Scientific Articles
In vitro evaluation mycoparasitic capacity of Irpex lacteus P7B against fungi and oomycetes associated with plant diseases
Abstract:
Background/Objective.
Diseases of agricultural crops affect the yields and quality of products. Synthetic chemical compounds are generally used to control them; these cause harmful impacts to the environment, as well as to human health. In this sense, beneficial microorganisms can be used in agriculture as biocontrol agents, and contribute to obtaining food in sufficient and safe quantities. The fungus Irpex lacteus has been reported as a potential biocontrol agent. The objective of this research work was to evaluate the in vitro mycoparasitic capacity of the endophytic fungus I. lacteus P7B against 22 fungi and one oomycete associated with plant diseases.
Materials and Methods.
The P7B isolate, previously detected as a mycoparasite, was used and molecularly identified by amplification and sequencing of the internal transcribed spacer (ITS) region of ribosomal DNA, using primers ITS1/ ITS4.The confrontations of the mycoparasite (P7B) against the phytopathogenic microorganisms were carried out in PDA culture medium. Three replicates were used for each microorganism, in addition to the controls, which consisted of placing the microorganisms individually.
Results.
Molecular analyses determined that isolate P7B corresponded to Irpex lacteus (GenBank: PP922180). The results of the in vitro assays indicated that I. lacteus P7B inhibited all the phytopathogenic agents with which it was confronted, 100% inhibition by I. lacteus occurred approximately in 14 days, except for Rhizopus spp., this was at 23 days after the confrontations.
Conclusion.
The present study demonstrates that the fungus I. lacteus presented 100% in vitro mycoparasitic capacity against the various fungi and an oomycete evaluated, so future work could focus on evaluating its mycoparasitic activity under field conditions.
Key words::
biocontrol agent, phytopathogens, mycoparasite
Introduction
Diseases caused by fungi and oomycetes currently cause important economic losses in crops (Meng et al., 2009). On the other hand, the world population increase demands greater amounts and quality of agricultural products, and consequently, a greater use of pesticides to fight diseases (Lahlali et al., 2022). However, in recent years, consumers have become more aware of the side effects of the use of pesticides such as chemical fungicides on human health and the environment (Hou and Wu, 2010). One of the alternatives to reduce dependence on pesticides is biological control (Compant et al., 2005; Barratt et al., 2018), which is defined, in general terms, as any living microorganism (including viruses) used to fight a pathogen or pest by parasitism, antibiosis, competition for space or resources (Eilenberg et al., 2001; Stenberg et al., 2021). In this regard, several species of basidiomycete mycoparasites have been reported as potential biocontrol agents (White and Traquair, 2006; Pineda-Suazo et al., 2021). Mycoparasitism is a lifestyle in which the fungus establishes parasitic interactions with other fungi (Karlsson et al., 2017). Mycoparasitic fungi are enzyme producers with the ability to degrade the cell walls of fungi, allowing them to penetrate into other fungi to extract nutrients for their development (Cao et al., 2009). In this sense, the fungus Irpex lacteus is characterized by its saprophytic habit, although it has been proven to have a mycoparasitic behavior under certain conditions, implying interactions in which
-
Meng et al., 2009Common processes in pathogenesis by fungal and oomycete plant pathogens, described with gene ontology termsBMC Microbiology, 2009
-
Lahlali et al., 2022Biological control of plant pathogens: a global perspectiveMicroorganisms, 2022
-
Hou and Wu, 2010Safety impact and farmer awareness of pesticide residuesFood and Agricultural Immunology, 2010
-
Compant et al., 2005Use of plant growth-promoting bacteria for biocontrol of plant diseases: principles, mechanisms of action, and future prospectsApplied and Environmental Microbiology, 2005
-
Barratt et al., 2018The status of biological control and recommendations for improving uptake for the futureBioControl, 2018
-
Eilenberg et al., 2001Suggestions for unifying the terminology in biological controlBioControl, 2001
-
Stenberg et al., 2021When is it biological control? A framework of definitions, mechanisms, and classificationsJournal of Pest Science, 2021
-
White and Traquair, 2006Necrotrophic mycoparasitism of Botrytis cinerea by cellulolytic and ligninocellulolytic BasidiomycetesCanadian Journal of Microbiology, 2006
-
Pineda-Suazo et al., 2021Growth inhibition of phytopathogenic fungi and oomycetes by Basidiomycete Irpex lacteus and identification of its antimicrobial extracellular metabolitesPolish Journal of Microbiology, 2021
-
Karlsson et al., 2017Necrotrophic mycoparasites and their genomesMicrobiology Spectrum, 2017
-
Cao et al., 2009Mycoparasitism of endophytic fungi isolated from reed on soilborne phytopathogenic fungi and production of cell wall-degrading enzymes in vitroCurrent Microbiology, 2009
I. lacteus colonizes and obtains nutrients from other fungi by secreting diverse hydrolytic enzymes (Metreveli et al., 2014; Mezule a d Civzele, 2020; Gafforov et al., 2023). The mycoparasitic abilities I. lacteus suggest possible applications in the biological control of phytopathogens in the agricultural context (White and Traquair, 2006; Sivanandhan et al., 2017; Yin et al., 2021). Due to this, the aim of this investigation was to evaluate the fungus I. lacteus (isolate P7B) in vitro with a dual confrontation against 22 fungi and one oomycete.
-
Mezule a d Civzele, 2020Bioprospecting white-rot Basidiomycete Irpex lacteus for improved extraction of lignocellulose-degrading enzymes and their further applicationJournal of Fungi (Basel), 2020
-
Gafforov et al., 2023Irpex lacteus (Fr.) Fr. - IRPICACEAEEthnobiology of Uzbekistan. Ethnobiology., 2023
-
White and Traquair, 2006Necrotrophic mycoparasitism of Botrytis cinerea by cellulolytic and ligninocellulolytic BasidiomycetesCanadian Journal of Microbiology, 2006
-
Sivanandhan et al., 2017Biocontrol properties of Basidiomycetes: An OverviewJournal of Fungi, 2017
-
Yin et al., 2021Antifeedant and antiphytopathogenic metabolites from co-culture of endophyte Irpex lacteus, phytopathogen Nigrospora oryzae, and entomopathogen Beauveria bassianaFitoterapia, 2021
This work was conducted in the Plant Physiology and Biotechnology Laboratory of the Facultad de Ciencias Agropecuarias y Ambientales of the Universidad Autónoma de Guerrero (FCAA-UAGro), located in Iguala de la Independencia, Guerrero, México.
For this study, isolate P7B was taken from an endophytic mycoparasitic fungus, which was isolated from the asymptomatic area of the Cedrus sp. rhizosphere, identified molecularly by DNA extraction, and for this purpose, the internal transcribed spacer (ITS) region of the ribosomal DNA as amplified by PCR using the ITS1/ITS4 primers (White et al., 1990). DNA extraction, PCR, and sequencing were performed by the sequencing service of the Macrogen company (Macrogen, Inc., Seoul, Korea). The sequences obtained were edited and aligned using the MEGA X® program, and a consensus sequence was obtained, which was compared with those available in the GenBank.
-
White et al., 1990Amplification and Direct sequencing of fungal ribosomal RNA genes for phylogeneticsPCR Protocols. A Guide to Methods and Applications, 1990
Isolate I. lacteus P7B underwent a dual confrontation against 22 fungi and one oomycete associated to diverse diseases (Table 1), belonging to the collection of phytopathogenic fungi of the Plant Physiology and Biotechnology Laboratory of the FCAA-UAGro. Strains of phytopathogenic fungi and the mycoparasitic agent (P7B) aged 12 days, developed in a PDA medium. For the confrontation, a disk, 0.5 cm in diameter and with mycelia, was placed 1.0 cm from the edge of the Petri dish and each isolation was placed on the opposite side of the dish, equidistantly. Three repetitions were used for each fungus or oomycete, along with control treatments, which consisted in placing a mycelium disk from each microorganism on one side of the Petri dish. The culture media were placed at a temperature of 28 °C, and the area of inhibition was recorded using a millimeter ruler when the control treatments covered the entire surface of the Petri dish with a PDA medium, which occurred approximately 14 days after cultured. Exceptionally, the treatments confronted with Rhizopus spp. were incubated for approximately 23 days, since a slow mycoparasitism was observed for I. lacteus P7B for this genus. Photographs were taken of the advancement of the dual confrontation every 24 hours (Sony camera, Vario-Tessar®). Additionally, the area of interaction between microorganisms was analyzed against the antagonistic fungus I. lacteus P7B, in order to observe possible damages in the structures of the parasitized microorganisms, using a compound microscope (LABOMED®).
Based on the values registered of the confrontations between microorganisms and the fungus I. lacteus P7B, the percentage of inhibition was estimated using the formula = (D1-D2)/D1*100.
where:
D1= Mycelial diameter of the control
D2= Mycelial diameter of the confronted microorganism
The analysis of the consensus sequence in the GenBank with the BLAST tool showed that isolate P7B had a percentage of identity of 99.85% with Irpex lacteus (accession number JX290579). The consensus sequence derived from this study was deposited in the GenBank with accession number PP922180.
Table 1
Microorganisms used in the evaluation for the confrontation with Irpex lacteus P7Bfrom the collection of pyhtopathogenic fungi of the Plant Physiology and Biotechnology Laboratory, FCAA-UAGro.
Microorganisms used in the evaluation for the confrontation with Irpex lacteus P7Bfrom the collection of pyhtopathogenic fungi of the Plant Physiology and Biotechnology Laboratory, FCAA-UAGro.
| Microorganisms | Identification Method | Reference | Host | Key | Disease |
|---|---|---|---|---|---|
| Corynespora cassiicola | Morphological and molecular | Ortega-Acosta et al., 2020 | Hibiscus sabdariffa | CC47GRO | Spotting of calyces |
| Colletotrichum gloeosporioides | Morphological and molecular | Cruz-Lagunas et al., 2020 | Citrus paradisi | COLTOR1 | Anthracnose on fruits |
| Rhizopus oryzae | Morphological and molecular | Palemón-Alberto et al., 2019 | Annona muricata | ANTRAGUB | Soft rot of fruits |
| Phytophthora sp. | Morphological | NA | Carica papaya | PAP-4 | Associated with basal rot |
| Macrophomina sp. | Morphological | NA | Arachis hypogaea | C4 | Associated with charcoal rot of fruits |
| Colletotrichum sp. | Morphological | NA | Plumeria rubra | CACALO1 | Associated with anthracnose on leaves |
| Rhizopus sp. | Morphological | NA | Stenocereus sp. | RIPITA | Associated with soft rot of fruits |
| Fusarium sp. | Morphological | NA | Musa sp. | FUPLA | Associated with fruit spotting |
| Alternaria sp. | Morphological | NA | Passiflora edulis | AL1 | Associated with fruit spotting |
| Colletotrichum sp. | Morphological | NA | Mangifera indica | COLMAN | Associated with fruit anthracnose |
| Rhizopus sp. | Morphological | NA | Carica papaya | RIZOPAP | Associated with soft rot of fruits |
| Penicillium sp. | Morphological | NA | Allium sativum | AJOPENI | Associated with bulb rot |
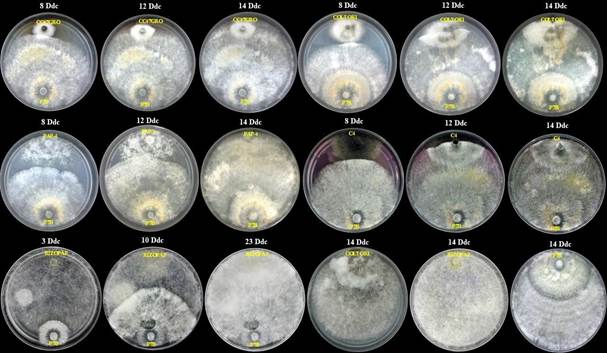
In the in vitro evaluation of 22 fungi and one oomycete against I. lacteus P7B (Table 1), approximately 14 days later, it displayed 100% mycoparasitism on all the microorganisms it was confronted with (Figure 1), except for the genus Rhizopus (23 days). Figure 2 shows some representative examples of the confrontation between I. lacteus P7B against fungi and one oomycete, in which a clear gradual mycelial invasion was observed. By the end of the experiment, it was determined that the fungus I. lacteus P7B induced an inhibition of 100% in all confrontations (Figure 1, 2).
Thumbnail
Figure 1
Effect of the confrontation in dual culture in PDA under in vitro conditions between I. lacteus P7B against fungi and an oomycete associated to plant diseases.
Effect of the confrontation in dual culture in PDA under in vitro conditions between I. lacteus P7B against fungi and an oomycete associated to plant diseases.
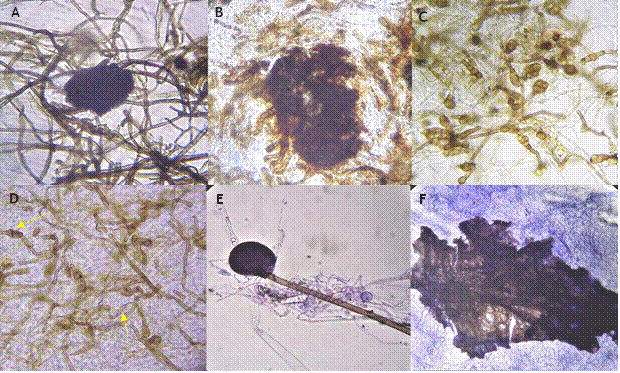
On the other hand, the fungi and an oomycete confronted with I. lacteus P7B all presented degradation of their structures when observed under the microscope. For example, Macrophomina sp. (isolate C4), in the zone of interaction, displayed degradation of sclerotia and hyphae (Figure 3B); Alternaria sp. (isolate AL1), it presented degraded conidia and hyphae (Figure 3D); for Rhizopus sp. (isolate RIZOPAP), degradation of sporangia was observed (Figure 3F); in control treatments, structures displayed no apparent damage (Figure 3A, C and E).
This work showed the mycoparasitic ability of I. lacteus P7B against 22 fungi and one oomycete associated to diverse phytosanitary problems. Literature on the potential of I. lacteus as a biocontrol agent is scarce. The fungus I. lacteus has the ability to produce diverse hydrolytic enzymes such as chitinases and glucanases, which degrade the cell walls of other fungi, facilitating the acquisition of nutrients (Qin et al., 2018; Roncero and Vázquez de Aldana, 2019). In a study carried out by White and Traquair (2006), by confronting I. lacteus against Botrytis cinerea in vitro, they proved that I. lacteus was able to parasite B. cinerea by degrading its structures such as conidiophores and conidia and parasiting its sclerotia, and reported a percentage of mycoparasitism of 100%, similar results reported in this study. On the other hand, in Mexico, I. lacteus has been evaluated against Fusarium pseudocircinatum, F. mexicanum, Colletotrichum coccodes, C. gloeosporioides, Phytophthora capsici and P. cinnamomi with a percentage of inhibition between 16.7 and 46.3% (Pineda-Suazo et al., 2021). In this investigation, I. lacteus P7B displayed a greater capacity for mycoparasiting diverse fungi and an oomycete, possibly due to the type of isolation. In addition, I. lacteus has been reported to belong to the group of necrotrophic mycoparasites, which are characterized for being highly destructive, scarcely specialized (Viterbo et al., 2007) and generally presenting a high range of hosts, including phytopathogens and extend to diverse taxonomic groups (Viterbo and Horwitz et al., 2010), as in this study, where I. lacteus parasite fungi and an oomycete of the divisions Ascomycota, Zygomycota and Oomycota. Additionally, compounds, derived from I. lacteus such as terpenes and aldehydes, have been detected which have an antifungal potential (Pineda- Suazo et al., 2021; Wang et al., 2021).
-
Qin et al., 2018Dye-decolorizing peroxidases in Irpex lacteus combining the catalytic properties of heme peroxidases and laccase play important roles in ligninolytic systemBiotechnology for Biofuels, 2018
-
White and Traquair (2006Necrotrophic mycoparasitism of Botrytis cinerea by cellulolytic and ligninocellulolytic BasidiomycetesCanadian Journal of Microbiology, 2006
-
Pineda-Suazo et al., 2021Growth inhibition of phytopathogenic fungi and oomycetes by Basidiomycete Irpex lacteus and identification of its antimicrobial extracellular metabolitesPolish Journal of Microbiology, 2021
-
Viterbo et al., 2007Plant disease biocontrol and induced resistance via fungal mycoparasitesThe Mycota, vol. IV, 2007
-
Wang et al., 2021The selective anti-fungal metabolites from Irpex lacteus and applications in the chemical interaction of Gastrodia elata, Armillaria sp., and endophytesFitoterapia, 2021
Thumbnail
Figure 2
Effect of the confrontation in dual culture in PDA under in vitro conditions between I. lacteus P7B against fungi and an oomycete associated to plant diseases. P7B = Irpex lacteus. CC47GRO = Corynespora cassiicola. COLTOR1 = Colletotrichum gloeosporioides. PAP-4= Phytophthora sp. C4 = Macrophomina sp. RIZOPAP= Rhizopus sp. Dac = Days after confrontation.
Effect of the confrontation in dual culture in PDA under in vitro conditions between I. lacteus P7B against fungi and an oomycete associated to plant diseases. P7B = Irpex lacteus. CC47GRO = Corynespora cassiicola. COLTOR1 = Colletotrichum gloeosporioides. PAP-4= Phytophthora sp. C4 = Macrophomina sp. RIZOPAP= Rhizopus sp. Dac = Days after confrontation.
Thumbnail
Figure 3
Effect of the confrontation in vitro of I. lacteus P7B against fungi and an oomycete. A= Macrophomina sp. (isolate C4) control; B= Macrophomina sp. (isolate C4) confronted with I. lacteus P7B, a degradation of sclerotia and hyphae can be observed. C = Alternaria sp. (isolate AL1) control; D = Alternaria sp. (isolate AL1) confronted with I. lacteus P7B, in which a degradation of conidia and hyphae can be observed. E = Rhizopus sp. (isolate RIZOPAP) control; F = Rhizopus sp. (isolate RIZOPAP) confronted with I. lacteus P7B, shows degraded sporangia. Images captured with an optic microscope with 10X (A, B, E and F), and 40X objective lens (C and D).
Effect of the confrontation in vitro of I. lacteus P7B against fungi and an oomycete. A= Macrophomina sp. (isolate C4) control; B= Macrophomina sp. (isolate C4) confronted with I. lacteus P7B, a degradation of sclerotia and hyphae can be observed. C = Alternaria sp. (isolate AL1) control; D = Alternaria sp. (isolate AL1) confronted with I. lacteus P7B, in which a degradation of conidia and hyphae can be observed. E = Rhizopus sp. (isolate RIZOPAP) control; F = Rhizopus sp. (isolate RIZOPAP) confronted with I. lacteus P7B, shows degraded sporangia. Images captured with an optic microscope with 10X (A, B, E and F), and 40X objective lens (C and D).
The fungus I. lacteus mycoparasited 100% in vitro 22 fungi and one oomycete evaluated in this study. Future investigations may focus on evaluating the antagonistic activity of I. lacteus under field conditions for the control of phytopathogens, as well as on the evaluation and determination of antifungal compounds derived from I. lacteus P7B.
Literatura citada
- Barratt, BIP Moran, VC Bigler, F Van Lenteren, JC (2018). The status of biological control and recommendations for improving uptake for the future. BioControl 63:155-167. 10.1007/s10526-017-9831-y Links
- Cao, R Liu, X Gao, K Mendgen, K Kang, Z Gao, J Dai, Y Wang, X (2009). Mycoparasitism of endophytic fungi isolated from reed on soilborne phytopathogenic fungi and production of cell wall-degrading enzymes in vitro. Current Microbiology. 10.1007/s00284-009-9477-9 Links
- Compant, S Duffy, B Nowak, J Clément, C Barka, EA (2005). Use of plant growth-promoting bacteria for biocontrol of plant diseases: principles, mechanisms of action, and future prospects. Applied and Environmental Microbiology 71. 10.1128/AEM.71.9.4951-4959.2005 Links
- Cruz-Lagunas, B Ortega-Acosta, SA Reyes-García, G Toribio-Jiménez, J Juárez-López, P Guillen-Sánchez, D Damián-Nava, A Romero-Ramírez, Y Palemón-Alberto, F (2020). Colletotrichum gloeosporioides causes anthracnose on grapefruit (Citrus paradisi) in Mexico. Australasian Plant Disease Notes 15: 31. 10.1007/s13314-020-00401-z Links
- Eilenberg, J Hajek, A Lomer, C (2001). Suggestions for unifying the terminology in biological control. BioControl 46: 387-400. 10.1023/A:1014193329979 Links
- Gafforov, Y Deshmukh, SK Tomšovský, M Yarasheva, M Wang, M Rapior, S (2023). Irpex lacteus (Fr.) Fr. - IRPICACEAE. Ethnobiology of Uzbekistan. Ethnobiology. Springer, Cham, Suiza. 1203-2217. 10.1007/978-3-031-23031-8_114 Links
- Hou, B Wu, L (2010). Safety impact and farmer awareness of pesticide residues. Food and Agricultural Immunology 21(3):191-200. 10.1080/09540105.2010.484858 Links
- Karlsson, M Atanasova, L Jensen, DF Zeilinger, S (2017). Necrotrophic mycoparasites and their genomes. Microbiology Spectrum 5(2):FUNK-0016-2016. 10.1128/microbiolspec.funk-0016-2016 Links
- Lahlali, R Ezrari, S Radouane, N Kenfaoui, J Esmaeel, Q El Hamss, H Belabess, Z Barka, EA (2022). Biological control of plant pathogens: a global perspective. Microorganisms 10(3):596. 10.3390/microorganisms10030596 Links
- Meng, S Torto-Alalibo, T Chibucos, MC Tyler, BM Dean, RA (2009). Common processes in pathogenesis by fungal and oomycete plant pathogens, described with gene ontology terms. BMC Microbiology 9 (Suppl 1):S7. 10.1186/1471-2180-9-S1-S7 Links
- Mezule, L Civzele, A (2020). Bioprospecting white-rot Basidiomycete Irpex lacteus for improved extraction of lignocellulose-degrading enzymes and their further application. Journal of Fungi (Basel) 6(4):256. 10.3390/jof6040256 Links
- Ortega-Acosta, SA Ochoa-Martínez, DL Hernández-Morales, J Palemón-Alberto, F (2020). Morphological and genetic characterization of Corynespora cassiicola isolates obtained from roselle and associated weeds. Mexican Journal of Phytopathology 38(1):62-78. 10.18781/R.MEX.FIT.1909-2 Links
- Palemón-Alberto, F Cruz-Lagunas, B Ortega-Acosta, SA Reyes-García, G Damián-Nava, A Toribio-Jiménez, J Romero-Ramírez, Y Vargas-Álvarez, D Bello-Martínez, A (2020). First report of rhizopus soft rot on soursop (Annona muricata) caused byRhizopus oryzaein México. Journal of Plant Disease and Protection 127:275-277. 10.1007/s41348-019-00287-9 Links
- Pineda-Suazo, D Montero-Vargas, JM Ordaz-Ortiz, JJ Vázquez-Marrufo, G (2021). Growth inhibition of phytopathogenic fungi and oomycetes by BasidiomyceteIrpex lacteusand identification of its antimicrobial extracellular metabolites. Polish Journal of Microbiology 70(1):131-136. 10.33073/pjm-2021-014 Links
- Qin, X Luo, H Zhang, X Yao, B Ma, F Su, X (2018). Dye-decolorizing peroxidases inIrpex lacteuscombining the catalytic properties of heme peroxidases and laccase play important roles in ligninolytic system. Biotechnology for Biofuels 11:302. 10.1186/s13068-018-1303-9 Links
- Sivanandhan, S Khusro, A Paulraj, MG Ignacimuthu, S AL-Dhabi, NA (2017). Biocontrol properties of Basidiomycetes: An Overview. Journal of Fungi 3(1):2. 10.3390/jof3010002 Links
- Viterbo, A Horwitz, BA (2010). Mycoparasitism. Cellular and Molecular Biology of Filamentous Fungi. ASM Press, Washington, DC., USA, 676-693. 10.1128/9781555816636.ch42 Links
- Wang, DL Yang, XQ Shi, WZ Cen, RH Yang, YB Ding, ZT (2021). The selective anti-fungal metabolites fromIrpex lacteusand applications in the chemical interaction ofGastrodia elata,Armillariasp., and endophytes. Fitoterapia 155:105035. 10.1016/j.fitote.2021.105035 Links
- Yin, HY Yang, XQ Wang, DL Zhao, TD Wang, CF Yang, YB Ding, ZT (2021). Antifeedant and antiphytopathogenic metabolites from co-culture of endophyteIrpex lacteus, phytopathogenNigrospora oryzae, and entomopathogenBeauveria bassiana. Fitoterapia 148:104781. 10.1016/j.fitote.2020.104781 Links
- White, GJ Traquair, JA (2006). Necrotrophic mycoparasitism ofBotrytis cinereaby cellulolytic and ligninocellulolytic Basidiomycetes. Canadian Journal of Microbiology 52(6):508-518. 10.1139/w05-141 Links
- White, TJ Bruns, T Lee, S Taylor, J (1990). Amplification and Direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR Protocols. A Guide to Methods and Applications. Academic Press, CA, USA, 315-322. Links
- Stenberg, JA Sundh, I Becher, PG Bjorkman, C Dubey, M Egan, PA Friberg, H Gil, JF Jensen, DF Jonsson, M Karlsson, M Khalil, S Ninkovic, V Rehermann, G Vetukuri, RR Viketoft, M (2021). When is it biological control? A framework of definitions, mechanisms, and classifications. Journal of Pest Science 94:665-676. 10.1007/s10340-021-01354-7 Links
- Viterbo, A Inbar, J Hadar, Y Chet, I (2007). Plant disease biocontrol and induced resistance via fungal mycoparasites. The Mycota, vol. IV. Springer, Berlin, Germany, 127-146. 10.1007/978-3-540-71840-6_8 Links
- Wang, DL Yang, XQ Shi, WZ Cen, RH Yang, YB Ding, ZT (2021). The selective anti-fungal metabolites fromIrpex lacteusand applications in the chemical interaction ofGastrodia elata,Armillariasp., and endophytes. Fitoterapia 155:105035. 10.1016/j.fitote.2021.105035 Links
- Yin, HY Yang, XQ Wang, DL Zhao, TD Wang, CF Yang, YB Ding, ZT (2021). Antifeedant and antiphytopathogenic metabolites from co-culture of endophyteIrpex lacteus, phytopathogenNigrospora oryzae, and entomopathogenBeauveria bassiana. Fitoterapia 148:104781. 10.1016/j.fitote.2020.104781 Links