 Address reprint requests to:
Address reprint requests to: Verna Mai, MD
Cancer Care Ontario. 505 ]]> University Ave. 18th Floor
Toronto, Ontario M5G1X3. Canada
E-mail:verna.mai@cancercare.on.ca
Breast cancer screening program in canada: successes and challenges
Programas de detección temprana de cáncer de mama en canadá: avances y obstáculos
Verna Mai, MD, MHSc, FRCPC; Terrence Sullivan, PhD; Anna M. Chiarelli, PhD
Cancer Care, Ontario, Canada
]]>
ABSTRACT
This paper describes breast screening program development in Canada and the current status of screening in Canada. Programs have been implemented in most of Canada, beginning in the late 1980's. Certain components are common to all the programs, but others, such as personal invitation letters for recruitment and clinical breast examination vary across the country. Key successes in organized breast screening in Canada include the development of a comprehensive set of screening performance indicators, which are reported on regularly, and the downward trend in mortality rates observed over the past 20 years. Challenges include the continued prevalence of opportunistic screening; the need to better manage follow-up; dealing with changing evidence; and supporting informed decision-making about screening. Approaches to breast screening are dependent on the health care services available in countries, but regardless of the approach, good evaluation is necessary.
Key words: mammography; utilization; secondary prevention; prevention and control; Canada
RESUMEN
Este artículo describe el desarrollo de la detección temprana de cáncer de mama en Canadá así como la situación actual de los programas de detección de cáncer en el mismo país. En su gran mayoría, estos programas de detección han sido implementados desde comienzos de los años ochenta. Algunos elementos de estos programas representan denominadores comunes en todos ellos. Sin embargo existen otros elementos -tales como invitaciones personales para reclutamiento y exámenes clínicos de mama, que difieren dependiendo de cada jurisdicción. Algunos de los avances en los programas organizados de detección temprana de cáncer de mama en Canadá incluyen la existencia de indicadores de evaluación de desempeño, sobre los cuales se reporta de forma regular. En base a estos indicadores se puede observar una tendencia descendente en los índices de mortalidad en los últimos 20 años. Algunas de las dificultades incluyen la persistencia de detección oportunística, la necesidad de gerenciar el efectivo seguimiento de pacientes, gerenciar el constante cambio de evidencia, así como el proveer asistencia en la toma de decisiones relacionadas a la detección temprana de cáncer. Las prácticas focalizadas en mejorar la detección temprana de cáncer dependen de los servicios de salud existentes en cada país. Sin embargo e independientemente de la orientación utilizada, la necesidad de evaluar el desempeño de los programas es un elemento vital.
Palabras clave: mamografía; utilización; prevención secundaria; prevención y control; Canadá
]]> Breast cancer trends in Canada
The most common cancer in Canadian women is breast cancer, with an estimated 22400 new cases were diagnosed in 2008, resulting in an incidence rate of 104 per 100000 women.1 Incidence rates increased steadily from 1969-1999, and have since stabilized. Breast cancer mortality trends show a very different pattern, with a decline of 25% from 1986 to 2003, from 32 per 100000 to 24 per 100000.2 Other developed countries have also experienced this trend in breast cancer mortality. This has been attributed to improvements in early detection and also to advances in treatment. While the relative contributions of screening and treatment are difficult to determine, modeling studies carried out on U.S. data have determined that both factors are almost equal in their impact.3 In Canada, earlier stage breast cancers, such as those found by mammography screening are associated with five year survival rates of greater than 90%.
Canadian breast screening recommendations
The Canadian Task Force on Preventive Health Care (CTFPHC) is a national body set up in 1976 to provide evidence-based recommendations on clinical prevention, including cancer screening.4 While a recommendation for breast screening was first published in 1986, the most current one for women aged 50-69 was published by the CTFPHC in 1998: "There is good evidence for screening women aged 50-69 years by clinical examination and mammography. The best available data support screening every 1-2 years." Both maneuvers were recommended as the relative contributions of each were not clear from the existing breast screening studies. An update was published in 2001 addressing women younger than 50 years of age: "Current evidence does not support the recommendation that screening mammography is included in or excluded from the periodic health examination of women aged 40-49 at average risk of breast cancer."
Finally, with regard to breast self examination, the CTFPHC has recommended against the inclusion of BSE teaching as part of the periodic health exam: "Because there is fair evidence of no benefit, and good evidence of harm, there is fair evidence to recommend that routine teaching of BSE be excluded from the periodic health examination of women aged 40-69."
The recommendations of the CTFPHC are consistent with those published by the International Agency for Research on Cancer (IARC) in 2002. The conclusions of the IARC after evaluation of all the available evidence were as follows: 1) The evidence is sufficient to demonstrate that screening women 50-69 with mammography reduces mortality from breast cancer by 25%. There is only limited evidence for women aged 40-49. 2) The evidence that clinical breast examination (CBE) or breast self-examination can reduce mortality from breast cancer is inadequate.5
Recent results were published in 2007 from a breast screening trial in the U.K. which focused specifically on screening impact for women starting at the age of 40. Between 1991-1996, 160921 women from England, Wales and Scotland aged 39-41 were randomized to annual screening intervention or control (usual medical care). After a follow-up period of 11 years, a reduction in breast cancer mortality of 17% in the intervention group, compared to the control group was observed, but this finding was not statistically significant.6
Breast screening program development in Canada
]]> The delivery of publicly funded health care services in Canada is the responsibility of the 10 provinces and three territories that exist as geopolitical entities. The breakdown of the estimated 33142610 Canadian population in 2008 varies greatly across jurisdictions with the largest population in the province of Ontario (12861940) and the smallest population in the territory of Nunavut (31143). To implement breast screening programs fully in Canada requires each of the 13 jurisdictions to implement their own provincial/territorial program. At a national level, there is pan-Canadian discussion and planning supported by the federal government, but moving forward with program implementation is largely a provincial/territorial decision.7The first province to start a breast screening program was British Columbia, in 1988. Most of the other jurisdictions implemented programs over the next 10 years. As of 2008, only the territory of Nunavut does not have a program.
The typical program model in Canada consists of the following components: a defined approach to identifying and inviting the target population to screening (in 8 of the 12 programs - personal invitations by mail are the method of recruitment); screening with bilateral 2 view mammography every two years; arrangements for follow-up of women with abnormal screening results and/or collection of follow-up diagnostic testing information from various sources; and sending reminder letters to participants with a normal screening result, when the next mammogram is due. There is a distinct program budget to support planning, implementation and evaluation, including some form of information system to track participants through the screening pathway to the point of reaching a cancer diagnosis.8
Aside from the key components of breast screening programs listed above, the approach varies across the country with regard to other program aspects. For example, the age eligibility criterion for program attendance varies (the target population for screening in Canada is the 50-69 year age group, but women aged 40-49 can participate in programs in all but three provinces). Another example is the provision of CBE- only three jurisdictions provide CBE as an adjunct to mammography screening. Some provinces transport mammography equipment and technologists by bus or plane to small rural communities to serve women who have pre-booked appointments. In Ontario, digital mammography is used on the travelling bus, allowing real time checks of the images by the medical radiation technologist or by radiologists located elsewhere at a major centre. This minimizes the need to rebook repeat procedures weeks or months later for those mammograms that turn out to be unsuitable for interpretation. These examples illustrate the autonomy that each province and territory has in determining how to provide breast screening services.
Opportunistic or non-programmatic breast screening
In all jurisdictions in Canada, women can be referred by a physician for mammograms outside of the breast screening programs. The amount of screening that takes place in a non -programmatic fashion varies across the country, and depends on the degree to which programs are population-based- i.e. whether all eligible women are included in program invitations and/or all mammography facilities are participating in the program. The usual method of funding bilateral mammograms outside of programs is by "fee for service billing" and there is usually a fee code which indicates the procedure of a mammogram. No other tracking of screen results or follow-up is usually available for non-programmatic screening, only the volumes and basic patient demographics (sex, age).
What has been successful in breast screening in Canada?
Mammography uptake
]]> As of 2008, 12 of the 13 jurisdictions in Canada have established breast screening programs, signifying a commitment across the country to provide access to quality screening. While the comprehensiveness of programs is variable, there has been steady growth in the number of women screened through organized breast screening programs. Although program participation rates in the programs are suboptimal (36.5% in 2003/04),8 the total uptake of mammography including both program and opportunistic screening is much higher. Sixty two percent of women aged 50-69 reported in the 2005 Canadian Community Health Survey that they had a mammogram in the previous two years.8 Although self reported rates tend to be higher than actual rates, this recent finding is approaching the Canadian target of 70% participation.A decline in breast cancer mortality since 1986
After a stable mortality trend for over 20 years, breast cancer mortality began to decline from approximately 1986 to the present. This timing spans the period during which there have been improved treatments for breast cancer and also introduction and growth of mammography screening. Screening has contributed to improved mortality rates through increased participation and also improved quality of mammography since the late 1980's.
Routine programmatic screening performance indicators
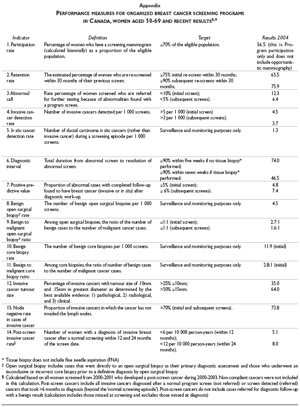
A common set of performance indicators has been adopted by a national committee with membership from all 13 provinces/territories and key breast cancer screening stakeholders including the Canadian Cancer Society, the Canadian Association of Radiologists and the Public Health Agency of Canada.9 The indicators cover participation and retention rates, screening results, diagnostic interventions and cancer detection rates (Appendix). All breast screening programs with information systems submit data to a national breast screening database and a national report on program performance is published biennially.8 It is posted on the website of the Public Health Agency of Canada. In addition, provinces have also started to report on system performance indicators on a routine basis. In Ontario, an annual Cancer System Quality Index is produced which includes indicators on breast screening participation rates by region and the time interval between an abnormal screen and breast surgery for breast cancer patients.10 Such public reporting can support a continuous cycle of quality improvement initiatives informed by the data.11
Targets have been set for most of the national breast screening indicators. Performance indicator targets have been achieved in Canada for the proportion of invasive cancers that are= or < 15 mm; node negative rate in detected cancers; and finally positive predictive value of mammography screens and the most recent data for 2004 are shown in the Appendix.
What are the key challenges in breast screening in Canada?
Dual systems for breast screening
Most Canadians have a family doctor. Opportunistic screening is a usual practice for many of the maneuvers that are performed for many types of screening. When the patient sees the doctor for an unrelated health concern, the opportunity for a screening referral arises and the doctor initiates referral for a mammogram (or distributes a fecal occult blood testing kit for colorectal screening, or performs a Pap test for cervical screening). This is adhoc, and not part of a program, which would provide routine reminders when screening is due. Because opportunistic screening already existed when breast screening programs were being implemented across Canada, it has continued to exist alongside of organized programs. Minimal evaluation of breast screening through the opportunistic system is possible, and the quality assurance programs for mammography that are implemented in programmatic facilities may also not be in place. In Canada, approximately half of screening mammography is provided by organized programs and half by opportunistic screening.8
]]> Aside from the issues of quality and monitoring, funding challenges have also arisen in recent years. While breast screening programs in many provinces have had their budgets capped at amounts lower than the demand for programmatic screening, most opportunistic mammography services, which are "fee for service", have not faced the same constraints. Whatever is billed for by the "fee for service" system is paid for by provincial governments.The challenge is to eliminate the dual systems and provide programmatic screening to all women eligible for breast screening. However, the fully organized program model used in some European countries is not easily achieved in Canada, given the reliance of Canada's medical system delivery models on primary care providers to be key players in providing preventive care to their practice populations. In Ontario, there are an estimated 10000 family physicians providing primary care services, and facilitating uptake of evidence-based screening through such a large number of practitioners is a challenge. However, some progress has been made in instilling a sense of "population" in family practice, and work has begun to formulate a hybrid type of screening delivery model whereby the provincial screening programs work with and support family doctors to achieve high levels of screening participation in their practice populations.
More emphasis on follow-up for abnormal screens
As screening programs were first established, their focus was on delivery of high quality screening. The next steps for women with an abnormal screen result were seen as the responsibility of the family doctor. Appropriate and timely follow-up assessment of screening abnormalities is needed to ensure maximal effectiveness of screening and minimize anxiety. Assessment procedures may include additional imaging and surgical procedures, in order to reach a diagnosis. In recent years, there has been a shift from the use of open surgical biopsy to less invasive core needle biopsies, guided by breast imaging, to obtain a tissue diagnosis prior to any surgical treatment for confirmed cancer cases. Data from the Canadian breast screening database show that from 2001/02 to 2003/04 core biopsy use increased from 9.6 to 12.3%, and open biopsy decreased from 7.2 to 5.6%.8
Timeliness targets have been established nationally for the diagnostic interval. These have been reported nationally since 2000, but there has only been minimal progress towards the achievement of these targets, particularly when a biopsy is required. The target is for 90% or more of women undergoing assessment with biopsy to be completed by seven weeks, but less than 50% were completed by this timeframe in 2004.8
The challenge of promoting evidence-based screening interventions, in the environment of constantly evolving evidence and development of new technologies
Scientific evidence is always evolving and new evidence may prove new screening tests to be of value or show that previously promoted tests are not as effective as once thought. It is necessary for screening programs to have the capacity to review new studies, create or update screening guidelines continuously and implement policies that are appropriate to the environment that the program exists in.
1. Changing evidence about established modalities
The breast screening program in Ontario (Ontario Breast Screening Program) started its operations in 1990 based on what was considered to be a comprehensive approach to screening. Three modalities were used for every screening visit: mammogram, clinical breast examination (CBE) by a trained nurse examiner, and breast self examination (BSE) teaching. Since then, emerging evidence and accumulating program data on clinical outcomes have raised serious questions about the utility of the clinical breast exam in the program, and the efficacy of breast self examination. Based on the 2001 recommendations of the CTFPHC, the program discontinued the teaching of BSE. However, despite the lack of evidence of effectiveness and the potential for harm related to unnecessary diagnostic procedures for benign lesions, this decision was not readily accepted by all stakeholders since it was such a departure from years of promotion of BSE by many groups. CBE is another maneuver that is used in only a few breast screening programs in Canada. Its effectiveness in reducing mortality from breast cancer has not been shown conclusively. Evidence of a reduction in breast cancer mortality rate through screening comes from the results of several randomized controlled trials.12,13 Of the trials conducted, four included CBE in addition to mammography. Studies showing mortality reduction in trials of CBE with mammography are similar to those in trials including mammography only.12 A recent study of four provinces (including Ontario) that provide CBE found that CBE contributes minimally to the early detection of breast cancer.14 Cancer detection rates were increased by CBE alone by 5-8% over those for mammography, however the clinically detected cancers were primarily invasive, and had a greater tumour size and nodal involvement.
2. New evidence about new screening technologies
]]> In addition to conventional mammography screening, evidence has emerged about the effectiveness of digital mammography and magnetic resonance imaging (MRI) for use in certain populations of women. Digital mammography performs better than conventional mammography in younger women and women with dense breasts.15 Magnetic Resonance Imaging (MRI) has been found to be more sensitive for detecting cancers in women who are BRCA1 and BRCA2 mutation carriers than mammography, ultrasound or CBE, with a sensitivity of 77% for cancer detection compared to 36% for mammography.16 This new, compelling evidence for women who are at increased risk of developing breast cancer is creating more complexity to breast screening, with implications of different screening recommendations for individuals, depending on risk profiles. This is not dissimilar to more recent colorectal screening recommendations, which recommend fecal occult blood testing for average risk individuals, and screening colonoscopy for individuals at increased risk due to familial risk factors.17 An important issue is whether breast screening programs that have targeted average risk, age-eligible women should also screen sub-populations at increased risk, incorporating the required screening modalities such as MRI.There are also a myriad of new technologies in the early stages of development. While some of them may prove to be effective, they are not currently recommended. Despite the lack of evidence, there is often media hype about their potential benefits and various commercial enterprises will promote their benefits to women. Some tests reported in the media that fall into this category include thermography scanning, laser detection, radioactive imaging techniques, and various protein marker tests of body fluids such as saliva. Providing clear messages about effective screening tests and educating the public about the attributes of a good screening test have become important roles for publicly funded screening programs in Canada. More research and development work is required to create frameworks that can help policy makers, health planners and the public distinguish between useless, promising and proven screening modalities.
Supporting informed decision-making about cancer screening
In most breast screening programs, the key message to eligible women has been that "breast screening saves lives". This is true, according to the best evidence available. However, a focus on persuading women to participate in screening, can omit important information about the limitations as well as benefits of breast screening. In recent years, screening programs have been challenged to provide more balanced information. Critics have proposed that the potential harms of screening may outweigh the benefits, yet screening programs continue to be promotional in their messaging. While promotion of mammography screening may arguably have been justified because there is sound evidence of efficacy, the promotional nature of health education efforts in the past may have reinforced the erroneous impression that all tests that can find cancer are worth considering. This is not the case, however. There are limitations to screening, including the fact that regular screening of a population will not reduce all mortality associated with the cancer being screened for, and there are imperfections of screening such as missed cancers, false positive results, and inability to distinguish between cancers that cannot be cured, despite being found earlier; cancers that are cured because they've been found earlier; and cancers that are more benign in their behavior and would not progress to cause death.
As an example of how informed participation is supported in the Ontario Breast Screening Program, the previous brief promotional message of "Breast Screening Saves Lives" has been supplemented with more factual information that notes that
"A mammogram is not a perfect test. It finds eight out of ten cancers. As well, not all cancers found at screening can be cured. Breast screening may not benefit women with aggressive cancers. However, many studies show that regular mammograms for women aged 50-69 reduce deaths from breast cancer by up to a third".
Work is underway by the national breast screening committee to complete a decision aid to assist women of all ages in reviewing the potential benefits and limitations of breast screening and take these into account in considering breast screening.
Conclusion
Canada has experienced some important successes in the implementation of breast screening programs. Almost all jurisdictions now have programs, and a national approach exists to collect data and report on key performance indicators across Canada as a whole and by province/territory.
]]> Many of the performance indicators show that the intermediate measures of a successful program have been achieved (Appendix). Screening programs detect a greater proportion of early stage cancers as shown by their small size and node negativity. There is also evidence of a decrease in the breast cancer mortality rate and the most recent data continue to show a declining trend. Participation rates in mammography are close to the 70% target, when we include the mammography that is delivered in an opportunistic manner.The key opportunities for further improvement are threefold:
1. Finding better ways to integrate the roles of screening programs and primary care providers so that the most effective methods of informing the eligible population about breast screening and recruiting them to programmatic screening can be implemented. The provision of all screening through a programmatic approach that provides recruitment, quality screening, facilitated assessment of abnormal screen results, tracking of diagnostic tests and outcomes and routine rescreening reminders is the goal. The introduction of better electronic records in primary care may facilitate integration between program information systems and physician patient records.
2. Improvements in methodology to evaluate and monitor the status of the many new breast screening modalities. This would help answer questions about when and how new tests should be introduced for widespread application in healthy women, and conversely, when older modalities of screening need to be retired.
3. Finally, in this age of growing consumer knowledge and sophistication, a continued shift in how screening information is presented is required. There are potential benefits and limitations for all screening tests and a balanced perspective must be presented, supporting informed decision-making about screening. Imparting such information, starting with breast screening, will assist decision-making about screening tests for other cancers as well, and may be most useful in helping individuals deal with the tests that have a lack of evidence of mortality benefit.
This is a brief synopsis of successes and challenges of breast screening in Canada. The Canadian experience may have limited utility in developing and middle income countries. The issues related to maximizing screening in Canada, a country with a publicly funded health care system and abundant mammography capacity will differ from those of other countries, such as Mexico, where certain realities and contextual issues will lead to different approaches. Many countries striving to improve early detection of breast cancer do not have the health care resources or infrastructure to provide accessible, high quality mammography for population-based screening. In the Canadian context, CBE is considered an adjunct only to mammography, if provided. Data from programs providing both modalities have confirmed the very minimal additional cancer detection offered by CBE when two view mammography is routinely performed.11 However, the data also show that a fairly large number of cancers are detected by both mammography and CBE. The proportion of cancers detected for women with a breast cancer on a first screen visit (n=800) was as follows: CBE only: 40 (5%); mammography only: 427 (53%); and both CBE and mammography detected: 323 (40%). Thus, while CBE alone detected only 5% of cancers, the results show CBE (including those that mammography also detected) detected 363/800 cancers or 45%. This provides a better estimate of the value of providing CBE in the context of "no mammography" and supports consideration of CBE as a viable screening modality in such circumstances, especially as a "transitional technology" while future capacity for mammography and other technology is under consideration. While technology needs are less with CBE, it would be essential in embarking on any CBE program to closely monitor and evaluate services (including training of practitioners and quality assurance) to maximize cancer detection rates and minimize false positive findings, since the positive predictive value is lower for CBE than for mammography.11 Finally, regardless of the screening test, well articulated assessment pathways must be planned to take care of those women with an abnormal screening result, or any screening endeavors will not achieve their health objectives.
References
1. Canadian Cancer Society/National Cancer Institute of Canada: Canadian Cancer Statistics 2008, Toronto, Canada, 2008. [ Links ]
2. Canadian Cancer Society/National Cancer Institute of Canada: Canadian Cancer Statistics 2007, Toronto, Canada;2007. [ Links ]
3. Berry DA, Cronin KA, Plevritis SK, Fryback DG, Clarke L, Zelen M, et al. Effect of Screening and Adjuvant Therapy on Mortality from Breast Cancer. N Eng J Med 2005;353:1784-92. [ Links ]
4. Canadian Task Force on Preventive Health Care. CTFPHC History/Methodology [online]. 1997 [cited 2008 Oct.13]. Available from URL:http://www.ctfphc.org/ [ Links ]
5. Vainio H, Bianchini F, editors. Breast Cancer Screening. Lyon, France: IARC Press; 2002. (IARC Handbooks of Cancer Prevention; Vol 7).
6. Moss SM, Cuckle H, Evans A, Johns L, Waller M, Bobrow L. Effect of mammographic screening from age 40 years on breast cancer mortality at 10 years' follow-up: a randomised controlled trial. Lancet 2006;368:2053-60. [ Links ]
7. Marchildon, GA. Health Systems in Transition: Toronto, Canada:University of Toronto Press;2006. [ Links ]
8. Public Health Agency of Canada. Organized Breast Screening Programs in Canada: Report on Program Performance in 2003 and 2004. Ottawa:Her Majesty the Queen in Right of Canada; 2008. [ Links ]
9. Public Health Agency of Canada. Guidelines for monitoring breast screening program performance: Report from the evaluation indicators working group: 2nd Edition. Ottawa; Her Majesty the Queen in Right of Canada; 2007. [ Links ]
10. Cancer Care Ontario [Online]. Cancer System Quality Index; 2008. [cited 2008 Oct.31]. Available from URL: http://www.cancercare.on.ca/english/csqi2008/csqiaccess/csqi/. [ Links ]
11. Sullivan T, Dobrow MJ, Schneider E, Newcomer L, Richards M, Wilkinson L, et al. Improving clinical accountability and performance in the cancer field. Pratiques et Organisation des Soins 2008; 39:207-15. [ Links ]
12. Humphrey LL, Helfand M, Chan BKS, Woolf SH. Breast cancer screening: A summary of the evidence for the U.S. Preventive Services Task Force. Ann Intern Med 2002;137:347-60. [ Links ]
13. Vainio H, Bianchini F, editors. Breast Cancer Screening. Lyon, France: (IARC Handbooks of Cancer Prevention; Vol 7:87-117) IARC Press; 2002. [ Links ]
14. Bancej C, Decker K, Chiarelli A, Harrison M, Turner D, Brisson J. Contribution of clinical breast examination to mammography screening in the early detection of breast cancer. J Med Screen 2003;10:16-21. [ Links ]
15. Pisano ED, Gatsonis C, Hendrick E, Yaffe M, Baum JK, Acharyya S, et al. Diagnostic Performance of Digital versus Film Mammography for Breast-Cancer Screening. N Eng J Med 2005;353:1773-83. [ Links ]
16. Warner E, Plewes DB, Hill KA, Causer PA, Zubovits JT, Jong RA, et al. Surveillance of BRCA1 and BRCA2 Mutation Carriers with Magnetic Resonance Imaging, Ultrasound, Mammography, and Clinical Breast Examination. JAMA 2004;292:1317-25. [ Links ]
17. Canadian Task Force on Preventive Health Care. Colorectal Cancer Screening: Recommendation Statement. CMAJ 2001;165:206-8. [ Links ]
 Address reprint requests to:
Address reprint requests to:
Verna Mai, MD
Cancer Care Ontario. 505 ]]>
University Ave. 18th Floor
Toronto, Ontario M5G1X3. Canada
E-mail:verna.mai@cancercare.on.ca
Received on: October 31, 2008
Accepted on: December 17, 2008
Appendix
]]>
]]>