Spectroscopic, chemical and morphological characterization and superficial properties of a Mexican montmorillonite
Yolanda Marina Vargas–Rodríguez1,*, Virginia Gómez–Vidales2, Eloy Vázquez–Labastida3, Arturo García–Bórquez4, Guillermo Aguilar–Sahagún5, Héctor Murrieta–Sánchez6 y Manuel Salmón2
1 Departamento de Ciencias Químicas, Facultad de Estudios Superiores Cuautitlán, Universidad Nacional Autónoma de México, Campo 1, 54740 Cuautitlán Izcalli, Estado de México, México.* ymjvargas@yahoo.com.mx
2 Instituto de Química, Universidad Nacional Autónoma de México, Ciudad Universitaria, 04510 México, D. F, México.
3 Departamento de Ingeniería Química Industrial, ESIQIE, Instituto Politécnico Nacional, Unidad Profesional Adolfo López Mateos, 07738 México, D. F, México.
4Departamento de Ciencias de Materiales, ESFM–IPN, Edif. 9–UPALM, 07738 México D.F., México.
]]> 5 Instituto de Investigación en Materiales, Universidad Nacional Autónoma de México, Ciudad Universitaria, Circuito Exterior, Del. Coyoacán, 04510 México D. F, México.6 Instituto de Física, Universidad Nacional Autónoma de México, Ciudad Universitaria, Circuito Exterior, Del. Coyoacán, 04510 México D. F, México. msuniversidad@gmail.com
Manuscrito recibido: Octubre 24, 2006
Manuscrito corregido recibido: Octubre 10, 2007
Manuscrito aceptado: Noviembre 12, 2007
RESUMEN
Se describe la caracterización mediante técnicas espectroscópicas de una montmorillonita natural colectada en Cuencamé, Durango. La composición de las capas tetraédricas y octaédricas fue analizada por Resonancia Magnética Nuclear (RMN) de ángulo mágico para los núcleos de 29Si y27Al. Las propiedades superficiales de la arcilla se cuantificaron utilizando técnicas de adsorción–desorción de nitrógeno y mediante la aplicación de los métodos de Brunahuer–Emmett–Teller (BET) y Barrett–Joyner–Halenda (BJH). Los resultados indican que la arcilla analizada es una montmorillonita sódica con una capa molecular de agua adsorbida entre sus láminas, cuyos átomos de Si en la estructura defilosilicato Q3presentan diferente composición Si–nAl (n =0, 1 y 2). El 3.3% de los átomos de Al correspondientes a la estructura se encuentran sustituyendo a los de Si en la capa. La presencia de los iones paramagnéticos Fe+3 y Mn+2 en la montmorillonita fue determinada por resonancia paramagnética electrónica (RPE), después de ser tratada con HCl y CF3SO3H. Adicionalmente, se discuten los resultados adquiridos por las técnicas de absorción atómica, microscopía de barrido electrónico, infrarrojo y análisis térmico gravimétrico.
Palabras clave: arcilla, montmorillonita, Resonancia Magnética Nuclear de ángulo mágico, 29Si, 27Al, resonancia paramagnética electrónica, difracción de rayos X, análisis térmico.
]]>ABSTRACT
The characterization by means of spectroscopic techniques of a natural montmorillonite collected at Cuencamé, Durango is described. The composition of the tetrahedral and octahedral layers was analyzed by magic angle spinning Nuclear Magnetic Resonance (NMR) for the nuclei of 29Si and 27Al. The superficial properties of the clay were quantified using the technique of adsorption–desorption of nitrogen as well as the Brunahuer–Emmett–Teller (BET) and Barrett–Joyner–Halenda (BJH) methods. The results indicate that the analyzed clay is a Na–montmorillonite with a molecular layer of water adsorbed between their laminae, where the Si atoms in the phyllosilicate Q3 structure display different composition of Si–nAl (n = 0, 1 and 2). About 3.3% of the Al atoms in the structure are replacing Si atoms in the layer. The presence of the paramagnetic Fe+3 and Mn +2 ions in the structure was determined by electronic paramagnetic resonance after the analysis of the acidified clay treated with HCl and CF3SO3H. Additionally, the results acquired by atomic absorption, scanning electron microscopy, infrared and gravimetric thermal analysis are discussed.
Key words: clay, montmorillonite, magic angle spinning (MAS), Nuclear Magnetic Resonance (NMR), 29Si, 27Al, electronic paramagnetic resonance, X–ray diffraction, thermal analysis.
INTRODUCCIÓN
Los minerales arcillosos del tipo montmorillonita, comunes en suelos y sedimentos, se presentan con iones sodio, calcio, magnesio o combinación de estos. En Durango (México), la especie dominante es la montmorillonita sódica baja en calcio y muy expansible (De Pablo–Galán, 1990).
Entre las aplicaciones industriales de las montmorillonitas se puede mencionar su uso como decolorantes de aceites (previamente tratadas con ácidos minerales), como adsorbentes de pesticidas o metales pesados en aguas residuales (Arvanitopyannis et al., 1989) y como catalizadores heterogéneos para promover reacciones químicas (Adams, 1987; Ballatine, 1992; Chitnis y Sharma, 1997; Salmón et al., 1997). Las propiedades catalíticas de las montmorillonitas se incrementan con los tratamientos ácidos y varían dependiendo de la fuerza acida, de los cambios en la estructura del aluminosilicato y de las propiedades superficiales (Rhodes y Brown, 1992; Breen et al., 1995, Vargas–Rodríguez et. al., 2007).
Generalmente, las arcillas industriales no son purificadas o caracterizadas apropiadamente y se expenden con variaciones en sus componentes químicos, con impurezas que modifican la reactividad y reproducibilidad de cada lote. De ahí se enfatiza la importancia de una caracterización apropiada de las arcillas naturales, tanto para su uso industrial como en investigación. La etapa de purificación para la remoción de impurezas insolubles y solubles es fundamental para que los resultados experimentales sean reproducibles y atribuibles a la arcilla, permitiendo su transformación en materiales más reactivos (con ácidos, superácidos, sales metálicas, pilareo, etc.) y utilización, por ejemplo como catalizadores heterogéneos. (Miranda et al., 1994, 2003a, 2003b; Salmón et al., 1997; Ruiz–Guerrero et al., 2006).
Durante las tres últimas décadas, la Resonancia Magnética Nuclear (RMN) de estado sólido se ha acreditado como una técnica válida para examinar la estructura y dinámica de materiales orgánicos, inorgánicos, biológicos, contaminantes ocluidos o soportados en diferentes materiales (Floúan et al., 1996; Schnelly Spiess, 2001; Castellani et al., 2002; Bradley y McLaughlin, 2007; Wind y Hu, 2007). En particular, la RMN de ángulo mágico (RMN–AM) permite diferenciar el ambiente químico local y puntual de los átomos que componen la estructura de las arcillas a través del estudio de 29Si y 27A1 (Fitzgerald, 1999). Adicionalmente, la difracción de Rayos X (DRX) de polvos, además de evidenciar la cristalinidad de montmorillonita a través de la medida del espaciamiento d001, permite evaluar el catión interlaminar a humedad relativa constante (Monsalvo et. al., 2006).
]]> La espectrofotometría de absorción en el infrarrojo (IR) permite adquirir información de la naturaleza de los sustituyentes isomórficos, distingue hidroxilos estructurales de los del agua molecular y reconoce las impurezas cristalinas y amorfas (Madejová y Komadel, 2001). La Resonancia Paramagnética Electrónica (RPE) reconoce la determinación de centros de imperfección sobre las redes de aluminosilicatos o sobre la superficie externa de la montmorillonita, y evidencia la presencia de iones paramagnéticos Fe3+, Mn2+, Cu2+, Cr3+, V4+, etc., hasta concentraciones 1 x 10–8 mol y se emplea para elucidar la estructura, confirmar la movilidad de los iones interlaminares o intercambiables, entre ellos, por ejemplo, Cu2+ y Mn2+ (McBride et al., 1975a, 1975b; McBride, 1995; Hall, 1980a, 1980b; Mabbs y Collison, 1992; Gehring et al., 1993; Gehring y Sposito,1995; Bensimon et al., 2000).El análisis térmico involucra una aproximación fenomenológica dinámica para el estudio de minerales que manifiestan una respuesta a los cambios de temperatura y a la fugacidad del agua que afectan la estabilidad de la arcilla. (Guggeheim y van Groos, 2001).
El conocimiento de la adsorción de gas por los materiales, es importante para determinar el área superficial y la distribución del tamaño de poro en sólidos utilizados como catalizadores, adsorbentes industriales (bentonitas), pigmentos cerámicos y materiales de construcción. La medición de adsorción de la interfase sólido–gas determina la naturaleza y comportamiento de las superficies de las arcillas (Sing et al, 1985).
En este trabajo se describen y discuten los resultados de la caracterización espectroscópica, química, morfológica y las propiedades superficiales de una montmorillonita de la región de Cuencamé, Durango (México), utilizando: Resonancia Magnética Nuclear de Ángulo Mágico (RMN–AM) de 29Si y 27A1, Resonancia Paramagnética Electrónica (RPE), Difracción de Rayos X (DRX), Infrarrojo (IR), absorción atómica (AA), Microscopía de Barrido Electrónico (MBE). Análisis Térmico Gravimétrico (TG) y adsorción–desorción de N2, aplicando los métodos de Brunauer–Emmett–Teller (BET) y Barrett– Joyner–Halenda (B JH) para el análisis de las isotermas.
METODOLOGÍA
Purificación de la montmorillonita
Purificación de la arcilla natural obtenida de la mina: 50 g de la arcilla bentonítica de la región de Cuencamé, Durango (México) se molió en un mortero y se suspendió en 1000 mL de agua desionizada. La mezcla se agitó por tres días y por decantación se separó la arcilla suspendida de los sólidos insolubles (el proceso se repitió cuatro veces). La suspensión resultante se centrifugó, eliminándose el líquido sobrenadante. La arcilla húmeda se secó a 80 °C con vacío, hasta obtener un sólido que se pulverizó y con este material (montmorillonita) se realizaron todos los análisis y experimentos descritos en este trabajo.
Tratamiento de la montmorillonita con los ácidos clorhídrico (HC1) y trifluorometansulfónico (CF3SO3H)
]]> La montmorillonita (10 g) fue suspendida en 300 mL de agua desionizada, la suspensión resultante fue agitada por 72 h. Posteriormente se adicionaron 0.18 moles del ácido correspondiente, continuándose la agitación por ocho horas. El sólido fue separado por centrifugación. El material obtenido se secó a 80 °C y fue caracterizado por RPE y MBE. Posteriormente 2.0 g de cada una de las muestras secas, se extrajeron en soxhlet con acetona por tres horas, los extractos se evaporaron, se secaron al vacío y se analizaron por RPE.
Métodos analíticos
Todas las muestras se analizaron por RPE y MBE. Las micrografías de MBE fueron obtenidas con un XL SFEG–Sirion equipado con un filamento de emisión y un detector especial para alta resolución (TLD). El microanálisis se llevó a cabo con un Sistema de Dispersión de Energía de Rayos X, marca EDAX, modelo Génesis acoplado al MBE. Adicionalmente se determinó la composición química por Absorción atómica con un equipo modelo Spectra AA. En este trabajo no se determinaron los límites de detección como fue sugerido por IUPAC (Longy Winefordner, 1983) y más recientemente por Verma y Santoyo (2005). Los espectros de RMN–AM para 27A1 y 29Si fueron obtenidos con un espectrómetro Bruker 300, con pulsos de 4.5 \xs y una repetición de 5s entre ellos para obtener la saturación, la frecuencia de giro se estableció en los límites de 4 – 4.5 kHz. El número de acumulaciones fue de 500. Todas las mediciones fueron realizadas a temperatura ambiente con Al(H20)3+ y tetrametilsilano como referencias para aluminio y silicio respectivamente. Se adquirió además, la desconvolución de las señales del espectro de RMN–AM 29Si. El espectro de difracción de rayos X fue obtenido con un difractómetro Philips PW–1730 a 35 KVy 30 mAutilizando radiación de CuKα y filtro de Ni, con monocromador secundario de grafito a una velocidad de barrido de 1o 2θ/min. El espectro de infrarrojo se obtuvo con un equipo Perkin Elmer 283, empleando pastillas de KBr. El estudio de RPE fue realizado en un espectrómetro Jeol JES–TE300, en onda continua y banda X, con una cavidad cilindrica en modo TE011. La calibración externa del campo magnético se realizó con un gaussmetro de precisión Jeol–ES–FC5. El análisis térmico gravimétrico se adquirió con un equipo TA Instruments Modelo 2160. El área específica de la arcilla fue determinada en un aparato Micromeritics ASAP 2000, Norcross GA, usando N2 como adsorbente a 77 K empleando el método de Brunauer–Emmett–Teller (BET). La distribución de tamaño de poro se calculó por el método Barrett–Joyner–Halenda (BJH) a partir de las isotermas de adsorción–desorción. Las muestras fueron evacuadas con vacío a 473 K por 12 horas, previamente a las mediciones de la adsorción–desorción.
RESULTADOS Y DISCUSIÓN Difracción de Rayos X
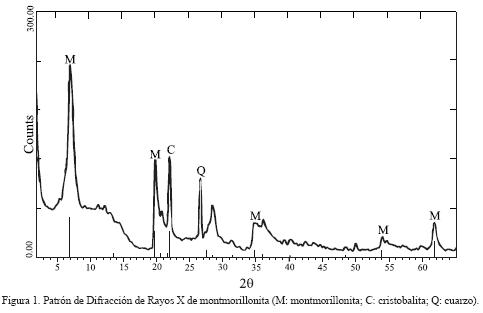
El espectro de DRX de la montmorillonita (Figura 1), exhibe la presencia de tres fases cristalinas que son: montmorillonita, cristobalita y cuarzo. Los datos indican un alto grado de cristalinidad para la montmorillonita que concuerdan con los del difractograma del estándar (JCPDS 29–1498). El primer pico, con intensidad del 100%, se asigna para d001; con un valor de 12.30 Å típico de montmorillonitas que presentan una capa molecular de agua adsorbida entre sus láminas. La cristobalita se identificó con los picos de reflexión d101 (4.044 Å), d111 (3.121 Å), d102 (2.%55 Å) y d112 (2.481 Å) y para cuarzo con las reflexiones en d100 (4.296 Å), dl0l (3.346 Å), d 110 (2.481 Å), d112(1.819 Å), d211 (1.544 Å), que corresponden a los estándares JCPDS: 39–1425 y 46–1045 para cada uno.

]]> Composición química
La composición química de la montmorillonita obtenida por MBE fue 5.16 % Na2O, 2.97 % K2O, 28.46 % A12O3, 54.72 % SiO2, 0.36 % CaO, 3.58 %Fe2O3, y 4.71 % MgO. En tanto que por AA se obtuvo 2.55 % Na2O, 0.34 %K2O, 21.14 % A12O3, 65.62 % SiO2, 0.70 %CaO, 5.04 % Fe2O3, 4.55 % MgO y 0.029% MnO. Es importante hacer notar que existe una diferencia en el resultado de los análisis entre las dos técnicas. Los valores para cada uno de los elementos son diferentes y por MBE no se detecta manganeso. Podemos atribuir esta diferencia entre las dos técnicas a que por AA se realiza un análisis total de la muestra, mientras que por MBE los análisis son puntuales. El manganeso, que se encuentra en cantidades mínimas, no fue detectado por MBE, debido probablemente a los límites de detección relativamente altos para esta técnica.
Las posibles causas de estas diferencias entre las dos técnicas podrían resolverse, en el futuro, mediante una mejor calibración de estos dos métodos analíticos (p. ej., Guevara et al., 2005) y el estudio de materiales internacionales de referencia apropiadamente seleccionados para este propósito (p. ej., Lozano y Bernal, 2005; Vermay Quiroz–Ruiz, 2006a, 2006 b).
En el análisis morfológico de la arcilla se observan cambios evidentes entre la montmorillonita sin tratamiento ácido y las tratadas con ácidos. La montmorillonita sin tratamiento presenta partículas sutilmente facetadas o con cantos agudos (Figura 2a). Es apropiado hacer evidente que en el grueso de los cantos se observa la estructura laminar (Figura 2b) que concuerda con el resultado de DRX y la cristalinidad confirmada por la reflexión en (001).
La montmorillonita, al ser tratada con CF3SO3H, presenta partículas con cantos más suaves (Figura 2c) y sin estructura laminar o muy escasa, en contraste, lo que presenta notoriamente es una abundancia de poros (Figura 2d). La montmorillonita tratada con HC1 exhibe escasas partículas y todas ellas muy deterioradas (Figura. 2e), material amorfo muy homogéneo, como se observa en detalle de alta resolución (Figura. 2f).
Resonancia magnética nuclear
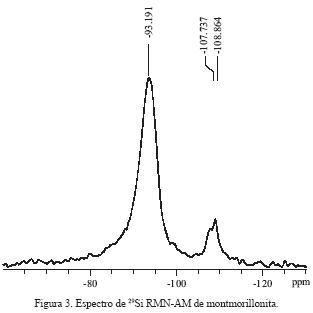
En el espectro de RMN–AM 29Si (Figura 3), se observa una resonancia ancha en –93.191 ppm; esta señal es asignada a silicio con estructura de filosilicato Q3 (Weiss et al, 1987). Existen, además, dos pequeñas resonancias en –107.737 y –108.864 ppm asignadas a silicio Si(OSi)4, con un desplazamiento característico para una estructura Q4 (Lippmaa et al, 1980). Después de realizar la descon–volución de las señales anteriores (Figura 4), se distinguen cinco señales en–84.81, –87.98, –93.18, –107.45 y –108.86 ppm, cuyas abundancias relativas y asignación se resumen en la Tabla 1. La señal centrada en –84.5 ppm corresponde a (Si–2A1), la de –87.98 se asigna a (Si–lAl) y la de –93.2 corresponde a (Si–0Al), coincidiendo con una estructura de filosilicatos (Kinsey et al, 1985; Barran et al, 1985). Las señales en –107.45 y –108.86 ppm se asignan a sílice y son característica para sus formas polimórficas de cuarzo y cristobalita (Lippmaa et al, 1980).


En el espectro de RMN–AM 27A1 (Figura 5) hay una señal en –3.8 ppm, típica para el aluminio del filosilicato y una en +55.0 ppm, asignada al aluminio tetraédrico (Müller et al, 1981; Ohkubo et al, 2003), correspondiendo además a los átomos de aluminio que sustituyen a los de silicio en la capa. La abundancia relativa de las señales corresponde al 96.7% y 3.3 % respectivamente.

Espectrofotometría de absorción infrarroja
Los picos de absorción observados en el espectro de IR de la montmorillonita (Figura 6) son característicos de los grupos hidroxilos, del silicato aniónico y de los cationes en la estructura de la montmorillonita (Madejová y Komadel, 2001). La señal más intensa a 1038 cm1, es atribuida a las vibraciones de alargamiento de las uniones Si–0 de la capa y la banda en 519 cm1 a Si–O–Al. La vibración de flexión del grupo OH coordinado a los cationes en 910 cm1 es establecida para A1A1OH y en 844 cm1 para AIMgOH. La señal en 791 cm1 se asigna a la vibración Si–0 y es característica de sílice en cualquier forma alotrópica. En este caso particular se establece para impurezas de cuarzo y cristobalita que concuerdan con los resultados obtenidos por RMN–AM de 29Si y por DRX.
]]>
Resonancia paramagnética electrónica
El espectro de resonancia paramagnética electrónica de la montmorillonita (Figura 7a) exhibe dos señales en g=4.3 y g=;2.0 (el parámetro g se define como la constante giromagnética, es adimensional y nos da características de qué tan libre se encuentra un electrón. Con la determinación de este parámetro se puede determinar las características de los átomos a los que está asociado el electrón). El trazo ancho que se encuentra a campo bajo es característico de iones Fe3+ (d5) en alto espin S= 5/2, en un campo cristalino ortorrómbico fuertemente distorsionado (Hall, 1980a). La señal pequeña que aparece en el campo central puede ser atribuida a diferentes causas, entre ellas a la resonancia del Fe3+ en bajo espin S=l/2; a defectos estructurales de la red cristalina, a impurezas de origen orgánico como semiquinonas (Cuttler y Banin, 1980), a manganeso o al traslape de estas señales. Entonces, la muestra de montmorillonita analizada por RPE no indica la presencia de manganeso, probablemente debido a que se encuentra en una forma diamagnética, al efecto dipolar generado por la cercanía e interacción de los átomos Mn–Mn o al traslape de señales.

El tratamiento de la montmorillonita con los ácidos CF3SO3H y HC1 produjo cambios importantes en la morfología del espectro de RPE, obteniéndose con ambos ácidos una disminución de la señal alrededor de g = 4.3 asignada a Fe3+ en alto espin, que sugiere un cambio en el estado de espin para este ion. Adicionalmente, en la región en la que se encuentra g≈2.0, con el tratamiento con ácidos aparecen seis señales.
En el espectro de RPE de la montmorillonita tratada con CF3SO3H (Figura 7b), las líneas están perfectamente definidas centradas en g= 2.0125, con una constante de acoplamiento A = 9.66 mT. Se pueden observar además señales dobles débiles entre los componentes hiperfinos principales que son asignados a transiciones prohibidas, causadas por distorsión axial del campo cristalino, sin acoplamiento dipolar. En el espectro de RPE de la muestra tratada con HC1 (Figura 7c), aparecen seis señales atenuadas en g = 2.0066 y constante de acoplamiento hiperfino (A) de 9.14 mT. En ambos casos la asignación de la señal corresponde a la transición central (+1/2 —> –1/2) de los iones Mn2+. Este espectro es similar al observado cuando se le intercalan iones manganeso en los espacios interlaminares a las esmectitas naturales (McBride et al, 1975a, 1975b), sugiriendo que en el tratamiento con ácido clorhídrico, los iones manganeso de la montmorillonita son disueltos y adsorbidos en la arcilla.
La excelente definición de las señales del espectro de RPE de la muestra tratada con CF3SO3H indica que los iones manganeso se encuentran diluidos magnéticamente, lo que implica una disolución parcial de estos iones en la montmorillonita.
]]> Después del tratamiento ácido, el análisis de RPE reveló la presencia de manganeso probablemente por conversión del material diamagnético a paramagnético o a una dilución magnética del manganeso sobre la superficie de la arcilla.El espectro de RPE del extracto acetónico del material tratado con el ácido CF3SO3H (Figura 8) exhibe un patrón de señales con estructura fina (cinco picos) y un desdoblamiento hiperfino (seis picos) con g=2.0125 y A=9.66 mT, característico para iones Mn2+ en alto espin S=5/2 con IMn=5/2. Adicionalmente, en g=4.3 aparece una señal sobrepuesta típica para iones Fe3+ (S=5/2). Con este análisis se establece los iones paramagnéticos contenidos en la montmorillonita por medio de un método sencillo de extracción.
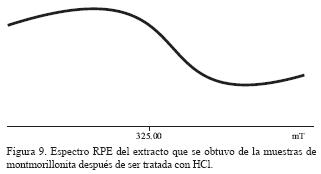
En el espectro de RPE correspondiente al extracto con acetona de la muestra tratada con HC1 (Figura 9) se observa una señal ancha centrada en g=2.0080, que evidencia interacciones dipolares fuertes, promovidas por una concentración alta de iones paramagnéticos extraídos de la montmorillonita por el tratamiento con HC1.
Análisis térmico
Las curvas experimentales del análisis térmico gra–vimétrico (TG) y de la derivada del análisis térmico gravi–métrico (DTG) para la montmorillonita en el intervalo de 20 a 800 °C se representan en la Figura 10. Analizando la curva de DTG se deducen tres reacciones endotérmicas, la primera a 81°C, con una pérdida del 6.67 % en peso, debida al agua que se encuentra adsorbida en la superficie de la arcilla. La segunda reacción se manifiesta en la gráfica a los 329 °C, con a una pérdida de peso de 0.24 % ocasionada por la eliminación de agua de los espacios interlaminares (Sarikaya et al., 2000). La tercera reacción se lleva a cabo a 721 °C, haciéndose evidente una pérdida del 2.44 % en peso del material asociado a la deshidroxilación de la capa.

Propiedades de superficie
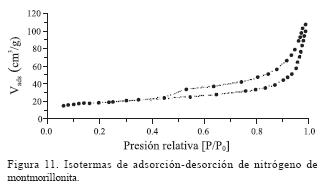
La montmorillonita fue caracterizada mediante la fisisorción de nitrógeno; la isoterma de adsorción y la curva de histéresis representadas en la Figura 11, muestra una isoterma de tipo IV atribuida a materiales mesoporosos, mientras que la histéresis que presenta es del tipo H3, asociada a aglomerados de partículas que presentan poros con forma de rendija, en donde ocurre condensación capilar (Leofanti et al., 1998).
Aplicando el método de BET (Sing et al, 1985), se determinó que la montmorillonita tiene una área superficial de 23 m2–g–1, siendo un valor intermedio comparado con otras montmorillonitas descritas en la literatura (Jozefaciuk y Bowanko, 2002). Con el método BJH, se determinó una distribución porosa monomodal (Figura 12), con un intervalo de diámetro amplio, encontrándose la mayor distribución porosa entre 20 y 1000 Å y con un máximo en 220 Å; los valores del máximo indican la presencia de material mesoporoso.

CONCLUSIONES
]]> Se determinó por métodos químicos, fisicoquímicos y espectroscópicos la composición mineral, química, estructural y morfológica de una montmorillonita natural de Cuencamé, Durango, México. La composición química se determinó por AA y por MBE, los elementos paramagnéticos se determinaron por RPE en la montmorillonita y en los extractos acetónicos de los materiales acidulados con HC1 y CF3SO3H, se confirmó la existencia de la formación de sales de Mn2+ y Fe3+.Con la técnica de adsorción–desorción de N2 y los métodos de BJH y BET se determinaron las propiedades superficiales de la montmorillonita, demostrando que el material es mesoporoso. Se discutió la descomposición mediante el análisis de RMN–AM 29Si y se definió el entorno químico del silicio debido a la sustitución de aluminio por silicio en la capa. Finalmente, se especificó la cantidad de aluminio en las capas del filosilicato.
AGRADECIMIENTOS
Agradecemos la asistencia técnica de Ma. Isabel Chávez, Leticia Baños, Raymundo Garduño, Porfirio García, Draucin Jiménez, Margarita de Pablo, Carlos Linares–López y Adolfo García. Esta investigación fue realizada con fondos de los proyectos DGAPA–UNAM (PAPIIT IN104305, IN104808) y CONACYT 059935.
REFERENCIAS
Adams, J.M., 1987, Synthetic organic chemistry using pillared, cation–exchanged and acid–treated montmorillonite catalysts– a review: Applied Clay Science, 2, 309–319. [ Links ]
Arvanitopyannis, I., Eleftheriadis I., Tsatsaroni, E., 1989, Influence of pH adsorption of dye–containing effluents with different bentonites: Chemosphere, 18(9–10), 1707–1711 [ Links ]
Ballatine, J.A., 1992, Solid supports and catalysts in organic synthesis: Chichester, Ellis Harwood Ltd., 100. [ Links ]
Barron, E, Slade, P., Frost, R., 1985, Solid state silicon–29 spin–lattice relaxation in several 2:1 phyllosicate minerals: Journal of Physical Chemistry, 9, 3305–3310. [ Links ]
Bensimon, Y., Deroide, B., Dijoux, F., Martineau, M., 2000, Nature and thermal stability of paramagnetic defects in natural clays: a study by electron spin resonance: Journal of Physics and Chemistry of Solids, 61, 1623–1632. [ Links ]
Bradley, S.A., McLaughlin, R.L., 2007, High–resolution magic–angle spinning NMR for the identification of reaction products directly from thin–layer chromatography spots: Journal of Magnetic Resonance, 45(10), 814–818. [ Links ]
Breen, C, Madejová, J., Komadel, P., 1995, Characterization of moderately acid–treated, size–fractionated montmorillonites using IR and MAS NMR spectroscopy and thermal analysis: Journal of Materials Chemistry, 5, 469–474. [ Links ]
Castellani, F., van Rossum, B., Diehl, A., Schubert, M., Rehbein, K., Oschkinat, H., 2002, Structure of a protein determined by solid–state magic–angle–spinning NMR spectroscopy: Nature, 420, 98–102. [ Links ]
Chitnis, S.R., Sharma, M.M., 1997, Industrial applications of acid–treated clays as catalyst: Reactive and Functional Polymers, 32, 93–115. [ Links ]
Cuttler, L.M., Banin, A., 1980, The behavior of synthetic 57Fe–doped kaolin: Mossbauer and electron paramagnetic resonance studies: Clay Minerals, 15, 429–444. [ Links ]
De Pablo–Galán, L., 1990, Diagenesis of Oligocene–Miocene vitric tuffs to montmorillonite and K–feldspar deposits, Durango, Mexico: Clays and Clay Minerals, 38, 426–436. [ Links ]
Fitzgerald, J.J., 1999, Solid–state NMR spectroscopy of inorganic materials: American Chemical Society, 48–57. [ Links ]
Florian, P., Vermillion, K.E., Grandinetti, P.J., Farnan, I., Stebbins, J. F., 1996, Cation distribution in mixed alkali disilicate glasses: Journal of the American Chemical Society, 118(14), 3493 –3497. [ Links ]
Gehring, A.U., Sposito, G., 1995, Reply residual manganese (II) speciation in montmorillonite: Clays and Clay Minerals, 43, 385–386. [ Links ]
Gehring, A.U., Fry, I. V, Lloyd, T, Sposito, G., 1993, Residual manganese (II) entrapped in single–layer–hydrate montmorillonite interlayers: Clays and Clay Minerals, 41, 565–569. [ Links ]
Guevara, M., Verma, S.P., Velasco–Tapia, F., Lozano–Santa Cruz, R., Girón, P., 2005, Comparison of linear regression models for quantitative geochemical analysis: Example of x–ray fluorescence spectrometry: Geostandards and Geoanalytical Research, 29(3), 271–284. [ Links ]
Guggeheim, S., van Groos, A.F.K., 2001 Baseline studies of the clay minerals society source clay: thermal analysis: Clays and Clay Minerals, 49, 433–443. [ Links ]
Hall, PL., 1980a, The application electron spin resonance spectroscopy to studies of clays minerals: I. Isomorphous substitutions and external surface properties: Clay Minerals, 15, 321–335. [ Links ]
Hall, PL., 1980b, The application electron spin resonance spectroscopy to studies of clays minerals: II. Interlaminar complexes–structure, dynamics and reactions: Clay Minerals, 15, 337–340. [ Links ]
Jozefaciuk, G., Bowanko, G., 2002, Effect of acid and alkali treatments on surface areas and adsorption energies of selected minerals: Clays Clay Minerals, 50, 771–779. [ Links ]
Kinsey, R., Kirkpatrick, R., Hower, J., Smith, K. and Olfield, E., 1985, High resolution aluminum–27 and silicon–29 nuclear magnetic resonance spectroscopy study of layer silicates including clay minerals: American Minealogist, 70, 533–548. [ Links ]
Leofanti, G., Padovan, M., Tozzola, G., Venturelli, B., 1998, Surface area and pore texture of catalysts: Catalysis Today, 41, 207–219. [ Links ]
Lippmaa, E., Magi, M., Samoson, A., Engelhardt, G., Grimmer A.R., 1980, Structural studies of silicates by solid state high resolution 29Si NMR spectroscopy: Journal of the American Chemical Society. 102, 4889–4893. [ Links ]
Long, G.L., Winefordner, J.D., 1983, Limit of detection. A closer look at the IUPAC definition: Analytical Chemistry, 55(7), 712–724. [ Links ]
Lozano, R., Bernal, J.P., 2005, Assessment of eight new geochemical reference materials for XRF major and trace element analysis: Revista Mexicana de Ciencias Geológicas, 22(3), 329–344. [ Links ]
Mabbs, F.E., Collison, D., 1992, Electron paramagnetic resonance of d transition metal compounds: Amsterdam, Elsevier, Studies in Inorganic Chemistry, 16, 1326 p. [ Links ]
Madejová, J., Komadel, P., 2001, Baseline studies of the clay minerals society source clays: infrared methods: Clays and Clay Minerals, 49, 410–432. [ Links ]
McBride, M., 1995, Comment on the natural Mn (II) EPR signal of Swy–1 montmorillonite: Clays and Clay Minerals, 43, 383–384. [ Links ]
McBride, M., Pinnavia, T.J., Mortland, M.M., 1975a, Electron spin resonance studies of cation orientation in restricted water layer on phyllosilicate (smectite) surface: Journal of Physical Chemistry, 79, 2430–2435. [ Links ]
McBride, M., Pinnavia, T.J., Mortland, M.M., 1975b, Electron spin relaxation and the mobility of manganese (II) exchange ions in smectite: American Mineralogist, 60, 66–72. [ Links ]
Miranda, R., Cortés, H., Aceves, J.M., Domínguez, J.M., Cabrera, A., Salmón, M., 1994, One pot synthesis of phenylmethanes using Al3+ doped and natural Clays: Synthetic Communications, 5, 727–732. [ Links ]
Miranda, R., Arroyo, G.A., Penieres, G., Salmón, M., Cabrera, A., Alvarez, C, Delgado, E, 2003a, Preparative heterocyclic chemistry using tonsil a bentonitic clay 1981 to 2003: Trends in Heterocyclic Chemistry, 9, 198–235. [ Links ]
Miranda, R., Ríos, H., Delgado, F., Castro, M., Cogordán, A., Salmón, M., 2003b, Characterization of a bentonitic clay and its application as catalyst in the preparation of benzyltoluenes and oligotoluenes: Applied Catalysis A: General, 244, 217–233. [ Links ]
Monsalvo, R., de Pablo L., Chavez, M. L., 2006, Hydration of Ca–montmorillonite at basin conditions: A Monte Carlo molecular simulation: Revista Mexicana de Ciencias Geológicas, 18(1), 1–12. [ Links ]
Müller, D., Gessner, W., Behrens, H., Scheler, G., 1981, Determination of the aluminium coordination in aluminium–oxygen compounds by solid–state high–resolution 27A1 NMR: Chemical Physics Letters, 79, 59–62. [ Links ]
Ohkubo, T, Kanehashi, K., Saito, K., Ikeda, Y., 2003, Observation of two 4–coordinated Al sites in montmorillonite using high magnetic field strength 27A1 MQMAS NMR: Clays and Clay Minerals, 51,513–518. [ Links ]
Rhodes, C.N., Brown, D.R., 1992, Structural characterization and optimization of acid–treated montmorillonite and high–porosity silica supports for ZnCl2 alkylation Catalyst: Journal of the Chemical Society, Faraday Transactions, 88, 2269–2274. [ Links ]
Ruiz–Guerrero, R., Cárdenas, J., Bautista, L., Vargas, M., Vázquez–Labastida, E., Salmón, M., 2006, Catalytic synthesis of 1,3,5–triphenylbenzenes, P–methylchalcones and 2,4,6–triphenylpyrylium salts, promoted by a super acid trifluoromethane sulfonic clay from acetophenones: Journal of Mexican Chemical Society, 50(3), 114–118. [ Links ]
Salmón, M., Pérez–Luna, N., López–Franco, C, Hernández, E., Álvarez–Ramírez, R.A., López–Ortega, A., Domínguez, J.M., 1997, Catalytic conversion of propylene oxide on a super acid sulfonic clay (SASC) system: Journal of Molecular Catalysis A, 122, 169–174. [ Links ]
Sarikaya, Y., Onal, M., Baran, B., Alemdaroglu, T, 2000, The effect of thermal treatment on some of the physicochemical properties of abentonite: Clays and Clay Minerals, 48, 557–562. [ Links ]
Schnell, I., Spiess, H.W., 2001, High–resolution 1H NMR spectroscopy in the solid state: very fast sample rotation and multiple–quantum coherences: Journal of Magnetic Resonance, 151, 153–227 [ Links ]
Sing, K.S.W., Everett, D.H., Haul, R.A.W., Moscow, L., Pierotti, R.A., Rouquérol, J., Siemieniewwska, T, 1985, Reporting physisorption data for gas/solid system: Pure & Applied Chemistry, 57, 603–619. [ Links ]
Vargas–Rodríguez, Y.M., Bertrán H.I., Vázquez–Labastida, E., Linares–López. C, Salmón, M., 2007, Synthesis and characterization of montmorillonite clays with modulable porosity induced with acids and superacids: Journal of Materials Research, 22 (3), 788–800. [ Links ]
Verma, S.P., Quiroz–Ruiz, A., 2006a, Critical values for six Dixon tests for outliers in normal samples up to sizes 100, and applications in science and engineering: Revista Mexicana de Ciencias Geológicas, 23(2), 133–161. [ Links ]
Verma, S.P., Quiroz–Ruiz, A., 2006b, Critical values for 22 discordancy test variants for outliers in normal samples up to sizes 100, and applications in science and engineering: Revista Mexicana de Ciencias Geológicas, 23(3), 302–319. [ Links ]
Verma, S.P., Santoyo, S., 2005, Is odd–even effect reflected in detection limits?: Accreditation and Quality Assurance, 10(4), 144–148. [ Links ]
Weiss, C, Altane, S., Kirkpatrick, R., 1987, High resolution 2'Si NMR spectroscopy of 2:1 layer silicates: correlation among chemical shifts, structure distortions and chemical variation: American Mineralogist, 72, 935–942. [ Links ]
Wind, R. A., Hu, J.Z., 2007, In vivo and ex vivo high–resolution 1H NMR in biological systems using low–speed magic angle spinning: Progress in Nuclear Magnetic Resonance Spectroscopy, 50(4) 207–259. [ Links ] ]]>