Neveen S. Ghaly1, F. R. Melek1* , Nayera A. M. Abdelwahed2
1 Chemistry of Natural Compounds Department, National Research Cenrtre, Dokki 12622, Giza, Egypt.
2 Chemistry of Natural and Microbial Products Department, National Research Centre, Dokki 12622, Giza.
Received August 2010.
Accepted December 2010.
]]> ABSTRACT
The flavonoids kaempferol–3–O–α–L–rhamnopyranoside, quercetin–3–O–α–L–rhamnopyranoside together with luteolin, kaempferol and quercetin, were isolated from the methanolic extract of the leaves of Albizia chinensis collected from Egypt. Identification of the flavonoid constituents was carried by analysing their spectroscopic data and/or by comparing these data with those reported in the literature. The first three isolates were tested for their antimicrobial activity and the results revealed that the tested compounds exhibited moderate inhibiting activity against gram–positive and gram–negative bacteria while no antifungal activity was observed.
Key words: Albizia chinensis, Leguminosa, flavonoids, antimicrobial activity.
RESUMEN
Se llevo a cabo el aislamiento de los flavonoides Kaemferol–3–O–α–L–ramnopirano–sida, quercetina–3–O–α–L–ramnopiranosida, luteolina, kaemferol y quercetina, del extracto metanolico de las hojas de Albizia chinensis colectada de Egipto. La identificación de los compuestos se llevo a cabo mediante el análisis espectroscópico. Se valoro la actividad antimicrobiana de los tres primeros compuestos mostrando actividad moderada contra bacterias gram–positivas y gram–negativas. No se observo actividad antifungicida de estos compuestos.
Palabras clave: Albizia chinensis, Leguminosa, flavonoides, actividad antimicrobiana.
INTRODUCTION
The genus Albizia (Leguminosa) comprises about 150 species widely distributed in Africa and Central and South America. In Africa, several Albizia species are used in folk medicine for the treatment of rheumatism, stomach trouble, cough, diarrhea, wounds and as anthelmintic (Watt and Breyer–brandwijk, 1962). In traditional Chinese medicine, Albizia members are used for the treatment of insominia, irritability, wounds and as antidysentric, antiseptic and antitubercular (Chadha, 1985). Phytochemical studies carried out on plants of genus Albizia, have revealed them as sources of different groups of natural products (triterpenoid saponins, di–terpenoids, lignans and pyridineglycoside). In particular, recently were reported eight compounds isolated from a 95% ethanolic extract from leaves of A. chinensis collected in China (Liu et al., 2009). The compounds were identified as quercetin 3'–O–β–D–glu–copyranosyl–3–O–rutinoside, kaempferol 3,7– di–O–β–D–glucopyranoside, rutin, D–pinitol, luteolin 7–O–β–D–glucopyranoside, (+)–lyoniresinol 3–α–O– β–D–glucopyranoside, (–)–lyoniresinol– 3–α–O– β – D–glucopyranoside and syringin.
]]> As a part of our continuous investigation on secondary metabolites from Albizia species (Melek et al., 2007; Miyase et al., 2010), we describe in this report the isolation of five flavonoids from the methanolic extract of A. chinensis leaves from Egypt.
MATERIALS AND METHODS
General experimental procedure
Column chromatography was run using silica gel 60 (Merck) and Sephadex LH–20 (Sigma). TLC and PTLC were carried out using silica gel (Merck) as stationary phase. Chromatograms were first visualized by observation under UV and then spraying with FeCl3 spray reagent. UV spectra were obtained on a Shimadzu UV–240 spectrometer. NMR spectra were recorded on a JEOL EX 500 MHz and on a Varian GEMINT 200 MHz spectrometers. Chemical shifts were given on 5–scale with TMS as internal standard.
Plant material
The leaves of A. chinensis were collected from the zoological garden in Giza, Egypt in June 2009. Plant identification was confirmed by Mrs. T. Labib, head specialist for plant identification in El–Orman public garden, Giza, Egypt. The herbarium voucher specimen was deposited in the Herbarium of NRC (CAIRC).
Extraction and isolation
Air–dried and powdered leaves (250g) of A. chinensis were extracted with MeOH (2L x 2) at room temperature. The total alcoholic extract was evaporated to dryness under reduced pressure. The residue (40 g) was suspended in distilled water (500 ml) and successively partitioned with CHCl3 (500 ml x 2), EtOAc (700 ml x 3) and BuOH (700 ml x 3). Part of EtOAc fraction (3g) was subjected to silica gel column chromatography. The column was eluted first with CHCl3 and then a CHCL3–MeOH mixture with increasing the amount of MeOH up to 20%. A total of 30 fractions (100 ml each) were collected. The fractions were monitored by TLC using the solvent system CHCL3–MeOH (5:1) and examined under UV followed by spraying with FeCl3 reagent. The fractions eluted with 2–5% MeOH were found to be similar, so they were combined and found to contain a mixture of compounds 3–5 after TLC examination. The combined fraction was repeatedly chromatographed on preparative silica gel TLC plates eluted with CHCL3–MeOH (5:1). Repeated purification of each component on Sephadex LH–20 column eluted with MeOH, afforded compounds 3 (37 mg), 4 (29 mg) and 5 (33 mg). The fractions eluted from the silica gel column with 6% MeOH, were combined after TLC analysis and contained compound 1 as a main component. The combined fraction was repeatedly chromatographed on Sephadex LH–20 column using methanol as eluent to yield compound 1 (42 mg). The fractions eluted with 8–20% MeOH were combined and found to contain a mixture of compounds 1 and 2 after TLC examination. The fraction was subjected to PTLC eluted with CHCl3–MeOH (5:1) to afford 2. Repeated purification of 2 on Sephadex LH–20 column eluted with MeOH yielded pure 2 (26 mg).
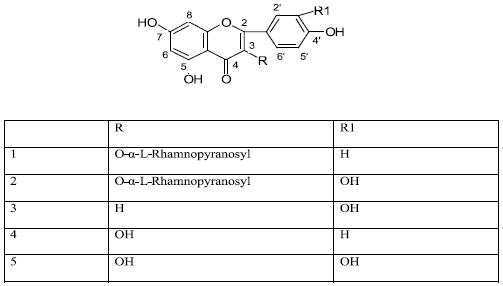
Kaempferol–3–O–a–L–rhamnopyranoside (1)
]]> Amorphous yellow powder, UV λmax nm (MeOH): 265, 340, + NaOMe: 275, 388; +AlCl3: 275, 300 sh, 347, 398; + AlCl3/HCl: 275, 300sh, 362; + NOAc: 274, 300sh, 362; + NaOAc/H3BO3: 262, 342. 1H–NMR (500 MHz, acetone–d6) δ: 12.67 (1H, s, OH–5), 7.82 (2H, d, J=8.4 Hz, H–2', H–6'), 6.99 (2H, d, J= 8.4 Hz, H–3', H–5'), 6.43 (1H, d, J=2.3Hz, H–8), 6.23 (1H, d, J=2.3 Hz, H–6), 5.49 (1H, d, J=1.5 Hz, H–1'' Rha), 2.29–3.31 (4H, H2"–H5"), 0.86 (3H, d, J=5.6 Hz, Me–6"). 13CNMR (500 MHz, acetone–d6) δ: 178.5 (C–4), 164.4 (C–7), 162.3 (C–5), 160.2 (C–4'), 157.7 (C–2), 157.2 (C–9), 134.9 (C–3), 130.8 (C–2'), 130.8 (C–6'), 121.5 (C–1'), 115.5 (C–3'), 115.5 (C–5'), 104.9 (C–10), 101.9 (C–1''), 98.8 (C–6), 93.7 (C–8), 71.3 (C–4''), 70.7 (C–3''), 70.6 (C–2''), 70.6 (C–5''), 16.9 (C–6'').Quercetin–3–O–a–L–rhamnopyranoside (2)
Amorphous yellow powder, UV λmax nm (MeOH): 256, 265 sh, 350; +NaOMe: 270, 327, 394; + AlCl3: 276, 304 sh, 334, 430; + AlCl3/HCl 272, 301 sh, 353, 401; + NaOAc: 271, 322 sh, 372; + NaOAc/H3BO3: 261, 301 sh, 367. 1HNMR (500 MHz, acetone–d6) δ: 12.68 (1H, s, OH–5), 7.46 (1H, d, J=2.3 Hz, H–2'), 7.34 (1H, dd, J=8.4, 2.3Hz, H–6'), 6.95 (1H, d, J=8.4 Hz, H–5'), 6.43 (1H, d, J=2.3 Hz, H–8), 6.22 (1H,d, J= 2.3 Hz, H–6), 5.45 (1H, d, J= 1.5 Hz, H–1" Rha), 3.30–3.32 (4H, H2" – H5"), 0.88 (3H, d, J= 5.6 Hz, Me–6"). 13CNMR (500 MHz, acetone–d6) δ: 178.5 (C–4), 164.5 (C–7), 162.3 (C–5), 157.6 (C–2), 157.2 (C–9), 148.5 (C–4'), 145.1 (C–3'), 135.0 (C–3), 121.9 (C–1'), 121.6 (C–6'), 115.9 (C–2'), 115.3 (C–5') 105.0 (C–10), 102.0 (C–1"), 98.7 (C–6), 93.7 (C–8), 71.3 (C–4"), 70.6 (C–2"), 70.5 (C–3"), 70.5 (C–5"), 16.9 (C–6").
Luteolin (3)
Amorphous yellow powder, UV λmax nm (MeOH): 254, 268, 349; +NaOMe: 230 sh, 266, 328, 403; +AlCl3: 273, 300 sh, 424; +AlCl3/HCl: 266 sh, 276, 294 sh, 355, 385; +NaOAc: 269, 384; +NaOAc/H3BO3: 260, 371. 1HNMR (500 MHz, acetone–d6) δ: 12.96 (1H, s, OH–5), 7.46 (1H, d, J=2.3 Hz, H–2'), 7.44 (1H, dd, J=8.4, 2.3 Hz, H–6'), 6.96 (1H, d, J= 8.4 Hz, H–5'), 6.55 (1H, s, H–3), 6.49 (1H, d, J= 2.3Hz, H–8), 6.21 (1H, d, J= 2.3 Hz, H–6).
Kaempferol (4)
Amorphous yellow powder, UV λmax nm (MeOH): 254 sh, 268, 322 sh, 365; +NaOMe: 275, 320, 412; +AlCl3: 262 sh, 270, 352, 426; +AlCl3/HCl 260, 271, 350, 426; +NaOAc: 275, 300, 385; +NaOAc/H3BO3: 269, 295 sh, 320 sh, 370. 1HNMR (200 MHz, DMSO–d6) δ: 8.00 (2H, d, J= 8.1 Hz, H–2', H–6'), 6.90 (2H, d, J=8.1 Hz, H–3', H–5'), 6.40 (1H, d, J= 2.5 Hz, H–8), 6.21 (1H, d, J= 2.5 Hz, H–6).
Quercetin (5)
Amorphous yellow powder, UV λmax nm (MeOH): 258, 266 sh, 299 sh, 360; +NaOMe: 272, 327, 416, +AlCl3: 275, 303 sh, 430; +AlCl3/HCl: 271, 300, 364 sh, 402; +NaOAc: 270, 325, 393; +NaOAc/H3BO3: 262, 298, 387. 1HNMR (200 MHz, DMSO–d6) δ: 7.28 (m, H–6', H–2'), 6.88 (1H, d, J= 8.0 Hz, H–5',6.40 (1H, d, J= 2.5 Hz, H–8), 6.20 (1H, d, J= 2.5 Hz, H–6).
General method for acid hydrolysis:
]]> Each glycoside (2 mg) in 3 ml 2N HCl and 3 ml methanol, was heated at 100°C for 2h. The mixture was left to cool, diluted with H2O and extracted twice with ethyl acetate. From the ethyl acetate layer, the aglycone was detected by TLC. The aqueous layer was repeatedly diluted with CH3OH and evaporated to dryness. The residue was investigated to detect the sugar L–rham–nose by paper chromatography (PC) using solvent system BAW (n–BuOH–HOAc–H2O, 4: 1: 5, top layer).Antimicrobial activity
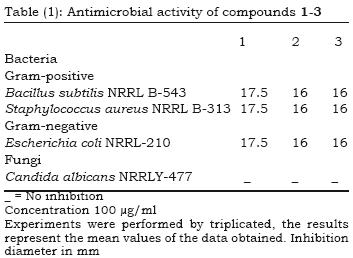
The antibacterial activity of the tested compounds were carried out against Bacillus subtilis NRRL B–543 and Staphylococcus aureus NRRL B–313 (Gram+ve bacteria) as well as Escherichia coli NRRL B–210 (Gram –ve bacteria) using nutrient agar medium. The antifungal activity of these compounds was also tested against Candida albicans NRRL Y–477 using Sabouraud dextrose agar medium.
RESULTS AND DISCUSSION
As described in the experimental section, from the methanolic extract of A. chinensis leaves, the flavonoids kaempferol 3–O–α–L–rhamnopyranoside (1 ), quercetin 3–O–α–L–rhamnopyranoside (2) luteolin (3) kaempferol (4) and quercetin (5), were isolated. The structural identification of the isolates was elucidated by acid hydrolysis, UV, 1H and 13CNMR spectroscopic analysis and/or comparison with published data.
Kaempferol 3–O–α–L–rhamnopyranoside (1) was obtained as a yellow powder. The 1HNMR spectrum of 1 showed one set of meta–coupled aromatic protons at 5 6.23 (1H, d, J=2.3 Hz) and δ 6.43 (1H, d, J= 2.3 Hz) and two sets of ortho–coupled aromatic protons at δ 7.82 (2H, d, J= 8.4 Hz) and δ 6.99 (2H, d, J=8.4 Hz). These signals were assigned to H–6, H–8, H–2' and H–6' and H–3' and H–5', respectively, of 4', 5, 7– tri–hydroxy flavonol skeleton. The two doublets at δ 5.49 (1H, J= 1.5 Hz, H–1") and δ 0.86 (3H, J= 5.6 Hz, H–6"), suggested the presence of a rhamnose moiety in the structure of 1 (Markham, 1978). The nature of the sugar was confirmed by acid hydrolysis of the glycoside and paper chromatography of the obtained rhamnose against authentic sample. The attachment of the rhamnose moiety to C–3 position of the aglycone was deduced from the two carbon signals at 5 134.9 assigned to C–3 and 5 157.7 due to C–2, in the 13CNMR spectrum of 1. The anomeric center of the rhamnose unit was determined to have a–configuration based on 3JH–1 H–2 value (1.5 Hz) and the pyra–nose form of the sugar was revealed from its 13CNMR chemical shift values. These values were consistent with those of other flavonol 3–O–rhamnoside. Based on the spectral data and comparison with literature values, 1 was identified as kaempferol 3–O–α–L–rhamnopyranoside.
Quercetin 3–O–α– L–rhamnopyranoside 2 (quercitrin) was obtained as a yellow powder. In the 1HNMR spectrum of 2, the presence of signals due to three meta – coupled protons at δ 6.22 (1H, d, J=2.3 Hz, H–6), δ 6.43 (1H, d, J= 2.3 Hz, H–8) and δ 7.46 (1H, d, J= 2.3 Hz, H–2') as well as two ortho–coupled protons at δ 6.95 (1H, d, J= 8.4 Hz, H–5') and δ 7.34 (1H, dd, J= 8.4, 2.3 Hz, H–6') along with carbon signal at δ 178.5 (C–4) in the 13CNMR spectrum, indicated a typical 3', 4', 5, 7– tetrahy–droxyflavonol skeleton. The two doublets in the 1HNMR spectrum of 2 at δ 5.45 (1H, J= 1.5 Hz) and δ 0.88 (3H, J= 5.6 Hz) were assigned to H–1" and H–6" of a rhamnose unit. The 13CNMR data of 2 and the result of acid hydrolysis indicated a–L–rhamnoside. The attachment of the moiety at C–3 position of the aglycone was revealed from the 5 value of C–3 (5 135.0) and that of C–2 (5 157.6) which are similar to the corresponding values of 1 . Compound 2 was then assigned the structure of quercetin 3–O–α–L–rhamnoside based on the above spectral evidences and comparison with literature values. The flavonoids luteolin (3), kaempferol (4) and quercetin (5) were characterized by comparison of their spectral data with the literature values (Harborne, 1993). Kaempferol was previously isolated from Albizia versicolor (Rukunga & Watermann, 1996) while luteolin was obtained from A. julibrissin (Jung et al., 2004). Quercetin 3–O–α–L– rhamnoside was reported also from A. julibrissin ( Lau et al., 2007). In the case of quercetin and kaempferol 3–O–α–L–rhamnoside, to our knowledge this is the first reported occurrence of these compounds in the genus Albizia.
The activities assigned to the flavonoids identified in this work could explain some of the medicinal properties of Albizia chi–nensis.
The antibacterial activity for compounds 1–3 against the micro–organisms Bacillus subtilis NRRL B–543 and Staphylococcus aureus NRRL B–313 (Gram +ve bacteria) and Escherichia coli NRRL B–210 (Gram –ve bacteria) together with the antifungal activity against Candida albicans NRRL Y–477, were studied using the agar diffusion method (Cruickshank et al., 1975). The results (Table 1) revealed that, in our experimental conditions, the tested compounds exhibited moderate inhibiting activity against gram–positive and gram–negative bacteria while no antifungal activity was observed.
]]>
REFERENCES
Chadha, Y.R. (1985). The Wealth of India, Raw Materials; Publication and information Directorate Vol.1A. Council of Scientific and Industrial Research, New Delhi, p. 126. [ Links ]
Cruickshank, R., Duguid, J.P., Marmion, B.P., Swain, R.H.A. (1975). Medical Microbiology, 12th Ed. Vol. II Churchill [ Links ]
Harborne, J.B. (1993). The Flavonoids Advances in Research". Chapman and Hall, London. [ Links ]
Jung, M.J., Kang, S.S., Jung, H.A., Kim, G.J., Choi, J.S. (2004) Isolation of flavonoids and a cerebroside from the stem bark of Albizia julibrissin. Archives of Pharmacal Research 27: 593–599. [ Links ]
Lau, C.S., Carrier, D.J., Beitle, R.R., Bransby, D.I., Howard, L.R., Lay, J. J.O., Liyanage, R., Clausen, E.C. (2007) Identification and quantification of glycoside flavonoids in the energy crop Albizia julibrissin. Bioresource Technology 98: 429–435. [ Links ]
Liu, R., Yu, S., Pei, Y. (2009) Chemical constituents from leaves of Albizia chinensis. Zhongguo Zhong Yao Za Zhi 34(16): 2063–2066. [ Links ]
Markham, K. R., Ternai, B., Stanley, R., Geiger, H., Mabry, T. J. (1978) Carbon 13 NMR studies of flavonoids –III: Naturally occurring flavonoid glycosides and their acylated derivatives. Tetrahedron 34: 1389–1397. [ Links ]
Melek, F.R., Miyase, T., Ghaly, N.S., Nabil, M. (2007) Triterpenoid saponins with N–acetyl sugar from the bark of Albizia procera. Phytochemistry 68: 1261–1266. [ Links ]
Miyase, T., Melek, F.R., Ghaly, N.S., Warashina, T., El–Kady, M., Nabil, M. (2010) Echinocystic acid 3, 16–O–bisglycosides from Albizia procera. Phytochemistry 71: 1375–1380. [ Links ]
Rukunga, G.M., Waterman, P.G. (1996) Kaempferol glycosides from Albizia versicolor. Bulletin of the Chemical Society of Ethiopia 10(1), 47–51. [ Links ]
Watt, J.M. Breyer–brandwijk, M.C. (1962). Medicinal and poisonous Plants of South and East Africa. Second ed. Livingstone, Edinburgh, p. 553. [ Links ]
]]>