Review articles
Rates and socioecological drivers of mangrove forest cover loss in Honduras: a systematic review
-
Publication dates-
January 06, 2026
Jan-Dec , 2025
- Article in PDF
- Article in XML
- Automatic translation
- Send this article by e-mail
- Share this article +
Abstract.
In Honduras, mangrove forests have experienced considerable decline, and to date, there is no synthesis addressing forest cover loss at the national level and its drivers. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines were used to conduct a systematic review to analyze forest cover loss in Honduran mangroves and the socioecological drivers that have influenced it over time. The literature review revealed that a total of 68,766.34 ha of mangrove forest cover were lost between 1965 and 2024. The areas most affected by deforestation were the Pacific coast of Honduras (PCH), with a loss of 54,409.75 ha and an annual rate of 1.51%, followed by the Atlantic coast of Honduras (ACH), particularly the departments of Cortés (loss: 1,377.83 ha; annual rate: 2.62%) and Gracias a Dios (loss: 10,260.23 ha; annual rate: 1.39%). Aquaculture and agriculture emerged as the main drivers, especially in the PCH (Gulf of Fonseca). On the other hand, urban development and industrialization played a predominant role in the ACH. Furthermore, the effects of climate change, particularly sea level rise, emerged as key drivers resulting in considerable mangrove losses in Cortés. The findings of this review highlight the importance of investigating national and regional variations in mangrove loss in Honduras and its underlying causes. With this information, more effective conservation measures tailored to the specific circumstances of each area can be designed to safeguard the sustainability of these valuable ecosystems.
Key words::
aquaculture, agriculture, climate change, coastal development, drivers, coastal wetlands
Introduction
Mangroves provide a unique connection between terrestrial and marine ecosystems and host a rich biodiversity of flora and fauna species that are critical to the livelihood of millions of people due to their ecological and economic importance (McKee et al. 2007, Bhowmik et al. 2022, Moore et al. 2022, Hamza et al. 2024). These coastal environments provide ecological and social benefits along tropical and subtropical coasts (Lee et al. 2014, Canty et al. 2022, Moore et al. 2022). Despite their crucial role in maintaining coastal biodiversity and providing essential ecosystem services, mangroves have suffered an alarming global loss, with estimates of at least 35% since the 1960s, due to their natural fragility and the impacts of human activity (Valiela et al. 2001, Bryan-Brown et al. 2020, Moore et al. 2022). The loss of cover and resulting fragmentation have also substantially affected the provision of ecosystem services (Feller et al. 2017).
-
McKee et al. 2007Caribbean mangroves adjust to rising sea level through biotic controls on change in soil elevationGlobal Ecol Biogeogr, 2007
-
Bhowmik et al. 2022Global mangrove deforestation and its interacting social-ecological drivers: a systematic review and synthesisSustainability, 2022
-
Moore et
al. 2022Mangrove cultural services and values: current status and knowledge gapsPeople Nat, 2022
-
Hamza et al. 2024Global patterns of mangrove resource utilization: a systematic reviewFront Sustain Resour Manag, 2024
-
Lee et al. 2014Ecological role and services of tropical mangrove ecosystems: a reassessmentGlob Ecol Biogeogr, 2014
-
Canty et al. 2022Mangrove diversity is more than fringe deepSci Rep, 2022
-
Moore et al. 2022Mangrove cultural services and values: current status and knowledge gapsPeople Nat, 2022
-
Valiela et al. 2001Mangrove forests: one of the world’s threatened major tropical environmentsBioScience, 2001
-
Bryan-Brown et al. 2020Global trends in mangrove forest fragmentationSci Rep, 2020
-
Moore
et al. 2022Mangrove cultural services and values: current status and knowledge gapsPeople Nat, 2022
-
Feller et al. 2017The state of the world’s mangroves in the 21st century under climate changeHydrobiologia, 2017
Mangroves worldwide face diverse threats ranging from natural, such as climate change encompassing sea level rise and hurricanes (Schaeffer-Novelli et al. 2016), to anthropogenic drivers such as the expansion of aquaculture and agriculture, overlogging, salt extraction, human settlements, and coastal development (Valiela et al. 2001, Canty et al. 2018). These drivers have the potential to exacerbate the rate of mangrove deforestation, which results in the loss of critical functions such as nutrient cycling, coastal protection, and habitat provision for biodiversity, as well as the release of large amounts of CO2 into the atmosphere due to the decomposition of organic matter (Lovelock et al. 2017, Alongi 2018).
-
Schaeffer-Novelli et al. 2016Climate changes in mangrove forests and salt marshesBrazil J Oceanogr, 2016
-
Valiela et
al. 2001Mangrove forests: one of the world’s threatened major tropical environmentsBioScience, 2001
-
Canty et al. 2018Dichotomy of mangrove management: a review of research and policy in the Mesoamerican reef regionOcean Coast Manag, 2018
-
Lovelock et al. 2017Mangrove dieback during fluctuating sea levelsSci Rep, 2017
-
Alongi 2018Impact of global change on nutrient dynamics in mangrove forestsForests, 2018
Between 1980 and 2005, approximately 3.6 million ha of mangroves were lost worldwide, representing 20% of global cover (FAO 2007). Current mangrove cover is estimated at 147,359 km² across 108 countries, with net losses of around 3.5% between 1996 and 2020 (Spalding and Leal 2021, Bunting et al. 2022). Specifically, Honduras has suffered one of the highest losses of mangrove cover (more than 20,000 ha; Carrasco and Caviedes 2014). On the Atlantic coast of Honduras (ACH), the main drivers of mangrove deforestation have been hurricanes, agriculture, urban expansion, and tourism infrastructure (Cahoon et al. 2003, Fickert and Grüninger 2010, Recio et al. 2016, Flores-Marin 2017, Tuholske et al. 2017, ICF 2022). On the Pacific coast of Honduras (PCH), aquaculture has been the main driver, as well as logging and salt production (Stanley 1998, Chen et al. 2013, ICF 2022).
-
FAO 2007The World’s Mangroves 1980-2005, 2007
-
Spalding and Leal 2021The State of the World’s Mangroves 2021, 2021
-
Bunting et al.
2022Global change in mangrove extent 1996-2020: Global Mangrove Watch, version 3.0Remote Sens, 2022
-
Carrasco and
Caviedes 2014Diagnóstico de los Ecosistemas Marino-Costeros y de Agua Dulce de Honduras: Basado en Análisis de Viabilidad, Amenazas y Situación, 2014
-
Cahoon et al. 2003Mass tree mortality leads to mangrove peat collapse at Bay Islands, Honduras, after Hurricane MitchJ Ecol, 2003
-
Fickert and Grüninger 2010Floristic zonation, vegetation structure, and plant diversity patterns within a Caribbean mangrove and swamp forest on the Bay Island of Utila (Honduras)Ecotropica, 2010
-
Recio et al. 2016Central America mangroves, tenure, and REDD+ assessment, 2016
-
Flores-Marin 2017Optimización espacial para la restauración ecológica de manglares en el Parque Nacional Blanca Jeannette Kawas Fernández, Honduras [dissertation], 2017
-
Tuholske et
al. 2017Thirty years of land use/cover change in the Caribbean: Assessing the relationship between urbanization and mangrove loss in Roatán, HondurasAppl Geogr, 2017
-
ICF 2022Actual ecosistemas de manglar en Honduras, 2022
-
Stanley 1998Explaining persistent conflict among resource users: the case of Honduran maricultureSoc Nat Res, 1998
-
Chen et al. 2013Multi-decadal mangrove forest change detection and prediction in Honduras, Central America, with landsat imagery and a markov Chain ModelRemote Sens, 2013
-
ICF 2022Actual ecosistemas de manglar en Honduras, 2022
The destruction of mangroves in Honduras has had serious consequences, such as altered drainage patterns, increased saline intrusion, and depletion of fish populations in the PCH and ACH (Thornton et al. 2003, Recio et al. 2016, Maya-Jariego et al. 2023). Ultimately, this loss has exposed the coast to erosion, flooding, and damage from extreme events. For example, mangrove responses to hurricanes on the Honduran islands of Roatán and Guanaja resulted in the disturbance and alteration of regeneration patterns (Cahoon et al. 2003; Fickert 2018, 2020). On the other hand, reports indicate that the presence of pollutants affects the soil of mangrove ecosystems and limits their development (Burgos-Bennett 2011, Davila-Chuga 2021). This phenomenon has been observed in fields of the African oil palm (Elaeis guineensis), which contribute chemical compounds with phosphorus and nitrogen to the environment. Furthermore, the arrival of cruise ships to Caribbean islands leads to the discharge of pollutants, which disrupt mangrove ecosystems (Burgos-Bennett 2011, Doiron and Weissenberger 2014, Canty et al. 2018).
-
Thornton et al.
2003From wetlands to wastelands: impacts of shrimp farmingWetl Sci Pract, 2003
-
Recio et al. 2016Central America mangroves, tenure, and REDD+ assessment, 2016
-
Maya-Jariego et al. 2023Children’s interaction with ecosystem services in a mangrove forest: perceptions of fisherfolks on the involvement of minors in fishing activities in the Gulf of Fonseca in HondurasOcean Coast Manag, 2023
-
Cahoon et al. 2003Mass tree mortality leads to mangrove peat collapse at Bay Islands, Honduras, after Hurricane MitchJ Ecol, 2003
-
Fickert 2018, 2020Better resilient than resistant-regeneration dynamics of storm-disturbed mangrove forests on the bay island of Guanaja (Honduras) during the first two decades after Hurricane Mitch (October 1998)Diversity, 2018
-
Burgos-Bennett 2011Revisión General de los Humedales Marino-Costeros del Área de La Bahía de Trujillo-Guaimoreto-Aguan; Colón, Honduras, 2011
-
Davila-Chuga 2021Implicaciones Socioeconómicas en los Servicios Ecosistémicos de los Bosques de Manglar en las Costas Pacífico y Caribe de Honduras [dissertation], 2021
-
Burgos-Bennett 2011Revisión General de los Humedales Marino-Costeros del Área de La Bahía de Trujillo-Guaimoreto-Aguan; Colón, Honduras, 2011
-
Doiron and
Weissenberger 2014Sustainable dive tourism: Social and environmental impacts. The case of Roatan, HondurasTour Manag Perspect, 2014
-
Canty et al.
2018Dichotomy of mangrove management: a review of research and policy in the Mesoamerican reef regionOcean Coast Manag, 2018
In the Gulf of Fonseca, the expansion of shrimp farming has increased land use changes and land conflicts, which have led to socioecological problems, such as deterioration of water quality, degradation of land, disappearance of seasonal lagoons, and decline in fish populations (Dewalt et al. 1996, Stanley 1998, Stonich 1992, Acharya 2002). Shrimp farming, along with demographic development, has indirectly affected mangroves through hydrological alterations caused by road construction, which modified mixing of fresh and salt water (Dewalt et al. 1996). In this regard, the objectives of this study were to identify the main drivers of mangrove deforestation in Honduras based on the scientific literature, analyze changes in historical forest cover, and assess the current status of mangrove forests.
-
Dewalt et
al. 1996Shrimp aquaculture development and the environment: people, mangroves and fisheries on the Gulf of Fonseca, HondurasWorld Dev., 1996
-
Stanley 1998Explaining persistent conflict among resource users: the case of Honduran maricultureSoc Nat Res, 1998
-
Stonich 1992Struggling with Honduran poverty: the environmental consequences of natural resource-based development and rural transformationsWorld Dev, 1992
-
Acharya 2002Life at the margins: the social, economic and ecological importance of mangrovesMad Bosq, 2002
-
Dewalt et al. 1996Shrimp aquaculture development and the environment: people, mangroves and fisheries on the Gulf of Fonseca, HondurasWorld Dev., 1996
Materials and methods
Protocol and information sources
To conduct this review, we used the guidelines established in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) for documenting systematic literature reviews (Moher et al. 2009). The main sources used for the search were Web of Science; Scopus; Directory of Open Access Journals (DOAJ); Red de Revistas Científicas de América Latina y el Caribe, España y Portugal (REDALYC) (REDALYC); and Scientific Electronic Library Online (SCIELO). In addition, Google Scholar was used to collect gray literature, such as theses, technical reports, and management plans, which were incorporated into the review. Following the recommendation of Pullin and Stewart (2006), the inclusion of this type of document in systematic reviews helps reduce publication bias. The search period was from 1 January 1980 to 30 July 2024, with the objective of covering the largest amount of available evidence on the drivers of mangrove deforestation in Honduras. During this period, original articles, commentaries, books, book chapters, reports, and management plans relevant to the study were collected.
-
Moher
et al. 2009Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statementPLoS Med, 2009
-
Pullin and Stewart (2006)Guidelines for systematic review in conservation and environmental managementConserv Biol, 2006
Eligibility criteria
The drivers of mangrove deforestation in Honduras were categorized using a systematic process based on a comprehensive analysis of the available scientific literature. This methodological approach allowed us to identify and classify the drivers that determine mangrove cover loss, using predefined criteria and thematic sets of keywords. The main classification criteria considered the intrinsic nature of the drivers, differentiating between those of natural origin and those of anthropogenic origin.
The initial bibliographic selection process identified 105 relevant sources, which were further filtered by applying 2 specific sets of keywords. These keyword sets were based on classifications defined by Bhowmik et al. (2022) and Hagger et al. (2022) and based on a priori knowledge of the main drivers of mangrove deforestation in Honduras.
-
Bhowmik et al. (2022)Global mangrove deforestation and its interacting social-ecological drivers: a systematic review and synthesisSustainability, 2022
-
Hagger et al. (2022)Drivers of global mangrove loss and gain in social-ecological systemsNat Comm, 2022
In this sense, the identification of the main groups of drivers was based on a systemic approach that considered 2 broad categories: natural and anthropogenic drivers. The group of natural drivers refers to drivers originating in the natural environment, although they can be influenced by human activities. Conversely, anthropogenic drivers originate from human and socioeconomic actions and processes.
The first set of words included natural drivers classified into 2 main categories: “Climate Change,” characterized by variations in sea level, precipitation patterns, and temperature patterns; and “Tropical Cyclones,” characterized by abrupt natural disturbances, such as hurricanes, that occur mostly in the PCH region. The second set of words included anthropogenic drivers, categorized through a hierarchical process into 4 main groups: (1) aquaculture and agriculture (shrimp farming, African oil palm cultivation, and saltpeter works); (2) urbanization and development (settlement expansion, tourism infrastructure, and logging); (3) industrialization and pollution; and (4) water flow modification (water diversion, sedimentation, and hydroelectric dam construction).
In addition, the keyword set used both Spanish and English terms such as “mangroves” or “coastal wetlands,” combined with location terms such as “Honduras,” “Gulf of Fonseca,” “Pacific,” “Caribbean,” “Gracias a Dios,” “Colón,” “Cortés,” “Cayos Cochinos,” “Bay Islands,” and “Atlantis,” as well as specific location names such as “Útila,” “Roatán,” “Guanaja,” “La Mosquitia,” “Jeannette Kawas National Park,” “Barra de Cuero y Salado,” “Bacalar Lagoon,” “Cuyamel-Omoa,” “Guaimoreto Lagoon,” “Punta Izopo,” and “Karataska Lagoon.”
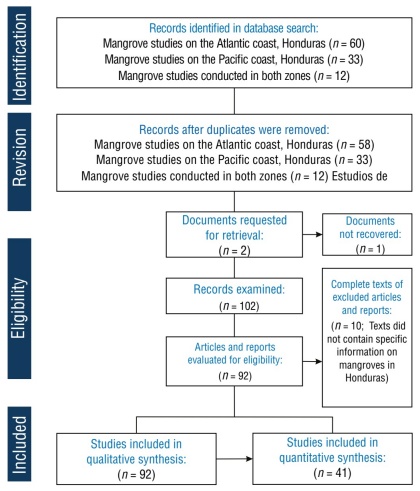
Studies and datasets containing information on the main drivers of mangrove deforestation in Honduras, as well as data on historical and current mangrove cover loss, were incorporated. Articles or documents that did not meet any of the selection criteria were excluded from further analysis. Specifically, documents that were unavailable or inaccessible, as well as those that were not relevant to the research objectives, were discarded. After the manual selection process, 92 documents that met the established criteria were included and considered for detailed analysis (Fig. 1).
Thumbnail
Figure 1
Flowchart based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines for the literature selection process in the systematic review to identify forest cover loss and its drivers in Honduran mangroves. Studies that included quantitative analyses were selected from the initial set of qualitative reviews.
Flowchart based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines for the literature selection process in the systematic review to identify forest cover loss and its drivers in Honduran mangroves. Studies that included quantitative analyses were selected from the initial set of qualitative reviews.
Regarding the above, a thorough qualitative analysis of all bibliographic documentation was conducted. However, for the quantitative analysis of mangrove cover, only 41 documents provided spatially explicit and methodologically consistent data for ACH and PCH. The number of studies reporting each of the identified drivers of change, as well as their distribution by driver group, was quantified in the seven locations where the mangrove ecosystems are located.
Data analysis process
Changes in the Honduran mangrove cover between 1965 and 2024 were analyzed, based on 41 studies reporting cover in the PCH and ACH (Fig. 1). Forest area values were compared, and changes in cover were calculated, determining losses at the national level. The average annual mangrove deforestation rate was calculated using the model proposed by the Food and Agriculture Organization of the United Nations (FAO 1996), which quantifies the annual percentage change in mangrove cover at the beginning of each period analyzed. This methodology is widely used in studies of forest ecosystem dynamics and is expressed in the following equation:
-
FAO 1996Forest Resources Assessment 1990: Survey of Tropical Forest Cover and Study of Change Orocesses, 1996
e1
A
n
n
u
a
l
r
a
t
e
%
=
1
-
S
2
1
/
t
S
1
100
(1)
where t is the time period analyzed, S 1 is the initial mangrove surface (older) and S 2 is the final mangrove surface (more recent).
The time periods analyzed were determined through an exhaustive review of the available scientific literature, directly extracting data reported in previous studies. For the PCH, the study period spanned from 1965 to 2024 (59 years); for the ACH, the study period spanned from 1985 and 2024 (39 years). This chronological selection was based on the availability of consistent and comparable data, thus ensuring the reliability of the temporal analyses performed. The disparity in the length of the periods between the 2 regions reflects differences in the historical coverage of mangrove studies in each area.
To assess changes in mangrove forest cover and the conservation status of species associated with this ecosystem, data from global reference platforms were systematically integrated and analyzed. Cover dynamics were assessed using records from Global Mangrove Watch (GMW 2025) and the geoportal of the Instituto de Conservación Forestal de Honduras (ICF 2024). In addition, to assess the distribution and conservation status of the characteristic species of this ecosystem, we consulted the International Union for Conservation of Nature (IUCN) Red List of Threatened Species (IUCN 2025) and the native distribution dataset from Plants of the World Online (POWO 2025).
-
ICF 2024Mapa de Cobertura Forestal 2024: Geoportal del Sector Forestal de Honduras, 2024
Mangrove ecosystems were mapped in 7 locations in Honduras, and natural and anthropogenic drivers of deforestation were identified and categorized. The thematic maps used in this study were created through a systematic classification process specifically designed for the objectives of the analysis. The cartographic databases used as the main input were obtained from the official geoportal of the Instituto Nacional de Conservación Forestal (ICF) using Quantum GIS v. 3.16.10. The number of research studies addressing each driver in the 7 locations with mangrove ecosystems was documented (Table 1), and the geographic zones in which these drivers interact were determined based on the literature. Finally, both natural and anthropogenic drivers were mapped at the national level using a chord dependency diagram.
Table 1
Natural and anthropogenic drivers of mangrove deforestation on the Pacific coast of Honduras (PCH) and on the Atlantic coast of Honduras (ACH). Values refer to the number of documents reviewed.
Natural and anthropogenic drivers of mangrove deforestation on the Pacific coast of Honduras (PCH) and on the Atlantic coast of Honduras (ACH). Values refer to the number of documents reviewed.
| Anthropogenic drivers | Natural drivers | ||||||
| Site | Aquaculture and agriculture | Urbanization and development | Industrialization and pollution | Flow modification | Climate change | Tropical cyclones | |
| PCH | |||||||
| Valley
and Choluteca |
44 | 11 | 7 | 8 | 3 | 6 | |
| ACH | |||||||
| Cortés | 12 | 13 | 8 | 7 | 5 | 5 | |
| Atlántida | 12 | 12 | 7 | 9 | 6 | 5 | |
| Colón | 6 | 9 | 8 | 7 | 4 | 5 | |
| Gracias a Dios | 9 | 4 | 2 | 7 | 6 | 3 | |
| Bay Islands | 2 | 17 | 10 | 17 | 8 | 14 | |
| Cayos Cochinos | 1 | 5 | 3 | 4 | 6 | 5 | |
| Total | 86 | 71 | 45 | 59 | 38 | 43 | |
Results
Spatiotemporal distribution studies developed for the loss of mangrove forest cover in Honduras
Initially, 105 documents were identified according to the established search criteria. However, 10 of these documents were deemed unsuitable and were excluded from the final analysis. Furthermore, of the initial selection of 105 documents, 12 were subsequently excluded because they did not contain relevant information, and 1 document could not be retrieved because downloading was not permitted, which resulted in a total of 92 documents that met the requirements (Fig. 1). Data related to the loss of mangrove forest cover were found to be limited, especially in the ACH region (Cortés, Colón, Atlántida, Gracias a Dios, and Cayos Cochinos).
Regarding the research on the mangrove species in the 7 localities of Honduras and their current distribution, the bibliographic review enabled the documentation of the conservation status of 8 mangrove species and their differential distribution between the coasts of the country: 7 species in the PCH and 5 in the ACH (Table 2). Note that only 1 article mentioned the presence of Tea Mangrove (Pelliciera rhizophorae) in Honduras; most of the previous literature did not refer to the presence of this species in the ACH, because its presence in Honduran territory was unknown until 2018 (Sutherland and Pérez 2018).
-
Sutherland and Pérez 2018La familia Tetrameristaceae descubierta en HondurasCeiba, 2018
Table 2
Number and distribution of mangrove species present on the Pacific coast of Honduras (PCH) and on the Atlantic coast of Honduras (ACH). The Valle and Choluteca sites together are considered the Gulf of Fonseca.
Number and distribution of mangrove species present on the Pacific coast of Honduras (PCH) and on the Atlantic coast of Honduras (ACH). The Valle and Choluteca sites together are considered the Gulf of Fonseca.
| PCH | ACH | |||||||
| Family | Species | Valley
and |
Gracias a Dios | Atlántida | Colón | Cortés | Bay Islands | Cayos Cochinos |
| Rhizophoraceae | Rhizophora mangle | x | x | x | x | x | x | x |
| Rhizophora racemosa | x | |||||||
| Rhizophora harrisonii | x | |||||||
| Combretaceae | Laguncularia racemosa | x | x | x | x | x | x | x |
| Conocarpus erectus | x | x | x | x | x | x | x | |
| Acanthaceae | Avicennia germinans | x | x | x | x | x | x | x |
| Avicennia bicolor | x | |||||||
| Tetrameristaceae | Pelliciera rhizophorae | x | ||||||
According to IUCN criteria, 5 of these species were classified as species of “least concern.” It is worth noting that, although not registered for Honduras in the IUCN database, the Red Mangrove (Rhizophora racemosa) was documented both in POWO and in previous scientific studies (Jiménez 1987, Lizano et al. 2001, Castañeda-Moya et al. 2006). On the other hand, the Zapatero or Caballero Mangrove (Rhizophora harrisonii) was not evaluated by the IUCN; however, its presence in the country was confirmed in POWO. Among the threatened species, 2 had a status of vulnerable: the Bicolor Mangrove (Avicennia bicolor) (in the PCH) (Sanchez-Paez 2000, Castañeda-Moya et al. 2006) and P. rhizophorae (in ACH) (Sutherland and Pérez 2018).
-
Jiménez 1987A clarification on the existence of Rhizophora species along the Pacific coast of Central AmericaBrenesia, 1987
-
Lizano et al. 2001Caracterización de manglares de Centroamérica con sensores remotosRev Biol Trop, 2001
-
Castañeda-Moya et al. 2006Mangrove zonation in the dry life zone of the Gulf of Fonseca, HondurasEstu Coast, 2006
-
Sanchez-Paez 2000Diagnóstico y Zonificación Preliminar de los Bosques de Mangle del Golfo de Fonseca, Honduras, 2000
-
Castañeda-Moya et al. 2006Mangrove zonation in the dry life zone of the Gulf of Fonseca, HondurasEstu Coast, 2006
-
Sutherland and Pérez 2018La familia Tetrameristaceae descubierta en HondurasCeiba, 2018
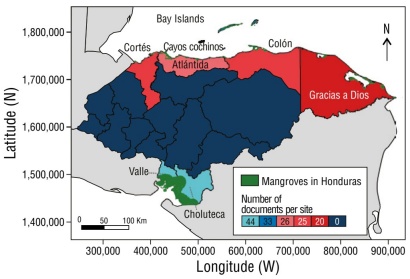
The detailed analysis of the geographic distribution, which focused on the drivers of mangrove deforestation, showed that the majority of research studies focused on 2 main areas: the Gulf of Fonseca (n = 44) in the PCH and the Bay Islands (n = 33) in the ACH. Research has primarily focused on the Gulf of Fonseca due to its location as the epicenter of anthropogenic mangrove deforestation in Honduras.
Studies in the Bay Islands have focused on land-use changes due to urbanization, coastal development, and the impacts of tourism. Abundant research has been conducted in this region due to the presence of the second-largest coral reef in the world, which is a UNESCO World Heritage Site (Tuholske et al. 2015). Diving activity, real estate development, and tourism infrastructure have been the subject of studies focused on understanding their effects on the environment. This has highlighted the growing interest of the scientific community and policymakers in understanding the protective role that mangrove forests and reefs play in the face of the impacts of climate change and human activities (Cahoon et al. 2003; Lebigre et al. 2003; Canty 2007; McKee et al. 2007; Vanselow et al. 2007; Fickert and Grüninger 2010; Doiron and Weissenberger 2014; Tuholske et al. 2017; Fickert 2018, 2020). Regarding the distribution of studies in Honduras, 13% (n = 12) covered research in all locations where mangroves are found (Fig. 3).
-
Tuholske et al. 2015Anthropogenic impacts on Roatán, Honduras: years of land-cover and land-use changePlurimondi, 2015
-
Cahoon et al. 2003Mass tree mortality leads to mangrove peat collapse at Bay Islands, Honduras, after Hurricane MitchJ Ecol, 2003
-
Lebigre et al.
2003Quel avenir pour les mangroves de l’archipel de la Bahía (Honduras)?Actes du Colloque. Espace littoraux en mutation, 2003
-
Canty 2007Positive and Negative Impacts of Dive Tourism: The Case Study of Utila, Honduras [dissertation], 2007
-
McKee et al. 2007Caribbean mangroves adjust to rising sea level through biotic controls on change in soil elevationGlobal Ecol Biogeogr, 2007
-
Vanselow et al. 2007Destruction and regeneration of terrestrial, littoral and marine ecosystems on the Island of Guanaja, Honduras, seven years after Hurricane MitchErdkunde, 2007
-
Fickert and Grüninger 2010Floristic zonation, vegetation structure, and plant diversity patterns within a Caribbean mangrove and swamp forest on the Bay Island of Utila (Honduras)Ecotropica, 2010
-
Doiron
and Weissenberger 2014Sustainable dive tourism: Social and environmental impacts. The case of Roatan, HondurasTour Manag Perspect, 2014
-
Tuholske et
al. 2017Thirty years of land use/cover change in the Caribbean: Assessing the relationship between urbanization and mangrove loss in Roatán, HondurasAppl Geogr, 2017
-
Fickert 2018Better resilient than resistant-regeneration dynamics of storm-disturbed mangrove forests on the bay island of Guanaja (Honduras) during the first two decades after Hurricane Mitch (October 1998)Diversity, 2018
-
2020To plant or not to plant, that is the question: reforestation vs. natural regeneration of hurricane-disturbed mangrove forests in Guanaja (Honduras)Forests, 2020
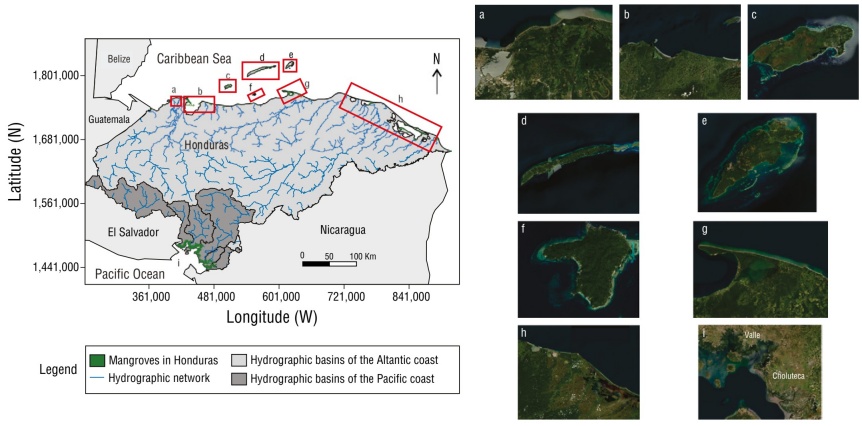
Thumbnail
Figure 2
Distribution of mangroves in 7 locations along the Pacific coast of Honduras (PCH) and the Atlantic coast of Honduras (ACH): Cortés (a), Atlántida (b), Útila (c), Roatán (d), Guanaja (e), Cayos Cochinos (f), Colón (g), Gracias a Dios (h), and Gulf of Fonseca (i) (Monserrate 2017).
Distribution of mangroves in 7 locations along the Pacific coast of Honduras (PCH) and the Atlantic coast of Honduras (ACH): Cortés (a), Atlántida (b), Útila (c), Roatán (d), Guanaja (e), Cayos Cochinos (f), Colón (g), Gracias a Dios (h), and Gulf of Fonseca (i) (Monserrate 2017).
Thumbnail
Figure 3
Geographic distribution of research studies of Honduran mangroves associated at the locality level.
Geographic distribution of research studies of Honduran mangroves associated at the locality level.
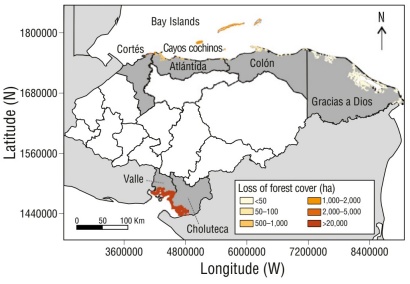
Historical and current loss of mangrove forest cover in Honduras
In Honduras, the loss of mangrove forest cover has exceeded 20,000 ha, with annual deforestation rates of 1.27%. Approximately 52% of the mangroves that once existed in Honduras have disappeared. Marked variability in deforestation rates was recorded along the PCH, with a particularly high impact in the Gulf of Fonseca (Fig. 4; Table 3). In this region, the mangrove deforestation rate reached 1.51% per year, similar to the value observed in Gracias a Dios (1.39% per year), but lower than that recorded in Cortés (2.62% per year).
Table 3
Rates of forest cover loss of mangrove forests on the Atlantic coast of Honduras (ACH) (1985-2024) and Pacific coast of Honduras (PCH) (1965-2024).
Rates of forest cover loss of mangrove forests on the Atlantic coast of Honduras (ACH) (1985-2024) and Pacific coast of Honduras (PCH) (1965-2024).
| Coast | Region | Historical surface (ha) | Current surface (ha) | Area of lost mangrove (ha) | Annual deforestation rate |
| PCH | Valle y
Choluteca |
91,800.00 | 37,390.25 | 54,409.75 | 1.51% |
| Islas de la Bahía | 2,874.00 | 2,318.72 | 555.28 | 0.54% | |
| Gracias a Dios | 23,995.00 | 13,735.00 | 10,260.23 | 1.39% | |
| ACH | Atlántida | 4,464.91 | 3,950.11 | 514.80 | 0.31% |
| Colón | 4,782.00 | 3,134.00 | 1,648.45 | 1.05% | |
| Cortés | 2,104.00 | 726.17 | 1,377.83 | 2.62% | |
| Total | 130,019.91 | 61,253.57 | 68,766.34 | 1.27% |
Thumbnail
Figure 4
Historical loss of the forest cover of Honduran mangroves by locality. Together, the localities of Valle and Choluteca are considered the Gulf of Fonseca.
Historical loss of the forest cover of Honduran mangroves by locality. Together, the localities of Valle and Choluteca are considered the Gulf of Fonseca.
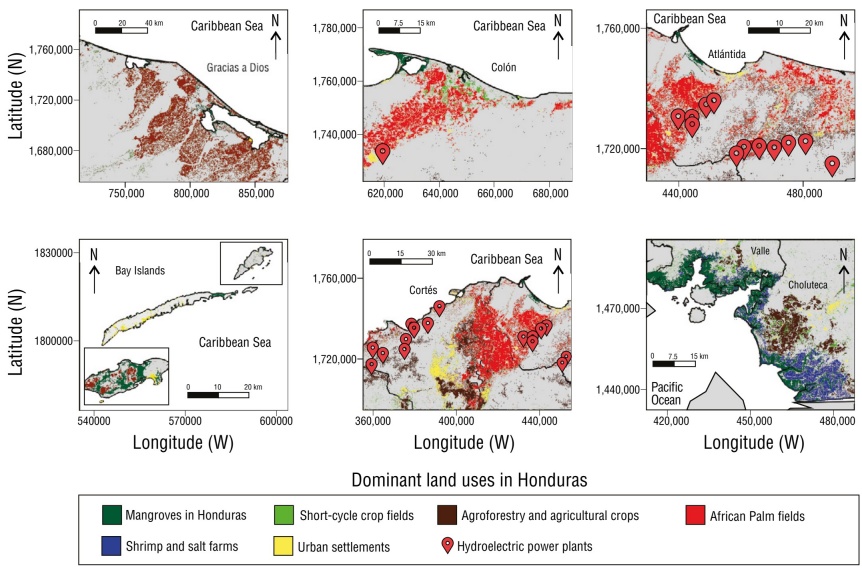
The main changes in land use that have led to mangrove replacement were identified based on the literature. In the PCH, shrimp farming and salt farms emerged as the most important land uses, as did the presence of other types of crops and urbanized areas. On the other hand, in the ACH, conversion to agriculture was important at the regional level. In Gracias a Dios, livestock and agricultural land and agroforestry occupied considerable areas. The towns of Atlántida, Colón, and Cortés, African palm fields covered large areas that, along with urban and industrial areas, dominated land use. It is important to mention that numerous hydroelectric plants were located in these towns, which have caused sediment retention and, consequently, coastal erosion (Carrasco and Caviedes 2014). In the Bay Islands, cultivated fields also occupied large areas, especially in Útila. Urbanized areas were larger in Roatán, where coastal development occurred as a result of the tourism industry (Figs. 5, 6).
-
Carrasco and Caviedes 2014Diagnóstico de los Ecosistemas Marino-Costeros y de Agua Dulce de Honduras: Basado en Análisis de Viabilidad, Amenazas y Situación, 2014
Thumbnail
Figure 5
Dominant land uses that have affected mangrove areas on the Pacific coast of Honduras (PCH) and the Atlantic coast of Honduras (ACH) in 2024.
Dominant land uses that have affected mangrove areas on the Pacific coast of Honduras (PCH) and the Atlantic coast of Honduras (ACH) in 2024.
Thumbnail
Figure 6
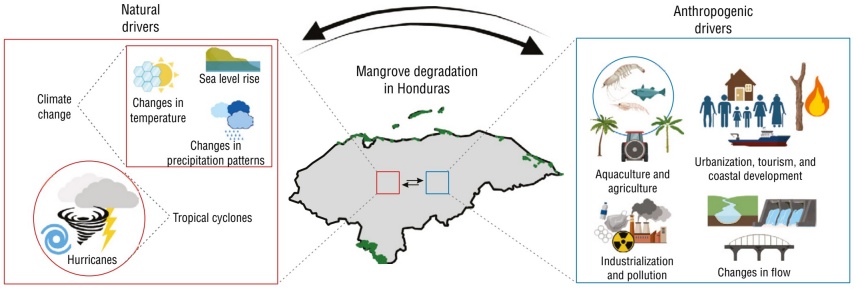
Group of identified natural and anthropogenic drivers of mangrove deforestation in Honduras.
Group of identified natural and anthropogenic drivers of mangrove deforestation in Honduras.
Socioecological drivers of mangrove deforestation in Honduras
The spatial patterns of natural and anthropogenic drivers varied markedly between the ACH and PCH regions. In the Bay Islands, deforestation was primarily associated with coastal tourism development and hurricane disturbances (Fickert 2018, 2020); in the continental areas of the ACH and PCH, aquaculture, agriculture, and urban expansion predominated (Fig. 7). These findings were consistent with the reviewed literature, which identified human activities as the most significant drivers of mangrove loss. The interaction between natural and anthropogenic drivers suggests a negative feedback scenario, where environmental disturbances increase ecosystem susceptibility to anthropogenic pressures, thus accelerating its degradation.
-
Fickert 2018, 2020Better resilient than resistant-regeneration dynamics of storm-disturbed mangrove forests on the bay island of Guanaja (Honduras) during the first two decades after Hurricane Mitch (October 1998)Diversity, 2018
Thumbnail
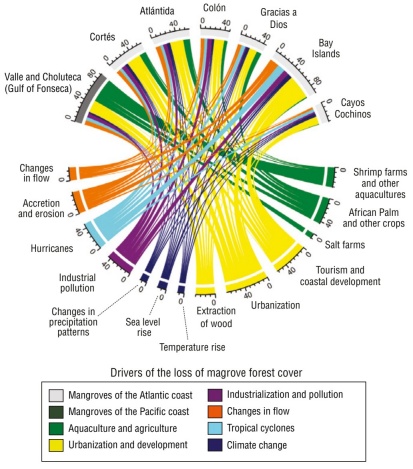
Figure 7
Chord dependency diagram among the groups and subgroups of drivers of mangrove deforestation in Honduras. The scale shows the number of studies that indicate the interaction between the driver and the location.
Chord dependency diagram among the groups and subgroups of drivers of mangrove deforestation in Honduras. The scale shows the number of studies that indicate the interaction between the driver and the location.
Discussion
Decrease in mangrove cover in Honduras
The literature on mangrove cover changes in Honduras (41 studies) spanned 5 decades for the PCH and 3 for the ACH, revealing a significant human footprint. Sixty-one percent of the mangroves were found in the PCH (Gulf of Fonseca) and the remaining 39% were distributed in the ACH (Cortés [1.19%], Atlántida [6.45%], Colón [5.12%], Gracias a Dios [22.42%], and Bay Islands [3.79%]). Of the literature reviewed, 62% studied ACH localities, which experienced a 38% loss of the national historical mangrove cover (Carrasco et al. 2013a, Carrasco et al. 2013b, Rivera-Monroy et al. 2013, Carrasco and Caviedes 2014, Flores-Marin 2017, Tuholske et al. 2017, Davila-Chuga 2021, Flores-Bueso 2022, ICF 2022). This is similar to what was reported for the PCH, which experienced the greatest loss of mangroves, with a 41% decrease of the historical cover, being the highest nationwide (Sanchez-Paez and Guevara-Mancera 2000, Wilburn-King 2008, Chen et al. 2013, Carrasco and Caviedes 2014, ICF 2022).
-
Carrasco et al. 2013aPlan de Conservación de la Reserva del Hombre y la Biósfera del Río Plátano: Basado en Análisis de Amenazas, Situación y del Impacto del Cambio Climático, y Definición de Metas y Estrategias, 2013
-
Carrasco et
al. 2013bPlan de Conservación del Parque Nacional Blanca Jeannette Kawas Fernández: Basado en Análisis de Amenazas, Situación y del Impacto del Cambio Climático, y Definición de Metas y Estrategias, 2013
-
Rivera-Monroy et al.
2013Análisis de Carbono Orgánico Total en Manglares del Parque Nacional Jeannette Kawas, 2013
-
Carrasco and Caviedes
2014Diagnóstico de los Ecosistemas Marino-Costeros y de Agua Dulce de Honduras: Basado en Análisis de Viabilidad, Amenazas y Situación, 2014
-
Flores-Marin 2017Optimización espacial para la restauración ecológica de manglares en el Parque Nacional Blanca Jeannette Kawas Fernández, Honduras [dissertation], 2017
-
Tuholske et al. 2017Thirty years of land use/cover change in the Caribbean: Assessing the relationship between urbanization and mangrove loss in Roatán, HondurasAppl Geogr, 2017
-
Davila-Chuga 2021Implicaciones Socioeconómicas en los Servicios Ecosistémicos de los Bosques de Manglar en las Costas Pacífico y Caribe de Honduras [dissertation], 2021
-
Flores-Bueso 2022Estructura poblacional, condición corporal de la iguana de cola espinosa de Útila (Ctenosaura bakeri) y análisis del cambio de cobertura de manglar en Útila, Islas de la Bahía, Honduras [dissertation], 2022
-
ICF 2022Actual ecosistemas de manglar en Honduras, 2022
-
Sanchez-Paez and
Guevara-Mancera 2000Diagnóstico y Zonificación Preliminar de los Bosques de Mangle del Golfo de Fonseca, Honduras, 2000
-
Wilburn-King
2008Political ecology of mangroves in southern Honduras: the emergence and evolution of environmental conflict in the Gulf of Fonseca 1973-2006 [dissertation], 2008
-
Chen et al. 2013Multi-decadal mangrove forest change detection and prediction in Honduras, Central America, with landsat imagery and a markov Chain ModelRemote Sens, 2013
-
Carrasco and Caviedes 2014Diagnóstico de los Ecosistemas Marino-Costeros y de Agua Dulce de Honduras: Basado en Análisis de Viabilidad, Amenazas y Situación, 2014
-
ICF 2022Actual ecosistemas de manglar en Honduras, 2022
The results revealed a 52.9% loss of Honduran mangroves in recent decades (~68,766 ha), with an average annual rate of 1.27% (~1,166 ha·y-1). These figures exceed the global average of annual mangrove loss (0.16-0.39%; Hamilton and Casey 2016), highlighting Honduras as a deforestation hotspot. Of the 52.9% mangrove loss, 79% corresponds to the Gulf of Fonseca mangroves in the PCH and the remaining 21% to the ACH mangroves (Table 3). At the national level, mangrove cover was reduced by 54,409.75 ha in the PCH at a rate of 922.19 ha·y-1 and 14,356.59 ha in the ACH at a rate of 368.11 ha·y-1 (Table 3).
-
Hamilton and Casey 2016Creation of a high spatio-temporal resolution global database of continuous mangrove forest cover for the 21st century (CGMFC-21)Glob Ecol Biogeogr, 2016
The loss of mangroves in Honduras reflects a more accelerated environmental crisis than in other tropical countries. For example, mangrove deforestation in the Colombian Pacific was estimated at a loss of 14% between 1984 and 2020 (Murillo-Sandoval et al. 2022), whereas in Honduras the loss was 52.9%. This discrepancy could be explained by the lack of effective conservation policies, such as those implemented in Brazil, where the establishment of protected areas has mitigated the pressures causing mangrove deforestation (Figueiroa et al. 2016, Medeiros et al. 2023). The data from Gracias a Dios (1.39% annually) are also worrying, as this region is home to the second largest mangrove area in the country. Here, mangrove degradation has been attributed to illegal logging and the expansion of livestock ranching (Burgos-Bennett 2011, Davila-Chuga 2021), which are recurring problems in areas with poor environmental governance (Supplementary Material Figure S7).
-
Murillo-Sandoval et al. 2022Mangroves cover change trajectories 1984-2020: the gradual decrease of mangroves in ColombiaFront Mar Sci, 2022
-
Figueiroa et al.
2016Evaluación de la efectividad de la integración de las unidades de conservación marino-costera federales en Santa CatarinaDesarro Medio Ambient, 2016
-
Medeiros et al. 2023Effectiveness of federal protected areas in the preservation of mangrove forests on the coast of the state of Paraíba, BrazilAn Acad Bras Cienc, 2023
-
Burgos-Bennett 2011Revisión General de los Humedales Marino-Costeros del Área de La Bahía de Trujillo-Guaimoreto-Aguan; Colón, Honduras, 2011
-
Davila-Chuga 2021Implicaciones Socioeconómicas en los Servicios Ecosistémicos de los Bosques de Manglar en las Costas Pacífico y Caribe de Honduras [dissertation], 2021
In general, the main drivers of mangrove loss in Honduras are anthropogenic: aquaculture in the PCH and agriculture and infrastructure in the ACH. Climate change and extreme events are also important factors, especially in Cortés and the Bay Islands (Cahoon and Hensel 2002, Vanselow et al. 2007, Fickert and Grüninger 2010, Fickert 2018, Fickert 2020). The loss of mangrove area due to human activities implies a reduction in ecosystem services specific to each location. If deforestation continues at an accelerated rate, the negative impacts on biodiversity and the provision of environmental services will be more evident.
-
Cahoon and Hensel 2002Hurricane Mitch: A Regional Perspective on Mangrove Damage, Recovery, and Sustainability, 2002
-
Vanselow et al. 2007Destruction and regeneration of terrestrial, littoral and marine ecosystems on the Island of Guanaja, Honduras, seven years after Hurricane MitchErdkunde, 2007
-
Fickert and Grüninger 2010Floristic zonation, vegetation structure, and plant diversity patterns within a Caribbean mangrove and swamp forest on the Bay Island of Utila (Honduras)Ecotropica, 2010
-
Fickert 2018Better resilient than resistant-regeneration dynamics of storm-disturbed mangrove forests on the bay island of Guanaja (Honduras) during the first two decades after Hurricane Mitch (October 1998)Diversity, 2018
-
Fickert 2020To plant or not to plant, that is the question: reforestation vs. natural regeneration of hurricane-disturbed mangrove forests in Guanaja (Honduras)Forests, 2020
Environmental drivers
Climate change
Of the literature reviewed, 9% identified environmental factors as drivers of mangrove decline in Honduras, including coastal erosion, increased salinity, ocean acidification, temperature changes, rising sea levels, and variations in precipitation (Table 3). These factors, associated with climate change, affect the hydroperiod, regeneration, growth, composition, and spatial distribution of mangroves (Field 1995). For example, rising temperatures have been found to increase water vapor deficits, reducing mangrove growth and survival (Adame et al. 2021). Furthermore, rising sea levels have caused flooding and erosion, which increases mangrove vulnerability. On the other hand, reduced precipitation has been observed to decrease photosynthesis, productivity, and reproduction, whereas high rainfall causes flooding that deposits sediments, suffocating mangroves. These changes in sedimentation, turbidity, and salinity significantly influence the growth and distribution of mangroves, which highlights the complex interaction between climatic factors and the health of these ecosystems (Friess et al. 2022).
-
Field 1995Impact of expected climate change on mangrovesHydrobiologia, 1995
-
Adame et al.
2021Mangroves in arid regions: ecology, threats, and opportunitiesEstuar Coast Shelf Sci, 2021
-
Friess
et al. 2022Mangrove forests under climate change in a 2°C world. WIREs ClimChange, 2022
Sea level rise has been identified as the primary threat to mangroves in Honduras. Studies in Puerto Cortés and Puerto Castilla revealed increases of 8.9 mm·y-1 and 3.2 mm·y-1, respectively, between 1940 and 1970 (Aubrey et al. 1988, Ellison and Farnsworth 1996). A USAID study (2012) confirmed these data, showing increases of 9.2 mm·y-1 in Puerto Cortés (1945-1975) and 3.1 mm·y-1 in Puerto Castilla (1954-1970). In Cuyamel-Omoa, mangrove cover showed an 80% decrease as a result of the impact of waves (ICF 2012). The most affected areas were Trujillo (Colón), Brus Laguna (Gracias a Dios), and the Bay Islands, with increased coastal erosion, except in the Bay Islands (USAID 2012, Carrasco et al. 2013a, b).
-
Aubrey et al. 1988Changing coastal levels of South America and the Caribbean region from tide-gauge recordsTectonophysics, 1988
-
Ellison and Farnsworth 1996Anthropogenic disturbance of caribbean mangrove ecosystems: past impacts, present trends, and future predictionsBiotropica, 1996
-
ICF 2012Plan de Manejo Parque Nacional Cuyamel-Omoa, 2012
-
USAID 2012Análisis de Vulnerabilidad al Cambio Climático del Caribe de Belice, Guatemala y Honduras. Programa Regional de USAID para el Manejo de Recursos Acuáticos y Alternativas Económicas, 2012
-
Carrasco et al.
2013aPlan de Conservación de la Reserva del Hombre y la Biósfera del Río Plátano: Basado en Análisis de Amenazas, Situación y del Impacto del Cambio Climático, y Definición de Metas y Estrategias, 2013
-
bPlan de Conservación del Parque Nacional Blanca Jeannette Kawas Fernández: Basado en Análisis de Amenazas, Situación y del Impacto del Cambio Climático, y Definición de Metas y Estrategias, 2013
These locations, with the exception of the Bay Islands, have shown an increased coastal erosion (Carrasco and Caviedes 2014). In the Gulf of Fonseca, sea level has risen approximately 50 cm, leaving the coast vulnerable due to mangrove deforestation (Fig. 8) (Del Cid-Gómez and Cáceres 2017). Although mangrove loss due to climate change in Honduras has not been quantified, it is likely significant, which creates the need for further research in this area.
-
Carrasco and
Caviedes 2014Diagnóstico de los Ecosistemas Marino-Costeros y de Agua Dulce de Honduras: Basado en Análisis de Viabilidad, Amenazas y Situación, 2014
-
Del Cid-Gómez and Cáceres 2017Variación de la línea de costa en la aldea de Cedeño, y cartografía de amenaza ante marejadas y ascenso del nivel del marPort Cienc, 2017
Thumbnail
Figure 8
Impact of sea level rise on the coastal infrastructure of Cedeño, Pacific coast of Honduras (PCH). Submerged infrastructure due to sea level rise (a) and coastal erosion labeled in red and local businesses destroyed by sea level rise (b).
Impact of sea level rise on the coastal infrastructure of Cedeño, Pacific coast of Honduras (PCH). Submerged infrastructure due to sea level rise (a) and coastal erosion labeled in red and local businesses destroyed by sea level rise (b).
Tropical cyclones
Honduran mangroves are particularly vulnerable to tropical cyclones due to their geographic location. These events affect mangroves through sediment deposition, wind damage, and submersion (Krauss and Osland 2020). Ten percent of the literature mentioned the impact of hurricanes, especially in the Bay Islands. Historically, 101 tropical storms and hurricanes have affected Honduras. Hurricane Mitch in 1998 was particularly devastating, causing significant losses in the mangroves of Trujillo, Colón. In the Bay Islands, especially Roatán and Guanaja, stands of mangrove trees with high mortality have been observed as a result of substrate collapse from the breakdown of mangrove peat (Doyle et al. 2002, Cahoon et al. 2003). Hurricane Mitch reduced mangrove forests by 37% in Roatán and by 97% in Guanaja, demonstrating the severe impact of these events on mangrove ecosystems (Supplementary Material Fig. S1) (Cahoon et al. 2003; Fickert 2018, 2020; Canty et al. 2022).
-
Krauss and Osland 2020Tropical cyclones and the organization of mangrove forests: a reviewAnn Bot, 2020
-
Doyle et al. 2002Hurricane Mitch: Landscape Analysis of Damaged Forest Resources of the Bay Islands and Caribbean Coast of Honduras, 2002
-
Cahoon et al. 2003Mass tree mortality leads to mangrove peat collapse at Bay Islands, Honduras, after Hurricane MitchJ Ecol, 2003
-
Cahoon et
al. 2003Mass tree mortality leads to mangrove peat collapse at Bay Islands, Honduras, after Hurricane MitchJ Ecol, 2003
-
Fickert 2018,
2020Better resilient than resistant-regeneration dynamics of storm-disturbed mangrove forests on the bay island of Guanaja (Honduras) during the first two decades after Hurricane Mitch (October 1998)Diversity, 2018
-
Canty et al.
2022Mangrove diversity is more than fringe deepSci Rep, 2022
Although Hurricane Mitch did not directly impact the Gulf of Fonseca, mangroves were affected by debris flows and sediment accumulation (McKee and McGinnis 2003). Some were buried under layers of sediment up to 100 cm deep, carried by river floods from the upper basin (Cahoon and Hensel 2002). In the central Gulf, there was massive tree mortality, canopy damage, and changes in elevation and soil properties (Cahoon et al. 2003, Hensel and Proffitt 2003, McKee and McGinnis 2003). In the southern region, a storm surge caused flooding and sediment deposition (Rivera-Monroy et al. 2002). In general, little attention has been paid to the consequences of hurricanes on mangroves, and the reduction in forest cover in ACH and PCH has not been quantified. The increased frequency of these events could limit the recovery time of mangroves, increasing the risk of permanent loss.
-
McKee and McGinnis 2003Hurricane Mitch: Effects on Mangrove Soil Characteristics and Root Contributions to Soil Stabilization, 2003
-
Cahoon and Hensel
2002Hurricane Mitch: A Regional Perspective on Mangrove Damage, Recovery, and Sustainability, 2002
-
Cahoon et al. 2003Mass tree mortality leads to mangrove peat collapse at Bay Islands, Honduras, after Hurricane MitchJ Ecol, 2003
-
Hensel and Proffitt 2003Hurricane Mitch: Acute Impacts on Mangrove Forest Structure and an Evaluation of Recovery Trajectories, 2003
-
McKee and McGinnis 2003Hurricane Mitch: Effects on Mangrove Soil Characteristics and Root Contributions to Soil Stabilization, 2003
-
Rivera-Monroy et al. 2002Hurricane Mitch: Integrative Management and Rehabilitation of Mangrove Resources to Develop Sustainable Shrimp Mariculture in the Gulf of Fonseca, Honduras, 2002
Anthropogenic drivers
Aquaculture and agriculture
Aquaculture and agriculture are the main drivers of mangrove deforestation, with aquaculture accounting for 47% of global mangrove loss in the 20th century (Friess et al. 2019, Goldberg et al. 2020, Bhowmik et al. 2022). In the PCH, the expansion of shrimp farming and saltpeter works has been the main cause of mangrove loss since 1965. In 1973, the expansion of shrimp farms resulted in the loss of 2,132.5 ha of dense forests (Dewalt et al. 1996). Between 1982 and 1992, shrimp farming occupied 11,515 ha, including 4,307 ha of mature, stressed, or dwarf mangroves (Ramirez 1994, Páez-Osuna 2001). Wilburn-King (2008) reported a loss of 3,733 ha of mangroves between 1989 and 1998 due to the production of salt and shrimp. Chen et al. (2013) reported that approximately 11.9% of mangroves were lost between 1985 and 2013, primarily due to shrimp farming (Supplementary Material Fig. S2) (Recio et al. 2016, Son et al. 2017). These data demonstrate the significant impact that human activities, such as shrimp farming, have had on the decline of mangrove forests over time in the PCH. Furthermore, sites in this area are considered to be particularly susceptible to the effects of climate change and human activities (Bhomia et al. 2016, Maya-Jariego et al. 2023).
-
Friess et al. 2019The state of the world’s mangrove forests: past, present, and futureAnnu Rev Environ Resourc, 2019
-
Goldberg et al. 2020Global declines in human-driven mangrove lossGlob Chang Biol, 2020
-
Bhowmik et al. 2022Global mangrove deforestation and its interacting social-ecological drivers: a systematic review and synthesisSustainability, 2022
-
Dewalt et al. 1996Shrimp aquaculture development and the environment: people, mangroves and fisheries on the Gulf of Fonseca, HondurasWorld Dev., 1996
-
Ramirez 1994CODDEFFAGOLF: Los defensores de los manglares del Golfo de Fonseca, HondurasRev Forest Centroamericana, 1994
-
Páez-Osuna 2001The environmental impact of shrimp aquaculture: causes, effects, and mitigating alternativesEnviron Manag, 2001
-
Wilburn-King (2008)Political ecology of mangroves in southern Honduras: the emergence and evolution of environmental conflict in the Gulf of Fonseca 1973-2006 [dissertation], 2008
-
Chen et al. (2013)Multi-decadal mangrove forest change detection and prediction in Honduras, Central America, with landsat imagery and a markov Chain ModelRemote Sens, 2013
-
Recio et al. 2016Central America mangroves, tenure, and REDD+ assessment, 2016
-
Son et al. 2017Mapping mangrove density from rapideye data in Central AmericaOpen Geosci, 2017
-
Bhomia et al. 2016Ecosystem carbon stocks of mangrove forests along the Pacific and Caribbean coasts of HondurasWetlands Ecol Manag, 2016
-
Maya-Jariego et al. 2023Children’s interaction with ecosystem services in a mangrove forest: perceptions of fisherfolks on the involvement of minors in fishing activities in the Gulf of Fonseca in HondurasOcean Coast Manag, 2023
Agricultural intensification has contributed to mangrove loss in the ACH. In particular, the main threats included the development of African palm plantations, land use change to pasture, and agriculture, which occupy approximately 140,000 ha, much of which used to be wetlands (Carrasco and Caviedes 2014, Flores-Marin 2017, Davila-Chuga 2021). A notable case is La Másica, Atlántida, where around 12 km2 of forested wetlands, including mangroves, were lost (Carrasco et al. 2013c). In the same area, socioeconomic activities have destroyed more than 500 hectares of mangroves. In Colón, the expansion of the African palm has led to the reduction of these forest areas; in Gracias a Dios and Cortés, livestock farming and technological agriculture have also had a negative impact (Burgos-Bennett 2011, Davila-Chuga 2021). In contrast, these activities have caused less damage in the PCH, although agriculture and aquaculture in the middle and upper basins produce pollutants that affect the development of mangrove ecosystems (Osorto-Nuñez 2022, Osorto-Nuñez et al. 2023).
-
Carrasco and Caviedes 2014Diagnóstico de los Ecosistemas Marino-Costeros y de Agua Dulce de Honduras: Basado en Análisis de Viabilidad, Amenazas y Situación, 2014
-
Flores-Marin 2017Optimización espacial para la restauración ecológica de manglares en el Parque Nacional Blanca Jeannette Kawas Fernández, Honduras [dissertation], 2017
-
Davila-Chuga 2021Implicaciones Socioeconómicas en los Servicios Ecosistémicos de los Bosques de Manglar en las Costas Pacífico y Caribe de Honduras [dissertation], 2021
-
Carrasco et
al. 2013cPlan de Conservación del Refugio de Vida Silvestre Barras de Cuero y Salado: Basado en Análisis de Amenazas, Situación y del Impacto del Cambio Climático, y Definición de Metas y Estrategias, 2013
-
Burgos-Bennett
2011Revisión General de los Humedales Marino-Costeros del Área de La Bahía de Trujillo-Guaimoreto-Aguan; Colón, Honduras, 2011
-
Davila-Chuga 2021Implicaciones Socioeconómicas en los Servicios Ecosistémicos de los Bosques de Manglar en las Costas Pacífico y Caribe de Honduras [dissertation], 2021
-
Osorto-Nuñez 2022Los modelos de cambio climático futuro como predictores de la reducción del área de distribución de dos especies de cíclidos endémicos de HondurasBiol Acuát, 2022
-
Osorto-Nuñez et al. 2023Evaluación de la calidad de agua mediante un análisis multivariante en los esteros del Golfo de Fonseca, HondurasBol Invest Mar Cost, 2023
Industrialization and pollution
Industrialization and pollution are key factors in the loss of mangrove cover (Zhang et al. 2014). Since 1995, organochlorine pesticides (e.g., carbofuran and propiconazole) have been detected in the Choluteca River, which flows into the Gulf of Fonseca, evidencing the damage that chemical pollutants cause to mangroves (Kammerbauer and Moncada 1998, Meyer 1999, Osorto-Nuñez 2022, Maya-Jariego et al. 2023). In the Bay Islands, discharges of organic matter from industrial waters have promoted the growth of calcareous green algae, which threaten these ecosystems. Furthermore, the cruise ships that arrive in this region have also contributed to pollution (Canty 2007, Carrasco et al. 2013d, Doiron and Weissenberger 2014). In Colón, the mining industry has caused mangrove destruction due to excessive sedimentation and the presence of heavy metals in the water and soil (Burgos-Bennett 2011). In the Sula Valley, approximately 150 of the 380 industries have produced problematic effluents that contaminate the Ulúa and Chamelecón rivers with mercury, lead, and arsenic (Marin and Hernandez 2002). Unfortunately, the accumulation of metals, such as copper and zinc, has become a global driver of mangrove deforestation (Sruthi et al. 2016, Bhowmik et al. 2022).
-
Zhang et al. 2014Heavy metal and organic contaminants in mangrove ecosystems of China: a reviewEnviron Sci Poll Res, 2014
-
Kammerbauer and Moncada 1998Pesticide residue assessment in three selected agricultural production systems in the Choluteca River Basin of HondurasEnviron Poll, 1998
-
Meyer 1999Presence of pesticide residues in water, sediment and biological samples taken from aquatic environments in Honduras, 1999
-
Osorto-Nuñez 2022Los modelos de cambio climático futuro como predictores de la reducción del área de distribución de dos especies de cíclidos endémicos de HondurasBiol Acuát, 2022
-
Maya-Jariego et al. 2023Children’s interaction with ecosystem services in a mangrove forest: perceptions of fisherfolks on the involvement of minors in fishing activities in the Gulf of Fonseca in HondurasOcean Coast Manag, 2023
-
Canty 2007Positive and Negative Impacts of Dive Tourism: The Case Study of Utila, Honduras [dissertation], 2007
-
Carrasco et al. 2013dPlan de Conservación del Parque Nacional Marino Islas de la Bahía: Basado en Análisis de Amenazas, Situación y del Impacto del Cambio Climático, y Definición de Metas y Estrategias, 2013
-
Doiron and Weissenberger 2014Sustainable dive tourism: Social and environmental impacts. The case of Roatan, HondurasTour Manag Perspect, 2014
-
Burgos-Bennett 2011Revisión General de los Humedales Marino-Costeros del Área de La Bahía de Trujillo-Guaimoreto-Aguan; Colón, Honduras, 2011
-
Marin and Hernandez 2002La contaminación marina en Honduras, 2002
-
Sruthi et al. 2016Heavy metal detoxification mechanisms in halophytes: an overviewWetlands Ecol Manag, 2016
-
Bhowmik et al. 2022Global mangrove deforestation and its interacting social-ecological drivers: a systematic review and synthesisSustainability, 2022
Urbanization and development
Of the studies reviewed, 37% indicated that urbanization and development are key drivers of mangrove deforestation in Honduras. Activities such as logging and the construction of settlements, roads, and tourism infrastructure have significantly reduced forest cover. In Roatán, between 1985 and 2015, the development of hotel infrastructure and the expansion of the airport increased the urban area by 982.8 ha, with 224.1 ha of mangroves converted to urban areas (Supplementary Material Fig. S3) (Carrasco et al. 2013d, Doiron and Weissenberger 2014, Tuholske et al. 2015, Tuholske et al. 2017). In Útila, the urban area grew from 118 ha in 2014 to 231 ha in 2021, affecting the southern mangroves (Flores-Bueso, 2022). In Tela, Atlántida, tourism development has put additional pressure on mangroves (Stovall 2015). In addition, population growth and hotel infrastructure have fragmented wetlands and altered the hydrological regime, exacerbating the loss of these vital ecosystems.
-
Carrasco et
al. 2013dPlan de Conservación del Parque Nacional Marino Islas de la Bahía: Basado en Análisis de Amenazas, Situación y del Impacto del Cambio Climático, y Definición de Metas y Estrategias, 2013
-
Doiron and Weissenberger
2014Sustainable dive tourism: Social and environmental impacts. The case of Roatan, HondurasTour Manag Perspect, 2014
-
Tuholske et al. 2015Anthropogenic impacts on Roatán, Honduras: years of land-cover and land-use changePlurimondi, 2015
-
Tuholske et al. 2017Thirty years of land use/cover change in the Caribbean: Assessing the relationship between urbanization and mangrove loss in Roatán, HondurasAppl Geogr, 2017
-
Flores-Bueso, 2022Estructura poblacional, condición corporal de la iguana de cola espinosa de Útila (Ctenosaura bakeri) y análisis del cambio de cobertura de manglar en Útila, Islas de la Bahía, Honduras [dissertation], 2022
-
Stovall 2015Carbon Storage, Forest Structure, and Fish Biodiversity Across Human-Impacted Mangrove Ecosystems in Tela Bay, Honduras [dissertation], 2015
The extraction of mangrove wood has had an important impact on the loss of forest cover, especially in the Gulf of Fonseca, where wood and bark are used as energy sources (Alfaro-Trejos 2011, Maya-Jariego et al. 2023). Between 1983 and 1986, the use of mangrove wood for fuel exceeded 80,000 m3·y-1 to 120,000 m3·y-1 (Jiménez 1992). From 1983 to 1989, 34,200 m³ of firewood and 5,340 m³ of timber were extracted, and consumption increased from 3,644 m3·y-1 to 4,212 m3·y-1 between 1986 and 1995 (Jiménez, 1999, Sanchez-Paez and Guevara-Mancera 2000, CONGESA 2001). According to Dewalt et al. (1996), this represented the use of 250 to 350 ha of mangroves annually. In the Gulf of Fonseca, 85% of households relied on wood for energy and construction of housing (Supplementary Material Fig. S4). In addition, industries such as bakeries and brickyards also use wood as a primary resource. In Roatán, logging has reduced the frequency of the Buttonwood Mangrove (Conocarpus erectus); this activity is also carried out in Cuyamel-Omoa for domestic and commercial purposes, highlighting the impact of timber extraction on these ecosystems (ICF 2012, Carrasco and Caviedes 2014).
-
Alfaro-Trejos 2011Cobertura y uso de la tierra en el ecosistema de mangle y zona ecotonal del corredor del mangle, desde la Bahía de Jiquilisco, El Salvador, hasta el Estero Padre Ramos, Jiquilillo, NicaraguaRev Geogr Amér Cent, 2011
-
Maya-Jariego et al. 2023Children’s interaction with ecosystem services in a mangrove forest: perceptions of fisherfolks on the involvement of minors in fishing activities in the Gulf of Fonseca in HondurasOcean Coast Manag, 2023
-
Jiménez 1992Mangrove Forests of the Pacific Coast of Central AmericaCoastal Plant Communities of Latin America, 1992
-
Jiménez, 1999Mangrove Forests of the Pacific Coast of Central AmericaCoastal Plant Communities of Latin America, 1992
-
Sanchez-Paez
and Guevara-Mancera 2000Diagnóstico y Zonificación Preliminar de los Bosques de Mangle del Golfo de Fonseca, Honduras, 2000
-
CONGESA
2001Valoración económica de los manglares del Golfo de Fonseca, Honduras, 2001
-
Dewalt et al.
(1996)Shrimp aquaculture development and the environment: people, mangroves and fisheries on the Gulf of Fonseca, HondurasWorld Dev., 1996
-
ICF
2012Plan de Manejo Parque Nacional Cuyamel-Omoa, 2012
-
Carrasco and Caviedes
2014Diagnóstico de los Ecosistemas Marino-Costeros y de Agua Dulce de Honduras: Basado en Análisis de Viabilidad, Amenazas y Situación, 2014
The expansion of human settlements and urbanization have significantly impacted mangroves in Honduras, especially in Útila (Supplementary Material Fig. S5). As observed in Atlántida, water pollution is a critical problem in Útila, where sewage contributes phosphorus to the mangroves (Carrasco et al. 2013d). In Colón, the accumulation of garbage and solid waste has severely affected these ecosystems (Burgos-Bennett 2011). In the Gulf of Fonseca, sewage discharges and solid waste accumulation have negatively impacted mangroves (Supplementary Material Fig. S4) (Maya-Jariego et al. 2023). In Cortés, settlements near Los Micos Lagoon have contaminated the soil and surface water (Carrasco et al. 2013b). Similarly, in the Bay Islands, especially in Útila, mangrove loss has been caused by sewage, logging, and the accumulation of waste from urban expansion (Carrasco et al. 2013d, Carrasco and Caviedes 2014). In Gracias a Dios, urbanization has contributed to logging and soil erosion, posing threats to mangroves (Davila-Chuga 2021). These factors highlight how urbanization has jeopardized the integrity of Honduran mangroves.
-
Carrasco et al. 2013dPlan de Conservación del Parque Nacional Marino Islas de la Bahía: Basado en Análisis de Amenazas, Situación y del Impacto del Cambio Climático, y Definición de Metas y Estrategias, 2013
-
Burgos-Bennett 2011Revisión General de los Humedales Marino-Costeros del Área de La Bahía de Trujillo-Guaimoreto-Aguan; Colón, Honduras, 2011
-
Maya-Jariego
et al. 2023Children’s interaction with ecosystem services in a mangrove forest: perceptions of fisherfolks on the involvement of minors in fishing activities in the Gulf of Fonseca in HondurasOcean Coast Manag, 2023
-
Carrasco et al. 2013bPlan de Conservación del Parque Nacional Blanca Jeannette Kawas Fernández: Basado en Análisis de Amenazas, Situación y del Impacto del Cambio Climático, y Definición de Metas y Estrategias, 2013
-
Carrasco et al.
2013dPlan de Conservación del Parque Nacional Marino Islas de la Bahía: Basado en Análisis de Amenazas, Situación y del Impacto del Cambio Climático, y Definición de Metas y Estrategias, 2013
-
Carrasco and Caviedes
2014Diagnóstico de los Ecosistemas Marino-Costeros y de Agua Dulce de Honduras: Basado en Análisis de Viabilidad, Amenazas y Situación, 2014
-
Davila-Chuga 2021Implicaciones Socioeconómicas en los Servicios Ecosistémicos de los Bosques de Manglar en las Costas Pacífico y Caribe de Honduras [dissertation], 2021
Flux modification
The modification of the water flux in the upper basin has been shown to decrease mangrove productivity (Bhowmik et al. 2022). According to projections based on previous scientific literature, dam construction in Honduras has had negative impacts on mangrove forests and has altered their ecological balance. The main consequences include sediment and nutrient retention, increased salinity in coastal areas, and the intensification of erosion processes (Morais and Pinheiro 2011). For example, in Balfate, Colón, a dam has altered the morphology and hydrodynamics of the river, which resulted in sediment retention (Carrasco and Caviedes 2014). In the Barra de Cuero and Salado Wildlife Refuge, hydroelectric projects are planned for 11 rivers, which could modify the natural hydrological regime and decrease the primary productivity of mangroves (Carrasco et al. 2013c). The Patuca III hydroelectric dam, in Gracias a Dios, could increase the salinity of the lagoons by reducing the contribution of water and sediment from the Patuca River, putting the mangroves at risk (Carrasco and Colindres 2011, Carrasco et al. 2013a). In Jeannette Kawas National Park, the hydrological regime has changed by 80% due to the channeling and draining of several rivers, highlighting the importance of considering these effects when planning hydroelectric projects to preserve these coastal ecosystems.
-
Bhowmik et
al. 2022Global mangrove deforestation and its interacting social-ecological drivers: a systematic review and synthesisSustainability, 2022
-
Morais and Pinheiro 2011The effect of semi-aridity and damming on sedimentary dynamics in estuaries-northeastern region of BrazilJ Coast Res, 2011
-
Carrasco and Caviedes 2014Diagnóstico de los Ecosistemas Marino-Costeros y de Agua Dulce de Honduras: Basado en Análisis de Viabilidad, Amenazas y Situación, 2014
-
Carrasco et al.
2013cPlan de Conservación del Refugio de Vida Silvestre Barras de Cuero y Salado: Basado en Análisis de Amenazas, Situación y del Impacto del Cambio Climático, y Definición de Metas y Estrategias, 2013
-
Carrasco and Colindres 2011Plan de manejo del sistema lagunar de Karataska. Programa de las Naciones Unidad para el Desarrollo, 2011
-
Carrasco et al. 2013aPlan de Conservación de la Reserva del Hombre y la Biósfera del Río Plátano: Basado en Análisis de Amenazas, Situación y del Impacto del Cambio Climático, y Definición de Metas y Estrategias, 2013
In general, our study, based on a synthesis of scientific literature, shows an intermediate value. The differences could reflect variations in the reference years, since the studies are not completely simultaneous, or in the spatial delimitation of the areas assessed. For example, the ICF (2024) could have incorporated recent restoration areas not accounted for in the GMW, whose version 3.0 dates from 2022. Likewise, the heterogeneity in the operational definitions of “mangrove” such as the inclusion or exclusion of associated salt marshes introduces variability.
-
ICF (2024)A clarification on the existence of Rhizophora species along the Pacific coast of Central AmericaBrenesia, 1987
Discrepancies in the current mangrove cover (61,253.57 ha) compared to that of the GMW (60,564 ha) and the ICF (61,639 ha) can also be attributed to methodological, technological, and temporal factors. First, the Sistema de Información para la Gestión y Monitoreo Forestal (SIGMOF 2024) reports that forest cover for 2024 was estimated with a declared accuracy of 92% and a margin of error of 8%, using multi-source data from the Sentinel-1 (radar) and Sentinel-2 (optical) satellites. This approach, which combines optical and radar sensors, has enabled a more robust classification in the face of adverse weather conditions (e.g., cloud cover), which could explain its slightly higher estimate.
-
SIGMOF 2024Cobertura y Uso de la Tierra, Honduras 2024, 2024
The GMW, on the other hand, uses L-band Synthetic Aperture Radar (SAR) data, developed under the Kyoto & Carbon initiative of JAXA. This technology is notable for its sensitivity to woody biomass and its ability to monitor mangroves in areas with frequent cloud cover (Lucas et al. 2014). However, as mentioned by Bunting et al. (2018), this technology has inherent limitations in discriminating mangroves from other woody vegetation covers (especially terrestrial forests and crop fields) on continental margins, which can lead to classification errors. Furthermore, the GMW uses a canopy cover threshold of 10% to define “mangrove,” whereas the ICF could apply more inclusive criteria, such as including areas undergoing regeneration.
-
Lucas et al. 2014Contribution of L-band SAR to systematic global mangrove monitoringMar Freshwater Res, 2014
-
Bunting et al. (2018)The Global Mangrove Watch-a new 2010 global baseline of mangrove extentRemote Sens, 2018
GMW version 2.0 (Bunting et al. 2019), currently considered the global reference standard due to its spatial resolution (25 m) and temporal update, optimized its methodology by integrating Landsat optical data to improve the accuracy in the delimitation of mangrove extent. However, as demonstrated by Thomas et al. (2014, 2018), L-band SAR sensors, while optimal for detecting temporal changes, have limited capabilities for accurately classifying absolute extent, particularly in regions with complex ecotonal gradients. This technical limitation adds to the global challenges documented in mangrove datasets, which include: (1) unmapped areas due to sensor failures (e.g., scan line error in Landsat 7 ETM+), (2) uneven temporal availability of images, and (3) interference from persistent cloud cover (Spalding 2010, Giri et al. 2011, Bunting et al. 2018).
-
Bunting et al. 2019Global Mangrove Watch (1996-2016), version 2.0, 2019
-
Thomas et al. (2014, 2018)An approach to monitoring mangrove extents through time-series comparison of JERS-1 SAR and ALOS PALSAR dataWetl Ecol Manag, 2014
-
Spalding 2010World Atlas of Mangroves, 2010
-
Giri et al. 2011Status and distribution of mangrove forests of the world using earth observation satellite dataGlob Ecol Biogeogr, 2011
-
Bunting et al. 2018The Global Mangrove Watch-a new 2010 global baseline of mangrove extentRemote Sens, 2018
The discrepancy in the results, although showing little difference in cover compared to GMW v. 2.0 (2020) and ICF (2024), could be attributed to differences in classification algorithms and variability in the quality of input data used by the various studies found in this research. This uncertainty underscores the need to implement standardized protocols that integrate complementary technologies to improve accuracy at local scales. The findings presented in this study emphasize that, while GMW represents a significant advance in global mangrove monitoring, its application to national-scale studies requires in situ validation and specific methodological adjustments that consider the biogeographical characteristics of each region.
-
ICF
(2024)A clarification on the existence of Rhizophora species along the Pacific coast of Central AmericaBrenesia, 1987
Conclusions
This review provides a synthesis of the drivers of mangrove forest cover loss in Honduras. The 52.9% decline in mangrove cover in Honduras over the past few decades significantly exceeded the global average rate of deforestation, positioning the country as a hotspot for mangrove loss. This severe decline, with an average annual rate of 1.27%, has been driven by a complex interplay of factors, with anthropogenic pressures emerging as the primary catalysts on both coasts. Specifically, the expansion of aquaculture in the PCH (Gulf of Fonseca) and, in parallel, tropical cyclones, agricultural intensification and infrastructure development in the ACH, represent the most important threats.
It is essential to implement strict monitoring and conservation measures, focusing on the most affected areas, while prioritizing comprehensive research in data-deficient regions, such as Gracias a Dios and Cayos Cochinos, to establish an accurate baseline of the extent and condition of their mangroves. This initial synthesis quantifies loss rates and elucidates the socioecological drivers of deforestation, laying the groundwork for future research employing advanced remote sensing methodologies and predictive models. Combined with high-resolution multispectral remote sensing and systematic field sampling, these approaches, which enable more precise and dynamic monitoring of mangrove cover, will reduce current margins of error and generate the robust information needed for the sustainable management of these critical ecosystems. Ultimately, integrating robust conservation policies with effective environmental governance will be fundamental to mitigate anthropogenic and natural impacts and ensure the resilience and continued provision of ecosystem services that these critical ecosystems provide to coastal communities and the biodiversity of the country.
Acknowledgments
We thank Alexandra Bonilla for her valuable assistance during the field visits to the mangroves in the Gulf of Fonseca area, Jorge Funez for his support in capturing photographs, María Fernanda Flores-Bueso for her collaboration with the images of the mangroves in Utila, and the reviewers who helped to improve the manuscript substantially. We thank the Mesoamerican Reef Fund (MAR Fund) for its financial support for the translation of this article.
Referencias
- Acharya G. 2002. Life at the margins: the social, economic and ecological importance of mangroves. Mad Bosq. 8:53-60. Links
- Adame MF, Reef R, Santini NS, Najera E, Turschwell MP, Hayes MA, Masque P, Lovelock CE. 2021. Mangroves in arid regions: ecology, threats, and opportunities. Estuar Coast Shelf Sci. 248:106796. https://doi.org/10.1016/j.ecss.2020.106796. Links
- Alfaro-Trejos S. 2011. Cobertura y uso de la tierra en el ecosistema de mangle y zona ecotonal del corredor del mangle, desde la Bahía de Jiquilisco, El Salvador, hasta el Estero Padre Ramos, Jiquilillo, Nicaragua. Rev Geogr Amér Cent. 2:1-19. Links
- Alongi DM. 2018. Impact of global change on nutrient dynamics in mangrove forests. Forests. 9(10):596. https://doi.org/10.3390/f9100596 Links
- Aubrey DG, Emery KO, Uchupi E. 1988. Changing coastal levels of South America and the Caribbean region from tide-gauge records. Tectonophysics. 154(3/4):269-284. https://doi.org/10.1016/0040-1951(88)90108-4 Links
- Bhomia RK, Kauffman JB, McFadden TN. 2016. Ecosystem carbon stocks of mangrove forests along the Pacific and Caribbean coasts of Honduras. Wetlands Ecol Manag. 24(2):187-201. https://doi.org/10.1007/s11273-016-9483-1 Links
- Bhowmik AK, Padmanaban R, Cabral P, Romeiras MM. 2022. Global mangrove deforestation and its interacting social-ecological drivers: a systematic review and synthesis. Sustainability. 14(8):4433. https://doi.org/10.3390/su14084433. Links
- Bryan-Brown DN, Connolly RM, Richards DR, Adame F, Friess DA, Brown CJ. 2020. Global trends in mangrove forest fragmentation. Sci Rep. 10(1):1-8. https://doi.org/10.1038/s41598-020-63880-1 Links
- Bunting P, Rosenqvist A, Lucas R, Rebelo L-M, Hilarides L, Thomas N, Hardy A, Itoh T, Shimada M, Finlayson C. 2018. The Global Mangrove Watch-a new 2010 global baseline of mangrove extent. Remote Sens. 10:1669. https://doi.org/10.3390/rs10101669 Links
- Bunting P, Rosenqvist A, Lucas R, Rebelo L-M, Hilarides L, Thomas N, Hardy A, Itoh T, Shimada M, Finlayson M. 2019. Global Mangrove Watch (1996-2016), version 2.0. Zenodo. https://doi.org/10.5281/zenodo.5658808 Links
- Bunting P, Rosenqvist A, Hilarides L, Lucas RM, Thomas N, Tadono T, Worthington TA, Spalding M, Murray NJ, Rebelo L-M. 2022. Global change in mangrove extent 1996-2020: Global Mangrove Watch, version 3.0. Remote Sens. 14(15):3657. Links
- Burgos-Bennett E. 2011. Revisión General de los Humedales Marino-Costeros del Área de La Bahía de Trujillo-Guaimoreto-Aguan; Colón, Honduras. Trujillo (Honduras): Fundación Calentura Guaimoreto/The Nature Conservancy. Technical report. 71 p. Links
- Cahoon DR, Hensel P. 2002. Hurricane Mitch: A Regional Perspective on Mangrove Damage, Recovery, and Sustainability. Reston (USA): United States Geological Survey. Technical report, No. 2003-183. 31 p. Links
- Cahoon DR, Hensel P, Rybczyk J, McKee KL, Proffitt CE, Perez BC. 2003. Mass tree mortality leads to mangrove peat collapse at Bay Islands, Honduras, after Hurricane Mitch. J Ecol. 91(6):1093-1105. https://doi.org/10.1046/j.1365-2745.2003.00841.x Links
- Canty SWJ. 2007. Positive and Negative Impacts of Dive Tourism: The Case Study of Utila, Honduras [dissertation]. [Suiza]: Lunds Universitet. 41 p. https://www.lumes.lu.se/sites/lumes.lu.se/files/steven_canty.pdf Links
- Canty SWJ, Preziosi R, Rowntree JK. 2018. Dichotomy of mangrove management: a review of research and policy in the Mesoamerican reef region. Ocean Coast Manag. 157:40-49. https://doi.org/10.1016/j.ocecoaman.2018.02.011 Links
- Canty SWJ, Kennedy JP, Fox G, Matterson K, González VL, Núñez-Vallecillo ML, Preziosi RF, Rowntree JK. 2022. Mangrove diversity is more than fringe deep. Sci Rep. 12(1):1-10. https://doi.org/10.1038/s41598-022-05847-y Links
- Carrasco JC, Caviedes V. 2014. Diagnóstico de los Ecosistemas Marino-Costeros y de Agua Dulce de Honduras: Basado en Análisis de Viabilidad, Amenazas y Situación. Tegucigalpa (Honduras): Instituto Nacional de Conservación y Desarrollo Forestal, Áreas Protegidas y Vida Silvestre/United States Agency for International Development ProParque. 102 p. Links
- Carrasco JC, Colindres I. 2011. Plan de manejo del sistema lagunar de Karataska. Programa de las Naciones Unidad para el Desarrollo. Karataska (Honduras): Comité de Gobernanza Pesquera del Sistema Lagunar de Karataska. Management Plan. 75 p. Links
- Carrasco JC, Portillo H, Estuardo S, Lara K. 2013a. Plan de Conservación de la Reserva del Hombre y la Biósfera del Río Plátano: Basado en Análisis de Amenazas, Situación y del Impacto del Cambio Climático, y Definición de Metas y Estrategias. Tegucigalpa (Honduras):Instituto Nacional de Conservación y Desarrollo Forestal, Áreas Protegidas y Vida Silvestre/United States Agency for International Development ProParque. Technical report. 86 p. Links
- Carrasco JC, Secaira E, Lara K. 2013b. Plan de Conservación del Parque Nacional Blanca Jeannette Kawas Fernández: Basado en Análisis de Amenazas, Situación y del Impacto del Cambio Climático, y Definición de Metas y Estrategias. Tegucigalpa (Honduras): Instituto Nacional de Conservación y Desarrollo Forestal, Áreas Protegidas y Vida Silvestre/United States Agency for International Development ProParque . Technical report. 55 p. Links
- Carrasco JC, Stuardo S, Lara K. 2013c. Plan de Conservación del Refugio de Vida Silvestre Barras de Cuero y Salado: Basado en Análisis de Amenazas, Situación y del Impacto del Cambio Climático, y Definición de Metas y Estrategias. Tegucigalpa (Honduras): Instituto Nacional de Conservación y Desarrollo Forestal, Áreas Protegidas y Vida Silvestre/United States Agency for International Development ProParque /Fundicion Acero y Hierro, Minería, Industria, Fundición. Techical Report. 49 p. Links
- Carrasco JC, Secaira E, Lara K. 2013d. Plan de Conservación del Parque Nacional Marino Islas de la Bahía: Basado en Análisis de Amenazas, Situación y del Impacto del Cambio Climático, y Definición de Metas y Estrategias. Tegucigalpa (Honduras): Instituto Nacional de Conservación y Desarrollo Forestal, Áreas Protegidas y Vida Silvestre/United States Agency for International Development ProParque. Technical report. 55 p. Links
- Castañeda-Moya E, Rivera-Monroy VH, Twilley RR. 2006. Mangrove zonation in the dry life zone of the Gulf of Fonseca, Honduras. Estu Coast. 29(5):751-764. https://doi.org/10.1007/BF02786526 Links
- Chen CF, Son NT, Chang N, Chen CR, Chang LY, Valdez M, Centeno G, Thompson CA, Aceituno JL. 2013. Multi-decadal mangrove forest change detection and prediction in Honduras, Central America, with landsat imagery and a markov Chain Model. Remote Sens. 5(12):6408-6426. https://doi.org/10.3390/rs5126408 Links
- CONGESA; Consultores en Gestión ambiental. 2001. Valoración económica de los manglares del Golfo de Fonseca, Honduras. Choluteca (Honduras): PROMANGLE AFE-COHDEFOR/OIMT. Technical report. 137 p. Links
- Davila-Chuga CL. 2021. Implicaciones Socioeconómicas en los Servicios Ecosistémicos de los Bosques de Manglar en las Costas Pacífico y Caribe de Honduras [dissertation]. [Honduras]: Universidad de Zamorano. 142 p. Links
- Del Cid-Gómez JA, Cáceres JD. 2017. Variación de la línea de costa en la aldea de Cedeño, y cartografía de amenaza ante marejadas y ascenso del nivel del mar. Port Cienc. 13:87-102. https://doi.org/10.5377/pc.v13i0.5969 Links
- Dewalt BR, Vergne P, Hardin M. 1996. Shrimp aquaculture development and the environment: people, mangroves and fisheries on the Gulf of Fonseca, Honduras. World Dev. 24(7):1193-1208. https://doi.org/10.1016/0305-750X(96)00033-2 Links
- Doiron S, Weissenberger S. 2014. Sustainable dive tourism: Social and environmental impacts. The case of Roatan, Honduras. Tour Manag Perspect. 10:19-26. https://doi.org/10.1016/j.tmp.2013.12.003 Links
- Doyle TW, Michot TC, Roetker F, Sullivan J, Melder M, Handley B, Balmat J. 2002. Hurricane Mitch: Landscape Analysis of Damaged Forest Resources of the Bay Islands and Caribbean Coast of Honduras. Reston (USA): United States Geological Survey . Technical report. 12 p. Links
- Ellison AM, Farnsworth EJ. 1996. Anthropogenic disturbance of caribbean mangrove ecosystems: past impacts, present trends, and future predictions. Biotropica. 28(4):549-565. Links
- FAO; Food and Agriculture Organization of the United Nations. 2007. The World’s Mangroves 1980-2005. Rome (Italy): FAO. Technical report. 78 p. http://www.fao.org/3/a1427e/a1427e00.pdf Links
- FAO; Food and Agriculture Organization of the United Nations. 1996. Forest Resources Assessment 1990: Survey of Tropical Forest Cover and Study of Change Orocesses. Rome (Italy): FAO . Forestry Paper, no. 130. 152 p. Links
- Feller IC, Friess DA, Krauss KW, Lewis RR. 2017. The state of the world’s mangroves in the 21st century under climate change. Hydrobiologia. 803(1):1-12. https://doi.org/10.1007/s10750-017-3331-z Links
- Fickert T, Grüninger F. 2010. Floristic zonation, vegetation structure, and plant diversity patterns within a Caribbean mangrove and swamp forest on the Bay Island of Utila (Honduras). Ecotropica. 16(2):73-92. Links
- Fickert T. 2018. Better resilient than resistant-regeneration dynamics of storm-disturbed mangrove forests on the bay island of Guanaja (Honduras) during the first two decades after Hurricane Mitch (October 1998). Diversity. 10(1):8. https://doi.org/10.3390/d10010008 Links
- Fickert T. 2020. To plant or not to plant, that is the question: reforestation vs. natural regeneration of hurricane-disturbed mangrove forests in Guanaja (Honduras). Forests. 11(10):1068. https://doi.org/10.3390/f11101068 Links
- Field CD. 1995. Impact of expected climate change on mangroves. Hydrobiologia. 295(1-3):75-81. https://doi.org/10.1007/BF00029113 Links
- Figueiroa AC, Brasil G, Pellin A, Scherer MEG. 2016. Evaluación de la efectividad de la integración de las unidades de conservación marino-costera federales en Santa Catarina. Desarro Medio Ambient. 38:361-375. https://doi.org/10.5380/dma.v38i0.46974 Links
- Flores-Marin AF. 2017. Optimización espacial para la restauración ecológica de manglares en el Parque Nacional Blanca Jeannette Kawas Fernández, Honduras [dissertation]. [Costa Rica]: Centro Agronómico Tropical de Investigación y Enseñanza. 83 p. https://repositorio.catie.ac.cr/handle/11554/8614 Links
- Flores-Bueso MF. 2022. Estructura poblacional, condición corporal de la iguana de cola espinosa de Útila (Ctenosaura bakeri) y análisis del cambio de cobertura de manglar en Útila, Islas de la Bahía, Honduras [dissertation]. [Honduras]: Universidad Nacional de Costa Rica. 71 p. Links
- Friess DA, Rogers K, Lovelock CE, Krauss KW, Hamilton SE, Lee SY, Lucas R, Primavera J, Rajkaran A, Shi S. 2019. The state of the world’s mangrove forests: past, present, and future. Annu Rev Environ Resourc. 44:89-115. https://doi.org/10.1146/annurev-environ-101718-033302 Links
- Friess DA, Adame MF, Adams JB, Lovelock CE. 2022. Mangrove forests under climate change in a 2°C world. WIREs Clim. Change. 13(4):e792. https://doi.org/10.1002/wcc.792 Links
- Giri C, Ochieng E, Tieszen LL, Zhu Z, Singh A, Loveland T, Masek J, Duke N. 2011. Status and distribution of mangrove forests of the world using earth observation satellite data. Glob Ecol Biogeogr. 20:154-159. https://doi.org/10.1111/j.1466-8238.2010.00584.x Links
- Goldberg L, Lagomasino D, Thomas N, Fatoyinbo T. 2020. Global declines in human-driven mangrove loss. Glob Chang Biol. 26(10):5844-5855. https://doi.org/10.1111/gcb.15275 Links
- Hagger V, Worthington TA, Lovelock CE, Adame MF, Amano T, Brown BM, Friess DA, Landis E, Mumby PJ, Morrison TH, et al. 2022. Drivers of global mangrove loss and gain in social-ecological systems. Nat Comm. 13:6373. https://doi.org/10.1038/s41467-022-33962-x Links
- Hamilton SE, Casey D. 2016. Creation of a high spatio-temporal resolution global database of continuous mangrove forest cover for the 21st century (CGMFC-21). Glob Ecol Biogeogr. 25(6):729-738. https://doi.org/10.1111/geb.12449 Links
- Hamza AJ, Esteves LS, Cvitanović M, Kairo JG. 2024. Global patterns of mangrove resource utilization: a systematic review. Front Sustain Resour Manag. 3:1395724. https://doi.org/10.3389/fsrma.2024.1395724 Links
- Hensel P, Proffitt E. 2003. Hurricane Mitch: Acute Impacts on Mangrove Forest Structure and an Evaluation of Recovery Trajectories. Reston (USA): United States Geological Survey . Technical report, no. 2003-182. 77 p. Links
- ICF; Instituto Nacional de Conservación y Desarrollo Forestal, Áreas Protegidas y Vida Silvestre. 2012. Plan de Manejo Parque Nacional Cuyamel-Omoa. Tegucigalpa (Honduras): ICF. Management plan. 179 p. Links
- ICF; Instituto Nacional de Conservación y Desarrollo Forestal, Áreas Protegidas y Vida Silvestrel. 2022. Actual ecosistemas de manglar en Honduras. Tegucigalpa (Honduras): ICF . Technical report. 79 p. Links
- ICF; Instituto Nacional de Conservación y Desarrollo Forestal, Áreas Protegidas y Vida Silvestre. 2024. Mapa de Cobertura Forestal 2024: Geoportal del Sector Forestal de Honduras; [accessed 2025 May 30]. https://geoportal.icf.gob.hn/geoportal/main Links
- Jiménez JA. 1987. A clarification on the existence of Rhizophora species along the Pacific coast of Central America. Brenesia. 28:25-32. Links
- Jiménez JA. 1992. Mangrove Forests of the Pacific Coast of Central America. In: Seeliger U (ed.), Coastal Plant Communities of Latin America. Cambridge (USA): Academic Press. p. 259-267. Links
- Jiménez JA. 1999. El Manejo de los Manglares en el Pacífico de Centroamérica: Usos Tradicionales y Potenciales. In: Yañez-Arancibia AA, Lara-Dominguez L (eds.). Ecosistemas de Manglar en América Tropical. Xalapa (Mexico): Instituto de Ecología A.C. p. 275-290. Links
- Kammerbauer J, Moncada J. 1998. Pesticide residue assessment in three selected agricultural production systems in the Choluteca River Basin of Honduras. Environ Poll. 103(2-3):171-181. https://doi.org/10.1016/S0269-7491(98)00125-0 Links
- Krauss KW, Osland MJ. 2020. Tropical cyclones and the organization of mangrove forests: a review. Ann Bot. 125(2):213-234. https://doi.org/10.1093/aob/mcz161 Links
- Lebigre JM, Portillo P, Thompson W. 2003. Quel avenir pour les mangroves de l’archipel de la Bahía (Honduras)? In: Hequette A (ed.), Actes du Colloque. Espace littoraux en mutation. Côte d’Opale (France): Commission de Géographie de la Mer et des Littoraux-Université du Littoral. p. 63-71. Links
- Lee SY, Primavera JH, Dahdouh-Guebas F, Mckee K, Bosire JO, Cannicci S, Diele K, Fromard F, Koedam N, Marchand C, et al. 2014. Ecological role and services of tropical mangrove ecosystems: a reassessment. Glob Ecol Biogeogr. 23(7):726-743. https://doi.org/10.1111/geb.12155 Links
- Lizano OG, Amador JA, Soto R. 2001. Caracterización de manglares de Centroamérica con sensores remotos. Rev Biol Trop. 49(S2):331-340. Links
- Lovelock CE, Feller IC, Reef R, Hickey S, Ball MC. 2017. Mangrove dieback during fluctuating sea levels. Sci Rep. 7(1):1-8. https://doi.org/10.1038/s41598-017-01927-6 Links
- Lucas R, Rebelo L-M, Fatoyinbo L, Rosenqvist A, Itoh T, Shimada M, Simard M, Souza-Filho PW, Thomas N, Trettin C, et al. 2014. Contribution of L-band SAR to systematic global mangrove monitoring. Mar Freshwater Res. 65:589-603. https://doi.org/10.1071/MF13177 Links
- Marin M, Hernandez GI. 2002. La contaminación marina en Honduras. Tegucigalpa (Honduras): MAR Fund. Technical report. 22 p. Links
- Maya-Jariego I, Holgado D, Castillo AC, Florido del Corral D, Gòmez Mestres S. 2023. Children’s interaction with ecosystem services in a mangrove forest: perceptions of fisherfolks on the involvement of minors in fishing activities in the Gulf of Fonseca in Honduras. Ocean Coast Manag. 236:106502. https://doi.org/10.1016/j.ocecoaman.2023.106502 Links
- McKee KL, McGinnis T. 2003. Hurricane Mitch: Effects on Mangrove Soil Characteristics and Root Contributions to Soil Stabilization. Reston (USA): United States Geological Survey . Technical report, no. 2003-178. 58. p. Links
- McKee KL, Cahoon DR, Feller IC. 2007. Caribbean mangroves adjust to rising sea level through biotic controls on change in soil elevation. Global Ecol Biogeogr. 16(5):545-556. https://doi.org/10.1111/j.1466-8238.2007.00317.x Links
- Medeiros IS, Santos SS, Rebelo VA, Almeida IC, Veloso TMG, Almeida NV, Borges JCG. 2023. Effectiveness of federal protected areas in the preservation of mangrove forests on the coast of the state of Paraíba, Brazil. An Acad Bras Cienc. 95(1):e20211079. https://doi.org/10.1590/0001-3765202320211079 Links
- Meyer DE. 1999. Presence of pesticide residues in water, sediment and biological samples taken from aquatic environments in Honduras. Vienna (Austria): International Atomic Energy Agency. p. 23-28. Links
- Moher D, Liberati A, Tetzlaff J, Altman D. 2009. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 6(7):e1000097. https://doi.org/10.1371/journal.pmed.1000097 Links
- Monserrate F, Valencia J, Leverón S, Pineda J, Cartagena C. 2017. Generación de los Mapas Oficiales de Cuencas, Subcuencas y Microcuencas para el Territorio Hondureño (Memoria Técnica). Tegucigalpa (Honduras): United States Agency for International Development, Centro Internacional de Agricultura Tropical, Dirección General de Recursos Hídricos. Technical report. 26 p. Links
- Moore AC, Hierro L, Mir N, Stewart T. 2022. Mangrove cultural services and values: current status and knowledge gaps. People Nat. 4:1083-1097. https://doi.org/10.1002/pan3.10375 Links
- Morais J, Pinheiro L. 2011. The effect of semi-aridity and damming on sedimentary dynamics in estuaries-northeastern region of Brazil. J Coast Res. 64:1540-1544. Links
- Murillo-Sandoval PJ, Fatoyinbo L, Simard M. 2022. Mangroves cover change trajectories 1984-2020: the gradual decrease of mangroves in Colombia. Front Mar Sci. 9:892946. https://doi.org/10.3389/fmars.2022.892946 Links
- Osorto-Nuñez MH. 2022. Los modelos de cambio climático futuro como predictores de la reducción del área de distribución de dos especies de cíclidos endémicos de Honduras. Biol Acuát. 38:e028-e028. https://doi.org/10.24215/16684869e028 Links
- Osorto-Nuñez MH, Rodríguez DM, Rodríguez VM. 2023. Evaluación de la calidad de agua mediante un análisis multivariante en los esteros del Golfo de Fonseca, Honduras. Bol Invest Mar Cost. 52(1):65-92. https://doi.org/10.25268/bimc.invemar.2023.52.1.1181 Links
- Páez-Osuna F. 2001. The environmental impact of shrimp aquaculture: causes, effects, and mitigating alternatives. Environ Manag. 28(1):131-140. https://doi.org/10.1007/s002670010212 Links
- Pullin AS, Stewart GB. 2006. Guidelines for systematic review in conservation and environmental management. Conserv Biol. 20(6):1647-1656. https://doi.org/10.1111/j.1523-1739.2006.00485.x Links
- Ramirez XA. 1994. CODDEFFAGOLF: Los defensores de los manglares del Golfo de Fonseca, Honduras. Rev Forest Centroamericana. 9:27-32. Links
- Recio ME, Kuper J, Valllejo M, Sommerville M, Jhaveri N. 2016. Central America mangroves, tenure, and REDD+ assessment. Washington (USA): United States Agency for International Development Tenure and Global Climate Change Program. Technical report. 28 p. Links
- Rivera-Monroy VH, Twilley RR, Castaneda E. 2002. Hurricane Mitch: Integrative Management and Rehabilitation of Mangrove Resources to Develop Sustainable Shrimp Mariculture in the Gulf of Fonseca, Honduras. Reston (USA): United States Geological Survey . Technical report. 120 p. Links
- Rivera-Monroy VH, Castañeda-Moya E, Carrasco JC, Caviedes V, Simard M. 2013. Análisis de Carbono Orgánico Total en Manglares del Parque Nacional Jeannette Kawas. Nairobi (Kenya): Programa de las Naciones Unidas para el Medio Ambiente (PNUMA). Technical report. 31 p. Links
- Sanchez-Paez H, Guevara-Mancera OA. 2000. Diagnóstico y Zonificación Preliminar de los Bosques de Mangle del Golfo de Fonseca, Honduras. Choluteca (Honduras): PROMANGLE/AFE-COHDEFOR/OIMT. Technical report. 149 p. Links
- Schaeffer-Novelli Y, Soriano-Sierra EJ, Do Vale CC, Bernini E, Rovai AS, Pinheiro MA, Schmidt AJ, De Almeida R, Coelho Júnior C, et al. 2016. Climate changes in mangrove forests and salt marshes. Brazil J Oceanogr. 64(2):37-52. Links
- SIGMOF; Sistema de Información para la Gestión y Monitoreo Forestal. 2024. Cobertura y Uso de la Tierra, Honduras 2024: SIGMOF; [accessed 2025 Jun 8]. https://sigmof.icf.gob.hn/cobertura-y-uso-de-la-tierra-2024 Links
- Son NT, Chen CF, Chen CR. 2017. Mapping mangrove density from rapideye data in Central America. Open Geosci. 9(1):211-220. https://doi.org/10.1515/geo-2017-0018 Links
- Spalding M. 2010. World Atlas of Mangroves. London (UK): Routledge. 336 p. https://doi.org/10.4324/9781849776608 Links
- Spalding M, Leal M. 2021. The State of the World’s Mangroves 2021. Gobal Mangrove Alliance. 198 p. Links
- Sruthi P, Shackira AM, Puthur JT. 2016. Heavy metal detoxification mechanisms in halophytes: an overview. Wetlands Ecol Manag. 25(2):129-148. https://doi.org/10.1007/s11273-016-9513-z Links
- Stanley DL. 1998. Explaining persistent conflict among resource users: the case of Honduran mariculture. Soc Nat Res. 11(3):267-278. https://doi.org/10.1080/08941929809381079 Links
- Stonich SC. 1992. Struggling with Honduran poverty: the environmental consequences of natural resource-based development and rural transformations. World Dev. 20(3):385-399. https://doi.org/10.1016/0305-750X(92)90031-P Links
- Stovall K. 2015. Carbon Storage, Forest Structure, and Fish Biodiversity Across Human-Impacted Mangrove Ecosystems in Tela Bay, Honduras [dissertation]. New Jersey (USA): Princeton University. 67 p. Links
- Sutherland CH, Pérez HI. 2018. La familia Tetrameristaceae descubierta en Honduras. Ceiba. 55(1):69-70. Links
- Thomas N, Lucas R, Itoh T, Simard M, Fatoyinbo L, Bunting P, Rosenqvist A. 2014. An approach to monitoring mangrove extents through time-series comparison of JERS-1 SAR and ALOS PALSAR data. Wetl Ecol Manag. 23:3-17. https://doi.org/10.1007/s11273-014-9370-6 Links
- Thomas N, Bunting P, Lucas R, Hardy A, Rosenqvist A, Fatoyinbo T. 2018. Mapping mangrove extent and change: a globally applicable approach. Remote Sens. 10:1466. https://doi.org/10.3390/rs10091466 Links
- Thornton C, Shanahan M, Williams J. 2003. From wetlands to wastelands: impacts of shrimp farming. Wetl Sci Pract. 20(1):48-53. https://doi.org/10.1672/0732-9393(2003)020[0048:FWTWIO]2.0.CO;2 Links
- Tuholske C, Lopéz-Carr D, Roberts D. 2015. Anthropogenic impacts on Roatán, Honduras: years of land-cover and land-use change. Plurimondi. 16(16):179-190. http://plurimondi.poliba.it/index.php/Plurimondi/article/view/71 Links
- Tuholske C, Tane Z, López-Carr D, Roberts D, Cassels S. 2017. Thirty years of land use/cover change in the Caribbean: Assessing the relationship between urbanization and mangrove loss in Roatán, Honduras. Appl Geogr. 88:84-93. https://doi.org/10.1016/j.apgeog.2017.08.018 Links
- USAID; Agencia de los Estados Unidos para el Desarrollo Internacional. 2012. Análisis de Vulnerabilidad al Cambio Climático del Caribe de Belice, Guatemala y Honduras. Programa Regional de USAID para el Manejo de Recursos Acuáticos y Alternativas Económicas. San Salvador (El Salvador): United States Agency for International Development/Centro Agronómico Tropical de Investigación y Enseñanza/The Nature Conservancy. Technical report. 87 p. Links
- Valiela I, Bowen JL, Joanna Y. 2001. Mangrove forests: one of the world’s threatened major tropical environments. BioScience. 51(10):807-815. https://doi.org/10.1641/0006-3568(2001)051[0807:MFOOTW]2.0.CO;2 Links
- Vanselow K, Kolb M, Fickert T. 2007. Destruction and regeneration of terrestrial, littoral and marine ecosystems on the Island of Guanaja, Honduras, seven years after Hurricane Mitch. Erdkunde. 61:358-371. Links
- Wilburn-King M. 2008. Political ecology of mangroves in southern Honduras: the emergence and evolution of environmental conflict in the Gulf of Fonseca 1973-2006 [dissertation]. [United Kigndom]: University of Cambridge. 375 p. Links
- Zhang Z, Xu XR, Sun, YX, Yu S, Shen YS, Peng, JX. 2014. Heavy metal and organic contaminants in mangrove ecosystems of China: a review. Environ Sci Poll Res. 21(20):11938-11950. https://doi.org/10.1007/s11356-014-3100-8 Links