Research articles
First bloom report of Polykrikos hartmannii (Dinophyceae) in the coastal waters of İzmir Bay, Aegean Sea (Eastern Mediterranean)
Primer reporte del florecimiento de Polykrikos hartmannii (Dinophyceae) en las aguas costeras de la Bahía de İzmir, Mar Egeo (Mediterráneo oriental)
Çolak Sabanci, Fatma1*
 http://orcid.org/0000-0001-9122-594X
http://orcid.org/0000-0001-9122-594X
Şahin, Fatih2
 http://orcid.org/0000-0003-0605-2672
http://orcid.org/0000-0003-0605-2672
Gárate-Lizárraga, Ismael3
 http://orcid.org/0000-0002-3835-183X
http://orcid.org/0000-0002-3835-183X
Erbaş, Edanur1
 http://orcid.org/0009-0001-5512-8904
http://orcid.org/0009-0001-5512-8904
Tan, İbrahim4
 http://orcid.org/0000-0002-4948-7687
http://orcid.org/0000-0002-4948-7687
 http://orcid.org/0000-0001-9122-594X
http://orcid.org/0000-0001-9122-594XŞahin, Fatih2
 http://orcid.org/0000-0003-0605-2672
http://orcid.org/0000-0003-0605-2672Gárate-Lizárraga, Ismael3
 http://orcid.org/0000-0002-3835-183X
http://orcid.org/0000-0002-3835-183XErbaş, Edanur1
 http://orcid.org/0009-0001-5512-8904
http://orcid.org/0009-0001-5512-8904Tan, İbrahim4
 http://orcid.org/0000-0002-4948-7687
http://orcid.org/0000-0002-4948-7687-
Publication dates-
November 28, 2025
Jan-Apr , 2025
- Article in PDF
- Article in XML
- Automatic translation
- Send this article by e-mail
- Share this article +
ABSTRACT
Background.
Harmful algal blooms have increased in frequency, intensity and distribution in the last decades around the world. This increase has also been observed for the waters of the Mediterranean Sea. In November 2023, several reddish patches were observed in İzmir Bay (Aegean Sea); the dinoflagellate Polykrikos hartmannii was the causative agent.
Goal.
The purpose of this study was to report for the first time a bloom of this species in İzmir Bay.
Methods.
During the bloom, surface samples of seawater were collected using 1 liter plastic bottles at eleven sampling stations and fixed with Lugol for cell counting, which were performed under an OLYMPUS BX-50 microscope, using 1 ml Sedgewick-Rafter chambers. Samples were collected for live analysis and correct identification of the responsible species.
Results.
The moderate bloom occurred in November 2023 was caused by Polykrikos hartmannii. During the bloom two-celled chains were the predominant species, with single cells occurring less frequently. The highest cell abundance (4.8 x 104 cells L-1) was observed at station 29 in the inner bay. Cell sizes were 29.7 - 34.6 μm long, and 39.6 - 44.5 μm wide.
Conclusions.
Living samples allowed correct identification of P. hartmannii. Cells tend to become round or disrupted when preserved with Lugol’s solution. This may be why this species has not been previously reported. The maximum abundance of P. hartmannii was recorded in İzmir Bay at a water temperature of 14.84 °C and a salinity of 38.02 ‰, coinciding with highest values of nutrients. The bloom occurred in the shallowest area with limited water circulation and significant riverine inflow.
Keywords::
phytoplankton, unarmored dinoflagellates, Polykrikos hartmannii, İzmir Bay, eastern Mediterranean
INTRODUCTION
Harmful algal blooms (HABs) represent a growing global threat that frequently occurs in bays and exhibits increased frequency, scale and duration. This phenomenon results in severe consequences for human health, economies, marine ecosystems and surrounding creatures (Anderson et al., 2008; Anderson et al., 2012; Grattan et al., 2016; Montes et al., 2018; Brown et al., 2020; Fire et al., 2021; Hallegraeff et al., 2021; Otero & Silva, 2022). The formation of HABs, particularly in coastal environments, occurs in response to changing physicochemical factors, such as tides, winds or available nutrients. These have different timescales of variability, and therefore, blooms can be short-term episodic events, recurrent seasonal phenomena, or rare events associated with exceptional climatic or hydrological conditions (Cloern, 1996). However, among the species causing HABs, there are several that can continue blooming over the long term even if environmental conditions have changed, making the characterization of the mechanisms causing HABs even more complex (Park et al., 2018).
-
Anderson et al., 2008Harmful algal blooms and eutrophication: Examining linkages from selected coastal regions of the United StatesHarmful Algae, 2008
-
Anderson et al., 2012Progress in understanding harmful algal blooms: paradigm shifts and new technologies for research, monitoring, and managementThe Annual Review of Marine Science, 2012
-
Grattan et al., 2016Harmful algal blooms and public healthHarmful Algae, 2016
-
Montes et al., 2018Quantifying harmful algal bloom thresholds for farmed salmon in southern ChileHarmful Algae, 2018
-
Brown et al., 2020Assessing risks and mitigating impacts of harmful algal blooms on mariculture and marine fisheriesReviews in Aquaculture, 2020
-
Fire et al., 2021An assessment of temporal, spatial and taxonomic trends in harmful algal toxin exposure in stranded marine mammals from the U.S. New England coastPlos One, 2021
-
Hallegraeff et al., 2021Perceived global increase in algal blooms isattributable to intensified monitoring and emergingbloom impactsCommunications Earth & Environment, 2021
-
Otero & Silva, 2022Emerging Marine Biotoxins in European Waters: Potential Risks and Analytical ChallengesMarine Drugs, 2022
-
Cloern, 1996Phytoplankton bloom dynamics in coastal ecosystems: a review with some general lessons from sustained investigation of San Francisco Bay, CaliforniaReviews of Geophysics, 1996
-
Park et al., 2018Intraspecific bloom succession in the harmful dinoflagellate Cochlodinium polykrikoides (Dinophyceae) extended the blooming period in Korean coastal waters in 2009Harmful Algae, 2018
Dinoflagellates, a diverse and ecologically significant group of marine phytoplankton, are the primary causative organisms in approximately 75% of documented harmful algal bloom events (Smayda, 1997) and are responsible for numerous red tide events, characterized by the discoloration of the sea surface (Holmes et al., 1967; Eppley & Harrison, 1975; Franks & Anderson, 1992; Anderson et al., 2002; Imai et al., 2006; Ferrante et al., 2013; Jeong et al., 2013; Park et al., 2013). Many dinoflagellates, including unarmored species, possess the adaptive ability to form resting cysts throughout their life cycles (Head, 1996). This ability not only enables their survival in adverse environmental conditions, but also is associated with various factors such as genetic recombination, continuation and cessation of blooms, recurrence of annual blooms, protection against viral, herbivorous or parasitic attacks, and geographical expansion of populations (Anderson & Wall, 1978; Hallegraeff & Bolch, 1991; Matsuoka & Fukuyo, 2002; Figueroa et al., 2010). This plays a crucial role in HAB dynamics, contributing to both the recurrence and geographic expansion of blooms (Bravo & Figueroa, 2014; Tang & Gobler, 2015; Yang et al., 2018).
-
Smayda, 1997Harmful algal blooms: Their ecophysiology and general relevance to phytoplankton blooms in the seaLimnology and Oceanography, 1997
-
Holmes et al., 1967Red water in La Jolla Bay, 1964-1966Limnology and Oceanography, 1967
-
Eppley & Harrison, 1975Physiological ecology of Gonyaulax polyedrum, a red tide water dinoflagellate of southern California1st Int. Conf. on Toxic Dinoflagellate Blooms, 1975
-
Franks & Anderson, 1992Alongshore transport of a toxic phytoplankton bloom in a buoyancy current: Alexandrium tamarense in the Gulf of MaineMarine Biology, 1992
-
Anderson et al., 2002Harmful algal blooms and eutrophication: nutrient sources, composition, and consequencesEstuaries, 2002
-
Imai et al., 2006Eutrophication and occurrences of harmful algal blooms in the Seto Inland Sea, JapanPlankton and Benthos Research, 2006
-
Ferrante et al., 2013Harmful algal blooms in the Mediterranean Sea: effects on human healthEuroMediterranean Biomedical Journal, 2013
-
Jeong et al., 2013Red tides in Masan Bay, Korea in 2004-2005: I. Daily variations in the abundance of red-tide organisms and environmental factorsHarmful Algae, 2013
-
Park et al., 2013Economic impact, management and mitigation of red tides in KoreaHarmful Algae, 2013
-
Head, 1996Modern dinoflagellate cysts and their biological affinities. Chapter 30Palynology, principles and applications, 1996
-
Anderson & Wall, 1978Potential importance of benthic cysts of Gonyaulax tamarensis and G. excavata in initiating toxic dinoflagellate bloomsJournal of Phycology, 1978
-
Hallegraeff & Bolch, 1991Transport of toxic dinoflagellate cysts via ships’ ballast waterMarine Pollution Bulletin, 1991
-
Matsuoka & Fukuyo, 2002Technical Guide for Modern Dinoflagellate Cyst StudyJapanese Society for the Promotion of Science, 2002
-
Figueroa et al., 2010From homothally to heterothally: mating preferences and genetic variation within clones of the dinoflagellate Gymnodinium catenatumDeep-Sea Research, 2010
-
Bravo & Figueroa, 2014Towards an Ecological Understanding of Dinoflagellate Cyst FunctionsMicroorganisms, 2014
-
Tang & Gobler, 2015Sexual resting cyst production by the dinoflagellate Akashiwo sanguinea: a potential mechanism contributing to the ubiquitous distribution of a harmful algaJournal of Phycology, 2015
-
Yang et al., 2018Solid sand particle addition can enhance the production of resting cysts in dinoflagellatesJournal of Oceanology and Limnology, 2018
The unarmored, gymnodinoid dinoflagellate Polykrikos hartmannii W.M.Zimmermann 1930 is a mixotrophic, cyst-producing and ichthyotoxic species that forms single-celled zooids or two-celled pseudocolonies (Hulburt, 1957; Tang et al., 2013; Lee et al., 2015). The species was first described by Zimmermann (1930) as P. hartmannii and then was subsequently placed in the genus Pheopolykrikos by Matsuoka & Fukuyo (1986) due to shared characteristics with Pheopolykrikos spp. Hoppenrath et al. (2010) revealed the presence of a nematocyst-taeniocyst complex, a diagnostic feature of the genus Polykrikos, leading to the reassignment of the species back to Polykrikos. Within current taxonomic nomenclature, the accepted name for this species is P. hartmannii (Guiry & Guiry, 2024).
-
Zimmermann 1930Neue und wenig bekannte Kleinalgen von Neapel IVZeitschrift für Botanik, 1930
-
Hulburt, 1957The Taxonomy of Unarmored Dinophyceae of Shallow Embayments on Cape Cod, MassachusettsBiological Bulletin, 1957
-
Tang et al., 2013Morphology, phylogeny, dynamics, and ichthyotoxicity of Pheopolykrikos hartmannii (Dinophyceae) isolates and blooms from New York, USAJournal of Phycology, 2013
-
Lee et al., 2015Mixotrophy in the nematocyst-taeniocyst complex-bearing phototrophic dinoflagellate Polykrikos hartmanniiHarmful Algae, 2015
-
Zimmermann (1930)Neue und wenig bekannte Kleinalgen von Neapel IVZeitschrift für Botanik, 1930
-
Matsuoka & Fukuyo (1986)Cyst and motile morphology of a colonial dinoflagellate Pheopolykrikos hartmannii (Zinunermann) comb. novJournal of Plankton Research, 1986
-
Hoppenrath et al. (2010)Re-classification of Pheopolykrikos hartmannii as Polykrikos (Dinophyceae) based partly on the ultrastructure of complex extrusomesEuropean Journal of Protistology, 2010
-
Guiry & Guiry, 2024AlgaeBase, 2024
Polykrikos hartmannii exhibits a global distribution pattern, based on its various life stages, including cyst, vegetative and motile stages that have been documented in diverse aquatic habitats, including lagoons, rivers, estuaries and marine environments. This species has been reported from Canada (Pospelova et al., 2010; Price & Pospelova, 2011), the USA (Hulburt, 1957; Steidinger & Tangen, 1997; Pospelova et al., 2004; Badylak & Phllips, 2004; Hoppenrath et al., 2009; Hoppenrath et al., 2010; Tang et al., 2013), Mexico (Morquecho & Lechuga-Devéze, 2003; Peña-Manjarrez et al., 2005, Gárate-Lizárraga et al., 2009, 2016; Gárate-Lizárraga, 2014; Escobar-Morales & Hernández-Becerril, 2015; Escarcega-Bata et al., 2023), Japan (Matsuoka & Fukuyo, 1986; Fuji & Matsuoka, 2006), China (Wang et al., 2004; Chai et al., 2020; Liu et al., 2023), Indonesia (Mizushima et al., 2007; Rukminasari & Tahir, 2021; Rachman et al., 2022), India (Godhe et al., 2000; D’costa et al., 2008), Malasia and Singapore (Hii et al., 2021),, Korea (Kim et al., 1990; Kim et al., 2008; Pospelova & Kim, 2010; Shin et al., 2011; Thangaraj et al., 2017; Kwak et al., 2022), Israel (Rubino et al., 2017), Bulgaria and Ukrain (Dzhembekova et al., 2017) and Turkey (Balkis et al., 2016; Aktan & Keskin, 2017). Despite its global distribution, there are few records of P. hartmannii causing significant blooms (Kim et al., 1990; Badylak & Phllips, 2004; Gárate-Lizárraga et al., 2009, 2016; Tang et al., 2013; Gárate-Lizárraga, 2014; Thangaraj et al., 2017).
-
Pospelova et al., 2010Organic-walled dinoflagellate cyst production, composition and flux from 1996 to 1998 in the central Strait of Georgia (BC, Canada): A sediment trap studyMarine Micropaleontology, 2010
-
Price & Pospelova, 2011High-resolution sediment trap study of organic-walled dinoflagellate cyst production and biogenic silica flux in Saanich Inlet (BC, Canada)Marine Micropaleontology, 2011
-
Hulburt, 1957The Taxonomy of Unarmored Dinophyceae of Shallow Embayments on Cape Cod, MassachusettsBiological Bulletin, 1957
-
Steidinger & Tangen, 1997DinoflagellatesIdentifying marine phytoplankton, 1997
-
Pospelova et al., 2004Environmental factors influencing the spatial distribution of dinoflagellate cyst assemblages in shallow lagoons of southern New England (USA)Review of Palaeobotany and Palynology, 2004
-
Badylak & Phllips, 2004Spatial and temporal patterns of phytoplankton composition in subtropical coastal lagoon, the Indian River Lagoon, Florida, USAJournal of Plankton Research, 2004
-
Hoppenrath et al., 2009Molecular phylogeny of ocelloid-bearing dinoflagellates (Warnowiaceae) as inferred from SSU and LSU rDNA sequencesBMC Ecology and Evolution, 2009
-
Hoppenrath et al., 2010Re-classification of Pheopolykrikos hartmannii as Polykrikos (Dinophyceae) based partly on the ultrastructure of complex extrusomesEuropean Journal of Protistology, 2010
-
Tang et al., 2013Morphology, phylogeny, dynamics, and ichthyotoxicity of Pheopolykrikos hartmannii (Dinophyceae) isolates and blooms from New York, USAJournal of Phycology, 2013
-
Morquecho & Lechuga-Devéze, 2003Dinoflagellate cysts in recent sediments from Bahía Concepción, Gulf of CaliforniaBotanica Marina, 2003
-
Peña-Manjarrez et al., 2005Dinoflagellate cysts and bloom events at Todos Santos Bay, Baja California, México, 1999-2000Continental Shelf Research, 2005
-
Gárate-Lizárraga et al., 2009A multi-species microalgae bloom in Bahía de La Paz, Gulf of California, Mexico (June 2008)CICIMAR Oceánides, 2009
-
2016Florecimientos algales nocivos en las costas del estado de GuerreroFlorecimientos Algales Nocivos en México, 2016
-
Gárate-Lizárraga, 2014Proliferation of Levanderina fissa and Polykrikos hartmannii (Dinophyceae: Gymnodiniales) in Bahía de La Paz, Gulf of California, MéxicoCICIMAR Oceánides, 2014
-
Escobar-Morales & Hernández-Becerril, 2015Free-living marine planktonic unarmoured dinoflagellates from the Gulf of Mexico and the Mexican PacificBotanica Marina, 2015
-
Escarcega-Bata et al., 2023Diversidad de dinoflagelados atecados del orden Gymnodiniales (Dinophyceae), con énfasis en aquellos formadores de florecimientos algales nocivos en las costas del Pacífico MexicanoActa Botanica Mexicana, 2023
-
Matsuoka & Fukuyo, 1986Cyst and motile morphology of a colonial dinoflagellate Pheopolykrikos hartmannii (Zinunermann) comb. novJournal of Plankton Research, 1986
-
Fuji & Matsuoka, 2006Seasonal change of dinoflagellates cyst flux collected in a sediment trap in Omura Bay, West JapanJournal of Plankton Research, 2006
-
Wang et al., 2004Dinoflagellate cyst records in recent sediments from Daya Bay, South China SeaPhycological Research, 2004
-
Chai et al., 2020Proof of homothally of Pheopolykrikos hartmannii and details of cyst germination processJournal of Oceanology and Limnology, 2020
-
Liu et al., 2023A combined approach detected novel species diversity and distribution of dinoflagellate cysts in the Yellow Sea, ChinaMarine Pollution Bulletin, 2023
-
Mizushima et al., 2007Vertical distribution of Pyrodinium bahamense var. compressum (Dinophyceae) cysts in Ambon Bay and Hurun Bay, IndonesiaPlankton and Benthos Research, 2007
-
Rukminasari & Tahir, 2021Species Assemblages and Distribution of Dinoflagellate Cysts from three Estuaries Sediment’s of Makassar Strait, Eastern IndonesiaOnLine Journal of Biological Sciences, 2021
-
Rachman et al., 2022Dinoflagellate Cyst Distribution in Relation to the Sediment Composition and Grain Size in the Coastal Area of Pangkajene, South Sulawesi, IndonesiaILMU KELAUTAN: Indonesian Journal of Marine Sciences, 2022
-
Godhe et al., 2000Dinoflagellate Cysts in Recent Marine Sediments from SW IndiaBotanica Marina, 2000
-
D’costa et al., 2008Dinoflagellates in a mesotrophic, tropical environment influenced by monsoonEstuarine, Coastal and Shelf Science, 2008
-
Hii et al., 2021Diverse harmful microalgal community assemblages in the Johor Strait and the environmental effects on its community dynamicsHarmful Algae, 2021
-
Kim et al., 1990Coastal algal blooms caused by the cyst-forming dinoflagellatesBulletin of The Korean Society, 1990
-
Kim et al., 2008Research Note: Molecular phylogenetic affiliations of Dissodinium pseudolunula, Pheopolykrikos hartmannii, Polykrikos cf. schwartzii and Polykrikos kofoidii to Gymnodinium sensu stricto species (Dinophyceae)Phycological Research, 2008
-
Pospelova & Kim, 2010Dinoflagellate cysts in recent estuarine sediments from aquaculture sites of southern South KoreaMarine Micropaleontology, 2010
-
Shin et al., 2011Dinoflagellate cysts in surface sediments from southern coast of KoreaEstuaries and Coasts, 2011
-
Thangaraj et al., 2017Molecular cloning reveals co-occurring species behind red tide blooms of the harmful dinoflagellate Cochlodinium polykrikoidesBiochemical Systematics and Ecology, 2017
-
Kwak et al., 2022Resting cysts and vegetative cells of Pheopolykrikos hartmannii (W. Zimmermann) Matsuoka & Fukuyo (Dinophyceae): Morphology, phylogeny, and effect of temperature on germinationKorean Journal of Environmental Biology, 2022
-
Rubino et al., 2017Plankton resting stages in recent sediments of Haifa port, Israel (Eastern Mediterranean)-Distribution, viability and potential environmental consequencesMarine Pollution Bulletin, 2017
-
Dzhembekova et al., 2017Applicability of massively parallel sequencing on monitoring harmful algae at Varna Bay in the Black SeaHarmful Algae, 2017
-
Balkis et al., 2016Dinoflagellate resting cysts in recent marine sediments from the Gulf of Gemlik (Marmara Sea, Turkey) and seasonal harmful algal bloomsPhycologia, 2016
-
Aktan & Keskin, 2017Second habitat record of Polykrikos hartmannii W. Zimm. (Dinophyceae) in the south Aegean Sea, Eastern MediterraneanTurkish Journal of Fisheries and Aquatic Sciences, 2017
-
Kim et al., 1990Coastal algal blooms caused by the cyst-forming dinoflagellatesBulletin of The Korean Society, 1990
-
Badylak & Phllips, 2004Spatial and temporal patterns of phytoplankton composition in subtropical coastal lagoon, the Indian River Lagoon, Florida, USAJournal of Plankton Research, 2004
-
Gárate-Lizárraga et al., 2009A multi-species microalgae bloom in Bahía de La Paz, Gulf of California, Mexico (June 2008)CICIMAR Oceánides, 2009
-
2016Florecimientos algales nocivos en las costas del estado de GuerreroFlorecimientos Algales Nocivos en México, 2016
-
Tang et al., 2013Morphology, phylogeny, dynamics, and ichthyotoxicity of Pheopolykrikos hartmannii (Dinophyceae) isolates and blooms from New York, USAJournal of Phycology, 2013
-
Gárate-Lizárraga, 2014Proliferation of Levanderina fissa and Polykrikos hartmannii (Dinophyceae: Gymnodiniales) in Bahía de La Paz, Gulf of California, MéxicoCICIMAR Oceánides, 2014
-
Thangaraj et al., 2017Molecular cloning reveals co-occurring species behind red tide blooms of the harmful dinoflagellate Cochlodinium polykrikoidesBiochemical Systematics and Ecology, 2017
This study provides the first documented bloom of P. hartmannii that occurred in the Mediterranean Sea, expanding our understanding of the distribution of this species and contributing to the known biodiversity of the region.
MATERIAL AND METHODS
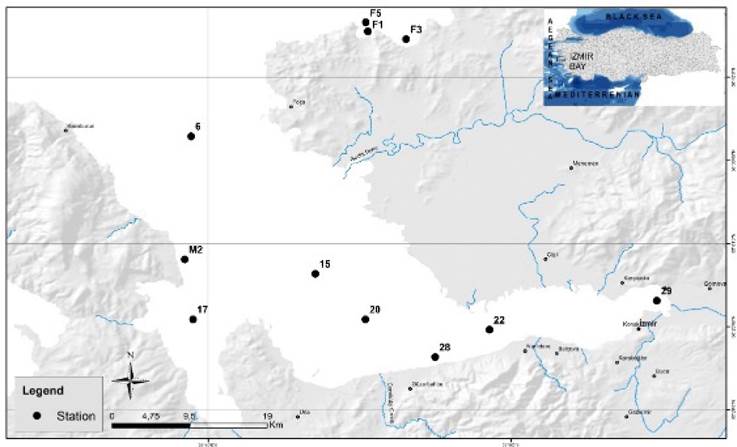
Phytoplankton samples were collected from 11 sites in İzmir Bay during a bloom event in November 2023 (Fig. 1). The sampling area is located in the Eastern Mediterranean (38° 20′ - 38°40′ N, 26° 30 ′-27° 10′ E). It is a closed bay opening into the Aegean Sea, with a surface area of 500 km², a water capacity of 11.5 million m³, and a total length of 64 km (Kontas et al., 2004). İzmir bay is divided topographically into three sections: inner bay (St. 29), middle bay (St. 22 and 28), and outer bay (St. 6, 15, 17, 20, M2, F1, F3 and F5). The wider and deeper section extending northwest-southeastward between the Karaburun Peninsula and the Gediz Delta is referred to as the outer bay. The width of the outer bay, which has a length of 45 km, reaches 24 km at its mouth. The depth in the outer bay varies between 45 and 70 m. The deepest point reaching 71 m is located at the mouth of the outer bay (between Foça and Karaburun). In the inner and middle bays, the depth increases from East to West. The deepest point (ca. 21 m) of the inner bay is in the central region.
-
Kontas et al., 2004Monitoring of eutrophication and nutrient limitation in the Izmir Bay (Turkey) before and after wastewater treatment plantEnvironment International, 2004
Thumbnail
Figure 1
Map of the sampling stations in Izmir Bay, Aegean Sea, in November 2023.
Map of the sampling stations in Izmir Bay, Aegean Sea, in November 2023.
Surface samples of the bloom were obtained using 1-liter plastic flasks. These samples without preservation were examined about one hour after the collection with an Olympus BX-50 (Olympus Optical Co. Ltd., Japan) microscope connected to a digital camera (ToupTek XCAM-1080). Some samples were fixed with Lugol’s solution for enumeration, and cell enumerations were conducted using a 1-ml Sedgewick-Rafter counting chamber. Live samples were used to identify Polykrikos hartmannii based on their distinctive morphology, including the cell shape, size, motility, and chloroplast color (Hulburt, 1957; Matsuoka & Fukuyo, 1986; Steidinger & Tangen, 1997; Hoppenrath et al., 2009; Hoppenrath et al., 2010; Tang et al., 2013; Gárate-Lizárraga, 2014; Escobar-Morales & Hernández-Becerril, 2015; Kwak et al., 2022).
-
Hulburt, 1957The Taxonomy of Unarmored Dinophyceae of Shallow Embayments on Cape Cod, MassachusettsBiological Bulletin, 1957
-
Matsuoka & Fukuyo, 1986Cyst and motile morphology of a colonial dinoflagellate Pheopolykrikos hartmannii (Zinunermann) comb. novJournal of Plankton Research, 1986
-
Steidinger & Tangen, 1997DinoflagellatesIdentifying marine phytoplankton, 1997
-
Hoppenrath et al., 2009Molecular phylogeny of ocelloid-bearing dinoflagellates (Warnowiaceae) as inferred from SSU and LSU rDNA sequencesBMC Ecology and Evolution, 2009
-
Hoppenrath et al., 2010Re-classification of Pheopolykrikos hartmannii as Polykrikos (Dinophyceae) based partly on the ultrastructure of complex extrusomesEuropean Journal of Protistology, 2010
-
Tang et al., 2013Morphology, phylogeny, dynamics, and ichthyotoxicity of Pheopolykrikos hartmannii (Dinophyceae) isolates and blooms from New York, USAJournal of Phycology, 2013
-
Gárate-Lizárraga, 2014Proliferation of Levanderina fissa and Polykrikos hartmannii (Dinophyceae: Gymnodiniales) in Bahía de La Paz, Gulf of California, MéxicoCICIMAR Oceánides, 2014
-
Escobar-Morales & Hernández-Becerril, 2015Free-living marine planktonic unarmoured dinoflagellates from the Gulf of Mexico and the Mexican PacificBotanica Marina, 2015
-
Kwak et al., 2022Resting cysts and vegetative cells of Pheopolykrikos hartmannii (W. Zimmermann) Matsuoka & Fukuyo (Dinophyceae): Morphology, phylogeny, and effect of temperature on germinationKorean Journal of Environmental Biology, 2022
Surface water samples were collected using 101 Teflon Niskin type bottles attached to a CTD (SeaBird SBE 25Plus/SBE 27 pH Sensor) Rosette System (SBE 32C 12 universal sampling bottle). Surface water was sampled to determine the temperature (ºC), salinity (‰), orthophosphate (μmol L-1), nitrite + nitrate nitrogen (μmol L-1), ammonium nitrogen (μmol L-1) and silicate (μmol L-1) at the time of sampling using a CTD and water sampler that makes real-time measurements at each station (MMG, 2017).
-
MMG, 2017Ministry of Environment and Urbanization of the Republic of Turkey, 2017
RESULTS
Characterization of the environment. An algal bloom and associated water discoloration in November were studied. This phenomenon followed a period of excessive rainfall. The bloom and subsequent color change dissipated during high wind conditions and re-emerged once the winds subsided. The bloom exhibited passive movement consistent with currents. Water discoloration ranged from reddish-brown to rust colored (Fig. 2) and was likely correlated with the cell abundance. In İzmir Bay, apart from the discoloration caused by excessive proliferation, no fish mortality or harmful effects were observed.
Thumbnail
Figure 2
Different views of the Polykrikos hartmannii bloom in Izmir Bay, Aegean Sea, Eastern Mediterranean, in November 2023.
Different views of the Polykrikos hartmannii bloom in Izmir Bay, Aegean Sea, Eastern Mediterranean, in November 2023.
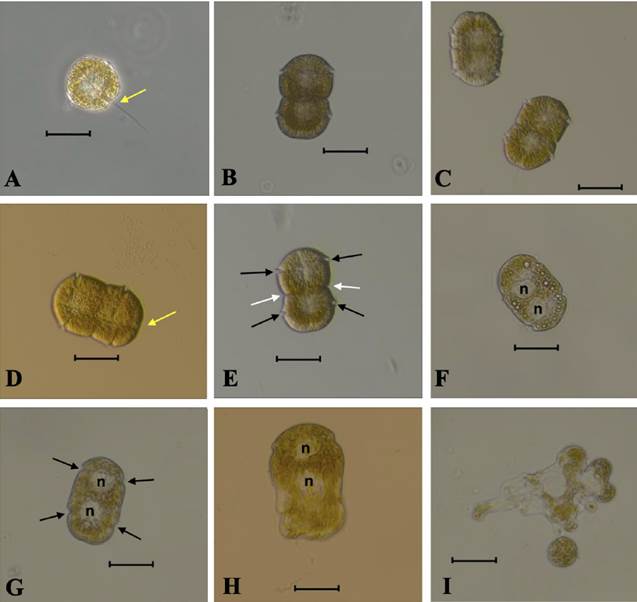
The morphological characteristics and abundance of P. hartmannii. Analysis of live samples allows us to identify a monospecific bloom caused by Polykrikos hartmannii. The two-celled colonies were the predominant form observed, with single cells occurring less frequently and never dorsoventrally compressed. The cell size was 29.7 - 34.6 μm long and 39.6 - 44.5 μm wide, with the round epicone often smaller than or sometimes equal to the cylindrical hypocone. In the two-celled colonies, the posterior zooid is slightly rounded at the antapex, whereas the anterior zooid was straight at the apex (Fig. 3C). Cells are often heavily pigmented, and numerous small chloroplasts of brown-greenish, yellow color are present (Figs. 3A; 3D). The nucleus of the anterior cell is in the hypocone, while that of the posterior cell is in the center of the epicone (Fig. 3E).
Thumbnail
Figure 3
Light microphotographs of Polykrikos hartmannii from Izmir Bay (A-I): single cells of Polykrikos hartmannii (A); ventral view of a two-celled chain showing general cell shape, cingulum, and the connection between the two zooids (B-E); a colony stressed by longer exposure in deeper focus, showing nucleus (n) (F, G) and lysis of the cell (H, I). Black arrows show two transverse furrows, white arrows show the visible border between the two zooids, and yellow arrows show the longitudinal flagellum. Scale bars: 40 μm
Light microphotographs of Polykrikos hartmannii from Izmir Bay (A-I): single cells of Polykrikos hartmannii (A); ventral view of a two-celled chain showing general cell shape, cingulum, and the connection between the two zooids (B-E); a colony stressed by longer exposure in deeper focus, showing nucleus (n) (F, G) and lysis of the cell (H, I). Black arrows show two transverse furrows, white arrows show the visible border between the two zooids, and yellow arrows show the longitudinal flagellum. Scale bars: 40 μm
In November 2023, the species exhibited a moderate bloom. The highest cell abundance (4.8 x 104 cells L-1, was observed at station 29 (the inner bay). The lowest cell abundance (11 cells L-1) was observed at station 6 (the outer bay).
Physical and chemical variables. The water temperature varied between 14.84 and 19.21 °C, while salinity levels ranged from 37.83 ‰ to 39.23 ‰. Nutrient concentrations during the bloom were 0.02 - 2.23 μM PO4-P, 0.05 - 12.06 μM NOx, 0.04 - 24.15 μM NH4-N and 0.45 - 22.25 μM Si (Table 1). The maximum abundance of P. hartmannii was recorded in İzmir Bay with a water temperature of 14.84 °C and salinity of 38.02 ‰.
Table 1
Physicochemical variables measured in Izmir Bay in November 2023. Temperature (C°), Salinity (‰), PO4-P: orthophosphate, NOx: nitrite + nitrate nitrogen, NH4-N: ammonium, Si: silicate.
Physicochemical variables measured in Izmir Bay in November 2023. Temperature (C°), Salinity (‰), PO4-P: orthophosphate, NOx: nitrite + nitrate nitrogen, NH4-N: ammonium, Si: silicate.
| Stations | Temperature (C°) | Salinity | PO4-P | NOx | NH4-N | Si |
|---|---|---|---|---|---|---|
| 6 | 19.21 | 38.56 | 0.02 | 0.34 | 0.47 | 1.15 |
| 15 | 18.26 | 38.42 | 0.07 | 0.26 | 1.36 | 1.78 |
| 17 | 16.78 | 38.56 | 0.02 | 0.05 | 0.04 | 0.84 |
| 20 | 18.13 | 38.34 | 0.04 | 1.13 | 0.65 | 3.52 |
| 22 | 15.25 | 37.83 | 0.15 | 2.03 | 3.47 | 4.14 |
| 28 | 15.87 | 37.92 | 0.18 | 1.82 | 2.50 | 4.12 |
| 29 | 14.84 | 38.02 | 2.23 | 12.06 | 24.15 | 22.25 |
| F1 | 17.37 | 39.23 | 0.02 | 0.05 | 0.19 | 0.58 |
| F3 | 17.43 | 39.23 | 0.02 | 0.05 | 0.70 | 0.45 |
| F5 | 17.35 | 39.23 | 0.02 | 0.05 | 0.72 | 0.56 |
| M2 | 17.46 | 38.71 | 0.02 | 0.05 | 0.04 | 1.18 |
DISCUSSION
The cell abundance is consistent with the previous reports of the P. hartmannii bloom in the Gulf of California, Mexico (Gárate-Lizárraga et al., 2009: max. 3.5 x 104 cells L-1; Gárate-Lizárraga, 2014: max. 3.3 x 104 cells L-1), but lower than others reported from Korea (Kim et al., 1990: 1 x 106 cells L-1; Thangaraj et al., 2017: 1.1 x 106 cells L-1) and the USA (Tang et al., 2013: max. 8.3 x 106 cells L-1). An exceptional bloom of P. hartmannii was reported in the northern part of the state of Guerrero, Mexico, which reached densities of 5263 x 103 cells L-1 (Gárate-Lizárraga et al., 2016).
-
Gárate-Lizárraga et al., 2009A multi-species microalgae bloom in Bahía de La Paz, Gulf of California, Mexico (June 2008)CICIMAR Oceánides, 2009
-
Gárate-Lizárraga, 2014Proliferation of Levanderina fissa and Polykrikos hartmannii (Dinophyceae: Gymnodiniales) in Bahía de La Paz, Gulf of California, MéxicoCICIMAR Oceánides, 2014
-
Kim et al., 1990Coastal algal blooms caused by the cyst-forming dinoflagellatesBulletin of The Korean Society, 1990
-
Thangaraj et al., 2017Molecular cloning reveals co-occurring species behind red tide blooms of the harmful dinoflagellate Cochlodinium polykrikoidesBiochemical Systematics and Ecology, 2017
-
Tang et al., 2013Morphology, phylogeny, dynamics, and ichthyotoxicity of Pheopolykrikos hartmannii (Dinophyceae) isolates and blooms from New York, USAJournal of Phycology, 2013
-
Gárate-Lizárraga et al., 2016Florecimientos algales nocivos en las costas del estado de GuerreroFlorecimientos Algales Nocivos en México, 2016
During the study period when the maximum cell abundances of P. hartmannii were observed, it is notable that the values of orthophosphate, nitrite + nitrate nitrogen, and silicate also reached their maximum levels. The presence of such high abundances in the inner bay is primarily a result of its “closeness”, characterized by limited water exchange with the open water of the İzmir Bay. This restricted exchange contributes to an elevated trophic state in the water. P. hartmannii bloom occurs in early fall in İzmir Bay following a heavy rain season. This is consistent with the observations of Gárate-Lizárraga (2014), who noted a similar pattern despite different temperatures, suggesting a potential link between bloom formation and post-rainy season conditions. P. hartmannii also blooms in other seasons, including summer (Gárate-Lizárraga et al., 2009; Thangaraj et al., 2017) and fall (Tang et al., 2013; Kim et al., 1990). While P. hartmannii blooms are typically associated with warmer waters (22 - 31°C; Badylak & Phlips, 2004; Gárate-Lizárraga, 2014, Gárate-Lizárraga et al., 2016; Thangaraj et al., 2017), the maximum abundances in İzmir Bay occurred at a notably lower temperature of 14.84 °C. This was accompanied by a wider temperature range (15 - 28°C) observed throughout the study period by Tang et al. (2013). Aktan and Keskin (2017) observed that the vegetative stage can occur at temperatures as low as 10°C, supporting the adaptability of the species to varying thermal conditions. This highlights the potential for P. hartmannii to bloom under diverse environmental conditions.
-
Gárate-Lizárraga (2014)Proliferation of Levanderina fissa and Polykrikos hartmannii (Dinophyceae: Gymnodiniales) in Bahía de La Paz, Gulf of California, MéxicoCICIMAR Oceánides, 2014
-
Gárate-Lizárraga et al., 2009A multi-species microalgae bloom in Bahía de La Paz, Gulf of California, Mexico (June 2008)CICIMAR Oceánides, 2009
-
Thangaraj et al., 2017Molecular cloning reveals co-occurring species behind red tide blooms of the harmful dinoflagellate Cochlodinium polykrikoidesBiochemical Systematics and Ecology, 2017
-
Tang et al., 2013Morphology, phylogeny, dynamics, and ichthyotoxicity of Pheopolykrikos hartmannii (Dinophyceae) isolates and blooms from New York, USAJournal of Phycology, 2013
-
Kim et al., 1990Coastal algal blooms caused by the cyst-forming dinoflagellatesBulletin of The Korean Society, 1990
-
Badylak & Phlips, 2004Spatial and temporal patterns of phytoplankton composition in subtropical coastal lagoon, the Indian River Lagoon, Florida, USAJournal of Plankton Research, 2004
-
Gárate-Lizárraga, 2014Proliferation of Levanderina fissa and Polykrikos hartmannii (Dinophyceae: Gymnodiniales) in Bahía de La Paz, Gulf of California, MéxicoCICIMAR Oceánides, 2014
-
Gárate-Lizárraga et al., 2016Florecimientos algales nocivos en las costas del estado de GuerreroFlorecimientos Algales Nocivos en México, 2016
-
Thangaraj et al., 2017Molecular cloning reveals co-occurring species behind red tide blooms of the harmful dinoflagellate Cochlodinium polykrikoidesBiochemical Systematics and Ecology, 2017
-
Tang et al. (2013)Morphology, phylogeny, dynamics, and ichthyotoxicity of Pheopolykrikos hartmannii (Dinophyceae) isolates and blooms from New York, USAJournal of Phycology, 2013
-
Aktan and Keskin (2017)Second habitat record of Polykrikos hartmannii W. Zimm. (Dinophyceae) in the south Aegean Sea, Eastern MediterraneanTurkish Journal of Fisheries and Aquatic Sciences, 2017
Laboratory studies have shown that P. hartmannii possesses several key features that likely contribute to its successful bloom formation. Lee et al. (2015) found evidence of a mixotrophic nature, allowing it to utilize both light and organic matter for nutrition. This mixotrophy probably provides a competitive advantage during blooms, potentially explaining the observed monospecific blooms. Chai et al. (2020) demonstrated its ability for homothallic reproduction, which simplifies reproduction and potentially aids in population maintenance and growth during blooms. Tang et al. (2013) showed acute ichtyotoxicity in a laboratory culture. Kwak et al. (2022) demonstrated a wide temperature tolerance for germination (10 - 30°C), with high germination rates exceeding 90 % at 15 - 20 °C in a laboratory setting. This flexibility in germination temperature likely contributes to the bloom-forming potential of this species. Overall, these studies conducted in a laboratory setting highlight several factors that may contribute to the success of P. hartmannii blooms.
-
Lee et al. (2015)Mixotrophy in the nematocyst-taeniocyst complex-bearing phototrophic dinoflagellate Polykrikos hartmanniiHarmful Algae, 2015
-
Chai et al. (2020)Proof of homothally of Pheopolykrikos hartmannii and details of cyst germination processJournal of Oceanology and Limnology, 2020
-
Tang et al. (2013)Morphology, phylogeny, dynamics, and ichthyotoxicity of Pheopolykrikos hartmannii (Dinophyceae) isolates and blooms from New York, USAJournal of Phycology, 2013
-
Kwak et al. (2022)Resting cysts and vegetative cells of Pheopolykrikos hartmannii (W. Zimmermann) Matsuoka & Fukuyo (Dinophyceae): Morphology, phylogeny, and effect of temperature on germinationKorean Journal of Environmental Biology, 2022
Globally, most records of P. hartmannii document its presence in cyst form (Matsuoka & Fukuyo, 1986; Morquecho & Lechuga-Devéze, 2003; Wang et al., 2004; Pospelova et al., 2004; Fuji & Matsuoka, 2006; Mizushima et al., 2007; D’costa et al., 2008; Pospelova & Kim, 2010; Pospelova et al., 2010; Price & Pospelova, 2011; Fertouna-Bellakhal et al., 2014; Balkis et al., 2016; Rubino et al., 2017; Di Poi et al., 2019; Dzhembekova et al., 2020; Rachman et al., 2022; Kwak et al., 2022; Liu et al., 2023). Records of its motile or vegetative form are less common (Hulburt, 1957; Matsuoka & Fukuyo, 1986; Peña-Manjarrez et al., 2005; Kim et al., 2008; Hoppenrath et al., 2009., 2010; Escobar-Morales & Hernández-Becerril, 2015; Aktan & Keskin, 2017; Chai et al., 2020). Blooms of this dinoflagellate occur in diverse coastal habitats worldwide, including the Mexican Pacific (Gárate-Lizárraga et al., 2009, 2016; Gárate-Lizárraga, 2014), the South Sea and East Sea in Korea (Kim et al., 1990; Thangaraj et al., 2017), and the USA (Badylak & Phlips, 2004; Tang et al., 2013).
-
Matsuoka & Fukuyo, 1986Cyst and motile morphology of a colonial dinoflagellate Pheopolykrikos hartmannii (Zinunermann) comb. novJournal of Plankton Research, 1986
-
Morquecho & Lechuga-Devéze, 2003Dinoflagellate cysts in recent sediments from Bahía Concepción, Gulf of CaliforniaBotanica Marina, 2003
-
Wang et al., 2004Dinoflagellate cyst records in recent sediments from Daya Bay, South China SeaPhycological Research, 2004
-
Pospelova et al., 2004Environmental factors influencing the spatial distribution of dinoflagellate cyst assemblages in shallow lagoons of southern New England (USA)Review of Palaeobotany and Palynology, 2004
-
Fuji & Matsuoka, 2006Seasonal change of dinoflagellates cyst flux collected in a sediment trap in Omura Bay, West JapanJournal of Plankton Research, 2006
-
Mizushima et al., 2007Vertical distribution of Pyrodinium bahamense var. compressum (Dinophyceae) cysts in Ambon Bay and Hurun Bay, IndonesiaPlankton and Benthos Research, 2007
-
D’costa et al., 2008Dinoflagellates in a mesotrophic, tropical environment influenced by monsoonEstuarine, Coastal and Shelf Science, 2008
-
Pospelova & Kim, 2010Dinoflagellate cysts in recent estuarine sediments from aquaculture sites of southern South KoreaMarine Micropaleontology, 2010
-
Pospelova et al., 2010Organic-walled dinoflagellate cyst production, composition and flux from 1996 to 1998 in the central Strait of Georgia (BC, Canada): A sediment trap studyMarine Micropaleontology, 2010
-
Price & Pospelova, 2011High-resolution sediment trap study of organic-walled dinoflagellate cyst production and biogenic silica flux in Saanich Inlet (BC, Canada)Marine Micropaleontology, 2011
-
Fertouna-Bellakhal et al., 2014Driving factors behind the distribution of dinocyst composition and abundance in surface sediments in a western Mediterranean coastal lagoon: report from a high resolution mapping studyMarine Pollution Bulletin, 2014
-
Balkis et al., 2016Dinoflagellate resting cysts in recent marine sediments from the Gulf of Gemlik (Marmara Sea, Turkey) and seasonal harmful algal bloomsPhycologia, 2016
-
Rubino et al., 2017Plankton resting stages in recent sediments of Haifa port, Israel (Eastern Mediterranean)-Distribution, viability and potential environmental consequencesMarine Pollution Bulletin, 2017
-
Di Poi et al., 2019Dinoflagellate resting cysts from surface sediments of the Adriatic Ports: Distribution and potential spreading patternsMarine Pollution Bulletin, 2019
-
Dzhembekova et al., 2020Comparative analysis of morphological and molecular approaches integrated into the study of the dinoflagellate biodiversity within the recently deposited Black Sea sediments - benefits and drawbacksBiodiversity Data Journal, 2020
-
Rachman et al., 2022Dinoflagellate Cyst Distribution in Relation to the Sediment Composition and Grain Size in the Coastal Area of Pangkajene, South Sulawesi, IndonesiaILMU KELAUTAN: Indonesian Journal of Marine Sciences, 2022
-
Kwak et al., 2022Resting cysts and vegetative cells of Pheopolykrikos hartmannii (W. Zimmermann) Matsuoka & Fukuyo (Dinophyceae): Morphology, phylogeny, and effect of temperature on germinationKorean Journal of Environmental Biology, 2022
-
Liu et al., 2023A combined approach detected novel species diversity and distribution of dinoflagellate cysts in the Yellow Sea, ChinaMarine Pollution Bulletin, 2023
-
Hulburt, 1957The Taxonomy of Unarmored Dinophyceae of Shallow Embayments on Cape Cod, MassachusettsBiological Bulletin, 1957
-
Matsuoka & Fukuyo, 1986Cyst and motile morphology of a colonial dinoflagellate Pheopolykrikos hartmannii (Zinunermann) comb. novJournal of Plankton Research, 1986
-
Peña-Manjarrez et al., 2005Dinoflagellate cysts and bloom events at Todos Santos Bay, Baja California, México, 1999-2000Continental Shelf Research, 2005
-
Kim et al., 2008Research Note: Molecular phylogenetic affiliations of Dissodinium pseudolunula, Pheopolykrikos hartmannii, Polykrikos cf. schwartzii and Polykrikos kofoidii to Gymnodinium sensu stricto species (Dinophyceae)Phycological Research, 2008
-
Hoppenrath et al., 2009Molecular phylogeny of ocelloid-bearing dinoflagellates (Warnowiaceae) as inferred from SSU and LSU rDNA sequencesBMC Ecology and Evolution, 2009
-
2010Re-classification of Pheopolykrikos hartmannii as Polykrikos (Dinophyceae) based partly on the ultrastructure of complex extrusomesEuropean Journal of Protistology, 2010
-
Escobar-Morales & Hernández-Becerril, 2015Free-living marine planktonic unarmoured dinoflagellates from the Gulf of Mexico and the Mexican PacificBotanica Marina, 2015
-
Aktan & Keskin, 2017Second habitat record of Polykrikos hartmannii W. Zimm. (Dinophyceae) in the south Aegean Sea, Eastern MediterraneanTurkish Journal of Fisheries and Aquatic Sciences, 2017
-
Chai et al., 2020Proof of homothally of Pheopolykrikos hartmannii and details of cyst germination processJournal of Oceanology and Limnology, 2020
-
Gárate-Lizárraga et al., 2009A multi-species microalgae bloom in Bahía de La Paz, Gulf of California, Mexico (June 2008)CICIMAR Oceánides, 2009
-
2016Florecimientos algales nocivos en las costas del estado de GuerreroFlorecimientos Algales Nocivos en México, 2016
-
Gárate-Lizárraga, 2014Proliferation of Levanderina fissa and Polykrikos hartmannii (Dinophyceae: Gymnodiniales) in Bahía de La Paz, Gulf of California, MéxicoCICIMAR Oceánides, 2014
-
Kim et al., 1990Coastal algal blooms caused by the cyst-forming dinoflagellatesBulletin of The Korean Society, 1990
-
Thangaraj et al., 2017Molecular cloning reveals co-occurring species behind red tide blooms of the harmful dinoflagellate Cochlodinium polykrikoidesBiochemical Systematics and Ecology, 2017
-
Badylak & Phlips, 2004Spatial and temporal patterns of phytoplankton composition in subtropical coastal lagoon, the Indian River Lagoon, Florida, USAJournal of Plankton Research, 2004
-
Tang et al., 2013Morphology, phylogeny, dynamics, and ichthyotoxicity of Pheopolykrikos hartmannii (Dinophyceae) isolates and blooms from New York, USAJournal of Phycology, 2013
Previous studies have identified both the cyst (Gómez, 2003; Fertouna-Bellakhal et al., 2014; Rubino et al., 2017) and vegetative forms (Aktan & Keskin, 2017) of P. hartmannii in the Mediterranean Sea. This study represents the first recorded bloom of the species in the Aegean Sea. Although the dinoflagellate cyst composition has been analyzed in İzmir Bay (Uzar et al., 2010; Aydin et al., 2011; Aydin & Uzar, 2013; Aydin et al., 2014; 2015), P. hartmannii was previously unrecorded in studies of the region’s dinoflagellate cyst composition. The inner bay, where the bloom occurred in November 2023, is the shallowest area with limited water circulation and significant riverine inflows. Sediment dredging activities carried out in this area to enhance limited water circulation may have introduced or reintroduced the P. hartmannii resting cysts into İzmir Bay. These disturbances, combined with the favorable temperature regime, potentially explain the sudden appearance of P. hartmannii in the bay. However, an alternative explanation for the previously undetected presence of P. hartmannii in İzmir Bay lies in the inherent fragility of its cells. Their delicate morphology could cause the cells to deform during the standard fixation procedures used for microscopic identification, potentially leading to cell shape changes. When combined with a lack of distinctive characteristics, identification becomes significantly more complex (Gómez et al., 2024). Because of the fragility of P. hartmannii cell wall, it is necessary for future studies to address molecular identification techniques (for example, Single-cell PCR), since these morphotypes could present genetic divergence compared to other morphotypes collected in distant regions. Furthermore, the rapid mobility and light sensitivity of dinoflagellates can hinder identification even when analyzing living samples (Gárate-Lizárraga et al., 2009). Microscopic examinations revealed a decrease in motility of live cells exposed to light for 20 minutes, followed by complete cessation of movement, cell swelling, and ultimately, cell lysis (Supplementary Material Video 1 and Video 2).
-
Gómez, 2003Checklist of Mediterranean Free-living DinoflagellatesBotanica Marina, 2003
-
Fertouna-Bellakhal et al., 2014Driving factors behind the distribution of dinocyst composition and abundance in surface sediments in a western Mediterranean coastal lagoon: report from a high resolution mapping studyMarine Pollution Bulletin, 2014
-
Rubino et al., 2017Plankton resting stages in recent sediments of Haifa port, Israel (Eastern Mediterranean)-Distribution, viability and potential environmental consequencesMarine Pollution Bulletin, 2017
-
Aktan & Keskin, 2017Second habitat record of Polykrikos hartmannii W. Zimm. (Dinophyceae) in the south Aegean Sea, Eastern MediterraneanTurkish Journal of Fisheries and Aquatic Sciences, 2017
-
Uzar et al., 2010Dinoflagellate cyst assemblages in surface sediments from Homa Lagoon (Izmir Bay, eastern Aegean Sea, the Mediterranean)Scientific Research and Essays, 2010
-
Aydin et al., 2011Distribution of dinoflagellate cysts in recent sediments from Izmir Bay (Aegean Sea, Eastern Mediterranean)Fuel and Energy Abstracts, 2011
-
Aydin & Uzar, 2013Some potentially toxic dinoflagellate cysts in recent sediments from İzmir BayEge Journal of Fisheries and Aquatic Sciences, 2013
-
Aydin et al., 2014Dinoflagellate cyst assemblages in surface sediments from Homa Lagoon (Izmir Bay, eastern Aegean Sea, the Mediterranean)Fresenius Environmental Bulletin, 2014
-
2015Impact of industrial pollution on recent dinoflagellate cysts in Izmir Bay (Eastern Aegean)Marine Pollution Bulletin, 2015
-
Gómez et al., 2024Misidentifications of the bloom-forming dinoflagellates Gymnodinium litorale and Margalefidinium polykrikoides in the Mediterranean SeaRegional Studies in Marine Science, 2024
-
Gárate-Lizárraga et al., 2009A multi-species microalgae bloom in Bahía de La Paz, Gulf of California, Mexico (June 2008)CICIMAR Oceánides, 2009
Further monitoring of the unarmored dinoflagellate P. hartmannii motile and cyst forms in İzmir Bay, including investigations into the physicochemical and biological factors driving its bloom formation, is crucial for understanding its ecological role and potential impacts. These efforts will yield invaluable insights for future management and mitigation strategies related to this species. This first observed P. hartmannii bloom highlights the need to investigate bloom-formation mechanisms and potential environmental impacts specific to the Mediterranean region.
ACKNOWLEDGMENTS
This study was supported by the “Izmir Bay, Yenifoça and Seferihisar Akarca Bay Oceanographic Monitoring Project” carried out by Izmir Metropolitan Municipality Directorate General of IZSU and coordinated by TUBITAK-Marmara Research Center. IGL thanks the Instituto Politécnico Nacional, Mexico (grant: SIP-20240611, Secretaría de Investigación y Posgrado); he is a COFAA (Comisión de Operación y Fomento de Actividades Académicas) fellow. We are grateful to Yuri B. Okolodkov (Universidad Veracruzana, Boca del Río, Veracruz, Mexico) and to Marcia M. Gowing (Seattle, WA, USA) for improving the English style.
REFERENCES
- Aktan, Y. & Ç. Keskin. 2017. Second habitat record of Polykrikos hartmannii W. Zimm. (Dinophyceae) in the south Aegean Sea, Eastern Mediterranean. Turkish Journal of Fisheries and Aquatic Sciences 17(5): 1077-1081. DOI:10.4194/1303-2712-v17_5_25 Links
- Anderson, D. M. & D. Wall. 1978. Potential importance of benthic cysts of Gonyaulax tamarensis and G. excavata in initiating toxic dinoflagellate blooms. Journal of Phycology 14: 224-234. DOI:10.1111/j.1529-8817.1978.tb02452.x Links
- Anderson, D. M. , P. M. Glibert & J. M, Burkholder. 2002. Harmful algal blooms and eutrophication: nutrient sources, composition, and consequences. Estuaries 25: 704-726. DOI:10.1007/BF02804901 Links
- Anderson, D. M., J. M. Burkholder, W. P. Cochlan, P. M. Glibert, C. J. Gobler, C. A. Heil, R. M. Kudela, M. L. Parsons, J. E. J. Rensel, D. W. Townsend, V. L. Trainer & G. A. Vargo. 2008. Harmful algal blooms and eutrophication: Examining linkages from selected coastal regions of the United States. Harmful Algae 8(1): 39-53. DOI:10.1016/j.hal.2008.08.017 Links
- Anderson, D. M., A. D. Cembella & G. M. Hallegraeff. 2012. Progress in understanding harmful algal blooms: paradigm shifts and new technologies for research, monitoring, and management. The Annual Review of Marine Science 4: 143-176. DOI:10.1146/annurev-marine-120308-081121 Links
- Aydin, H., K. Matsuoka & E. Minareci. 2011. Distribution of dinoflagellate cysts in recent sediments from Izmir Bay (Aegean Sea, Eastern Mediterranean). Fuel and Energy Abstracts 80: 44-52. DOI:10.1016/j.marmicro.2011.03.004 Links
- Aydin, H. & S. Uzar. 2013. Some potentially toxic dinoflagellate cysts in recent sediments from İzmir Bay. Ege Journal of Fisheries and Aquatic Sciences 30(3): 109-114. DOI:10.12714/egejfas.2013.30.3.04 Links
- Aydin, H., E. E. Yurur & S. Uzar . 2014. Dinoflagellate cyst assemblages in surface sediments from Homa Lagoon (Izmir Bay, eastern Aegean Sea, the Mediterranean). Fresenius Environmental Bulletin 23(8): 1795-1801. Links
- Aydin, H., E. E. Yurur, S. Uzar & F. Kucuksezgin. 2015. Impact of industrial pollution on recent dinoflagellate cysts in Izmir Bay (Eastern Aegean). Marine Pollution Bulletin 94(1-2): 144-152. DOI:10.1016/j.marpolbul.2015.02.038 Links
- Badylak, S. & E. J. Phlips. 2004. Spatial and temporal patterns of phytoplankton composition in subtropical coastal lagoon, the Indian River Lagoon, Florida, USA. Journal of Plankton Research 26(10): 1229-1247. DOI:10.1093/plankt/fbh114 Links
- Balkis, N., M. Balci, A. Giannakourou, A. Venetsanopoulou & P. Mudie. 2016. Dinoflagellate resting cysts in recent marine sediments from the Gulf of Gemlik (Marmara Sea, Turkey) and seasonal harmful algal blooms. Phycologia 55(2): 187-209. DOI:10.2216/15-93.1 Links
- Bravo, I. & R. I. Figueroa. 2014. Towards an Ecological Understanding of Dinoflagellate Cyst Functions. Microorganisms 2(1): 11-32. DOI:10.3390/microorganisms2010011 Links
- Brown, A. R., M. Lilley, J. Shutler, C. Lowe, Y. Artioli, R. Torres, E. Berdalet & C. R. Tyler. 2020. Assessing risks and mitigating impacts of harmful algal blooms on mariculture and marine fisheries. Reviews in Aquaculture 12(3): 1663-1688. DOI:10.1111/raq.12403 Links
- Chai, Z., Z. Hu, Y. Liu & Y. Z. Tang. 2020. Proof of homothally of Pheopolykrikos hartmannii and details of cyst germination process. Journal of Oceanology and Limnology 38: 114-123. DOI:10.1007/s00343-019-9077-x Links
- Cloern, J. E. 1996. Phytoplankton bloom dynamics in coastal ecosystems: a review with some general lessons from sustained investigation of San Francisco Bay, California. Reviews of Geophysics 34(2): 127-168. DOI:10.1029/96RG00986 Links
- D’Costa, P. M., A. C. Anil. J. S. Patil, S. Hegde, M. S. D’Silva & M. Chourasia. 2008. Dinoflagellates in a mesotrophic, tropical environment influenced by monsoon. Estuarine, Coastal and Shelf Science 77(1): 77-90. DOI:10.1016/j.ecss.2007.09.002 Links
- Di Poi, E., R. Kraus, M. Cabrini, S. Finotto, V. Flander-Putrle, M. Grego, N. Kužat, Ž. Ninčević-Gladan, L. Pezzolesi, E. Riccardi, F. B. Aubry & M. Bastianini. 2019. Dinoflagellate resting cysts from surface sediments of the Adriatic Ports: Distribution and potential spreading patterns. Marine Pollution Bulletin 147: 185-208. DOI:10.1016/j.marpolbul.2019.01.014 Links
- Dzhembekova, N., S. Urusizaki, S. Moncheva, P. Ivanova & S. Nagai. 2017. Applicability of massively parallel sequencing on monitoring harmful algae at Varna Bay in the Black Sea. Harmful Algae 68: 40-51. DOI:10.1016/j.hal.2017.07.004 Links
- Dzhembekova, N. F. Rubino, S. Nagai, I. Zlateva, N. Slabakova, P. Ivanova, V. Slabakova & S. Moncheva. 2020. Comparative analysis of morphological and molecular approaches integrated into the study of the dinoflagellate biodiversity within the recently deposited Black Sea sediments - benefits and drawbacks. Biodiversity Data Journal 8: e55172. DOI:10.3897/BDJ.8.e55172 Links
- Escarcega-Bata, A. J., M. L. Núñez Resendiz, M. C. Ruiz-de la Torre, K. M. Dreckmann, M. E. Zamudio-Resendiz & A. Sentíes. 2023. Diversidad de dinoflagelados atecados del orden Gymnodiniales (Dinophyceae), con énfasis en aquellos formadores de florecimientos algales nocivos en las costas del Pacífico Mexicano. Acta Botanica Mexicana 130: e2126. DOI: 10.21829/abm130.2023.2126 Links
- Escobar-Morales, S. & D. Hernández-Becerril. 2015. Free-living marine planktonic unarmoured dinoflagellates from the Gulf of Mexico and the Mexican Pacific. Botanica Marina 58(1): 9-22. DOI:10.1515/bot-2014-0049 Links
- Eppley, R. W. & W. G. Harrison. 1975. Physiological ecology of Gonyaulax polyedrum, a red tide water dinoflagellate of southern California. In: Le Cicero VR (Eds.). 1st Int. Conf. on Toxic Dinoflagellate Blooms, Massachusetts Science and Technology Foundation, Wakefield, MA. pp. 11-22. Links
- Ferrante, M., S. Sciacca, R. Fallico, M. Fiore, G. O. Conti & C. Ledda. 2013. Harmful algal blooms in the Mediterranean Sea: effects on human health. EuroMediterranean Biomedical Journal 8(6): 25-34. Links
- Fertouna-Bellakhal, M., A. Dhib, B. Béjaoui, S. Turki & L. Aleya. 2014. Driving factors behind the distribution of dinocyst composition and abundance in surface sediments in a western Mediterranean coastal lagoon: report from a high resolution mapping study. Marine Pollution Bulletin 84(1-2): 347-362. DOI:10.1016/j.marpolbul.2014.04.041 Links
- Figueroa, R. I., K. Rengefors, I. Bravo & S. Bensch. 2010. From homothally to heterothally: mating preferences and genetic variation within clones of the dinoflagellate Gymnodinium catenatum. Deep-Sea Research II 57: 190-198. DOI:10.1016/j.dsr2.2009.09.016 Links
- Fire, S. E., A. Bogomolni, R. A. DiGiovanni Jr, G. Early, T. A. Leighfield, K. Matassa, G. A. Miller, K. M. T. Moore, M. Moore, M. Niemeyer, K. Pugliares, Z. Wang & F. W. Wenzel. 2021. An assessment of temporal, spatial and taxonomic trends in harmful algal toxin exposure in stranded marine mammals from the U.S. New England coast. Plos One 16(1): e0243570. DOI:10.1371/journal.pone.0243570 Links
- Franks, P. J. S. & D. M. Anderson. 1992. Alongshore transport of a toxic phytoplankton bloom in a buoyancy current: Alexandrium tamarense in the Gulf of Maine. Marine Biology 112: 153-164. DOI:10.1007/BF00349739 Links
- Fujii, R. & K. Matsuoka. 2006. Seasonal change of dinoflagellates cyst flux collected in a sediment trap in Omura Bay, West Japan. Journal of Plankton Research 28(2): 131-147. DOI:10.1093/plankt/fbi106 Links
- Gárate-Lizárraga, I., C. J. Band-Schmidt, F. Aguirre-Bahena & T. Grayeb del Alamo. 2009. A multi-species microalgae bloom in Bahía de La Paz, Gulf of California, Mexico (June 2008). CICIMAR Oceánides 24(1): 15-29. DOI:10.37543/oceanides.v24i1.50 Links
- Gárate-Lizárraga, I. 2014. Proliferation of Levanderina fissa and Polykrikos hartmannii (Dinophyceae: Gymnodiniales) in Bahía de La Paz, Gulf of California, México. CICIMAR Oceánides 29(2): 25-35. DOI:10.37543/oceanides.v29i2.137 Links
- Gárate-Lizárraga, I., B. Pérez-Cruz, J. A. Diaz-Ortiz, Y. B. Okolodkov & S. López-Silva. 2016. Florecimientos algales nocivos en las costas del estado de Guerrero. In: García-Mendoza, E., S. I. Quijano-Scheggia, A. Olivos-Ortiz & E. J. Núñez-Vázquez (Eds.). Florecimientos Algales Nocivos en México. CICESE, Ensenada, México, Pp. 228-241. Links
- Godhe, A., I. Karunasagar & B. Karlson. 2000. Dinoflagellate Cysts in Recent Marine Sediments from SW India. Botanica Marina 43: 39-48. DOI:10.1515/BOT.2000.004 Links
- Gómez, F. 2003. Checklist of Mediterranean Free-living Dinoflagellates. Botanica Marina 46: 215-242. DOI:10.1515/BOT.2003.021 Links
- Gómez, F., L. Roselli, H. Zhang & S. Lin. 2024. Misidentifications of the bloom-forming dinoflagellates Gymnodinium litorale and Margalefidinium polykrikoides in the Mediterranean Sea. Regional Studies in Marine Science 70: 103376. DOI:10.1016/j.rsma.2024.103376 Links
- Grattan, L. M., S. Holobaugh & J. G. Morris. 2016. Harmful algal blooms and public health. Harmful Algae 57: 2-8. DOI:10.1016/j.hal.2016.05.003 Links
- Guiry, M. D. & G. M. Guiry. 2024. AlgaeBase. World-wide electronic publication, National University of Ireland, Galway. Available online at: https://www.algaebase.org (accessed 7 May 2024). Links
- Hallegraeff, G. M. & C. J. Bolch. 1991. Transport of toxic dinoflagellate cysts via ships’ ballast water. Marine Pollution Bulletin 22: 27-30. DOI:10.1016/0025-326X(91)90441-T Links
- Hallegraeff, G., D. M. Anderson, C. Belin, M. Y. Dechraoui Bottein, E. Bresnan, M. Chinain, H. Enevoldsen, M. Iwataki, B. Karlson, C. H. McKenzie, I. Sunesen, G. C. Pitcher, P. Provoost, A. Richardson, L. Schweibold, P. A. Tester, V. L Trainer, A. T. Yñiguez & A. Zingone. 2021. Perceived global increase in algal blooms isattributable to intensified monitoring and emergingbloom impacts. Communications Earth & Environment 2:117. DOI:10.1038/s43247-021-00178-8 Links
- Head, M. J. 1996. Modern dinoflagellate cysts and their biological affinities. Chapter 30 In: Jansonius J & D. C. McGregor, (Eds.). Palynology, principles and applications. American Association of Stratigraphic Palynologists Foundation, Vol 3. pp 1197-1248. Links
- Hii, K. S., M. Mohd-Din, Z. Luo, S. N. Tan, Z. F. Lim, L. K. Lee, S. C. Y. Leong, S. T. Teng, H. Gu, X. Cao, P. T. Lim & C. P. Leaw. 2021. Diverse harmful microalgal community assemblages in the Johor Strait and the environmental effects on its community dynamics. Harmful Algae 107: 102077. DOI:10.1016/j.hal.2021.102077 Links
- Holmes, R. W., P. M. Williams & R. W. Eppley. 1967. Red water in La Jolla Bay, 1964-1966. Limnology and Oceanography 12: 503-512. DOI:10.4319/lo.1967.12.3.0503 Links
- Hoppenrath, M., T. R. Bachvaroff, S. M. Handy, C. F. Delwiche & B. S. Leander. 2009. Molecular phylogeny of ocelloid-bearing dinoflagellates (Warnowiaceae) as inferred from SSU and LSU rDNA sequences. BMC Ecology and Evolution 9(1): 116. DOI:10.1186/1471-2148-9-116 Links
- Hoppenrath, M., N. Yubuki, T. R. Bachvaroff & B. S. Leander. 2010. Re-classification of Pheopolykrikos hartmannii as Polykrikos (Dinophyceae) based partly on the ultrastructure of complex extrusomes. European Journal of Protistology 46(1): 29-37. DOI:10.1016/j.ejop.2009.08.003 Links
- Hulburt, E. M. 1957. The Taxonomy of Unarmored Dinophyceae of Shallow Embayments on Cape Cod, Massachusetts. Biological Bulletin 112(2): 196-219. DOI:10.2307/1539198 Links
- Imai, I., M. Yamaguchi & Y. Hori. 2006. Eutrophication and occurrences of harmful algal blooms in the Seto Inland Sea, Japan. Plankton and Benthos Research 1(2): 71-84. DOI:10.3800/pbr.1.71 Links
- Jeong, H. J., Y. D. Yoo, K. H. Lee, T. H. Kim, K. A. Seong, N. S. Kang, S. Y. Lee, J. S. Kim, S. Kim & W. H. Yih. 2013. Red tides in Masan Bay, Korea in 2004-2005: I. Daily variations in the abundance of red-tide organisms and environmental factors. Harmful Algae 30 (Suppl.1): 75-88. DOI:10.1016/j.hal.2013.10.008 Links
- Kim, H. G., J. S. Park & S. G. Lee. 1990. Coastal algal blooms caused by the cyst-forming dinoflagellates. Bulletin of The Korean Society 23(6): 468-474. Links
- Kim, K.Y., M. Iwataki & C. H. Kim. 2008. Research Note: Molecular phylogenetic affiliations of Dissodinium pseudolunula, Pheopolykrikos hartmannii, Polykrikos cf. schwartzii and Polykrikos kofoidii to Gymnodinium sensu stricto species (Dinophyceae). Phycological Research 56(2): 89-92. DOI:10.1111/j.1440-1835.2008.00489.x Links
- Kontas, A., F. Kucuksezgin, O. Altay & E. Uluturhan. 2004. Monitoring of eutrophication and nutrient limitation in the Izmir Bay (Turkey) before and after wastewater treatment plant. Environment International 29: 1057-1062. DOI:10.1016/S0160-4120(03)00098-9 Links
- Kwak, K. Y., J. Y. Youn, H. J. Kim, K. H. Han, Z. Li & H. H. Shin. 2022. Resting cysts and vegetative cells of Pheopolykrikos hartmannii (W. Zimmermann) Matsuoka & Fukuyo (Dinophyceae): Morphology, phylogeny, and effect of temperature on germination. Korean Journal of Environmental Biology 40(4): 387-397. DOI:10.11626/KJEB.2022.40.4.387 Links
- Lee, M. J., H. J. Jeong, K. H. Lee, S. H. Jang, J. H. Kim & K. Y. Kim. 2015. Mixotrophy in the nematocyst-taeniocyst complex-bearing phototrophic dinoflagellate Polykrikos hartmannii. Harmful Algae 49: 124-134. DOI:10.1016/j.hal.2015.08.006 Links
- Liu, X., Y. Liu, Z. Chai, Z. Hu & Y. Z. Tang . 2023. A combined approach detected novel species diversity and distribution of dinoflagellate cysts in the Yellow Sea, China. Marine Pollution Bulletin 187:114567. DOI:10.1016/j.marpolbul.2022.114567 Links
- Matsuoka, K. & Y. Fukuyo. 1986. Cyst and motile morphology of a colonial dinoflagellate Pheopolykrikos hartmannii (Zinunermann) comb. nov. Journal of Plankton Research 8(4): 811-818. DOI:10.1093/plankt/8.4.811 Links
- Matsuoka, K. & Y. Fukuyo. 2002. Technical Guide for Modern Dinoflagellate Cyst Study. WESTPAC-HAB/WEATPAC/IOC, Japanese Society for the Promotion of Science, pp. 29. Links
- Mizushima, K., K. Matsuoka & Y. Fukuyo. 2007. Vertical distribution of Pyrodinium bahamense var. compressum (Dinophyceae) cysts in Ambon Bay and Hurun Bay, Indonesia. Plankton and Benthos Research 2(4):163-174. DOI:10.3800/pbr.2.163 Links
- Montes, R., X. Rojas, P. Artacho, A. Tello & R. Quinones. 2018. Quantifying harmful algal bloom thresholds for farmed salmon in southern Chile. Harmful Algae 77: 55-65. DOI:10.1016/j.hal.2018.05.004 Links
- Morquecho, L. & C. Lechuga-Devéze. 2003. Dinoflagellate cysts in recent sediments from Bahía Concepción, Gulf of California. Botanica Marina 46: 132-141. DOI:10.1515/BOT.2003.014 Links
- MMG. Marine Monitoring Guidelines. 2017. Ministry of Environment and Urbanization of the Republic of Turkey, Directorate General Environmental Impact Assessment, Permit and Inspection, TÜBİTAKMAM Press. ISBN: 978-605-5294-84-7, pp. 442. Links
- Otero, P. & M. Silva. 2022. Emerging Marine Biotoxins in European Waters: Potential Risks and Analytical Challenges. Marine Drugs 20(3): 199. DOI:10.3390/md20030199 Links
- Park, T. G., W. A. Lim, Y. T. Park, C. K. Lee & H. J. Jeong. 2013. Economic impact, management and mitigation of red tides in Korea. Harmful Algae 30: 131-143. DOI:10.1016/j.hal.2013.10.012 Links
- Park, B. S., J. H. Kim, J. H. Kim, S. H. Baek & M. S. Han. 2018. Intraspecific bloom succession in the harmful dinoflagellate Cochlodinium polykrikoides (Dinophyceae) extended the blooming period in Korean coastal waters in 2009. Harmful Algae 71: 78-88. DOI:10.1016/j.hal.2017.12.004 Links
- Peña-Manjarrez, J., J. Helenes, G. Gaxiola-Castro & E. Orellana-Cepeda. 2005. Dinoflagellate cysts and bloom events at Todos Santos Bay, Baja California, México, 1999-2000. Continental Shelf Research 25: 1375-1393. DOI:10.1016/j.csr.2005.02.002 Links
- Pospelova, V., G. L. Chmura & H. A. Walker. 2004. Environmental factors influencing the spatial distribution of dinoflagellate cyst assemblages in shallow lagoons of southern New England (USA). Review of Palaeobotany and Palynology 128(1-2): 7-34. DOI:10.1016/S0034-6667(03)00110-6 Links
- Pospelova, V. & S. J. Kim. 2010. Dinoflagellate cysts in recent estuarine sediments from aquaculture sites of southern South Korea. Marine Micropaleontology 76(1-2): 37-51. DOI:10.1016/j.marmicro.2010.04.003 Links
- Pospelova, V., S. Esenkulova, S. C. Johannessen, M. C. O’Brien & R. W. Macdonald. 2010. Organic-walled dinoflagellate cyst production, composition and flux from 1996 to 1998 in the central Strait of Georgia (BC, Canada): A sediment trap study. Marine Micropaleontology 75(1-4): 17-37. DOI:10.1016/j.marmicro.2010.02.003 Links
- Price, A. M. & V. Pospelova. 2011. High-resolution sediment trap study of organic-walled dinoflagellate cyst production and biogenic silica flux in Saanich Inlet (BC, Canada). Marine Micropaleontology 80(1-2): 18-43. DOI:10.1016/j.marmicro.2011.03.003 Links
- Rachman, A., H. Thoha, M. D. B. Intan, O. R. Sianturi, Y. Witasari, S. P. A. Wibowo & M. Iwataki. 2022. Dinoflagellate Cyst Distribution in Relation to the Sediment Composition and Grain Size in the Coastal Area of Pangkajene, South Sulawesi, Indonesia. ILMU KELAUTAN: Indonesian Journal of Marine Sciences 27(2): 111-123. DOI:10.14710/ik.ijms.27.2.111-123 Links
- Rubino, F., M. Belmonte & B. S. Galil. 2017. Plankton resting stages in recent sediments of Haifa port, Israel (Eastern Mediterranean)-Distribution, viability and potential environmental consequences. Marine Pollution Bulletin 116(1-2): 258-269. DOI:10.1016/j.marpolbul.2016.12.078 Links
- Rukminasari, N. & A. Tahir. 2021. Species Assemblages and Distribution of Dinoflagellate Cysts from three Estuaries Sediment’s of Makassar Strait, Eastern Indonesia. OnLine Journal of Biological Sciences 21(2): 232-244. DOI:10.3844/ojbsci.2021.232.244 Links
- Shin, H. H., Y. H. Yoon, Y. O. Kim & K. Matsuoka. 2011. Dinoflagellate cysts in surface sediments from southern coast of Korea. Estuaries and Coasts 34: 712-725. DOI:10.1007/s12237-011-9373-y Links
- Smayda, T. J. 1997. Harmful algal blooms: Their ecophysiology and general relevance to phytoplankton blooms in the sea. Limnology and Oceanography 42: 1137-1153. DOI:10.4319/lo.1997.42.5_part_2.1137 Links
- Steidinger, K. A. & K. Tangen. 1997. Dinoflagellates. In: Tomas C.R. (Eds.) Identifying marine phytoplankton. Academic Press, San Diego, U.S.A. pp 448-449. DOI:10.1016/B978-012693018-4/50005-7 Links
- Tang, Y. Z., M. J. Harke & C. J. Gobler. 2013. Morphology, phylogeny, dynamics, and ichthyotoxicity of Pheopolykrikos hartmannii (Dinophyceae) isolates and blooms from New York, USA. Journal of Phycology 49(6): 1084-1094. DOI:10.1111/jpy.12114 Links
- Tang, Y. Z. & C. J. Gobler. 2015. Sexual resting cyst production by the dinoflagellate Akashiwo sanguinea: a potential mechanism contributing to the ubiquitous distribution of a harmful alga. Journal of Phycology 51(2): 298-309. DOI:10.1111/jpy.12274 Links
- Thangaraj, P., T. G. Park & J. S. Ki. 2017. Molecular cloning reveals co-occurring species behind red tide blooms of the harmful dinoflagellate Cochlodinium polykrikoides. Biochemical Systematics and Ecology 70: 29-34. DOI:10.1016/j.bse.2016.10.021 Links
- Uzar, S., H. Aydin & E. Minareci. 2010. Dinoflagellate cyst assemblages in surface sediments from Homa Lagoon (Izmir Bay, eastern Aegean Sea, the Mediterranean). Scientific Research and Essays 5(3): 285-295. Links
- Wang, Z., K. Matsuoka, Y. Qi, J. Chen & S. Lu. 2004. Dinoflagellate cyst records in recent sediments from Daya Bay, South China Sea. Phycological Research 52(4): 396-407. DOI:10.1111/j.1440-1835.2004.tb00348.x Links
- Yang, A., Z. Hu & Y. Tang. 2018. Solid sand particle addition can enhance the production of resting cysts in dinoflagellates. Journal of Oceanology and Limnology 36(2): 273-280. DOI:10.1007/s00343-018-6291-x Links
- Zimmermann, W. 1930. Neue und wenig bekannte Kleinalgen von Neapel IV. Zeitschrift für Botanik 23: 421-442. Links