Revisiones de literatura
The goat udder: mechanism of milk secretion, and protein/fat synthesis
Valencia-Magaña, Favio*1
 http://orcid.org/0000-0001-8390-5224
http://orcid.org/0000-0001-8390-5224
Prado-Rebolledo, Omar2
 http://orcid.org/0000-0001-8802-0177
http://orcid.org/0000-0001-8802-0177
Hernández-Rivera, Juan2
 http://orcid.org/0000-0003-1805-5264
http://orcid.org/0000-0003-1805-5264
Cruz-Tamayo, Alvar3
 http://orcid.org/0000-0002-5509-3430
http://orcid.org/0000-0002-5509-3430
García-Casillas, Arturo**2
 http://orcid.org/0000-0002-7716-210X
http://orcid.org/0000-0002-7716-210X
 http://orcid.org/0000-0001-8390-5224
http://orcid.org/0000-0001-8390-5224Prado-Rebolledo, Omar2
 http://orcid.org/0000-0001-8802-0177
http://orcid.org/0000-0001-8802-0177Hernández-Rivera, Juan2
 http://orcid.org/0000-0003-1805-5264
http://orcid.org/0000-0003-1805-5264Cruz-Tamayo, Alvar3
 http://orcid.org/0000-0002-5509-3430
http://orcid.org/0000-0002-5509-3430García-Casillas, Arturo**2
 http://orcid.org/0000-0002-7716-210X
http://orcid.org/0000-0002-7716-210X-
Publication dates-
October 27, 2023
Jan-Dec , 2023
- Article in PDF
- Article in XML
- Automatic translation
- Send this article by e-mail
- Share this article +
ABSTRACT
In recent years, the shift in milk marketing towards a standardized price structure based on dairy components has placed greater emphasis on lipid and protein concentration over the quantity of L or kg of milk produced. The nutritional content of goat's milk exceeds the nutritional content of cow's milk, in terms of proteins and fats, and it is the lactocytes of the caprine mammary gland, which must replicate and synthesize these components dairy products. Therefore, this review initially considers the understanding of the anatomy and histology of the mammary gland as responsible for all the activities related to the milk ejection mechanism. The development of the mammary gland determines all aspects of cell behavior, so its development is reviewed through four stages: i) mammogenesis, ii) lactogenesis, iii) galactopoiesis, and iv) involution. The remainder of the review emphasizes milk lipogenesis and proteogenesis, due to their various roles within cellular metabolism and the production of the lipid fraction and protein fraction of milk.
Keywords::
mammary gland, goat milk, lipogenesis, proteogenesis
aminoacyl site
amino acids
acyl-CoA:cholesterol acyltransferase
antidiuretic hormone
deoxyribonucleic
messenger ribonucleic acid
transfer ribonucleic ribonucleic acid
adenosine triphosphate
pyruvate
tricalcium phosphate
phosphocholine cytidylyl transferase
carbon dioxide
diacylglycerol
acyl-CoA:diacylglycerol acyltransferase
estrone
17 β-estradiol
estriol
fatty acid synthase I
growth hormone
glycerol-3-phosphate acyltransferase
hydrogenocarbonate anion
lysophospholipid acyltransferase
nicotinamide adenine dinucleotide phosphate
peptide bonding
oxytocin
peptidyl site
postpartum
progesterone
phosphatidic acid phosphatase
phospholipase A
perilipin
prolactin
triacylglycerols
uracil-adenine-adenine
uracil-adenine-guanine
uracil-guanine-guanine-adenine
INTRODUCTION
Goat milk has positioned itself as an important element in the human diet (Bauman et al., 2006). Its nutritional relevance lies mainly in two components: i) the lipid fraction, formed by fatty acids (Harvatine et al., 2009) and ii) the protein fraction, where caseins, whey proteins and fat globule membrane proteins are distinguished (Swaisgood, 2003). The nutritional content of goat milk exceeds that of cow milk in terms of protein (goat milk: 3.40 g/100 mL-1 vs cow milk: 3.30 g/100 mL-1) and fat (goat milk: 4.30 g/100 mL-1 vs cow milk: 3.95 g/100 mL-1) content (Davidson & Stabenfeldt, 2020). Current trends in milk production and consumption enhance lipid and protein concentration over the amount of L or kg of milk produced (Manterola, 2011), taking into account the eating habits of a growing urban population (Vidal, 2013). Goat milk collectors use this product mainly for cheese production (National Chamber of Industrial Milk, 2021). For this reason, it is necessary to increase our understanding of the metabolism involved in goat milk production and its lipid and protein contents (Heid & Keenan, 2005; Kumar et al., 2009). Therefore, this review discusses the anatomy and histology of the mammary gland. Its development as a milk-producing organ through four stages: i) mammogenesis, ii) lactogenesis, iii) galactopoiesis, and iv) involution. Continuing with milk ejection and its hormonal control, and to substantiate the basic biochemistry of milk lipid and protein synthesis, information on the processes of milk lipogenesis and proteogenesis is presented at the end.
-
Bauman et al., 2006Major advances associated with the biosynthesis of milkJournal of Dairy Science, 2006
-
Harvatine et al., 2009Recent advances in the regulation of milk fat synthesisAnimal, 2009
-
Swaisgood, 2003Protein composition of milk: identification, structure, and chemical compositionAdvanced dairy chemistry: proteins part A, 2003
-
Davidson & Stabenfeldt, 2020The mammary gland and lactationCunningham’s Textbook of Veterinary Physiology, 2020
-
Manterola, 2011Estrategias nutricionales y alimenticias para modificar los sólidos totales de la leche, 2011
-
Vidal, 2013Lácteos: oferta y demanda en el contexto regional y mundial, 2013
-
National Chamber of Industrial Milk, 2021CANILEC. Markets and statistics, 2021
-
Heid & Keenan, 2005Intracellular origin and secretion of milk fat globulesEuropean Journal of Cell Biology, 2005
-
Kumar et al., 2009Factors affecting rumen methanogens and methane mitigation strategiesWorld Journal of Microbiology & Biotechnology, 2009
I. Anatomy and Histology of the Mammary Gland
Goats have two independent mammary glands, their location in the body of the animal is inguinal and they are: i) pear or elongated type, ii) oval or Alpine type, and iii) globular or Saanen type (Menzies, 2021). Histologically, each mammary gland is composed of two tissues: i) the parenchyma whose origin is embryonic ectoderm, and which includes lactocytes or lacteal exocrineocytes and myoepithelial cells (Lawhead & Baker, 2017), and ii) the stroma whose origin is embryonic mesoderm, and which includes blood vessels, lymphatic vessels, adipose tissue, connective tissue and nervous tissue (Baljit, 2017).
-
Menzies, 2021Udder health for dairy goatsVeterinary Clinics of North America: Food Animal Practice, 2021
-
Lawhead & Baker, 2017The endocrine system/endocrine glandsIntroduction to Veterinary Science, 2017
-
Baljit, 2017Ruminants / the udderDyce, Sack and Wensing's Textbook of Veterinary Anatomy, 2017
The parenchyma develops through the proliferation of lactocytes arising from the primary mammary cord (Menzies, 2021). Lactocytes present receptors for prolactin (PRL) (Baljit, 2017), and eventually form hollow circular structures with a length of 100 to 500 µm called alveoli (Lawhead & Baker, 2017). The outer wall of each alveolus is surrounded by arterial capillaries and venous capillaries along with a layer of myoepithelial cells with receptors for oxytocin (OXT) (Davidson & Stabenfeldt, 2020). The internal structure of the mammary gland consists of: i) parenchyma, ii) lactiferous ducts: intra and inter lobular and lobular depending on their connection within the mammary gland (Reese et al., 2020), iii) glandular lobules (Figure 1), formed by lobules with 150 to 220 lacteal alveoli, iv) myoepithelial cells, v) venules, vi) arterioles, vii) capillaries, viii) gland cisternae (Figure 2), ix) teat cistern and x) nipple canal (Davidson & Stabenfeldt, 2020).
-
Menzies, 2021Udder health for dairy goatsVeterinary Clinics of North America: Food Animal Practice, 2021
-
Baljit, 2017Ruminants / the udderDyce, Sack and Wensing's Textbook of Veterinary Anatomy, 2017
-
Lawhead & Baker, 2017The endocrine system/endocrine glandsIntroduction to Veterinary Science, 2017
-
Davidson & Stabenfeldt, 2020The mammary gland and lactationCunningham’s Textbook of Veterinary Physiology, 2020
-
Reese et al., 2020Mammary gland (mamma, uber, mastos)Veterinary Anatomy of Domestic Animals, 2020
Thumbnail
Figure 1
Representation of the glandular lobe in the mammary gland of a goat
Representation of the glandular lobe in the mammary gland of a goat
Thumbnail
Figure 2
Representation of the mammary gland of a goat
Representation of the mammary gland of a goat
The irrigation of the mammary gland is carried out by the external pudendal artery, which passes through the inguinal canal and divides into cranial and caudal branches (Davidson & Stabenfeldt, 2020). Venous circulation is mainly by the venous circle formed by the external pudendal vein, the caudal superficial epigastric vein and the perineal vein (Lérias et al., 2014). Innervation of the mammary gland is mainly carried out by sympathetic nerve fibers in the first and second lumbar nerves and the inguinal nerves, their function is the control of blood flow in the udder and innervation of the smooth muscle tissue surrounding the lactiferous ducts, gland cistern muscles, teat cistern muscles and teat canal (Dee & Magee, 2018). Milk contained within the alveolar lumen empties into small intralobulillar ducts (Figure 3) that empty into a central collecting space, from which the interlobular ducts emerge (Davidson & Stabenfeldt, 2020).
-
Davidson & Stabenfeldt, 2020The mammary gland and lactationCunningham’s Textbook of Veterinary Physiology, 2020
-
Lérias et al., 2014The mammary gland in small ruminants: major morphological and functional events underlying milk production-a reviewJournal of Dairy Research, 2014
-
Dee & Magee, 2018Anatomy and physiology of the mammary glandAnatomy and Physiology of Farm Animals, 2018
-
Davidson & Stabenfeldt, 2020The mammary gland and lactationCunningham’s Textbook of Veterinary Physiology, 2020
Thumbnail
Figure 3
Lactiferous ducts of the mammary gland
Lactiferous ducts of the mammary gland
Within the lobule the interlobular ducts join to form an intralobular duct, which upon exiting the lobule acquires the name interlobular duct; these ducts may lead directly into the cistern of the gland or join other interlobular ducts before reaching it (Dee & Magee, 2018). Reece & Rowe (2017b). noted that the ductal system connects the gland cistern to the teat cistern, which allows milk to pass from the formation area to the delivery area or nipple canal. The gland cistern in goats is larger in volume compared to that of cattle, and allows for almost 70 % of the milk produced between each milking (Martínez & Suárez, 2018). The anatomy and histology of the mammary gland are modified throughout lactation, by changes associated mainly with the neuroendocrine system (Dee & Magee, 2018). Therefore, there are three stages in mammary biology, characterized by gestation/lactation cycles: i) proliferation, ii) secretion and iii) involution (Lawhead & Baker, 2017). Although most of the proliferation occurs during gestation and most of the involution occurs after milk production, both stages overlap: parenchymal proliferation continues during early lactation (1/3 lactation) and its involution begins during late lactation (3/3 lactation) (Lérias et al., 2014). Event that in goats is reached from 180 to 280 d, with peak production between 8 and 12 weeks postpartum (p.p.) (Menzies, 2021) (Figure 4).
-
Dee & Magee, 2018Anatomy and physiology of the mammary glandAnatomy and Physiology of Farm Animals, 2018
-
Reece & Rowe (2017b)Lactation / functional anatomy of female mammary glands. Mammogenesis, lactogenesis and lactationFunctional Anatomy and Physiology of Domestic Animals, 2017
-
Martínez & Suárez, 2018The mammary gland: morphology and development. Synthesis of milk componentsGoat dairying: production, management, health, quality of milk and products, 201
-
Dee & Magee, 2018Anatomy and physiology of the mammary glandAnatomy and Physiology of Farm Animals, 2018
-
Lawhead & Baker, 2017The endocrine system/endocrine glandsIntroduction to Veterinary Science, 2017
-
Lérias et al., 2014The mammary gland in small ruminants: major morphological and functional events underlying milk production-a reviewJournal of Dairy Research, 2014
-
Menzies, 2021Udder health for dairy goatsVeterinary Clinics of North America: Food Animal Practice, 2021
Thumbnail
Figure 4
Parenchymal development and milk and solids production curve in goats
Parenchymal development and milk and solids production curve in goats
Reese et al. (2020) indicated that when the mammary gland is in a resting state, the lactocytes have a cubic appearance, whereas when the mammary gland is in milk production, their shape is cylindrical (Davidson & Stabenfeldt, 2020). Furthermore, it is important to note that higher milk production is negatively correlated with milk fat and protein, i.e. a decrease in milk L is equivalent to a higher milk solids content and vice versa (Martínez & Suárez, 2018).
-
Reese et al. (2020)Mammary gland (mamma, uber, mastos)Veterinary Anatomy of Domestic Animals, 2020
-
Davidson & Stabenfeldt, 2020The mammary gland and lactationCunningham’s Textbook of Veterinary Physiology, 2020
-
Martínez & Suárez, 2018The mammary gland: morphology and development. Synthesis of milk componentsGoat dairying: production, management, health, quality of milk and products, 201
II. Mammary gland development
Lactation proceeds through a cycle consisting of four stages: i) mammogenesis, ii) lactogenesis, iii) galactopoiesis, and iv) involution (Baljit, 2017). Mammogenesis initiates during fetal life in the embryonic ectoderm, forming the mammary band in the inguinal region after 30 d of conception (Reese et al., 2020), the mammary gland at two months and the nipple cistern at three months of fetal life (Lawhead & Baker, 2017).
-
Baljit, 2017Ruminants / the udderDyce, Sack and Wensing's Textbook of Veterinary Anatomy, 2017
-
Reese et al., 2020Mammary gland (mamma, uber, mastos)Veterinary Anatomy of Domestic Animals, 2020
-
Lawhead & Baker, 2017The endocrine system/endocrine glandsIntroduction to Veterinary Science, 2017
From birth to puberty, the mammary gland exhibits isometric growth with increased connective tissue and fat deposition (Dee & Magee, 2018). Cyclic ovarian activity results in the production of estrogens e.g., estrone (E 1), 17 β-estradiol (E 2) and estriol (E 3). E1, E2 and E3 (Reece & Rowe, 2017a) (Figure 5), together with growth hormone (GH) and adrenal androstenedione, are responsible for the growth of lactiferous ducts (Maldonado et al., 2018). In this regard Lérias et al. (2014) stated that the use of plastic implants with estrogens e.g., E1, E2 and E3, directly and locally stimulates the growth of the lactiferous ducts and on the contrary the application of implants with anti-estrogenic activity, inhibits the growth of the lactiferous ducts within circumscribed areas (Reese et al., 2020).
-
Dee & Magee, 2018Anatomy and physiology of the mammary glandAnatomy and Physiology of Farm Animals, 2018
-
Reece & Rowe, 2017aLactation / functional anatomy of female mammary glands. Mammogenesis, lactogenesis and lactationFunctional Anatomy and Physiology of Domestic Animals, 2017
-
Maldonado et al., 2018Factors influencing milk production of local goats in the Comarca Lagunera, MexicoLivestock Research for Rural Development, 2018
-
Lérias et al. (2014)The mammary gland in small ruminants: major morphological and functional events underlying milk production-a reviewJournal of Dairy Research, 2014
-
Reese et al., 2020Mammary gland (mamma, uber, mastos)Veterinary Anatomy of Domestic Animals, 2020
Thumbnail
Figure 5
Hormones involved in mammary gland development
Hormones involved in mammary gland development
After puberty, the mammary gland shows allometric growth (Goff, 2015) and with each estrus there is a slight development of alveoli influenced by E1, E2 and E3, progesterone (P 4) (Reece & Rowe, 2017a), GH and adrenal androstenedione (Lawhead & Baker, 2017). Information consistent with that reported by Reece & Rowe (2017b) who noted a synergistic stimulation of PRL, androstenedione, E1, E2 and E3, and P4 on mammary gland growth and alveolar lobe development (Neville et al., 2002). Most parenchymal growth happens during gestation (Goff, 2015), induced by P4, PRL and adrenal androstenedione (Reese et al., 2020). By day 35 (Figure 6) stroma is abundant, by day 92 glandular lobules form with several lobes clustered together; milk secretion is present within the alveolar lumen in some lobules, and considerable stroma is still present, by day 120 the lobes of the alveoli are almost fully developed (Svennersten & Olsson, 2005); the alveoli are filled with milk secretion and stroma is reduced to thin bands (Lawhead & Baker, 2017).
-
Goff, 2015Endocrinology, reproduction, and lactation/The mammary gland and lactationDukes' Physiology of Domestic Animals, 2015
-
Reece & Rowe, 2017aLactation / functional anatomy of female mammary glands. Mammogenesis, lactogenesis and lactationFunctional Anatomy and Physiology of Domestic Animals, 2017
-
Lawhead & Baker, 2017The endocrine system/endocrine glandsIntroduction to Veterinary Science, 2017
-
Reece &
Rowe (2017b)Mammary gland (mamma, uber, mastos)Veterinary Anatomy of Domestic Animals, 2020
-
Neville et al.,
2002Hormonal regulation of mammary differentiation and milk secretionJournal of Mammary Gland Biology and Neoplasia, 2002
-
Goff, 2015Endocrinology, reproduction, and lactation/The mammary gland and lactationDukes' Physiology of Domestic Animals, 2015
-
Reese
et al., 2020Mammary gland (mamma, uber, mastos)Veterinary Anatomy of Domestic Animals, 2020
-
Svennersten & Olsson,
2005Endocrinology of milk productionDomestic Animal Endocrinology, 2005
-
Lawhead & Baker, 2017The endocrine system/endocrine glandsIntroduction to Veterinary Science, 2017
Thumbnail
Figure 6
Goat mammary gland parenchyma during gestation
Goat mammary gland parenchyma during gestation
After parturition and with the placenta expulsion, P4 levels fall, initiating lactogenesis (Saipin et al., 2020). The initial phase of lactation is characterized by the positive regulation of milk production, cell proliferation, and a decrease in the apoptosis process in the mammary gland (Henna et al., 2021). During this process the adenohypophyseal endocrine tissue intervenes with the secretion of PRL in its lactotrope cells (Dee & Magee, 2018). PRL is a peptide of 199 amino acids (aa) and atomic mass of 23,000 Da, which binds to its tyrosine kinase family receptors located on lactocytes and activates signal transducers and activators of transcription associated with proliferation, differentiation, and lactogenesis (Lawhead & Baker, 2017). Therefore, PRL is indispensable in milk production (Svennersten & Olsson, 2005). In relation to the topic, an investigation that aimed to evaluate the effect of long-term inhibition of PRL, reported that administration for nine weeks of a dopaminergic agonist called quinagolide decreased milk production, confirming the importance of PRL in the functioning of the mammary gland (Lacasse et al., 2011).
-
Saipin et al., 2020Effect of incubation temperature on lactogenic function of goat milk-derived mammary epithelial cellsIn Vitro Cellular & Developmental Biology, 2020
-
Henna et al., 2021Endocrine, energy, and lipid status during parturition and early lactation in indigenous goats native to the Algerian SaharaVeterinary World, 2021
-
Dee & Magee, 2018Anatomy and physiology of the mammary glandAnatomy and Physiology of Farm Animals, 2018
-
Lawhead & Baker, 2017The endocrine system/endocrine glandsIntroduction to Veterinary Science, 2017
-
Svennersten & Olsson, 2005Endocrinology of milk productionDomestic Animal Endocrinology, 2005
-
Lacasse et al., 2011Effect of the prolactin-release inhibitor quinagolide on lactating dairy cowsJournal of Dairy Science, 2011
III. Milk ejection
Milk from the cistern (70 % of the milk produced between each milking), can be extracted independent of hormonal processes, by a passive mechanism (only by gravity) (Menzies, 2021). For its part, alveolar milk ejection begins with afferent induction (Lérias et al., 2014), by sensory cells in the teat skin and udder base (Martínez & Suárez, 2018) and mechanical stimuli on the teat (Reece & Rowe, 2017b). It can also be triggered by visual stimuli e.g., milking from other females, auditory stimuli e.g., noise from buckets or the milking machine, olfactory stimuli e.g., the milk itself, and even become a conditioned reflex (Lawhead & Baker, 2017).
-
Menzies, 2021Udder health for dairy goatsVeterinary Clinics of North America: Food Animal Practice, 2021
-
Lérias et al., 2014The mammary gland in small ruminants: major morphological and functional events underlying milk production-a reviewJournal of Dairy Research, 2014
-
Martínez
& Suárez, 2018The mammary gland: morphology and development. Synthesis of milk componentsGoat dairying: production, management, health, quality of milk and products, 201
-
Reece & Rowe, 2017bLactation / functional anatomy of female mammary glands. Mammogenesis, lactogenesis and lactationFunctional Anatomy and Physiology of Domestic Animals, 2017
-
Lawhead & Baker, 2017The endocrine system/endocrine glandsIntroduction to Veterinary Science, 2017
The ejecto-lacteal reflex, becomes an electrical impulse that travels up the inguinal somatic nerves to the spinal cord (Figure 7), reaching the paraventricular nucleus of the hypothalamus (Davidson & Stabenfeldt, 2020), where action potentials are produced in intermittent pulses, releasing 9 aa OXT [peptide (cysteine-tyrosine-isoleucine-glutamineasparagine-asparagine-cysteine-proline-leucin e-glycine)] (Svennersten & Olsson, 2005) stored in the neurohypophysis (Dee & Magee, 2018).
-
Davidson & Stabenfeldt,
2020The mammary gland and lactationCunningham’s Textbook of Veterinary Physiology, 2020
-
Svennersten & Olsson,
2005Endocrinology of milk productionDomestic Animal Endocrinology, 2005
-
Dee & Magee, 2018Anatomy and physiology of the mammary glandAnatomy and Physiology of Farm Animals, 2018
Thumbnail
Figure 7
Mechanism of milk ejection and inhibition
Mechanism of milk ejection and inhibition
The electrical impulse that travels up the somatic inguinal nerves to the spinal cord also reaches the supraoptic nucleus of the hypothalamus (Reese et al., 2020),, where action potentials are produced in intermittent pulses, which release the antidiuretic hormone (ADH) also called vasopressin peptide of 9 aa (cysteine-tyrosine-tyrosine-phenylalanineglutamine-asparagine-asparagine-cysteine-proline-arginine-glycine) stored in the neurohypophysis (Thul et al., 2020).
-
Reese et al., 2020Mammary gland (mamma, uber, mastos)Veterinary Anatomy of Domestic Animals, 2020
-
Thul et al., 2020Oxytocin and postpartum depression: A systematic reviewPsychoneuroendocrinology, 2020
Both hormones travel via the external pudendal artery to the mammary gland (Davidson & Stabenfeldt, 2020). Main functions of OXT in the mammary gland are: i) to cause contraction of the myoepithelial cells surrounding the alveoli, to empty milk from the lactocytes into the alveolar lumen (Belo & Bruckmaier, 2010), and ii) to cause contraction of the intra- and inter-lobular milk ducts, forcing the flow of milk into the cistern of the gland (Neville et al., 2002; Svennersten & Olsson, 2005).
-
Davidson & Stabenfeldt, 2020The mammary gland and lactationCunningham’s Textbook of Veterinary Physiology, 2020
-
Belo & Bruckmaier, 2010Suitability of low-dosage oxytocin treatment to induce milk ejection in dairy cowsJournal of Dairy Science, 2010
-
Neville et al., 2002Hormonal regulation of mammary differentiation and milk secretionJournal of Mammary Gland Biology and Neoplasia, 2002
-
Svennersten & Olsson, 2005Endocrinology of milk productionDomestic Animal Endocrinology, 2005
For its part, ADH acts on vascular smooth muscle causing vasoconstriction and osmotic and oncotic pressure changes in the lactiferous ducts facilitating milk outflow (Goff, 2015). The increase in pressure within the mammary gland is evident at the minute of the ejection reflex, as milk is expelled from the alveoli and lactiferous ducts due to contraction of the myoepithelial cells (Lérias et al., 2014). The term used in mammals to describe this phenomenon is milk "let-down" (Davidson & Stabenfeldt, 2020).
-
Goff, 2015Endocrinology, reproduction, and lactation/The mammary gland and lactationDukes' Physiology of Domestic Animals, 2015
-
Lérias et al., 2014The mammary gland in small ruminants: major morphological and functional events underlying milk production-a reviewJournal of Dairy Research, 2014
-
Davidson & Stabenfeldt, 2020The mammary gland and lactationCunningham’s Textbook of Veterinary Physiology, 2020
Milk flow increases the gland cistern size, originating an increase in pressure (Lawhead & Baker, 2017). Thus, the ejection rate presents an autocrine control at the glandular level by lactation inhibitory factor (Dee & Magee, 2018). This protein is produced by the same lactocytes of the glandular parenchyma and it is secreted together with the milk into the lacteal alveoli (Davidson & Stabenfeldt, 2020). In this regard Bruckmaier & Wellnitz (2008) indicated that lactation inhibitory factor exhibits two modes of action: i) it accumulates in milk until it reaches a critical concentration that inhibits ejection, and ii) when milk accumulates within a lacteal alveolus, it extends its surface exposing potential receptors for lactation inhibitory factor, allowing its binding and triggering inhibition.
-
Lawhead & Baker, 2017The endocrine system/endocrine glandsIntroduction to Veterinary Science, 2017
-
Dee & Magee, 2018Anatomy and physiology of the mammary glandAnatomy and Physiology of Farm Animals, 2018
-
Davidson & Stabenfeldt, 2020The mammary gland and lactationCunningham’s Textbook of Veterinary Physiology, 2020
-
Bruckmaier & Wellnitz (2008Induction of milk ejection and milk removal in different production systemsJournal of Animal Science, 2008
Simultaneously, the ejecto-lacteal reflex can be temporarily inhibited by the release into the bloodstream of adrenaline also called epinephrine (Svennersten & Olsson, 2005) (Figure 7), as a result of increased pressure generating stress (Reese et al., 2020). Adrenaline constricts blood vessels including the external pudendal artery, making it impossible for OXT to reach the myoepithelial cells surrounding the lacteal alveoli and indirectly inhibiting their contraction (Reece & Rowe, 2017b).
-
Svennersten & Olsson, 2005Endocrinology of milk productionDomestic Animal Endocrinology, 2005
-
Reese et al.,
2020Mammary gland (mamma, uber, mastos)Veterinary Anatomy of Domestic Animals, 2020
-
Reece & Rowe,
2017bLactation / functional anatomy of female mammary glands. Mammogenesis, lactogenesis and lactationFunctional Anatomy and Physiology of Domestic Animals, 2017
The maintenance period, or galactopoiesis, occurs when constant suckling at the teat continues to stimulate milk production (Bruckmaier & Wellnitz, 2008), main hormones controlling this physiological stage are PRL and GH (Lawhead & Baker, 2017). Both hormones are important for galactopoiesis, but one predominates in importance relative to the other depending on the species (Baljit, 2017). In rodents as in humans PRL is more important and in ruminants GH has a more active participation (Goff, 2015).
-
Bruckmaier & Wellnitz, 2008Induction of milk ejection and milk removal in different production systemsJournal of Animal Science, 2008
-
Lawhead & Baker, 2017The endocrine system/endocrine glandsIntroduction to Veterinary Science, 2017
-
Baljit, 2017Ruminants / the udderDyce, Sack and Wensing's Textbook of Veterinary Anatomy, 2017
-
Goff, 2015Endocrinology, reproduction, and lactation/The mammary gland and lactationDukes' Physiology of Domestic Animals, 2015
IV. Milk lipogenesis
Milk lipogenesis takes place in different cellular compartments (Gartner, 2018). It starts in the mitochondria of lactocytes (Friedman & Nunnari, 2014), with the production of acetylCoA from fatty acid oxidation (Nelson & Cox, 2017b), pyruvate (C 3 H 3 O 3) oxidation (McDonald et al., 2011) and catabolism of aa carbonaceous skeletons (Rodwell, 2018) (Figure 8).
-
Gartner, 2018The cell and the organellesColor atlas and text of histology, 2018
-
Friedman & Nunnari, 2014Mitochondrial form and functionNature, 2014
-
Nelson & Cox, 2017Protein metabolismLehninger. Principles of Biochemistry, 2017
-
McDonald et al., 2011LipidsAnimal Nutrition, 2011
-
Rodwell, 2018Catabolism of proteins and amino acid nitrogenHarper’s Illustrated Biochemistry, 2018
Thumbnail
Figure 8
Subcellular localization of lipid metabolism
Subcellular localization of lipid metabolism
Like other metabolic pathways, fatty acid synthesis is endergonic and reductive (Botham & Mayes, 2018b). Therefore, the process uses adenosine triphosphate (ATP as an energy source (Botham & Mayes, 2018a) and nicotinamide adenine dinucleotide phosphate (NADPH) as a reduced electron carrier (Madigan et al., 2019a). Consequently, fatty acid synthesis continues in the cytoplasm (Appleton et al., 2013d), where NADPH is available for reductive synthesis [i.e., where the (NADPH)/(NADP+) ratio is high] (Cooper, 2019a). However, the inner mitochondrial membrane is impermeable to the passage of acetyl-CoA (Ellis et al., 2015; Mas, 2018), so a shuttle for transfer of acetyl-CoA (acetyl groups) from the mitochondrial matrix to the cytoplasm is required (Nunes et al., 2013) (Figure 9).
-
Botham & Mayes, 2018bLipids of physiological significanceHarper’s Illustrated Biochemistry, 2018
-
Botham & Mayes, 2018aBioenergetics: the role of ATPHarper’s Illustrated Biochemistry, 2018
-
Madigan et
al., 2019aProtein synthesis: translationBrock Biology of Microorganisms, 2019
-
Appleton
et al., 2013dTransporte y metabolismo de los lípidosLo Esencial en Metabolismo y Nutrición, 2013
-
Cooper, 2019aEukaryotic RNA polymerases and general transcription factorsThe Cell: A Molecular Approach, 2019
-
Ellis et al., 2015Metabolic and tissue-specific regulation of acyl-CoA metabolismPLOS ONE, 2015
-
Mas, 2018Metabolismo de los lípidosBioquímica de Laguna y Piña, 2018
-
Nunes et
al., 2013Regulation of the mitochondrial tricarboxylic acid cycleCurrent Opinion in Plant Biology, 2013
Thumbnail
Figure 9
Shuttle for acetyl group transfer
Shuttle for acetyl group transfer
Intra-mitochondrial acetyl-CoA first reacts with oxaloacetate to form citrate (Nelson & Cox, 2017a), in the citric acid cycle reaction catalyzed by citrate synthase (Appleton et al., 2013b). Citrate passes through the mitochondrial membrane on its transporter (Nunes et al., 2013). In the cytoplasm, citrate cleavage catalyzed by citrate lyase regenerates acetylCoA and oxaloacetate in an ATP-dependent reaction (Ellis et al., 2015; Verschueren et al., 2019). Oxaloacetate cannot return directly to the mitochondrial matrix, as there is no transporter for it (Nelson & Cox, 2017a). So, malate dehydrogenase catalyzes its reduction to malate, and this passes through the mitochondrial membrane on its transporter (Nunes et al., 2013).
-
Nelson & Cox, 2017aProtein metabolismLehninger. Principles of Biochemistry, 2017
-
Appleton et al., 2013bTransporte y metabolismo de los lípidosLo Esencial en Metabolismo y Nutrición, 2013
-
Nunes et al., 2013Regulation of the mitochondrial tricarboxylic acid cycleCurrent Opinion in Plant Biology, 2013
-
Ellis et al., 2015Metabolic and tissue-specific regulation of acyl-CoA metabolismPLOS ONE, 2015
-
Verschueren et al., 2019Structure of ATP citrate lyase and the origin of citrate synthase in the Krebs cycleNature, 2019
-
Nelson & Cox,
2017aProtein metabolismLehninger. Principles of Biochemistry, 2017
-
Nunes et al.,
2013Regulation of the mitochondrial tricarboxylic acid cycleCurrent Opinion in Plant Biology, 2013
In the mitochondrial matrix, malate is re-oxidized to oxaloacetate catalyzed by malate dehydrogenase to complete the shuttle (Friedman & Nunnari, 2014). The pyruvate produced is sent to the mitochondrion by its transporter, and then converted back to oxaloacetate catalyzed by pyruvate carboxylase (Nelson & Cox, 2017a). In the resulting cycle, two molecules of ATP are consumed (by citrate lyase and pyruvate carboxylase) for every molecule of acetyl-CoA supplied for lactate lipogenesis (Appleton et al., 2013c).
-
Friedman & Nunnari, 2014Mitochondrial form and functionNature, 2014
-
Nelson & Cox, 2017aProtein metabolismLehninger. Principles of Biochemistry, 2017
-
Appleton et al., 2013cTransporte y metabolismo de los lípidosLo Esencial en Metabolismo y Nutrición, 2013
In the cytoplasm, fatty acid biosynthesis begins with the participation of a three-carbon intermediate called malonyl-CoA (Mas, 2018) (Figure 10).
-
Mas, 2018Metabolismo de los lípidosBioquímica de Laguna y Piña, 2018
Thumbnail
Figure 10
Malonyl-CoA
Malonyl-CoA
The formation of malonyl-CoA is from acetyl-CoA in an irreversible process catalyzed by biotin carboxylase (Nunes et al., 2013). In this two-step reaction a carboxyl group derived from the hydrogenocarbonate anion (HCO 3 -), binds to a nitrogen of the biotin ring in an ATP-dependent reaction (Botham & Mayes, 2018a), activating carbon dioxide (CO 2) (Mas, 2018). The biotinyl group serves as a temporary CO2 carrier (Nelson & Cox, 2017a), and part of the transporter protein and the long flexible biotin arm turn to transport activated CO2 from biotin to acetyl-CoA producing malonyl-CoA (Cooper, 2019a).
-
Nunes et al., 2013Regulation of the mitochondrial tricarboxylic acid cycleCurrent Opinion in Plant Biology, 2013
-
Botham & Mayes, 2018aLipids of physiological significanceHarper’s Illustrated Biochemistry, 2018
-
Mas, 2018Metabolismo de los lípidosBioquímica de Laguna y Piña, 2018
-
Nelson & Cox, 2017Protein metabolismLehninger. Principles of Biochemistry, 2017
-
Cooper, 2019aEukaryotic RNA polymerases and general transcription factorsThe Cell: A Molecular Approach, 2019
From malonyl-CoA lipogenesis is performed by the fatty acid synthase I (FAS I) protein complex (Suburu et al., 2014). This system performs synthesis, reduction, dehydration and again reduction by concentrating malonyl-CoA groups with acetyl-CoA, with loss of CO2 at each step (Belew et al., 2019). After each two-carbon addition, reductions convert the growing chain to a four-carbon fatty acid, then six, then eight carbons, and so on (Song et al., 2018) (Figure 11). Fatty acid synthesis in FAS I always reaches 16 carbons (palmitic, C16:0) and no intermediates are released (Chandel, 2021).
-
Suburu et al., 2014Fatty acid synthase is required for mammary gland development and milk production during lactationAmerican Journal of Physiology-Endocrinology and Metabolism, 2014
-
Belew et al., 2019Transfer of glucose hydrogens via acetyl-CoA, malonyl-CoA, and NADPH to fatty acids during de novo lipogenesisJournal of Lipid Research, 2019
-
Song et al., 2018Regulation and metabolic significance of de novo lipogenesis in adipose tissuesNutrients, 2018
-
Chandel, 2021Lipid metabolismCold Spring Harbor Perspectives in Biology, 2021
Thumbnail
Figure 11
General process for the synthesis of palmitic acid
General process for the synthesis of palmitic acid
Palmitic, leaves FAS I and can enter the endoplasmic reticulum (Olarte et al., 2020), to be elongated (coupling new carbons to lengthen the chain) (Balla et al., 2019) and desaturated (introducing cis double bonds between its carbons) to form polyunsaturated fatty acids (Rowland & Voeltz, 2012). At this time the mammary gland parenchyma undergoes a functional and morphological differentiation termed lactogenesis I (Reese et al., 2020; Menzies, 2021), and initiates the accumulation of lipid microdroplets (Ashdown & Done, 2011). These droplets are obtained from NEFA, released from circulating lipoproteins by lipoprotein lipase located in the vascular bed of the mammary gland (Davidson & Stabenfeldt, 2020) or by circulating fatty acids derived from adipose tissue bound to ALB (Fox et al., 2015).
-
Olarte et al., 2020Determinants of endoplasmic reticulum-to-lipid droplet protein targetingDevelopmental Cell, 2020
-
Balla et al., 2019Lipid dynamics at contact sites between the endoplasmic reticulum and other organellesAnnual Review of Cell and Developmental Biology, 2019
-
Rowland & Voeltz, 2012Endoplasmic reticulum-mitochondria contacts: function of the junctionNature Reviews Molecular Cell, 2012
-
Reese et al., 2020Mammary gland (mamma, uber, mastos)Veterinary Anatomy of Domestic Animals, 2020
-
Menzies, 2021Udder health for dairy goatsVeterinary Clinics of North America: Food Animal Practice, 2021
-
Ashdown & Done, 2011La ubreAtlas en color de anatomía veterinaria. Rumiantes, 2011
-
Davidson & Stabenfeldt, 2020The mammary gland and lactationCunningham’s Textbook of Veterinary Physiology, 2020
-
Fox et al., 2015Structure and development of mammary tissueDairy Chemistry and Biochemistry, 2015
After parturition lactogenesis II begins (Baljit, 2017), this process requires: i) coordination and activation of fatty acids by acyl-CoA synthetases (Fernandez & Ellis, 2020), ii) de novo synthesis of medium-chain fatty acids from GLU (Jones, 2016; Cooper, 2019a), and iii) synthesis of neutral lipids e.g., triacylglycerol (TAG), COL and diacylglycerol (DAG) esters (Sanhueza et al., 2012; Nelson & Cox, 2017b), which provide binding for accessory proteins (Reece & Rowe, 2017b), e.g., CTP:phosphocholine cytidylyl transferase (CCT) and perilipins (Plin) (Henry et al., 2015).
-
Baljit, 2017Ruminants / the udderDyce, Sack and Wensing's Textbook of Veterinary Anatomy, 2017
-
Fernandez & Ellis, 2020Acyl-CoA synthetases as regulators of brain phospholipid acyl-chain diversityProstaglandins Leukot Essent Fatty Acids, 2020
-
Jones, 2016Hepatic glucose and lipid metabolismDiabetologia, 2016
-
Cooper, 2019Eukaryotic RNA polymerases and general transcription factorsThe Cell: A Molecular Approach, 2019
-
Sanhueza et al., 2012Cholesterol metabolism: increasingly complexGrasas y Aceites, 2012
-
Nelson & Cox, 2017Protein metabolismLehninger. Principles of Biochemistry, 2017
-
Reece & Rowe, 2017Lactation / functional anatomy of female mammary glands. Mammogenesis, lactogenesis and lactationFunctional Anatomy and Physiology of Domestic Animals, 2017
-
Henry et al., 2015Phosphoproteomics of the goat milk fat globule membrane: New insights into lipid droplet secretion from the mammary epithelial cellProteomics, 2015
In general, milk fat globules are thought to form from tubular microdomains of the smooth endoplasmic reticulum (Pol et al., 2014). However, unlike other lipogenic cells e.g., hepatocytes and adrenocortical cells (Saheki & De Camilli, 2017), mammary gland parenchyma is highly enriched in rough endoplasmic reticulum with enzymes for neutral lipid synthesis (Sturley & Hussain, 2012).
-
Pol et al., 2014Review: biogenesis of the multifunctional lipid droplet: lipids, proteins and sitesJournal of Cell Biology, 2014
-
Saheki & De Camilli, 2017Endoplasmic Reticulum-Plasma Membrane Contact SitesAnnual Review of Biochemistry, 2017
-
Sturley & Hussain, 2012Lipid droplet formation on opposing sides of the endoplasmic reticulumJournal of Lipid Research, 2012
This process begins with the esterification of fatty acids to a glycerol molecule to form TAG (Tortora et al., 2019), in four reactions catalyzed by members of glycerol-3phosphate acyltransferase (GPAT), phosphatidic acid phosphatase (PAP) and acylCoA:diacylglycerol acyltransferase (DGAT) enzyme families (Monks et al., 2020). Monks et al., 2020). The final step in this pathway is the esterification of DAG to TAG (Chandel, 2021). In turn, the synthesis of COL esters is mediated by acyl-CoA:cholesterol acyltransferase (ACAT) (Sanhueza et al., 2012).
-
Tortora et al., 2019Organic compounds. Structure and chemistry: carbohydrates, lipids, proteins, nucleic acidsMicrobiology: An Introduction, 2019
-
Monks et al., 2020Organellar contacts of milk lipid dropletsThousand Oaks Journal, 2020
-
Chandel, 2021Lipid metabolismCold Spring Harbor Perspectives in Biology, 2021
-
Sanhueza et al., 2012Cholesterol metabolism: increasingly complexGrasas y Aceites, 2012
Once these elements are structured, they are incorporated into the fat globule (Figure 12), together with carotenoids, fat-soluble vitamins and phosphatidylcholine (Mas, 2018). Two main pathways contribute to phosphatidylcholine synthesis: i) the Kennedy pathway for de novo synthesis of phospholipids, a reaction catalyzed by CCT and ii) the Lands cycle (Appleton et al., 2013d; John et al., 2022). In the latter, the remodeling of phospholipids in the fat globule membrane takes place in deacylation/reacylation reactions (Henry et al., 2015; Guoyao, 2017a), catalyzed by phospholipase A (PLA) and lysophospholipid acyltransferase (LFLAT) (Seoane et al., 2018). Botham & Mayes (2018b) established that, thanks to these phospholipids, the apolar tails project to glycerides and the polar heads project to water.
-
Mas, 2018Metabolismo de los lípidosBioquímica de Laguna y Piña, 2018
-
Appleton et al., 2013Transporte y metabolismo de los lípidosLo Esencial en Metabolismo y Nutrición, 2013
-
ohn et al., 2022Rewiring phospholipid biosynthesis reveals resilience to membrane perturbations and uncovers regulators of lipid homeostasisJournal of the European Molecular Biology Organization, 2022
-
Henry et al., 2015Phosphoproteomics of the goat milk fat globule membrane: New insights into lipid droplet secretion from the mammary epithelial cellProteomics, 2015
-
Guoyao, 2017Nutrition and metabolism of protein and amino acidsPrinciples of Animal Nutrition, 2017
-
Seoane et al., 2018Biomimetic generation and remodeling of phospholipid membranes by dynamic imine chemistryJournal of the American Chemical Society, 2018
-
Botham & Mayes (2018Lipids of physiological significanceHarper’s Illustrated Biochemistry, 2018
Thumbnail
Figure 12
Electron micrograph of a milk fat globule (asterisk) attached to the apical plasma membrane; casein micelles (red)
Electron micrograph of a milk fat globule (asterisk) attached to the apical plasma membrane; casein micelles (red)
Regulation of lipolysis appears to be the main function of the five Plin proteins (Lundquist et al., 2020), which prevent access of lipases to the fat globule (Zhang & Liu, 2019). Unlike lipoproteins, milk lipids are not packaged into vesicles in the Golgi apparatus (Wilson et al., 2011), nor are they secreted by an exocytic mechanism (Lowe, 2011). Instead, they advance unidirectionally toward the apical pole of the lactocyte (Davidson & Stabenfeldt, 2020) and once there, they pass into the alveolar lumen via an apocrine mechanism (Figure 12), in the form of Plin-coated milk fat globules (Lundquist et al., 2020), to continue their transit into the intralobulillar ducts and flow into a central collecting space (Davidson & Stabenfeldt, 2020).
-
Lundquist et al., 2020Lipid droplets throughout the evolutionary treeProgress in Lipid Research, 2020
-
Zhang & Liu, 2019The new face of the lipid droplet: lipid droplet proteinsProteomics, 2019
-
Wilson et al., 2011The Golgi apparatus: an organelle with multiple complex functionsBiochemical Journal, 2011
-
Lowe, 2011Structural organization of the Golgi apparatusCurrent Opinion in Cell Biology, 2011
-
Davidson & Stabenfeldt, 2020The mammary gland and lactationCunningham’s Textbook of Veterinary Physiology, 2020
V. Milk proteogenesis
Usually a portion of the dietary protein resists bacterial proteolysis in the rumen and passes into the abomasum without being catabolized (Appleton et al., 2013a), along with ruminal bacteria attached to the fermented feed (Guoyao, 2017b). Pancreatic acinar cells translate hydrolases e.g., peptidase or protease, aminotransferase and nuclease (Philipps, 2018). In the duodenum, these enzymes soak the food bolus and its proteins lose their peptide bonds by hydrolysis (Lozano et al., 2005). This process, releases the aa from their polymeric structure to be absorbed at the intestinal level (Piña & Flores, 2018), transported to the liver and redirected to the cytoplasm of lactocytes (Ahern, 2019).
-
Appleton et al., 2013aTransporte y metabolismo de los lípidosLo Esencial en Metabolismo y Nutrición, 2013
-
Guoyao, 2017Nutrition and metabolism of protein and amino acidsPrinciples of Animal Nutrition, 2017
-
Philipps, 2018Proteases-general aspectsEnzymes in Human and Animal Nutrition: Principles and Perspectives, 2018
-
Lozano et al., 2005Metabolismo nitrogenadoBioquímica y Biología Molecular para Ciencias de la Salud, 2005
-
Piña & Flores, 2018Metabolismo de los compuestos nitrogenadosBioquímica de Laguna y Piña, 2018
-
Ahern, 2019Amino acids: 20 building blocks of lifeBiochemistry and Molecular Biology: How Life Works, 2019
Milk proteogenesis begins in the lactocyte nucleus with transfer ribonucleic acid (tRNA) transcription (Madigan et al., 2019b). RNA polymerase carries out the transcription of messenger ribonucleic acid (mRNA) (Cooper, 2019b), starting from a segment of deoxyribonucleic acid (DNA) that serves as a template (Singh & Rajeev, 2020). This DNA segment contains exons (coding regions) and introns (non-coding regions) (Nelson & Cox, 2017c). Before leaving the nucleus, segments corresponding to introns are cut (Weil, 2018b) and segments corresponding to coding exons are spliced (Madigan et al., 2019b).
-
Madigan et al., 2019Protein synthesis: translationBrock Biology of Microorganisms, 2019
-
Cooper, 2019Eukaryotic RNA polymerases and general transcription factorsThe Cell: A Molecular Approach, 2019
-
Singh & Rajeev, 2020DNA: an important component of lifeBiochemistry, Biophysics, and Molecular Chemistry: Applied Research and Interactions, 2020
-
Nelson & Cox, 2017cProtein metabolismLehninger. Principles of Biochemistry, 2017
-
Weil, 2018RNA synthesis, processing and modificationHarper’s Illustrated Biochemistry, 2018
Next, tRNA and mRNA exit the nucleus and enter the cytoplasm (Weil, 2018a). At this point, protein translation is promoted on ribosomes with three main steps: i) initiation, ii) elongation and iii) termination (Nelson & Cox, 2017c) (Figure 13). 2
-
Weil, 2018RNA synthesis, processing and modificationHarper’s Illustrated Biochemistry, 2018
-
Nelson & Cox, 2017Protein metabolismLehninger. Principles of Biochemistry, 2017
Thumbnail
Figure 13
Proteogenesis, transcription and protein translation
Proteogenesis, transcription and protein translation
tRNA transports aa from the cytoplasm to the ribosomes (Weil, 2018b) and to ensure that the tRNA carries the correct aa, each tRNA contains a specific sequence of three nitrogenous bases called an anticodon (Lozano et al., 2005). In the case of mRNA its specific sequence of three nitrogenous bases is called a codon (Angov, 2011). Ribosomes contained in the lactocytes of the glandular parenchyma are the organelles responsible for the translation of different types of caseins e.g., αS1-CN, αS2-CN, β-CN, and κ-CN (Doherty & Doudna, 2000). A lactocyte can have thousands of ribosomes (Ingolia, 2014), and their number increases with the proliferation of the glandular parenchyma during early lactation (1/3 lactation) (Reese et al., 2020) and their involution begins during late lactation (3/3 lactation) along with the decrease in milk (Davidson & Stabenfeldt, 2020).
-
Weil, 2018bRNA synthesis, processing and modificationHarper’s Illustrated Biochemistry, 2018
-
Lozano et al.,
2005Metabolismo nitrogenadoBioquímica y Biología Molecular para Ciencias de la Salud, 2005
-
Angov,
2011Codon usage: nature's roadmap to expression and folding of proteinsBiotechnology Journal, 2011
-
Doherty & Doudna, 2000Ribozyme structures and mechanismsAnnual Review of Biochemistry, 2000
-
Ingolia,
2014Ribosome profiling: new views of translation, from single codons to genome scaleNature Reviews Genetics, 2014
-
Reese et al., 2020Mammary gland (mamma, uber, mastos)Veterinary Anatomy of Domestic Animals, 2020
-
Davidson &
Stabenfeldt, 2020The mammary gland and lactationCunningham’s Textbook of Veterinary Physiology, 2020
Each ribosome consists of two subunits the peptidyl (P) site and the aminoacyl (A) site (Ingolia, 2014), which associate for codon-anticodon base pairing (Weil, 2018a) and dissociate during translation termination (Swaisgood, 2003). The A site is where the first aa-loaded tRNA is docked (Madigan et al., 2019b). P site is where the growing polypeptide chain binds to the preceding tRNA (Piña & Flores, 2018). During peptide bond formation (O=C-N-H) the growing chain moves toward the tRNA at the A site (Nelson & Cox, 2017c). After elongation the tRNA containing the polypeptide translocates from the A site to the P site (Madigan et al., 2019b), thus freeing the A site for a new aa-loaded tRNA (Lozano et al., 2005) (Figure 13). In each translocation, the ribosome advances three nucleotides (one codon) along the mRNA (Appleton et al., 2013a), exposing a new codon at the A site (Weil, 2018a). Accuracy in translocation is essential for the accuracy of dairy proteogenesis (Weil, 2018a). That is, the ribosome must move exactly one codon at each step or the fidelity of translation would be compromised (Angov, 2011).
-
Ingolia,
2014Ribosome profiling: new views of translation, from single codons to genome scaleNature Reviews Genetics, 2014
-
Weil, 2018aRNA synthesis, processing and modificationHarper’s Illustrated Biochemistry, 2018
-
Swaisgood,
2003Protein composition of milk: identification, structure, and chemical compositionAdvanced dairy chemistry: proteins part A, 2003
-
Madigan et al.,
2019bProtein synthesis: translationBrock Biology of Microorganisms, 2019
-
Piña & Flores,
2018Metabolismo de los compuestos nitrogenadosBioquímica de Laguna y Piña, 2018
-
Nelson & Cox, 2017cProtein metabolismLehninger. Principles of Biochemistry, 2017
-
Madigan et al.,
2019bProtein synthesis: translationBrock Biology of Microorganisms, 2019
-
Lozano et al., 2005Metabolismo nitrogenadoBioquímica y Biología Molecular para Ciencias de la Salud, 2005
-
Appleton et al.,
2013aTransporte y metabolismo de los lípidosLo Esencial en Metabolismo y Nutrición, 2013
-
Weil, 2018aRNA synthesis, processing and modificationHarper’s Illustrated Biochemistry, 2018
-
Weil, 2018aRNA synthesis, processing and modificationHarper’s Illustrated Biochemistry, 2018
-
Angov, 2011Codon usage: nature's roadmap to expression and folding of proteinsBiotechnology Journal, 2011
Protein translation ends when the ribosome reaches a termination codon e.g. uraciladenine-guanine (UAG), uracil-adenine-adenine (UAA) and uracil-guanine-adenine (UGA) (Weil, 2018a), as no tRNA binds to a codon of these (Piña & Flores, 2018). Instead, release factors recognize UAG, UAA and UGA (Nelson & Cox, 2017c) and cleave the attached polypeptide from the final tRNA (Lozano et al., 2005), releasing the finished caseins (Davidson & Stabenfeldt, 2020). mRNA is then released and can be re-read by other ribosomes (Madigan et al., 2019b), and the ribosomal subunits dissociate and become free to form new initiation complexes and repeat the process (Pacheco et al., 2021). Finally, in the Golgi apparatus, glycosylation of caseins (binding with lactose) takes place (Wilson et al., 2011) and during their movement through the cytoplasm tricalcium phosphate [Ca 3 (PO 4 )2] and other ions are coupled to form a structure called a micelle (Reese et al., 2020), which will be exported to the alveolar lumen (Dee & Magee, 2018), to continue its transit to the intralobulillar ducts and flow into a central collecting space (Davidson & Stabenfeldt, 2020).
-
Weil, 2018RNA synthesis, processing and modificationHarper’s Illustrated Biochemistry, 2018
-
Piña & Flores,
2018Metabolismo de los compuestos nitrogenadosBioquímica de Laguna y Piña, 2018
-
Nelson & Cox, 2017cProtein metabolismLehninger. Principles of Biochemistry, 2017
-
Lozano et al., 2005Metabolismo nitrogenadoBioquímica y Biología Molecular para Ciencias de la Salud, 2005
-
Davidson & Stabenfeldt,
2020The mammary gland and lactationCunningham’s Textbook of Veterinary Physiology, 2020
-
Madigan et al.,
2019bProtein synthesis: translationBrock Biology of Microorganisms, 2019
-
Pacheco et al., 2021Biochemistry and metabolic pathways of polysaccharides, lipids, and proteinsAbanico Veterinario, 2021
-
Wilson et al.,
2011The Golgi apparatus: an organelle with multiple complex functionsBiochemical Journal, 2011
-
Reese et al.,
2020Mammary gland (mamma, uber, mastos)Veterinary Anatomy of Domestic Animals, 2020
-
Dee & Magee, 2018Anatomy and physiology of the mammary glandAnatomy and Physiology of Farm Animals, 2018
-
Davidson & Stabenfeldt, 2020The mammary gland and lactationCunningham’s Textbook of Veterinary Physiology, 2020
The protein fraction is divided into 20 % for soluble or serum proteins where the following stand out: i) β-lactoglobulin (retinol and fatty acid binding and possible antioxidant) (McKerchar et al., 2023), ii) α-lactoalbumin (lactose production, calcium transport, immunomodulatory and anticancer) (Diao et al., 2022), iii) immunoglobulins IgA, IgM and IgG (immune protection) (Nayik et al., 2022) iv) lactoferrin (antibacterial, antioxidant, immunomodulatory, iron absorption and anticarcinogenic) (Sansi et al., 2022), and v) lactoperoxidase (antibacterial) (Lérias et al., 2014). All soluble proteins present a higher proportion of leucine, isoleucine and valine (Ahern, 2019).
-
McKerchar et al., 2023The protein dynamics of bovine and caprine β-lactoglobulin differ as a function of pHFood Chemistry, 2023
-
Diao et al., 2022Complexation of ellagic acid with αlactalbumin and its antioxidant propertyFood Chemistry, 2022
-
Nayik et al., 2022Nutritional profile, processing and potential products: A comparative review of goat milkDairy, 2022
-
Sansi et al., 2022Antimicrobial bioactive peptides from goat Milk proteins: In silico prediction and analysisJournal of Food Biochemistry, 2022
-
Lérias et al., 2014The mammary gland in small ruminants: major morphological and functional events underlying milk production-a reviewJournal of Dairy Research, 2014
-
Ahern, 2019Amino acids: 20 building blocks of lifeBiochemistry and Molecular Biology: How Life Works, 2019
Eighty percent of the protein fraction corresponds to insoluble proteins or caseins whose function is to transport and bind minerals, mainly calcium and phosphorus (Dhasmana et al., 2022). The concentration of αs1-casein and αs2-casein is lower in goat milk than in cow milk, the fraction of β-casein is higher and the amount of and κ-casein is equal to that of cow milk (Saikia et al., 2022). The αs-casein is the main protein found in cow's milk, whereas the main protein factor found in goat's milk is β-casein (Dhasmana et al., 2022). All insoluble proteins have a higher proportion of histidine, methionine and phenylalanine (Ahern, 2019).
-
Dhasmana et al., 2022Potential nutraceuticals from the casein fraction of goat's milkJournal of Food Biochemistry, 2022
-
Saikia et al., 2022Goat milk and its nutraceutical propertiesInternational Journal of Applied Research, 2022
-
Dhasmana et al., 2022Potential nutraceuticals from the casein fraction of goat's milkJournal of Food Biochemistry, 2022
-
Ahern, 2019Amino acids: 20 building blocks of lifeBiochemistry and Molecular Biology: How Life Works, 2019
CONCLUSIONS
In recent years, the shift in milk marketing towards a standardized price structure based on lipid and protein concentration requires a better understanding of the anatomical and physiological processes occurring in the mammary gland. Goats appear to be the least affected species with respect to emotional stress and milk ejection. Because of the morphological and physiological characteristics of the goat mammary gland, 70 % of the milk produced between milkings can be extracted independently of hormonal processes. The development of the mammary gland through its four stages: i) mammogenesis, ii) lactogenesis, iii) galactopoiesis, and iv) involution, determines all aspects of the behavior of the lacteal glandular parenchyma. Lactocytes possess a high metabolic complexity and a robust microscopic organization to develop their systems of extraction, utilization of polysaccharides, lipids and proteins and transformation into milk fat and protein. Knowledge of the anabolic and catabolic processes of these molecules will allow understanding the basic biochemistry of milk production.
ACKNOWLEDGMENTS
This work was supported by the National Council of Science and Technology (CONACyTMexico) and the project: Relationship between blood biochemical analytes and milk fat/protein in goat and cow (University of Colima)
LITERATURA CITADA
- Ahern K. 2019. Amino acids: 20 building blocks of life. In: Ahern K, Biochemistry and Molecular Biology: How Life Works. Virginia, United States: The Teaching Company. Pp. 29-40. ISBN: 978-1-25-983793-7. https://www.thegreatcourses.com/courses/biochemistry-and-molecular-biology-how-lifeworks Links
- Angov E. 2011. Codon usage: nature's roadmap to expression and folding of proteins. Biotechnology Journal. 6(6):650-659. ISSN: 1860-7314. https://doi.org/10.1002/biot.201000332 Links
- Appleton A, Vanbergen O, Dominiczak MH. 2013a. Metabolismo de las proteínas. En: Horton-Szar D, Lo Esencial en Metabolismo y Nutrición. Barcelona, España: Elsevier Health Sciences. Pp. 71-82. ISBN: 978-0-7234-3626-3. https://www.elsevier.com/books/lo-esencial-en-metabolismo-y-nutricion/978-84-9113537-1 Links
- Appleton A, Vanbergen O, Dominiczak MH. 2013b. Metabolismo energético I: ciclo ATC. En: Horton-Szar D, Lo Esencial en Metabolismo y Nutrición. Barcelona, España: Elsevier Health Sciences. Pp. 13-17. ISBN: 978-0-7234-3763-5. https://www.elsevier.com/books/lo-esencial-en-metabolismo-y-nutricion/978-84-9113537-1 Links
- Appleton A, Vanbergen O, Dominiczak MH. 2013c. Metabolismo energético II: generación de ATP. En: Horton-Szar D, Lo Esencial en Metabolismo y Nutrición. Barcelona, España: Elsevier Health Sciences. Pp. 17-23. ISBN: 978-0-7234-3763-5. https://www.elsevier.com/books/lo-esencial-en-metabolismo-y-nutricion/978-84-9113537-1 Links
- Appleton A, Vanbergen O, Dominiczak MH. 2013d. Transporte y metabolismo de los lípidos. En: Horton-Szar D, Lo Esencial en Metabolismo y Nutrición. Barcelona, España: Elsevier Health Sciences. Pp. 45-70p. ISBN: 978-0-7234-3626-3. https://www.elsevier.com/books/lo-esencial-en-metabolismo-y-nutricion/978-84-9113537-1 Links
- Ashdown RR, Done SH. 2011. La ubre. En: Ashdown RR, Atlas en color de anatomía veterinaria. Rumiantes. Barcelona, España: Elsevier Health Science Division. Pp. 219-230. ISBN: 9788480865418. https://catalogo.udes.edu.co/cgi-bin/koha/opacdetail.pl?biblionumber=31192 Links
- Baljit S. 2017. Ruminants / the udder. In: Baljit S, Dyce, Sack and Wensing's Textbook of Veterinary Anatomy. New York, EE. UU.: Elsevier Health Science Division. Pp. 1252-1264. ISBN: 978-0323442640. https://www.elsevier.com/books/dyce-sack-andwensings-textbook-of-veterinary-anatomy/singh/978-0-323-44264-0 Links
- Balla T, Kim YJ, Alvarez-Prats A, Pemberton J. 2019. Lipid dynamics at contact sites between the endoplasmic reticulum and other organelles. Annual Review of Cell and Developmental Biology. 35(1):85-109. ISSN: 1530-8995. https://doi.org/10.1146/annurevcellbio-100818-125251 Links
- Bauman DE, Mather IH, Wall RJ, Lock AL. 2006. Major advances associated with the biosynthesis of milk. Journal of Dairy Science. 89(4):1235-1243. ISSN: 1525-3198. https://doi.org/10.3168/jds.S0022-0302(06)72192-0 Links
- Belew GD, Silva J, Rito J, Tavares L, Viegas I, Teixeira J, Oliveira PJ, Macedo MP, Jones JG. 2019. Transfer of glucose hydrogens via acetyl-CoA, malonyl-CoA, and NADPH to fatty acids during de novo lipogenesis. Journal of Lipid Research. 60(12):2050-2056. ISSN: 1539-7262. https://doi.org/10.1194/jlr.RA119000354 Links
- Belo CJ, Bruckmaier RM. 2010. Suitability of low-dosage oxytocin treatment to induce milk ejection in dairy cows. Journal of Dairy Science. 93(1):63-69. ISSN: 1525-3198. https://doi.org/10.3168/jds.2009-2084 Links
- Botham MK, Mayes AP. 2018a. Bioenergetics: the role of ATP. In: Rodwell WV, Bender AD, Botham MK, Kennelly JP , Weil AP, Harper’s Illustrated Biochemistry. United States: McGraw-Hill Education / Medical. Pp. 277-291. ISBN: 978-1-25-983793-7. https://accessmedicine.mhmedical.com/book.aspx?bookID=2386 Links
- Botham MK, Mayes AP. 2018b. Lipids of physiological significance. In: Rodwell WV, Bender AD, Botham MK, Kennelly JP , Weil AP, Harper’s Illustrated Biochemistry. United States: McGraw-Hill Education / Medical. Pp. 483-511. ISBN: 978-1-25-983793-7. https://accessmedicine.mhmedical.com/book.aspx?bookID=2386 Links
- Bruckmaier RM, Wellnitz O. 2008. Induction of milk ejection and milk removal in different production systems. Journal of Animal Science. 86(13Suppl):15-20. ISSN: 15253163. https://doi.org/10.2527/jas.2007-0335 Links
- Chandel NS. 2021. Lipid metabolism. Cold Spring Harbor Perspectives in Biology. 13(9):34-41. ISSN: 1943-0264https://doi.org/10.1101/cshperspect.a040576 Links
- Cooper GM. 2019a. The biosynthesis of cell constituents. Carbohydrates, lipids, proteins, adn nucleic acids. In: Cooper GM, The Cell: A Molecular Approach. Oxford, New York: Oxford University Press. Pp. 102-111. ISBN: 978-1-60535-707-2. https://learninglink.oup.com/access/cooper8e Links
- Cooper GM. 2019b. Eukaryotic RNA polymerases and general transcription factors. In: Cooper GM, The Cell: A Molecular Approach. Oxford, New York: Oxford University Press. Pp. 258-276. ISBN: 978-1-60535-707-2. https://learninglink.oup.com/access/cooper8e Links
- Davidson PA, Stabenfeldt HG. 2020. The mammary gland and lactation. In: Klein BG, Cunningham’s Textbook of Veterinary Physiology. St. Louis, Missouri: Elsevier Health Science Division.Pp. 458-471. ISBN: 978-0-323-55227-1. https://www.elsevier.com/books/cunningham's-textbook-of-veterinary-physiology/978-0323-55227-1 Links
- Dee FA, Magee C. 2018. Anatomy and physiology of the mammary gland. In: Iowa State University Press, Anatomy and Physiology of Farm Animals. Iowa, EE. UU.: Wiley Blackwell. Pp. 501-515. ISBN: 9780813813943. https://www.wiley.com/enie/Anatomy+and+Physiology+of+Farm+Animals%2C+8th+Edition-p-9781119239765 Links
- Dhasmana S, Das S, Shrivastava S. 2022. Potential nutraceuticals from the casein fraction of goat's milk. Journal of Food Biochemistry. 46(6):e13982. ISSN: 1745-4514. https://doi.org/10.1111/jfbc.13982 Links
- Diao M, Liang Y, Zhao J, Zhang J, Zhang T. 2022. Complexation of ellagic acid with αlactalbumin and its antioxidant property. Food Chemistry. 372(1):131307. ISSN: 1873-7072. https://doi.org/10.1016/j.foodchem.2021.131307 Links
- Doherty EA, Doudna JA. 2000. Ribozyme structures and mechanisms. Annual Review of Biochemistry. 69:597-615. ISSN: 0066-4154. https://doi.org/10.1146/annurev.biochem.69.1.597 Links
- Ellis JM, Bowman CE, Wolfgang MJ. 2015. Metabolic and tissue-specific regulation of acyl-CoA metabolism. PLOS ONE. 10(3):e0116587. ISSN: 1932-6203. https://doi.org/10.1371/journal.pone.0116587 Links
- Fernandez RF, Ellis JM. 2020. Acyl-CoA synthetases as regulators of brain phospholipid acyl-chain diversity. Prostaglandins Leukot Essent Fatty Acids. 161(1):102-115. ISSN: 1532-2823. https://doi.org/10.1016/j.plefa.2020.102175 Links
- Fox PF, Lowe UT, McSweeney PLH, O'Mahony JA. 2015. Structure and development of mammary tissue. In: Fox PF , McSweeney PLH, Dairy Chemistry and Biochemistry. New York, United States: Springer International Publishing. Pp.1-7. ISBN: 978-3-319-14892-2. https://link.springer.com/book/10.1007/978-3-319-14892-2 Links
- Friedman JR, Nunnari J. 2014. Mitochondrial form and function. Nature. 505(7483):335343. ISSN: 1476-4687. https://doi.org/10.1038/nature12985 Links
- Gartner LP. 2018. The cell and the organelles. In: Vosburgh A , Horvath K, Color atlas and text of histology. Philadelphia, United States: Lippincott Williams & Wilkins. Pp. 1726. ISBN: 9781496346735 https://meded.lwwhealthlibrary.com/book.aspx?bookid=2066 Links
- Goff PJ. 2015. Endocrinology, reproduction, and lactation/The mammary gland and lactation. In: Reece OW, Erickson HH, Goff PJ , Uemura EE, Dukes' Physiology of Domestic Animals. New York, EE. UU.: John Wiley & Sons. Pp. 617-727. ISBN: 9780118501399/2015. https://www.wiley.com/enus/Dukes%27+Physiology+of+Domestic+Animals%2C+13th+Edition-p-9781118501399 Links
- Guoyao W. 2017a. Chemistry of lipids. In: Guoyao W, Principles of Animal Nutrition. New York, United States: CRC Press. Pp. 109-142. ISBN: 978-1-4987-2160-8. https://www.routledge.com/Principles-of-Animal-Nutrition/Wu/p/book/9781032095998 Links
- Guoyao W. 2017b. Nutrition and metabolism of protein and amino acids. In: Guoyao W, Principles of Animal Nutrition. New York, United States: CRC Press. Pp. 349-411. ISBN: 978-1-4987-2160-8. https://www.routledge.com/Principles-of-AnimalNutrition/Wu/p/book/9781032095998 Links
- Harvatine KJ, Boisclair YR, Bauman DE. 2009. Recent advances in the regulation of milk fat synthesis. Animal. 3(1):40-54. ISSN: 1751-732X. https://doi.org/10.1017/S1751731108003133 Links
- Heid HW, Keenan TW. 2005. Intracellular origin and secretion of milk fat globules. European Journal of Cell Biology. 84(2-3):245-258. ISSN: 0171-9335. https://doi.org/10.1016/j.ejcb.2004.12.002 Links
- Henna K, Boudjellaba S, Khammar F, Amirat Z, Chesneau D, Charallah S. 2021. Endocrine, energy, and lipid status during parturition and early lactation in indigenous goats native to the Algerian Sahara. Veterinary World. 14(9):2419-2426. ISSN: 0972-8988. https://doi.org/10.14202/vetworld.2021.2419-2426 Links
- Henry C, Saadaoui B, Bouvier F, Cebo C. 2015. Phosphoproteomics of the goat milk fat globule membrane: New insights into lipid droplet secretion from the mammary epithelial cell. Proteomics. 15(13):2307-2317. ISSN: 1615-9861. https://doi.org/10.1002/pmic.201400245 Links
- Ingolia NT. 2014. Ribosome profiling: new views of translation, from single codons to genome scale. Nature Reviews Genetics. 15(3):205-213. ISSN: 1471-0064. https://doi.org/10.1038/nrg3645 Links
- John PAT, van Schie SNS, Cheung NJ, Michel AH, Peter M, Kornmann B. 2022. Rewiring phospholipid biosynthesis reveals resilience to membrane perturbations and uncovers regulators of lipid homeostasis. Journal of the European Molecular Biology Organization. 41(7):e109998. ISSN: 1460-2075. https://doi.org/10.15252/embj.2021109998 Links
- Jones JG. 2016. Hepatic glucose and lipid metabolism. Diabetologia. 59(6):1098-1103. ISSN: 1432-0428 https://doi.org/10.1007/s00125-016-3940-5 Links
- Kumar S, Puniya AK, Puniya M, Dagar SS, Sirohi SK, Singh K, Griffith GW. 2009. Factors affecting rumen methanogens and methane mitigation strategies. World Journal of Microbiology & Biotechnology. 25(9):1557-1566. ISSN: 0959-3993. https://doi.org/10.1007/s11274-009-0041-3 Links
- Lacasse P, Lollivier V, Bruckmaier RM, Boisclair YR, Wagner GF, Boutinaud M. 2011. Effect of the prolactin-release inhibitor quinagolide on lactating dairy cows. Journal of Dairy Science. 94(3):1302-1309. ISSN: 1525-3198. https://doi.org/10.3168/jds.2010-3649 Links
- Lawhead BJ, Baker M. 2017. The endocrine system/endocrine glands. In: Lawhead BJ, Baker M, Introduction to Veterinary Science. Wisconsin Madison, EE. UU.: Cengage Learning. Pp. 169-179. ISBN: 978-1-1115-4279-5. https://www.cengagebrain.com.mx/shop/isbn/9781111542795 Links
- Lérias RJ, Hernández CLE, Suárez TA, Castro N, Pourlis A, Almeida AM. 2014. The mammary gland in small ruminants: major morphological and functional events underlying milk production-a review. Journal of Dairy Research. 81(3):304-318. ISSN: 1469-7629. https://doi.org/10.1017/S0022029914000235 Links
- Lowe M. 2011. Structural organization of the Golgi apparatus. Current Opinion in Cell Biology. 23(1):85-93. ISSN: 1879-0410. https://doi.org/10.1016/j.ceb.2010.10.004 Links
- Lozano JA, Galindo JD, García BJC, Martínez LJH, Peñafiel GR, Solano MF. 2005. Metabolismo nitrogenado. En: Lozano JA, Bioquímica y Biología Molecular para Ciencias de la Salud. Barcelona, España: McGraw-Hill Interamericana. Pp. 275-302. ISBN: 4-4860642-68. https://jabega.uma.es/discovery/fulldisplay/alma991000078319704986/34CBUA_UMA:VU1 Links
- Lundquist PK, Shivaiah KK, Espinoza-Corral R. 2020. Lipid droplets throughout the evolutionary tree. Progress in Lipid Research. 78(1):101-109. ISSN: 1873-2194. https://doi.org/10.1016/j.plipres.2020.101029 Links
- Madigan TM, Bender SK, Buckley HD, Sattley WM, Stahl AD. 2019a. Biosyntheses. Sugars and polysaccharides. Amino acids and nucleotides. Fatty acids and lipids. In: Madigan TM, Brock Biology of Microorganisms. New York, United States: Pearson. Pp. 130-137. ISBN: 978-1-292-23510-3. https://www.pearson.com/en-us/subjectcatalog/p/brock-biology-of-microorganisms/P200000006867/9780135860717 Links
- Madigan TM, Bender SK, Buckley HD, Sattley WM, Stahl AD. 2019b. Protein synthesis: translation. In: Madigan TM, Brock Biology of Microorganisms. New York, United States: Pearson. Pp. 156-170. ISBN: 978-1-292-23510-3 https://www.pearson.com/enus/subject-catalog/p/brock-biology-of-microorganisms/P200000006867/9780135860717 Links
- Maldonado JJA, Salinas GH, Torres HG, Becerril PCM, P. DR.2018. Factors influencing milk production of local goats in the Comarca Lagunera, Mexico. Livestock Research for Rural Development. 30(7):2-7. ISSN: 0121-3784. https://www.cabdirect.org/cabdirect/abstract/20183317336 Links
- Manterola BH. 2011. Estrategias nutricionales y alimenticias para modificar los sólidos totales de la leche. En Seminario sobre productividad en sistemas pastoriles lecheros. Departamento de Producción Animal, (Ed.). Págs. 1-20. Circular de Extensión. Facultad de Ciencias Agronómicas, Universidad de Chile. https://www.paislobo.cl/2011/10/seminario-sobre-productividad-en.html Links
- Martínez GM, Suárez VH. 2018. The mammary gland: morphology and development. Synthesis of milk components. In: Martínez GM , Suárez VH, Goat dairying: production, management, health, quality of milk and products. Buenos Aires, Argentina: Ediciones INTA. Pp. 37-41. ISBN: 978-987-521-972-4. https://repositorio.inta.gob.ar/xmlui/handle/20.500.12123/5408?locale-attribute=en Links
- Mas OJ. 2018. Metabolismo de los lípidos. En: Hernández MMT, Bioquímica de Laguna y Piña. Ciudad de México, México: El Manual Moderno. Pp. 660-713. ISBN: 978-607-448708-4. https://libros.facmed.unam.mx/index.php/2021/07/22/bioquimica-de-laguna-ypina/ Links
- McDonald P, Edwards RA, Greenhalgh JFD, Morgan CA, Sinclair LA, Wilkinson RG. 2011. Lipids. In: McDonald P, Animal Nutrition. New York, United States: Prentice Hall. Pp. 32-52. ISBN: 978-1408204238.https://www.pearson.com/en-gb/search.html?aq=Mc%20Donald-Animal-Nutrition-7th-Edition Links
- McKerchar HJ, Lento C, Bennie RZ, Crowther JM, Dolamore F, Dyer JM, Clerens S, Mercadante D, Wilson DJ, Dobson RCJ. 2023. The protein dynamics of bovine and caprine β-lactoglobulin differ as a function of pH. Food Chemistry. 408(1):135229. ISSN: 1873-7072. https://doi.org/10.1016/j.foodchem.2022.135229 Links
- Menzies P. 2021. Udder health for dairy goats. Veterinary Clinics of North America: Food Animal Practice. 37(1):149-174. ISSN: 1558-4240. https://doi.org/10.1016/j.cvfa.2020.12.002 Links
- Monks J, Ladinsky MS, McManaman JL. 2020. Organellar contacts of milk lipid droplets. Thousand Oaks Journal. 3(1):2-12. ISSN: 2515-2564. https://doi.org/10.1177/2515256419897226 Links
- National Chamber of Industrial Milk. 2021. CANILEC. Markets and statistics. http://www.canilec.org.mx/index.html Links
- Nayik GA, Jagdale YD, Gaikwad SA, Devkatte AN, Dar AH, Ansari MJ. 2022. Nutritional profile, processing and potential products: A comparative review of goat milk. Dairy. 3(3):622-647. ISSN: 2624-862X. https://doi.org/10.3390/dairy3030044 Links
- Nelson LD, Cox MM. 2017a. The citric acid cycle. In: Nelson LD , Cox MM, Lehninger. Principles of Biochemistry. New York, United States: Freeman, W. H. Pp. 1668-1743. ISBN: 9781464126116. https://link.springer.com/book/9781319381493 Links
- Nelson LD, Cox MM. 2017b. Lipids. In: Nelson LD , Cox MM, Lehninger. Principles of Biochemistry. New York, United States: Freeman, W. H. Pp. 989-1056. ISBN: 9781464126116. https://link.springer.com/book/9781319381493 Links
- Nelson LD, Cox MM. 2017c. Protein metabolism. In: Nelson LD , Cox MM, Lehninger. Principles of Biochemistry. New York, United States: Freeman, W. H. Pp. 2820-2941. ISBN: 9781464126116. https://link.springer.com/book/9781319381493 Links
- Neville MC, McFadden TB, Forsyth I. 2002. Hormonal regulation of mammary differentiation and milk secretion. Journal of Mammary Gland Biology and Neoplasia. 7(1):49-66. ISSN: 1083-3021. https://doi.org/10.1023/A:1015770423167 Links
- Nunes NA, Araujo WL, Obata T, Fernie AR. 2013. Regulation of the mitochondrial tricarboxylic acid cycle. Current Opinion in Plant Biology. 16(3):335-343. ISSN: 18790356. https://doi.org/10.1016/j.pbi.2013.01.004 Links
- Olarte MJ, Kim S, Sharp ME, Swanson JMJ, Farese RV Jr ., Walther TC. 2020. Determinants of endoplasmic reticulum-to-lipid droplet protein targeting. Developmental Cell. 54(4):471-487 e477. ISSN: 1878-1551. https://doi.org/10.1016/j.devcel.2020.07.001 Links
- Pacheco GV, Caballero ZA, Martínez GS, Prado ROF, García CAC. 2021. Biochemistry and metabolic pathways of polysaccharides, lipids, and proteins. Abanico Veterinario. 11(1):1-26. ISSN: 2448-6132. https://doi.org/10.21929/abavet2021.47 Links
- Philipps WP. 2018. Proteases-general aspects. In: Simões NC , Kumar V, Enzymes in Human and Animal Nutrition: Principles and Perspectives. New York, United States: Academic Press. Pp. 257-264. ISBN: 9780128094266. https://shop.elsevier.com/books/enzymes-in-human-and-animal-nutrition/nunes/978-012-805419-2 Links
- Piña GE, Flores HO. 2018. Metabolismo de los compuestos nitrogenados. En: Hernández MMT, Bioquímica de Laguna y Piña. Ciudad de México, México: El Manual Moderno. Pp. 714-763. ISBN: 978-607-448-708-4. https://libros.facmed.unam.mx/index.php/2021/07/22/bioquimica-de-laguna-y-pina/ Links
- Pol A, Gross SP, Parton RG. 2014. Review: biogenesis of the multifunctional lipid droplet: lipids, proteins and sites. Journal of Cell Biology. 204(5):635-646. ISSN: 15408140. https://doi.org/10.1083/jcb.201311051 Links
- Reece OW, Rowe WE. 2017a. Endocrine system. In: Reece OW, Rowe WE, Functional Anatomy and Physiology of Domestic Animals. New York, United States: John Wiley & Sons. Pp. 242-262. ISBN: 9781119270843. https://www.wiley.com/enus/Functional+Anatomy+and+Physiology+of+Domestic+Animals%2C+5th+Edition-p9781119270867 Links
- Reece OW, Rowe WE. 2017b. Lactation / functional anatomy of female mammary glands. Mammogenesis, lactogenesis and lactation. In: Reece OW, Rowe WE, Functional Anatomy and Physiology of Domestic Animals. New York, EE. UU.: John Wiley & Sons. Pp. 660-678. ISBN: 9781119270843.https://www.wiley.com/en-us/Functional+Anatomy+and+Physiology+of+Domestic+Animals%2C+5th+Edition-p9781119270867 Links
- Reese OW, Budras KD, Mülling C, Bragulla H, Hagen J, Witter K, König HE. 2020. Mammary gland (mamma, uber, mastos). In: König HE , Liebich GG, Veterinary Anatomy of Domestic Animals. Stuttgart, Germany: Georg Thieme Verlag KG. Pp. 642-648. ISBN: 978-3-13-242933-8 https://vetbooks.ir/veterinary-anatomy-of-domestic-animals-textbookand-colour-atlas-7th-edition/ Links
- Rodwell WV. 2018. Catabolism of proteins and amino acid nitrogen. In: Rodwell WV, Bender AD, Botham MK, Kennelly JP , Weil AP, Harper’s Illustrated Biochemistry. United States: McGraw-Hill Education / Medical. Pp. 661-686. ISBN: 978-1-25-983793-7 https://accessmedicine.mhmedical.com/book.aspx?bookID=2386 Links
- Rowland AA, Voeltz GK. 2012. Endoplasmic reticulum-mitochondria contacts: function of the junction. Nature Reviews Molecular Cell. 13(10):607-625. ISSN: 1471-0080. https://doi.org/10.1038/nrm3440 Links
- Saheki Y, De Camilli P. 2017. Endoplasmic Reticulum-Plasma Membrane Contact Sites. Annual Review of Biochemistry. 86:659-684. ISSN: 1545-4509. https://doi.org/10.1146/annurev-biochem-061516-044932 Links
- Saikia D, Hassani MI, Walia A. 2022. Goat milk and its nutraceutical properties. International Journal of Applied Research. 8(4):119-122. ISSN: 2394-5869. https://doi.org/10.22271/allresearch.2022.v8.i4b.9639 Links
- Saipin N, Thuwanut P, Thammacharoen S, Rungsiwiwut R. 2020. Effect of incubation temperature on lactogenic function of goat milk-derived mammary epithelial cells. In Vitro Cellular & Developmental Biology. 56(10):842-846. ISSN: 1543-706X. https://doi.org/10.1007/s11626-020-00529-3 Links
- Sanhueza J, Valenzuela R, Valenzuela A. 2012. Cholesterol metabolism: increasingly complex. Grasas y Aceites. 63(4):373-382. ISSN: 1988-4214. https://doi.org/10.3989/gya.035512 Links
- Sansi MS, Iram D, Zanab S, Vij S, Puniya AK, Singh A, Ashutosh Meena S. 2022. Antimicrobial bioactive peptides from goat Milk proteins: In silico prediction and analysis. Journal of Food Biochemistry. 45(10):e14311. ISSN: 1745-4514. https://doi.org/10.1111/jfbc.14311 Links
- Seoane A, Brea RJ, Fuertes A, Podolsky KA, Devaraj NK. 2018. Biomimetic generation and remodeling of phospholipid membranes by dynamic imine chemistry. Journal of the American Chemical Society. 140(27):8388-8391. ISSN: 1520-5126. https://doi.org/10.1021/jacs.8b04557 Links
- Singh A , Rajeev S. 2020. DNA: an important component of life. In: Torrens F, Mahapatra KD , Haghi AK, Biochemistry, Biophysics, and Molecular Chemistry: Applied Research and Interactions. Florida, United States: Apple Academic Press, Inc. Pp. 195-208. ISBN: 9781771888165. https://www.routledge.com/Biochemistry-Biophysics-and-MolecularChemistry-Applied-Research-and/Torrens-Mahapatra-Haghi/p/book/9781774635100 Links
- Song Z, Xiaoli AM, Yang F. 2018. Regulation and metabolic significance of de novo lipogenesis in adipose tissues. Nutrients. 10(10):1-10. ISSN: 2072-6643. https://doi.org/10.3390/nu10101383 Links
- Sturley SL, Hussain MM. 2012. Lipid droplet formation on opposing sides of the endoplasmic reticulum. Journal of Lipid Research. 53(9):1800-1810. ISSN: 1539-7262. https://doi.org/10.1194/jlr.R028290 Links
- Suburu J, Shi L, Wu J, Wang S, Samuel M, Thomas MJ, Kock ND, Yang G, Kridel S, Chen YQ. 2014. Fatty acid synthase is required for mammary gland development and milk production during lactation. American Journal of Physiology-Endocrinology and Metabolism. 306(10):E1132-E1143. ISSN: 1522-1555. https://doi.org/10.1152/ajpendo.00514.2013 Links
- Svennersten SK, Olsson K. 2005. Endocrinology of milk production. Domestic Animal Endocrinology. 29(2):241-258. ISSN: 18790054. https://doi.org/10.1016/j.domaniend.2005.03.006 Links
- Swaisgood HE. 2003. Protein composition of milk: identification, structure, and chemical composition. In: Fox PF , McSweeney PLH, Advanced dairy chemistry: proteins part A. Cork, Ireland: Springer Science. Pp. 140-225. ISBN: 9780306472718. https://link.springer.com/book/10.1007/978-1-4419-8602-3 Links
- Thul TA, Corwin EJ, Carlson NS, Brennan PA, Young LJ. 2020. Oxytocin and postpartum depression: A systematic review. Psychoneuroendocrinology. 120(1):104-109. ISSN: 1873-3360. https://doi.org/10.1016/j.psyneuen.2020.104793 Links
- Tortora JG, Funke RB, Case LC. 2019. Organic compounds. Structure and chemistry: carbohydrates, lipids, proteins, nucleic acids. In: Beauparlant S, Microbiology: An Introduction. New York, United States: Pearson. Pp. 33-47. ISBN: 978-0-13-460518-0. https://www.pearson.com/en-us/subject-catalog/p/microbiology-anintroduction/P200000006850/9780135789377 Links
- Verschueren KHG, Blanchet C, Felix J, Dansercoer A, De Vos D, Bloch Y, Van Beeumen J, Svergun D, Gutsche I, Savvides SN, Verstraete K. 2019. Structure of ATP citrate lyase and the origin of citrate synthase in the Krebs cycle. Nature. 568(7753):571575. ISSN: 1476-4687. https://doi.org/10.1038/s41586-019-1095-5 Links
- Vidal ME. 2013. Lácteos: oferta y demanda en el contexto regional y mundial. En 1º Foro de Agricultura de América del Sur. Oficina de Programación y Política Agropecuaria, (Ed.). Consejo Agropecuario del Sur. Foz de Iguazú, Brasil. https://www.odepa.gob.cl/publicaciones/noticias/noticias-institucionales/primer-foro-dela-agricultura-en-america-del-sur Links
- Weil AP. 2018a. Protein synthesis and the genetic code. In: Rodwell WV, Bender AD, Botham MK, Kennelly JP , Weil AP, Harper’s Illustrated Biochemistry. United States: McGraw-Hill Education / Medical. Pp. 955-991. ISBN: 978-1-25-983793-7. https://accessmedicine.mhmedical.com/book.aspx?bookID=2386 Links
- Weil AP. 2018b. RNA synthesis, processing and modification. In: Rodwell WV, Bender AD, Botham MK, Kennelly JP , Weil AP, Harper’s Illustrated Biochemistry. United States: McGraw-Hill Education / Medical. Pp. 911-954. ISBN: 978-1-25-983793-7. https://accessmedicine.mhmedical.com/book.aspx?bookID=2386 Links
- Wilson C, Venditti R, Rega LR, Colanzi A, D'Angelo G, De Matteis MA. 2011. The Golgi apparatus: an organelle with multiple complex functions. Biochemical Journal. 433(1):19. ISSN: 1470-8728. https://doi.org/10.1042/BJ20101058 Links
- Zhang C, Liu P. 2019. The new face of the lipid droplet: lipid droplet proteins. Proteomics. 19(10):e1700223. ISSN: 1615-9861. https://doi.org/10.1002/pmic.201700223 Links

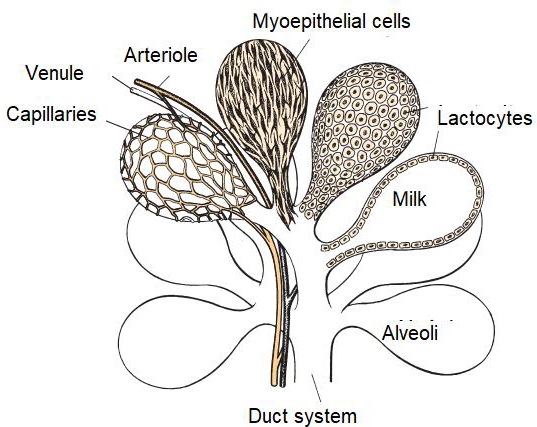
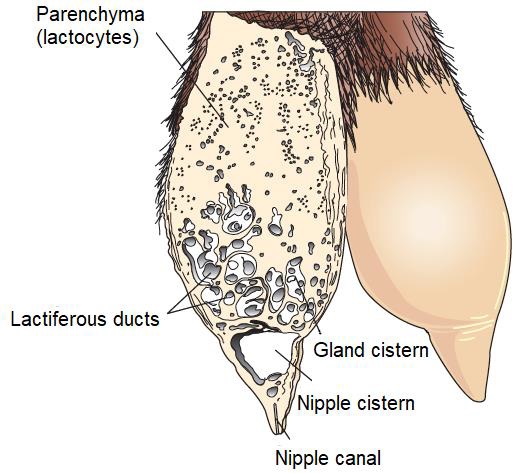


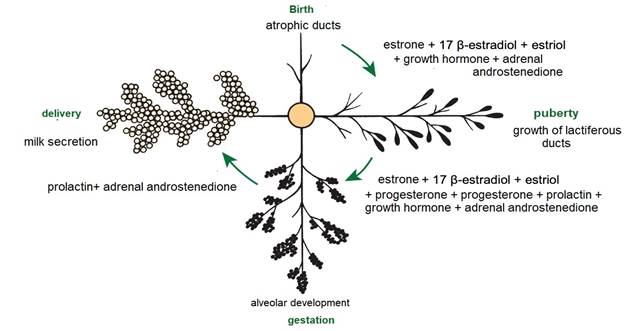
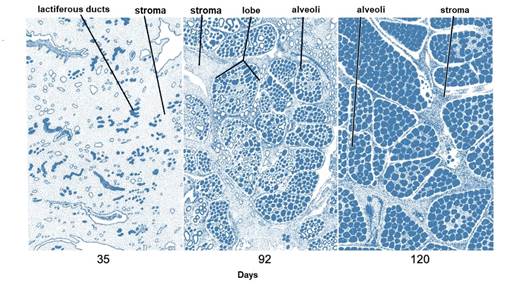 Source: (Dee & Magee, 2018)
Source: (Dee & Magee, 2018)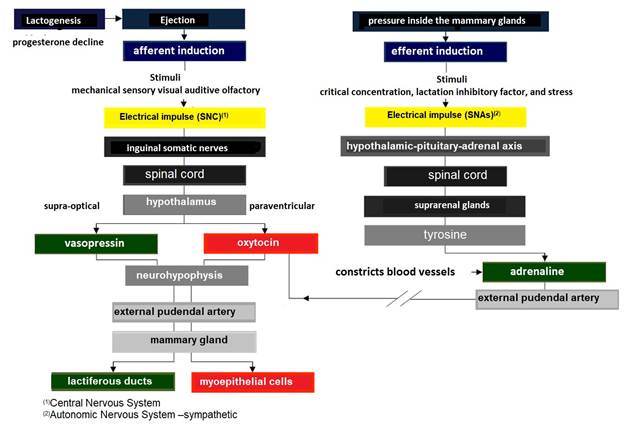
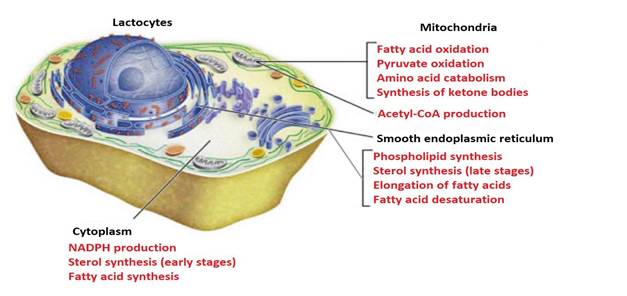
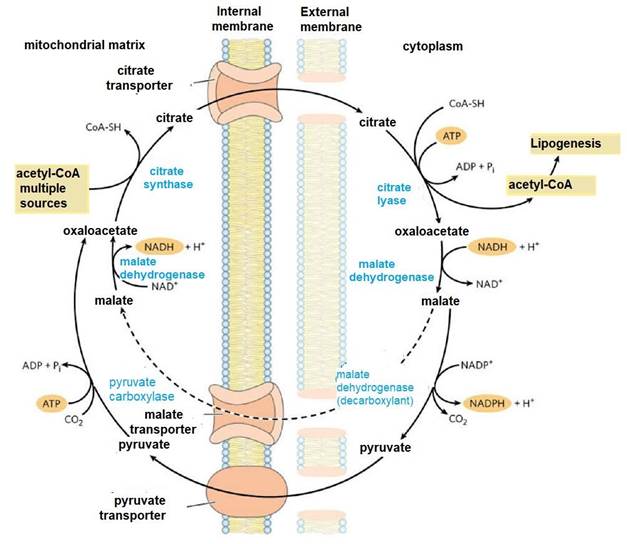
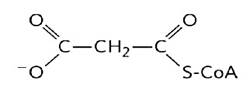

 Fuente: (Dee & Magee, 2018)
Fuente: (Dee & Magee, 2018)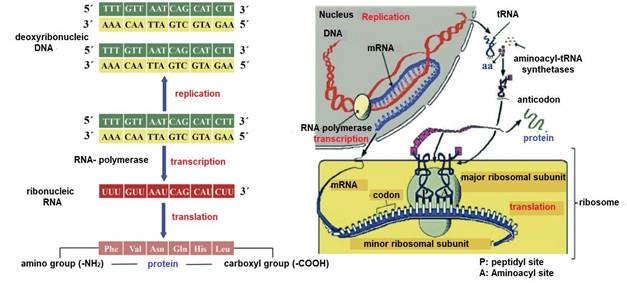 Source: (Pacheco et al., 2021)
Source: (Pacheco et al., 2021)