranti P, Ercolini D, Mauriello G. 2010. Changes in the proteome of Salmonella enterica serovar Thompson as stress adaptation to sublethal concentrations of thymol. Proteomics. 10(5):1040-1049. ISSN: 1876-7737. https://doi.org/10.1002/pmic.200900568 Links
- Du E, Gan L, Li Z, Wang W, Liu D, Guo Y. 2015. In vitro antibacterial activity of thymol and carvacrol and their effects on broiler chickens challenged with Clostridium perfringens. Journal of animal science and biotechnology. 6(58):1-12. ISSN: 2049-1891. https://doi.org/10.1186/s40104-015-0055-7 Links
- Duda MA, Kozłowska J, Krzyżek P, Anioł M, Seniuk A, Jermakow K, Dworniczek E. 2020. Antimicrobial O-alkyl derivatives of naringenin and their oximes against multidrugresistant bacteria. Molecules. 25(16-3642):1-15. ISSN: 1420-3049. https://doi.org/10.3390/molecules25163642 Links
- Erazo GMJ, Arroyo BFA, Arroyo BDA, Castro GMR, Santacruz TSG, Armas VADC. 2017. Efecto antimicrobiano del cinamaldehído, timol, eugenol y quitosano sobre cepas de Streptococcus mutans. Revista Cubana de Estomatología. 54(4):1-9. ISSN: 1561297X. http://scielo.sld.cu/scielo.php?script=sci_arttext&pid=S003475072017000400005&lng=es&tlng=es Links
- Galarza GMI, Yarzábal RLA. 2021. Staphylococcus aureus resistentes à meticilina em animais de fazenda em américa do Sul: uma revisão sistemática. Vive Revista de Salud. 4(11):246-265. ISSN:2664-3243 https://doi.org/10.33996/revistavive.v4i11.99 Links
- Gallegos FPI, Bañuelos VR, Delgadillo RL, Meza LC, Echavarría CF. 2019. Actividad antibacteriana de cinco compuestos terpenoides: carvacrol, limoneno, linalool, αterpineno y timol. Tropical and Subtropical Agroecosystems. 22(2):241-248. ISSN:18700462. http://dx.doi.org/10.56369/tsaes.2838 Links
- Gallegos FPI, Delgadillo RL, Bañuelos VR, Echavarría CF, Valladares CB, Meza LC. 2022. Inhibition of bacterial mobility by terpenoid compounds and plant essential oils. Tropical and Subtropical Agroecosystems. 25(1):1-10. ISSN:1870-0462. http://dx.doi.org/10.56369/tsaes.3914 Links
- Garcia CM, Picos CLA, Gutiérrez GEP, Angulo EMA, Licea CA, Heredia JB. 2022. Loading and release of phenolic compounds present in Mexican oregano (Lippia graveolens) in different chitosan bio-polymeric cationic matrixes. Polymers. 4(17):3609. ISSN: 2073-4360. https://doi.org/10.3390/polym14173609 Links
- García PJR; Marroquín DC; Pérez GMI. 2019. Inclusión de extracto de Lippia graveolens (Kunth) en la alimentación de Oreochromis niloticus (Linnaeus, 1758) para la prevención de estreptococosis por Streptococcus agalactiae. AquaTIC. 1(54):15-24. ISSN:1578-4541. https://www.redalyc.org/journal/494/49464451002/html/ Links
- González TME, Hernández SLY, Muñoz OV, Dorazco GA, Guevara FP, Aguirre HE. 2017. Pharmacological evaluation of the anxiolytic-like effects of Lippia graveolens and bioactive compounds. Pharmaceutical biology. ISSN: 1744-5116. 55(1):1569-1576. https://doi.org/10.1080/13880209.2017.1310908 Links
- Mar, Escobedo BC. 2022. In vitro evaluation of the antimicrobial activity of two types of oregano (Lippia berlandieri) essential oils against bacteria from shrimp ponds. Revista bio ciencias. 9(2022):1-15. ISSN: 2007-3380. https://doi.org/10.15741/revbio.09.e1344 Links
- Guo Y, Huang C, Su H, Zhang Z, Chen M, Wang R, Liu, M. 2022. Luteolin increases susceptibility to macrolides by inhibiting MsrA efflux pump in Trueperella pyogenes. Veterinary Research. 53(2022):1-11. ISSN: 1297-9716. https://doi.org/10.1186/s13567-021-01021-w Links
- Gupta R, Kumar S, Khurana R. 2020. Essential oils and mastitis in dairy animals: a review. Haryana Veterinarian. 2020(59):1-9. ISSN: 0033-4359. https://www.researchgate.net/publication/340004579 Links
- Hernandez T, Canales M, Avila JG, García AM, Meraz S, Caballero J, Rafael, LIRA. 2009. Composition and antibacterial activity of essential oil of Lippia graveolens HBK (Verbenaceae). Boletín Latinoamericano y del Caribe de Plantas Medicinales y Aromáticas. 8(4):295-300. ISSN: 0717-7917. https://www.redalyc.org/pdf/856/85611265010.pdf Links
- Hooda H, Singh P, Bajpai S. 2020. Effect of quercitin impregnated silver nanoparticle on growth of some clinical pathogens. Materials Today: Proceedings. 2020(31):625-630. ISSN: 2214-7853. https://doi.org/10.1016/j.matpr.2020.03.530 Links
- Ke JY, Banh T, Hsiao YH, Cole RM, Straka SR, Yee LD, Belury MA. 2017. Citrus flavonoid naringenin reduces mammary tumor cell viability, adipose mass, and adipose inflammation in obese ovariectomized mice. Molecular nutrition & food research. 61(9): 1600934. ISSN: 1613-4133. https://doi.org/10.1002/mnfr.201600934 Links
- Kovačević Z, Samardžija M, Horvat O, Tomanić D, Radinović M, Bijelić K, Kladar N. 2022. Is there a relationship between antimicrobial use and antibiotic resistance of the most common mastitis pathogens in dairy cows?. Antibiotics. 12(3):1-15. ISSN: 20796382. https://doi.org/10.3390/antibiotics12010003 Links
- Kozłowska J, Grela E, Baczyńska D, Grabowiecka A, Anioł M. 2019. Novel O-alkyl derivatives of naringenin and their oximes with antimicrobial and anticancer activity. Molecules. 24(4-679):1-15. ISSN: 1420-3049. https://doi.org/10.3390/molecules24040679 Links
- Lee JH, Kim YG, Lee J. 2017. Carvacrol‐rich oregano oil and thymol‐rich thyme red oil inhibit biofilm formation and the virulence of uropathogenic Escherichia coli. Journal of applied microbiology. 123(6):1420-1428. ISSN: 1364-5072. https://doi.org/10.1111/jam.13602 Links
- Lee JH, Park JH, Cho HS, Joo SW, Cho MH, Lee J. 2013. Anti-biofilm activities of quercetin and tannic acid against Staphylococcus aureus. Biofouling. 29(5):491-499. ISSN: 1029-2454. https://doi.org/10.1080/08927014.2013.788692 Links
- Leyva LN, Nair V, Bang WY, Cisneros ZL, Heredia JB. 2016. Protective role of terpenes and polyphenols from three species of Oregano (Lippia graveolens, Lippia palmeri and Hedeoma patens) on the suppression of lipopolysaccharide-induced inflammation in RAW 264.7 macrophage cells. Journal of Ethnopharmacology. 2016(187):302-312. ISSN: 0378-8741. https://doi.org/10.1016/j.jep.2016.04.051 Links
- Li X, Xu C, Liang B, Kastelic JP, Han B, Tong X, Gao J. 2023. Alternatives to antibiotics for treatment of mastitis in dairy cows. Frontiers in Veterinary Science. 10(1160350):1-13. ISSN: 2297-1769. https://doi.org/10.3389/fvets.2023.1160350 Links
- Lin LZ, Mukhopadhyay S, Robbins RJ, Harnly JM. 2007. Identification and quantification of flavonoids of Mexican oregano (Lippia graveolens) by LC-DAD-ESI/MS analysis. Journal of food composition and analysis. 20(5):361-369. ISSN: 0889-1575. https://doi.org/10.1016/j.jfca.2006.09.005 Links
- Llamas TI, Grijalva AR, Porter BL, Calvo ILM. 2022. Impact of the in situ-ex situ management of Mexican oregano Lippia origanoides Kunth in northwestern Yucatan. Botanical Sciences. 100(3):610-630. ISSN: 2007-4476. https://doi.org/10.17129/botsci.2994 Links
- Martínez NDA, Parra TV, Ferrer OMM, Calvo ILM. 2014. Genetic diversity and genetic structure in wild populations of Mexican oregano (Lippia graveolens HBK) and its relationship with the chemical composition of the essential oil. Plant systematics and evolution. 300(2014):535-547. ISSN: 1615-6110. https://doi.org/10.1007/s00606-0130902-y Links
- Memar MY, Raei P, Alizadeh N, Aghdam MA, Kafil HS. 2017. Carvacrol and thymol: strong antimicrobial agents against resistant isolates. Reviews and Research in Medical Microbiology. 28(2):63-68. ISSN: 2770-3150. https://doi.org/10.1097/MRM.0000000000000100 Links
- Morales UAL, Rivero PN, Valladares CB, Velázquez OV, Delgadillo RL, Zaragoza BA. 2023. Bovine mastitis, a worldwide impact disease: prevalence, antimicrobial resistance, and viable alternative approaches. Veterinary and Animal Science. 21(100306):1-14. ISSN: 2451-943X. https://doi.org/10.1016/j.vas.2023.100306 Links
- Nonato CDFA, Camilo CJ, Leite DOD, da Nobrega MGLA, Ribeiro FJ, de Menezes IRA, da Costa JGM. 2022. Comparative analysis of chemical profiles and antioxidant activities of essential oils obtained from species of Lippia L. by chemometrics. Food Chemistry. 2022(384):1-8. ISSN: 0308-8146. https://doi.org/10.1016/j.foodchem.2022.132614 Links
- Ocampo VRV, Malda BGX, Suárez RG. 2009. Biología reproductiva del orégano mexicano (Lippia graveolens Kunth) en tres condiciones de aprovechamiento. Agrociencia. 43(5):475-482. ISSN: 1405-3195. http://www.scielo.org.mx/scielo.php?script=sci_arttext&pid=S140531952009000500003 &lng=es&nrm=iso Links
- Pascu C, Herman V, Iancu I, Costinar L. 2022. Etiology of mastitis and antimicrobial resistance in dairy cattle farms in the western part of Romania. Antibiotics. 11(1):57. ISSN: 2079-6382. https://doi.org/10.3390/antibiotics11010057 Links
- Picos SMA, Gutiérrez GEP, Valdez TB, Angulo EMA, López MLX, Delgado VF, Heredia JB. 2021. Supercritical CO2 extraction of oregano (Lippia graveolens) phenolic compounds with antioxidant, α-amylase and α-glucosidase inhibitory capacity. Journal of Food Measurement and Characterization. 15(4):3480-3490. ISSN: 2193-4134. https://doi.org/10.1007/s11694-021-00928-4 Links
- Pinheiro LG, dos Santos FRO, Rodrigues THS, Pinto VDPT, Barbosa FCB. 2022. Inhibitory and bactericidal activities of Lippia origanoides essential oil against Escherichia coli, Klebsiella pneumoniae and Pseudomonas aeruginosa multidrug resistant. Research, Society and Development. 11(9):1-12. ISSN: 2525-3409. https://doi.org/10.33448/rsdv11i9.31478 Links
- Plaper AGM, Hafner I, Oblak M, Šolmajer T, Jerala R. 2003. Characterization of quercetin binding site on DNA gyrase. Biochemical and biophysical research communications. 306(2):530-536. ISSN: 0006-291X. https://doi.org/10.1016/S0006291X(03)01006-4 Links
- Qian WD, Fu YT, Liu M, Zhang JN, Wang WJ, Li JY, Li YD. 2020. Mechanisms of Action of Luteolin Against Single-and Dual-Species of Escherichia coli and Enterobacter cloacae and Its Antibiofilm Activities. Applied Biochemistry and Biotechnology. 193(5):1397-1414. ISSN: 1559-0291. https://doi.org/10.1007/s12010-020-03330-w Links
- Rani S, Verma S., Singh H, Ram C. 2022. Antibacterial activity and mechanism of essential oils in combination with medium‐chain fatty acids against predominant bovine mastitis pathogens. Letters in Applied Microbiology. 74(6):959-969. ISSN: 1472-765X. https://doi.org/10.1111/lam.13675 Links
- Rastrelli L, Caceres A, Morales C., De Simone F, Aquino R. 1998. Iridoids from Lippia graveolens. Phytochemistry. 49(6):1829-1832. ISSN: 1873-3700. https://doi.org/10.1016/S0031-9422(98)00196-4 Links
- Reyes JF, Munguía PR, Cid PTS, Hernández CP, Ochoa VCE, Avila SR. 2020. Inhibitory Effect of Mexican Oregano (Lippia berlandieri Schauer) Essential Oil on Pseudomonas aeruginosa and Salmonella Thyphimurium Biofilm Formation. Frontiers in Sustainable Food Systems. 4(36):1-6. ISSN: 2571-581X. https://doi.org/10.3389/fsufs.2020.00036 Links
- Shapira R, Mimran E. 2007. Isolation and characterization of Escherichia coli mutants exhibiting altered response to thymol. Microbial Drug Resistance. 13(3):157-165. ISSN: 1931-8448. https://doi.org/10.1089/mdr.2007.731 Links
- Sharun K, Dhama K, Tiwari R, Gugjoo MB, Iqbal YM, Patel SK, Chaicumpa W. 2021. Advances in therapeutic and managemental approaches of bovine mastitis: a comprehensive review. Veterinary Quarterly. 41(1):107-136. ISSN:1875-5941. https://doi.org/10.1080/01652176.2021.1882713 Links
- Soto ALC, Sacramento RJC, Ruiz MCA, Lope NMC, Rocha UJA. 2019. Extraction yield and kinetic study of Lippia graveolens with supercritical CO2. The Journal of Supercritical Fluids. 2019(145):205-210. ISSN: 0896-8446. https://doi.org/10.1016/j.supflu.2018.12.018 Links
- Suarez BJM, Suarez MC, Calvo MA, Parada F, Cortés F, Tobón F, Toro S. 2024. Screening of essential oils against oxacillin-resistant Staphylococcus aureus strains isolated from bovine mastitis. Boletín Latinoamericano y del Caribe de Plantas Medicinales y Aromáticas. 23(3):401-409. ISSN: 0717-7917. https://doi.org/10.37360/blacpma.24.23.3.28 Links
- Tapia RMR, Hernandez MA, Gonzalez AGA, a TMA, Martins CM, Ayala ZJF. 2017. Carvacrol as potential quorum sensing inhibitor of Pseudomonas aeruginosa and biofilm production on stainless steel surfaces. Food Control. 2017(75):255-261. ISSN: 09567135. https://doi.org/10.1016/j.foodcont.2016.12.014 Links
- Torres SLM, Pérez CA, Torregroza EA, Vitola RD. 2022. Chemical comparison of the essential oils of Lippia Origanoides in two agroclimatic zones of the Colombian Caribbean coast. Dyna. 89(220):172-177. ISSN: 0012-7353. https://doi.org/10.15446/dyna.v89n220.95739 Links
- Ultee A, Kets EP, Alberda M, Hoekstra FA, Smid EJ. 2000. Adaptation of the food-borne pathogen Bacillus cereus to carvacrol. Archives of microbiology. 174(4):233-238. ISSN: 1432-072X. https://doi.org/10.1007/s002030000199 Links
- Vernin G, Lageot C, Gaydou EM, Parkanyi C. 2001. Analysis of the essential oil of Lippia graveolens HBK from El Salvador. Flavour and fragrance journal. 16(3):219-226. ISSN: 1099-1026. https://doi.org/10.1002/ffj.984 Links
- Wang LH, Zeng XA, Wang MS, Brennan CS, Gong D. 2018. Modification of membrane properties and fatty acids biosynthesis-related genes in Escherichia coli and Staphylococcus aureus: Implications for the antibacterial mechanism of naringenin. Biochimica et Biophysica. Acta (BBA)-Biomembranes. 1860(2):481-490. ISSN: 18792642. https://doi.org/10.1016/j.bbamem.217.11.007 Links
- Wang Z, Xue Y, Gao Y, Guo M, Liu Y, Zou X, Yan Y. 2021. Phage vB_PaeS-PAJD-1 rescues murine mastitis infected with multidrug-resistant Pseudomonas aeruginosa. Frontiers in Cellular and Infection Microbiology. 689770(11):1-13. ISSN: 2235-2988 https://doi.org/10.3389/fcimb.2021.689770 Links
- Wu C, Xu Q, Chen X, Liu J. 2019. Delivery luteolin with folacin-modified nanoparticle for glioma therapy. International Journal of Nanomedicine. 2019(14):7515-7531. ISSN: 11782013. https://doi.org/10.2147/IJN.S214585 Links