H. HANDT, R. FERNÁNDEZ,
Instituto de Ciencias de la Tierra, Facultad de Ciencias, Universidad Central de Venezuela,
Apdo. Postal 47325, Caracas 1041–Venezuela
Corresponding author: R. Fernández; e–mails: raizadelvalle@yahoo.com, rfernan@ciens.ucv.ve
Z. BENZO, C. GÓMEZ, E. MARCANO, F. GALÁRRAGA,
Laboratorio de Química Analítica, Centro de Química,
Instituto Venezolano de Investigaciones Científicas,
Apdo. Postal 21827, Caracas 1020–A, Venezuela
Received November 16, 2007; accepted April 30, 2008
RESUMEN
Un método de extracción secuencial fue aplicado para analizar Cd, Cr, Cu, Mn, Ni, Pb, V y Zn en muestras de polvo urbano provenientes de colegios ubicados en zonas con diferentes concentraciones de partículas suspendidas totales. El objetivo de este trabajo fue estudiar el fraccionamiento químico de los elementos en este tipo de muestras. Esta información permite entender los procesos relacionados con el origen, la movilización y el transporte de los metales. Una vez realizada la digestión de las muestras fueron determinadas las concentraciones totales y parciales de los metales. Los resultados obtenidos tanto para la digestión total como para la extracción secuencial son comparables. Los metales determinados están asociados a diversas formas químicas. Pb, Zn y Cu están asociados principalmente a las fases carbonato y óxido, Ni a la fase orgánica y V, Mn, Cr y Cd están presentes principalmente en la fase óxido, aunque también están presentes, en menor proporción, en la fase residual. El análisis multivariado (componentes principales) permitió establecer el fraccionamiento y posibles fuentes de estos elementos.
ABSTRACT
]]> A sequential extraction approach has been applied to analyze Cd, Cr, Cu, Mn, Ni, Pb, V and Zn in dust samples from different schools chosen according with their location in areas with different concentrations of total suspended particles (TSP). The aim of this work is to provide information about the chemical fractionation of metal content. Hence, information about the origin, mode of occurrence, mobilization and transport of metals could be obtained. Total and partial metal concentrations were further determined by analyzing the samples after acid digestion procedure. The results compare very well for both methods. The metals are associated with various chemical forms as distinguished by sequential chemical extraction. The carbonate and Fe–Mn oxide phases dominate for Pb, Zn and Cu, whereas for Ni the organic phase is most important. V, Mn, Cr, Cd are mainly present in the oxide phases, however they are too in a residual phase but in minor proportion. The different sources for the elements studied are supported by the results from multivariate (PCA) analysis.Keywords: Operational speciation, metals, dust, schools.
1. Introduction
Many heavy metals are very common in industrial and domestic usage. Consequently, dust containing heavy metals have been dispersed into the environment and could cause human health problems through the atmosphere. Heavy metals have an effect on biochemical mechanisms, especially because they can be bioaccumulated. The extent of bioaccumulation and bioavailability depends on the characteristic chemical composition of the particles (Fernández and Ternero–Rodríguez, 2004). Thus, quantification of the chemical form of metals present in those particles is necessary. Determination of total concentration is often insufficient and speciation is a mean of determining the real activity of a metal in the environment. Most studies (Orlova et al.,1995; Tong, 1998; Tong et al., 2000; Shorteen and Hooven, 2000) on metal atmospheric contamination have reported total metal concentration. The need for speciation studies in environmental studies is a new trend in analytical chemistry (Szpunar and Lobinski, 1999). Sequential extraction methods using a series of chemicals of increasing strength are widely used for metal fractionation and evaluation of metal leaching (Tongtavee et al., 2005). Metal speciation in airborne particles and dust has been reported (Fernández et al., 2002). Sequential extraction approach can provide detailed information about the origin, mode of occurrence, biological and physicochemical availability, mobilization and transport of metals (Das et al., 1995). Thus, the information obtained by applying this approach is of a great utility in environmental research.
The purpose of this study was to determine the chemical speciation pattern of dust in seven schools at different locations according with the concentration of total auspended particles (TSP) and hence to asses the degree of environmental hazard of children attending to these schools.
2. Methods
2.1 Sampling
Dust samples were collected (7 schools samples, one for each school) from the floor site by careful sweeping of dust using a conventional clean brush and stored in polyethylene bags at room temperature. The samples used in this study were selected from those collected in two campaign samplings in 1998 and 2003, in Caracas, Venezuela. Sample A comes from a school, previously classified as a contaminated area related to the TSP concentration; samples B, D, E and F from schools located in moderated contaminated areas, and C and G from schools in low contaminated areas (Morales et al., 2003).
]]>2.2 Sample treatment
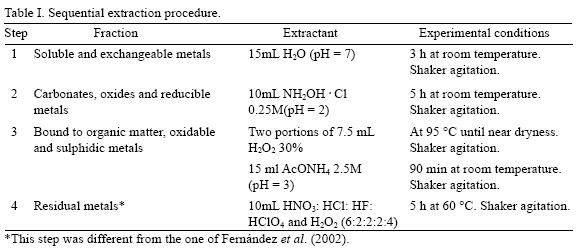
Indoor dust samples collected from classroom places in the schools were passed through a 35 meshes sieve to remove all large particles. This was followed by subsequent passing through a 230 mesh sieve (63 µm) and through a 325 mesh sieve (45 µm). Only the fractions of particle size between 45–63 µm and < 45 µm were analyzed. This was followed by the sequential extraction procedure outlined in Table I. For the purpose of this publication just the results from the 325 mesh sieve (45 µm) are showed.

2.3 Standard reference material (SRM)
SRM 1648 Urban Particulate Matter from the National Institute of Standards and Technology (NIST) (Gaithersburg, MD) was used to validate the acid digestion procedure.
2.4 Preparation of standard solutions and glassware
]]> All chemical reagents were of analytical grade. Deionized water from a Milli Q water purification unit (Millipore, Bedford, MA) was used throughout this work. All glass and plastic ware used was cleaned and soaked in HNO3 (5% v/v) and rinse several times with deionized water before use. Multi–element (Cd, Cr, Cu, Mn, Ni, Pb, V and Zn) standard stock solution (1000 mg L–1) Merck (Darmstadt, Germany) were used to prepare the working standard solutions for calibration purposes.
2.5 Analytical fractionation scheme
The fractionation scheme used was based on Fernández et al. (2002) because it simulates the pH condition found through the human respiratory tract.
The geochemical phase at each extraction step is operationally defined according to the reagents used as shown in Table I. The last step was modified because the presence of a persistent black residue, probably soot.
Dust samples were weighed accurately (0.5 g), transferred into a centrifuge tube and the extractions at each step were carried out using the conditions shown in Table I. A water bath was used for the extraction steps when required. Centrifugation (5000 rpm for 20 min for the first two extracts and 30 min for two last ones), decantation and filtration steps (Whatman filter paper No. 52, metal composition was checked), were used for separation of the liquid and solid phases at each step. The original dust samples were treated using in both total digestion and sequential extraction to examine the efficiency of the two methods.
2. 6 Original sample and residue digestion
Original samples digestion was performed using a mixture of HNO3: HCl: HF: HClO4 and H2O2 (6:2:2:2:4). Amounts of 0.5 g of original dust samples were transferred to plastic centrifuge tubes together with 6 mL HNO3,2 mL HCl,2 mL HF and 2 mL HClO4, heated to 60 °C on a heating plate for 8 hours, and then boric acid 5% (m/v) was added (to mask free fluoride ions in the solution and redissolve fluoride precipitates). Finally, 4 mL of H2O2 were added and heated up to additional 2 hours. The digested solutions cooled to room temperature, centrifuged and decanted were then made up to volume in a 50 mL volumetric flask. Total metal concentrations were determined by both a single digestion of each two granulometric fractions of dust and by summation of extractable metals in each fraction of the exchangeable, carbonate and oxides, oxidable and residual fractions.
The same acid digestion procedure was applied to the residual fraction from step 3 in the sequential extraction procedure.
]]>2. 7 Analysis of extracts and acid digests
Analytes concentrations in extracts and acid digests were determined using inductively coupled plasma optical emission spectroscopy (ICP–OES). The ICP–OES measurements were performed with a Perkin Elmer (Norwalk, CT) spectrometer, model Optima 3000. Concentrations of the metals were obtained by calibration against matrix matching aqueous standards after checking matrix interferences if any, by means of standards addition calibration curves.
2.8 Complementary analysis
X–ray diffraction (XRD) analysis (Siemens D5005) were carried out on the samples in order to determine the mineralogical composition, using Phillips equipment with dust camera and Co anode.
2.9 Statistic analysis
The statistical analysis was based on principal components multivariate statistic program (PCA) often used in environmental studies, Multi–Variate Statistical Package (MVSP) Version 3.1; Varimax rotation was not used (Miranda et al., 1996).
Metals concentration (Cd, Cr, Cu, Mn, Ni, Pb, V and Zn) in each two granulometric fractions from seven schools were evaluated using a PCA on a matrix correlation from eighty four (84) cases (7 school × 3 granulometric fractions × 4 chemical fractions). The results obtained can be visualized in a two dimensional plot (Fig. 1).
]]>
3. Results and discussion
3.1 Sequential extraction
The total concentration of metals are easily computed from a simple summation of the data obtained by each extraction step for all dust samples, and then compared with the results given by the total digestion. The results obtained from the sequential extraction experiments provide an interesting insight into the variability of metals association in dusts at schools located in different areas. Table II shows these results, expressed as mean concentration for three replicates. Most of lead is associated with the carbonate and iron/manganese hydrous oxide fraction, followed by the organic and residual fractions. An interested observation lies in the high concentrations level found for lead (43–1027 µg/g) in this chemical fraction (S2). For school's location F, the total loadings in this fraction account for 51% of the total lead found in this location. This school is located by a highway. This fact, presumably gives rise to the greater build up of lead in those dust samples. These results are similar to those obtained by Evans et al. (1992) and Monaci et al. (2000). The association of lead with the iron/manganese hydrous oxide fraction of dusts gives rise to a pool of lead which though relatively immobile may create long term contamination problems. Interestingly, the pattern of association is not repeated in location A which has been classified as a contaminated area, respect to the total suspended particles (Morales et al., 2003).
The speciation patterns obtained for the elements studied in the different locations are quite different. The results indicate that most of lead, zinc, and copper are tied up with carbonate phase (S2). A good proportion of zinc and copper are also extracted from the organic phase (S3). These metals may have similar patterns of association with the phases in dusts. Nickel is mostly tied up with the organic phase, and an association may exist between the organic content of dust and the amount of nickel extracted from the organic phase. Vanadium, manganese, chromium, cadmium and a proportion of lead remain in the residual material (S4).
Nickel and vanadium come usually from fuel oil (Kowalczyk et al., 1982). Cadmium is associated with motor traffic emissions as a constituent of motor alloys or with the electronic components and batteries of cars. Because of cadmium impurities in zinc materials, cadmium also accompanies zinc oxides used in tire manufacture. Other metals contained in dust that are considered to be a health hazard also come from vehicle emissions. These are: zinc, present in tire wear, brake emissions and corrosion of galvanized parts; iron in auto body rust and engine parts; copper in brake emissions and engine parts; chromium in air conditioning coolants and engine parts; and nickel in diesel fuel and gasoline (Monaci et al., 2000).
Therefore, these metals (Cd, Cr, Cu, Mn, Ni, Pb, V and Zn) can be considered as pollutants of potentially wide dispersion.
Data on metal speciation of dust of different particle size is essential for considering the processes of metal bioavailability as well as environmental risk assessment. Particle size < 45 µm were chosen because they remain in the atmospheric particulate matter which represents an important risk to human health by inhalation, ingest or contact (Tong, 1998; Orlova et al., 1995).
]]> In this study, the fraction of particle size between 45–63 µm was also analyzed following the same fractionation procedure. The results demonstrate that there are slight differences between the two fractions, having in general a slight increase in metal concentration in the smaller particles (< 45 µm) when compared with the 45–63 urn fraction. The particle fraction greater than 63 µm was not analyzed.
in metal concentration in the smaller particles (< 45 µm) when compared with the 45–63 urn fraction. The particle fraction greater than 63 µm was not analyzed.
3.2 Multivariate analysis results
Principal components analysis (PCA) was used to assist in the identification of pollution sources in this kind of samples. Table III shows the factors loadings as well as the eigenvalues. The loading plot of the variables on PC 1 vs PC2 shows in two dimensions the correlation between eight heavy metals in each extract fraction for all the samples. Two (2) factors were obtained, accounting for 72.45% of the total variance.

The elements are distributed in three groups. Group one, containing the elements Cr, Cu, Mn, Ni, Pb, V and Zn, represent all the soluble and exchangeable elements. Group two has a variety of elements (Cd, Cr, Cu, Mn, Ni, Pb, V and Zn) associated to the carbonate/oxides and organic matter fraction, and Group three includes all the elements (Cd, Cr, Cu, Mn, Ni, Pb, V and Zn) associated to the silicate phase (Fig. 1). This separation or distribution is related to the form in which each element is present in the samples. Most of the elements are associated to the oxide/carbonate/ organic matter phases (group two) with the highest concentration for Cu, Zn and Pb.
The loadings of the eigenvalues corresponding to factor one (Table III), show a strong correlation among the elements and all the variables have positive loading on PC1 with 49.06 as percentage of variance accounting for 72.45 of the total variance in the graph. The results may suggest that these groups of elements are mainly from vehicular emission source, mainly Pb, Zn, Ni and Cd. In addition, some variables have similar positive loading on PC2 with 23.39 percent of variance and the elements Cu, Zn and Pb are likely to be derived from an industrial source additionally to the vehicular source (Yongming et al., 2006).
The loading for Ni and Mn falls in the PC 1, which suggests that these elements separate group two and three from group one.
Finally Cr, Cd and V have positive strong correlation in PC1 and negative correlation in PC2. These results suggest that the elements come from two main sources (vehicular and industrial).
]]>3.3 Total sample digestion
In order to test the accuracy of the digestion method applied to the dust samples considered in this work, the certified reference material urban particulate matter (NIST 1648) was analyzed by the same procedure. Results are shown in Table III. The values reported are the average of three replicate samples. The concentrations found are in good agreement with the certified values at 95% confidence level, according to Student–t test. The recovery limits shown in Table IV, fall within those recommended by the AOAC's single Lab validation document (Thompson et al., 2002).

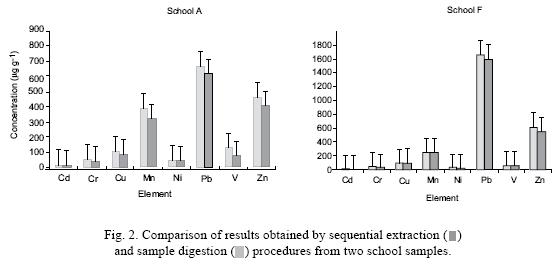
Figure 2 shows the total metal concentration of the digested original dust samples, together with the concentrations found by the sequential extraction methodology. Similar patterns were obtained for all samples considered in this work. Only two of them are shown for this purpose.
3.4 XRD analysis
The XRD analysis revealed five minerals: quartz (SiO2), calcite (CaCO3), plagioclase [(Na,Ca) AlSi3O8], kaolinite [Al2SiO5(OH)4] and in minor proportion potassic feldspar (KAlSi3O8). These apparently relate to the patterns of speciation in only one sense: the generally higher abundance of calcite (CaCO3) in dusts as opposed to surrounding soils where schools are located is reflected in a greater proportion of metal in the carbonate phase in these samples. It may be supposed that some calcium (CaO) in the school dust arises from abrasion of the road surface materials (such as cement). The apparent lack of iron oxides is probably due to their presence in an amorphous form which is not amenable to XRD analysis.
]]>4. Conclusions
A sequential extraction procedure was applied to dust samples from schools located in different areas of the city of Caracas. The metals examined are associated with various chemical forms as distinguished by sequential chemical extraction. The carbonate and Fe–Mn oxide phases dominate for Pb, Zn and Cu, whereas for Ni the organic phase is most important. Vanadium, Mn, Cr, Cd and a proportion of Pb remain in the residual material. This chemical speciation scheme has made possible to identify the chemical forms in particles that can provide detailed information about the origin, mode of occurrence, biological and physicochemical availability, mobilization and transport of metals, while leaving total metal contents as a pollution indicator. Good agreement between simple summation of the data obtained by the extraction method for each dust sample and the results given by the total digestion of each sample was obtained. Probable anthropogenic sources for the metals present in the different fractions, such us industrial and vehicular emissions, have been identified.
It is imperative to conduct further characterization of the magnitude of schools dust contamination and that major pollution sources could be identified.
A large period of sampling with many samples collected in different situations is necessary to obtain sufficient environmental information.
The different sources for the elements studied are supported by the results from multivariate (PCA) analysis.
Acknowledgements
The authors would like to gratefully thank CDCH–UCV, Project N0 PG 03–00–6054–2005 for funding this project and to FONACIT through the project Lab–19998003690. Also to Kizzy de Freitas and Jenny Bracho for their transcription help, and to two anonymous referees for their valuable comments.
]]> References
Das A. K., R. Chakraborty, M. L. Cervera and M. de la Guardia, 1995. Metal speciation in solid matrices. Talanta 42, 1007–1030. [ Links ]
Evans E. M., M. Kingston and L. Lechame, 1992. The speciation pattern of lead in street dusts and soils in the vicinity of two London schools. Environ. Inter. 18, 153–162. [ Links ]
Fernández A., M. Ternero, F. Barragán and J. Jiménez, 2002. A chemical speciation of trace for fine urban particles. Atmos. Environ. 36, 773–780. [ Links ]
Fernández A. and M. Ternero–Rodríguez, 2004. Study of traffic pollution by metals in Sevilla–España by physical and chemical speciation methods. Anal. Bioanal. Chem. 379, 684–699. [ Links ]
Kowalczyk G. S., G. E. Gordon and S. W. Rhelngrovers, 1982. Identification of atmospheric particulate sources in Washington, D. C. using chemical element balances. Environ. Sci. Technol. 16, 79–90. [ Links ]
Miranda J., E. Andrade, A. López, T. Ledesma, A. Cahill and P. Wakabayashi, 1996. A receptor model for atmospheric aerosols from a southwestern site in México City. Atmos. Environ. 30, 3471–3479. [ Links ]
Monaci F., F. Moni, E. Lanciotti, D. Grechi and R. Bargagli, 2000. Biomonitoring of airborne metals in urban environments: new tracers of vehicle emission, in place of lead. Environ. Pollut. 107, 321–327. [ Links ]
Morales F., R. Fernández and Z. Benzo, 2003. Lead exposure in day care centers in the Caracas Valley–Venezuela. Int. J. Environ. Hlth Res. 13, 3–9. [ Links ]
Orlova A. O., D. I. Bannon, M. R. Farfel, V. M. Thomas, L. V. Aleschukin, V. V. Kudashov, J. P. Shine and G. I. Kruchkov, 1995. Pilot study of sources of lead exposure in Moscow, Russia. Eviron. Geochem. Health 17, 200–210. [ Links ]
Szpunar J. and R. Lobinski, 1999. Speciation in the environmental field: trends in analytical chemistry. Fresenius J. Anal. Chem. 363, 550–557. [ Links ]
Thompson M., S. Ellison, and R. Wood, 2002. Harmonized guidelines for single–laboratory validation of methods of analysis. Pure Appl. Chem. 74, 835–855. [ Links ]
Tong S., 1998. Indoor and outdoor household dust contamination in Cincinnati, Ohio, USA. Environ. Geochem. Health 20, 123–133 [ Links ]
Tong S., Y. Schirnding and T. Prapamontol, 2000. Environmental lead exposure: a public health problem of global dimensions, Australia. Bull. World Health Org. 78, 1068–1077. [ Links ]
Tongtavee N., J. Shiowastana and R. G. McLaren, 2005. Fractionation of lead in soils affected by smelter activities using a continuous–flow sequential extraction system. Int. J. Environ. An. Ch. 85, 567–583. [ Links ]
Yongming H., D. Peixvan, C. Junji and E. Posmentier, 2006. Multivariate analysis of heavy metal contamination in urban dusts of Xi'an, Central China. Sci. Total Environ. 355, 176–186. [ Links ] ]]>