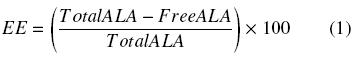
Preparation and properties of alpha-lipoic acid-loaded chitosan nanoparticles
Preparación y propiedades de nanopartículas de quitosano conteniendo ácido alfa lipoico
V. Velasco-Rodríguez1, M. Cornejo-Mazón1, J.O. Flores-Flores2, G.F. Gutiérrez-López1 and H. Hernández-Sánchez1*
1 Escuela Nacional de Ciencias Biológicas del Instituto Politécnico Nacional, Carpio y Plan de Ayala, Col. Sto.Tomás, CP 11340, México, DF, México.* Corresponding author. E-mail: hhernan1955@yahoo.com Tel. (55)57296000 ext. 62461, Fax: ext 62463.
]]> 2 Centro de Ciencias Aplicadas y Desarrollo Tecnológico, Universidad Nacional Autónoma de México, México, DF, México.
Recibido 30 de Agosto 2011
Aceptado 16 de Diciembre 2011
Abstract
The aim of this study was to prepare alpha-lipoic acid (ALA) loaded chitosan nanoparticles by the ionotropic gelation between chitosan and sodium pyrophosphate or tripolyphosphate and the use of the surfactant poloxamer 188 as a stabilizer. Their particle size and distribution along with their encapsulation efficiency and zeta potential was determined. The smallest particle size (180 nm) were obtained when sodium pyrophosphate was used as the polyanion. The addition of the poloxamer incresased the particle size independently of the polyanionic compound used. All the polydispersity indexes were > 0 .2, indicating that all the chitosan nanoparticle systems were polydisperse. The highest encapsulation efficiency (62.4%) was obtained in the case of the tripolyphosphate and poloxamer system. The isoelectric point (pI) for all the chitosan nanoparticles lied in the 6.8-7.4 range. All the chitosan nanopartides, with the exception of the ones prepared with TPP and poloxamer 188, were stable at pH values < 5.3, indicating a good potential for use in foods.
Keywords: alpha-lipoic acid, chitosan, nanoparticles, nanoencapsulation, ionic gelation.
Resumen
]]> El objetivo de este trabajo fue la preparación de ácido alfa lipoico inmovilizado en nanopartsculas de quitosano preparadas por la gelificación ionotropica entre el quitosano y pirofosfato y tripolifosfato de sodio y utilizando poloxámero 188 como estabilizante. Se determinó el tamaño, distribución de tamaños, potencial zeta y eficiencia de encapsulación para las nanopartículas. El tamaño más pequeño de partícula (180 nm) se obtuvo cuando se uso pirofosfato de sodio como polianión. La adición del poloxámero incremento el tamaño de las partículas independientemente del polianión usado. Todos los índices de polidispersidad fueron > 0.2, indicando que todos los sistemas de nanopartículas preparados fueron polidispersos. La mayor eficiencia de encapsulación (62.4%) se obtuvo en el caso del sistema tripolifosfato - poloxámero. El punto isoelectrico (pI) de todas las nanopartículas de quitosano estuvo en el intervalo 6.8 a 7.4 y todas las preparaciones fueron estables (excepto la que incluyo TPP y poloxámero 188) a valores de pH < 5.3. Esto indica un buen potencial para su uso en alimentos.Palabras clave: ácido alfa-lipoico, quitosano, nanopartículas, nanoencapsulación, gelificación ionica.
1 Introduction
α-Lipoic acid (ALA) or 6,8-thioctic acid is a necessary cofactor for the α-ketoacid dehydrogenases in mitochondria which are related to energy production (Shay et al., 2009). It is also a powerful lipophilic free-radical scavenger and antioxidant capable of recycling glutathione and an enhancer of the activities of vitamins C and E (Bernkop-SchnUrch et al., 2004). It has been prescribed for the amelioration of amanita mushroom poisoning, diabetic neuropathy, metabolic syndrome, peripheral artery disease, and for the treatment of a diversity of skin and liver diseases (Carlson et al., 2007). ALA is easily absorbed and transported across cell membranes and consequently it is widely used as a drug for the ailments mentioned above and as a diet and antiaging supplement (Kofuji et al., 2008). Nevertheless, ALA is a very unstable compound which decomposes under light or heat or even at room temperature. Insoluble unusable ALA polymers are generated when the monomer is heated beyond its melting point (59-62ºC). This kind of reactions is usually accompanied by an unpleasant odor (Kofuji et al., 2008). For that reason, the protection or stabilization of ALA is necessary to produce an adequate food supplement.
Chitosan is an aminopolysaccharide mainly produced from chitin, which is the earth's second most abundant carbohydrate polymer (after cellulose) and is the structural component of the exoskeletons of arthropods and some fungal cell walls. Chemically, chitosan is a high-molecular-weight linear polycationic heteropolysaccharide consisting of N-acetyl-D-glucosamine and D-glucosamine linked by β-(1 → 4) glycosidic bonds. The amount of each monosaccharide will depend on the degree of deacetylation (75-95%) of the polymer (Raafat and Sahl, 2009). Due to its distinctive biological characteristics, including nontoxicity and biodegradability, chitosan has many applications in the food and pharmaceutical industries (Kong et al., 2010). Some applications in regenerative medicine and tissue engineering have also been reported (Olivas-Armendariz et al., 2009). Chitosan has also been used in the preparation of controlled drug release systems based on ionic interactions with anionic drugs such as ALA. In vitro release studies with tablets with 80% ALA and 20% chitosan acetate showed a controlled ALA release over a period of time of 24 h (Bernkop-Schnurch et al., 2004). This ionic interaction method has also been used to stabilize ALA in the form of a complex with chitosan (Kofuji et al., 2008).
Encapsulation is another method that is very commonly used to protect ALA. The use of the same ionic interaction of chitosan with a polyanionic compound such as alginic acid and its salts can be used to form the capsules. Controlled release of ALA has been achieved through the incorporation of this compound into chitosan-alginate gel beads (Kofuji et al., 2009). The use of chitosan as wall material for the spray dried encapsulation of bioactive compounds such as ascorbic acid (Pulido and Beristain, 2010) and also ALA (Weerakody et al., 2008) has been successfully carried out. Particle sizes of the loaded microspheres varied around 7 µm. Nanotechnology has the potential to modify many macroscale characteristics including stability during shelf life. One of the most interesting applications of this technology is the change in pharmacokinetics and biodistribution through encapsulation in nanoparticles (Huang et al.,2010).
In the case of chitosan, there are at least four methods existing for nanoparticle formation: ionotropic gelation, microemulsion, emulsification solvent diffusion and polyelectrolyte complex (Tiyaboonchai, 2003). The oral bioavailability of the highly lipophilic antibiotic cyclosporin-A has been improved through its incorporation in chitosan nanoparticles using the emulsification solvent diffusion method (El-Shabouri, 2002). In this method, an o/w emulsion is obtained by injecting an organic phase into chitosan solution containing a stabilizing agent (generally poloxamer) under stirring, followed by high-pressure homogenization (Tiyaboonchai, 2003). Many reports involve the use of ionotropic gelation to produce chitosan nanoparticles. In this technique, the particle formation is based on the electrostatic interaction between the amino groups of chitosan and the negatively charged groups of polyanions such as tripolyphosphate. This method involves the dissolution of chitosan in acetic acid followed by the addition of the polyanion and a stabilizer such as poloxamer (Tiyaboonchai, 2003). Poloxamer 188 is a difunctional block copolymer surfactant approved by the FDA under the trade name Lutrol® F 68. This technique has been used to produce antimicrobial chitosan nanoparticles loaded with metal ions (Qi et al., 2004; Du et al., 2009) or with nisin (Zohri et al., 2010). The purpose of this work was to evaluate the ionotropic gelation method to produce chitosan nanoparticles to encapsulate and protect ALA.
2 Materials and methods
]]> 2.1 MaterialsLipoic acid (T5625), chitosan 75% deacetylated (C3646) and sodium tripolyphosphate (238503) were purchased from Sigma-Aldrich (St. Louis, USA). Lutrol®R F 68 Poloxamer 188 was a gift from BASF México. All the other reagents were of analytical grade and used as obtained.
2.2 Preparation of the nanocapsules of ALA and chitosan
Chitosan nanoparticles were prepared based on the ionotropic gelation between sodium pyrophosphate (SPP) or sodium tripolyphosphate (TPP) and chitosan (Du et al., 2009). Chitosan was dissolved in 1% acetic acid to obtain a 0.3% (w/v) chitosan solution in the absence or presence of 1% of the steric stabilizing agent poloxamer 188. This mixture was added to a 0.3% (w/v) ALA solution in absolute ethanol in a weight ratio of 1:1 and the mixture stirred to form a solution (Weerakody et al., 2008). SPP or TPP were dissolved in water to a concentration of 1%. Under constant stirring at room temperature, 1 ml of SPP or TPP solution was added into 25 ml of chitosan-ALA solution and the stirring continued for 20 min and then the mixture was sonicated at 1.5 kW for 30 min. The process was performed with the sonicator 30sec "on", 30 sec "off" so the temperature in the water bath should remain around 25ºC. This suspension was centrifugated at 12,000Xg for 10 min and resuspended in water for further use and characterization. All the experiments were conducted in triplicate.
2.3 Transmission electron microscopy (TEM) studies
The morphology was examined using a JEOL JEM1010 TEM at a voltage of 60 kV. The aqueous dispersion at pH 6.5 of the nanoparticles (5 μl) was drop-casted onto a formvar carbon- coated copper grid (200 mesh) and then stained with OsO4 vapor (evaporating from a 1 wt% aqueous solution).
2.4 ALA encapsulation efficiency
The encapsulation efficiency of the ALA-loaded chitosan nanoparticles was analyzed using ultracentrifugation of the freshly prepared suspension at 44,400Xg. The amount of ALA in the supernatant was determined by HPLC. In brief, 1 ml of supernatant was extracted with the same volume of a mixture of 2% aqueous acetic acid and absolute ethanol (1:1) and filtered (0.2 μm Acrodiscs) prior to analysis to remove solid residues. The lipoic acid in the ethanolic extracts was measured using HPLC method adapted from Weerakody et al. (2008). The HPLC system (Beckman) consisted of a Gold System Programmable Solvent Module 126 / Binary Pump, degasser, autosampler and diode array detector (DAD Gold 168). The lipoic acid samples were analysed by reversed-phase HPLC on a Nova-Pak C18 Column, 4 µm, 3.9 x 150 mm. A mobile phase of methanol:water (80:20 v/v) adjusted to pH 3 with phosphoric acid with a flow rate of 0.8 ml/min was employed to separate the lipoic acid from other components in the samples. The lipoic acid was detected at 329 nm with the DAD. With the knowledge of the initial (total) concentration of ALA used to prepare the nanoparticles and the free ALA (the ALA which was not incorporated in the particles and that remains in the supernatant), the encapsulation efficiency (EE) could be calculated as:
The results are the average of three determinations.
]]> 2.5 Chitosan nanoparticle size and distributionThe size anddistribution of the prepared nano particles was analyzed by dynamic light scattering (DLS) using a Zetasizer Nano ZS90 (Malvern Instruments Ltd., Malvern, UK). This technique is widely used as an adequate way of measuring particle sizes in the range from a few nanometers up to 1 or 2 µm. Since DLS measures the apparent hydrodynamic radius of particles, it is suitable to determine the basic size of particles (Alexander and Dalgleish, 2006). The nanoparticle samples were diluted 1:400 (v/v) with deionized water and measured at a temperature of 25ºC and a scattering angle of 90º. Size distribution was evaluated by the Zetasizer software as a polydispersity index (PdI). All the results are the average of 100 readings performed automatically by the equipment.
2.6 Zeta potential
This parameter is used to describe the surface charge property of nanoparticles. It indicates the electrical potential of the nanoparticles and is influenced by the composition of the particle and the medium in which it is dispersed. Nanoparticles with a zeta potential outside the range of ±30 mV have been shown to be stable in suspension, as the surface charge avoids aggregation of the particles (Mohanraj and Chen, 2006). Titration experiments on the nanoparticles were performed over a pH range between 3 and 10 using the Malvern Zetasizer Nano ZS90. For the analysis, the samples (original suspensions) were diluted 1:200 (v/v) with deionized water and the pH value of the suspension was adjusted by the titration unit and the zeta potential measured by the Zetasizer Nano. All the results are the average of 10 determinations.
3 Results and discussion
3.1 Encapsulation efficiency
With an initial concentration of ALA of 1.5 g/l (7.27 µmol/l), encapsulation efficiencies of 57.69, 51.81, 49.03 and 62.4% were obtained for the chitosan systems with SPP, SPP + poloxamer, TPP and TPP + poloxamer respectively. In general, these values are similar to those obtained by Weerakody et al. (2008) for the encapsulation of ALA in chitosan microspheres by spray drying (55.2%). However, a higher efficiency of 62.4% was obtained in our study in the case of the system chitosan - TPP -poloxamer 188.
3.2 Nanoparticle size and distribution
We studied the effect of the kind of polyanion and the presence or absence of the stabilizer poloxamer 188 on the particle size and the polydispersity index (Pdl), where the polydispersity, in the field of light scattering, is used to describe the width of the nanoparticle size distribution. A lower polydispersity index indicates a narrower size distribution and usually, samples with a Pdl <~ 0.2 are considered to be monodisperse. Particle size and distribution analysis of the ALA loaded chitosan nanoparticles are shown in Table 1. The smaller sizes (180 nm) were obtained when SPP was used as the polyanion. The addition of the poloxamer 188 incresased the particle size independently of the polyanionic compound used. All the polydispersity indexes were > 0.2, indicating the formation of polydisperse mixtures of nanoparticles. These particle sizes were similar to those reported for the chitosan-polyethylene oxide with TPP nanoparticles (200-1000 nm) prepared by Calvo et al. (1997). The average size of the chitosan TPP nanoparticles prepared by Du et al. (2009) are smaller (53.99 nm), however the polydispersity of their preparations is higher (1.00) than the values in our study. These authors used also a Zetasizer Nano for their determinations. In the case of the synthesis of iron oxide nanoparticles on chitosan templates, sizes between 265 and 279 nm with PdI between 0.13 and 0.48 were obtained (Nidhin et al., 2008).
]]>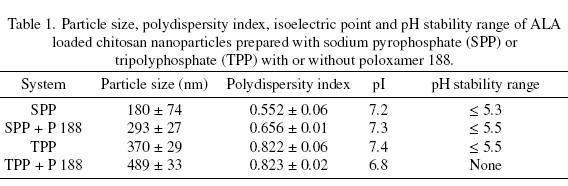
Particle size is a very important feature of nanoparticles since it has been observed that the uptake efficiency of nanoparticles (around 100 nm size) by the intestinal tissue is up to 250 times that of larger size microparticles (Desai et al., 1996). The size of the ALA loaded chitosan nanoparticles is in the range of 180 to 489 nm. On the other hand, nanoparticles can also be administered intravenously since the diameter of the smallest blood capillary is around 4000 nm. Particles greater than 100 nm in size are quickly taken up by the reticuloendothelial system while smaller sized particles have longer circulation times (Tiyaboonchai, 2003). In the case of this study, the ALA loaded chitosan nanoparticles would be more effective if administered intravenously.
The morphology of the ALA loaded chitosan nanoparticles was examined by TEM (Fig. 1). It can be observed that the nanoparticles have irregular spherical shapes and that they have a tendency to agglomerate at pH 6.5. It can then be concluded that the sizes measured by dynamic light scattering are the sizes of these clusters and not those of individual nanoparticles, which according to the TEMs, are around 20 to 40 nm. The applicability of many kinds of nanoparticles is commonly limited by a low stability and a tendency to create agglomerates. Stabilization of nanoparticles can be achieved by changing their surface properties. Electrostatic stabilization is the most common method used to steady the nanoparticle colloid dispersions. Surfactants such as the poloxamers are known as the most effective electrostatic stabilizing agents, so it is possible that an increase in their concentration could be effective in reducing the agglomeration and the particle size.
3.3 Zeta potential
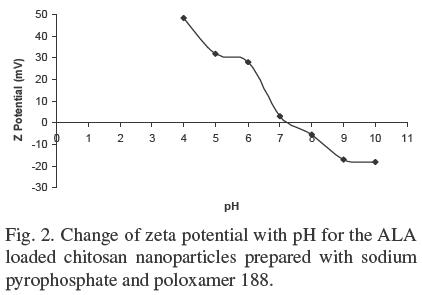
Figure 2 shows, as an example, the typical plot of zeta potential (in mV) versus pH for the ALA loaded chitosan nanoparticles prepared with SPP and poloxamer 188. It can be observed that the zeta potential decreases when pH increases. The surface of the chitosan nanoparticles is charged positively at acidic and neutral conditions and negatively at basic conditions with the transition occurring at its pI. The isoelectric point for all the chitosan nanoparticles lied in the 6.8 - 7.4 pH range. All the nanoparticles, with the exception of the particles prepared with TPP and poloxamer 188, were stable at pH values < 5.3, indicating a good potential for use in acid foods, however, considering their effective size, they could be assimilated more easily if administered intravenously, provided the pH could be adjusted to avoid agglomeration. The zeta potential values are similar to those reported by Du et al. (2009) for chitosan nanoparticles prepared with TPP and no surfactant. The nanoparticles prepared with TPP and poloxamer 188 were not stable since they were out of the range of zeta potential of ± 30 mV at any pH and showed a tendency to agglomerate as shown in Figure 1a. The last phenomenon could be explained by the fact that the TEM preparations were done at pH 6.5, which is very near the pI of the nanoparticles.
Conclusions
]]> Encapsulation of ALA in chitosan nanoparticles is more efficient than in microspheres. Depending on the system, nanoparticles in the size range of 180 to 489 nm can be obtained. The size and distribution of the nanoparticles can be controlled by changing the polyanionic compound and by using a surfactant such as poloxamer 188, which could also decrease the tendency to agglomerate of the particles if an adequate concentration could be found. The nanoparticles with the smallest size and polydispersity were obtained with SPP as the polyanion in the absence of surfactant. These particles could be administered in foods or intravenously. This is the first report of pI values and stability ranges for chitosan nanoparticles.
Acknowledgements
This work was partially financed by the Instituto Politécnico Nacional (México City, México). Verónica Velasco Rodríguez thanks the Consejo Nacional de Ciencia y Tecnología (Mexico City, Mexico) for the awarding of their scholarship. Authors Gutiérrez-López and Hernández-Sánchez also acknowledge the fellowship from COFAA-IPN (Mexico City, Mexico). The help of María Esther Sánchez Espíndola (IPN) in the TEM experiments is greatly appreciated.
References
Alexander, M. and Dalgleish, D.G. (2006). Dynamic light scattering techniques and theirapplications in food science. Food Biophysics 1, 2-13. [ Links ]
Bernkop-Schnürch, A., Schuhbauer, H., Clausen, A.E. and Hanel, R. (2004). Development of a sustained release dosage form for a-lipoic acid. I. Design and in vitro evaluation. Drug Development and Industrial Pharmacy 30, 27-34. [ Links ]
]]>Calvo, P., Remunan-Lopez, C., Vila-Jato, J.L. and Alonso, M.J. (1997). Novel hydrophilic chitosan-polyethylene oxide nanoparticles as protein carriers. Journal of Applied Polymer Science 63, 125-132. [ Links ]
Carlson, D.A., Smith, A.R., Fischer, S.J., Young, K.L. and Packer, L. (2007). The plasma pharmacokinetics of R-(+)-lipoic acid admisnistered as sodium r-(+)-lipoate to healthy human subjects. Alternative Medicine Review 12, 343-351. [ Links ]
Desai, M.P., Labhasetwar, V., Amidon, G.L. and Levy, R.J. (1996). Gastrointestinal uptake of biodegradable microparticles: effect of particle size. Pharmaceutical Research 13, 1838-1845. [ Links ]
Du, W.L., Niu, S.S., Xu, Y.L., Xu, Z.R. and Fan, C.L. (2009). Antibacterial activity of chitosan tripolyphosphate nanoparticles loaded with various metal ions. Carbohydrate Polymers 75, 385-389. [ Links ]
El-Shaboury, M.H. (2002). Positively charged nanoparticles for improving the oral bioavailability of cyclosporin-A. International Journal of Pharmaceutics 249, 101-108. [ Links ]
]]>Huang, Q., Yu, H. and Ru, Q. (2010). Bioavailability and delivery of nutraceuticals using nanotechnology. Journal of Food Science 75, 50-57. [ Links ]
Kofuji, K., Nakamura, M., Isobe, T., Murata, Y. and Kawashima, S. (2008). Stabilization of α-lipoic acid by complex formation with chitosan. Food Chemistry 109, 167-171. [ Links ]
Kofuji, K., Isobe, T. and Murata, Y. (2009). Controlled release of alpha-lipoic acid through incorporation into natural polysaccharide-based gel beads. Food Chemistry 115, 483-487. [ Links ]
Kong, M., Chen, X.G., Xing, K. and Park, H.J. (2010). Antimicrobial properties of chitosan and mode of action: A state of the art review. International Journal of Food Microbiology 144, 51-63. [ Links ]
Mohanraj, V.J. and Chen, Y. (2006). Nanoparticles -A review. Tropical Journal of Pharmaceutical Research 5, 561-573. [ Links ]
]]>Nidhin, M., Indumathy, R., Sreeram, K.J. and Naur, B.U. (2008). Synthesis of iron oxide nanoparticles of narrow size distribution on polysaccharide templates. Bulletin of Material Science 31, 93-96. [ Links ]
Olivas-Armendáriz, I., García-Casillas, P., Martel-Estrada, A., Martinez-Sánchez, R., Martínez-Villafañe, A. and Martínez-Pérez, C.A. (2009). Preparation and characterization of chitosan/carbon nanotubes composites. Revista Mexicana de Ingeniería Química 8, 205-211. [ Links ]
Pulido, A. and Beristain, C.I. (2010). Spray dried encapsulation of ascorbic acid using chitosan as wall material. Revista Mexicana de Ingeniería Química 9, 189-195. [ Links ]
Raafat, D. and Sahl, H.G. (2009). Chitosan and its antimicrobial potential - a critical literature survey. Microbial Biotechnology 2, 186-201. [ Links ]
Shay, K.P., Moreau, R.F., Smith, E.J., Smith, A.R. and Hagen, T.M. (2009). Alpha-lipoic acid as a dietary supplement: Molecularmechanisms and therapeutic potential. Biochimica et Biophysica Acta 1790, 1149-1160. [ Links ]
]]>Tiyaboonchai, W. (2003). Chitosan nanoparticles: a promising system for drug delivery. Naresuan University Journal 11, 51-66. [ Links ]
Weerakody, R., Fagan, P. and Kosaraju, S.L. (2008). Chitosan microspheres for encapsulation of α-lipoic acid. International Journal of Pharmaceutics 357, 213-218. [ Links ]
]]>