A simple method for the isolation of lycopene from Lycopersicon esculentum
Un método simple para aislar licopeno, a partir de Lycopersicon esculentum
Myong-Kyun Roh1, Min-Hee Jeon1, Jin-Nam Moon1, Woi-Sook Moon1, Sun-Mee Park2 and Jae-Suk Choi3,4
1 Department of Research and Development, Ecomine Co., Ltd., Nam-gu, Busan, Republic of Korea.
]]> 2 Hazard Analysis Critical Control Point Training Center, Silla University, Sasang-gu, Busan, Republic of Korea.3 Regional Innovation System Center, Industry-Academic Cooperation Foundation, Silla University, Sasang-gu, Busan, Republic of Korea.
4 Corresponding author: bioengchoi@korea.com
Received: July 31st, 2012
Accepted: October 25th, 2012
Abstract
The purpose of this study was to develop a simple and effective method using conventional solvent extraction and anti-solvent precipitation for the isolation of lycopene from Lycopersicon esculentum. A total of 100 grams of freeze-dried L. esculentum powder was weighed into a glass tube with a glass filter at the bottom. The material was extracted for 1 hour using 1 L of the following solvents: hexane, ethyl acetate, and ethanol. The extraction yields were 3.58, 4.39, and 1.25 mg/g, respectively, indicating that ethyl acetate was the most effective of the studied extraction solvents. Next, all carotenoids, except lycopene, were removed from the crude extract using an anti-solvent (methanol) salting-out method. The precipitated and isolated lycopene totaled 3.50 mg/g, indicating a 77.43% lycopene recovery rate. The lycopene isolation method developed in this study was more effective than the previous reported methods for the large-scale preparative isolation of lycopene.
Key words: anti-solvent, lycopene, precipitation.
]]>Resumen
El objetivo de este estudio fue desarrollar un método sencillo y eficaz mediante la extracción de un disolvente convencional y precipitación anti-disolvente para el aislamiento de licopeno a partir de Lycopersicon esculentum. Un total de 100 gramos de polvo liofilizado de L. esculentum se pesaron en un tubo de vidrio con un filtro de vidrio en el fondo. El material se extrajo durante una hora con un litro de los disolventes hexano, acetato de etilo y etanol. Los rendimientos de extracción fueron 3.58, 4.39 y 1.25 mg/g, respectivamente, lo que indica que el acetato de etilo fue el más eficiente de los disolventes de extracción estudiados. Todos los carotenoides, excepto licopeno, fueron separados del extracto crudo utilizando un método de salificación anti-disolvente (metanol). El licopeno precipitado y aislado fue de 3.50 mg/g, que es una tasa de recuperación del 77.43%. El método de aislamiento de licopeno desarrollado en este estudio fue más eficaz que los métodos ya disponibles para el aislamiento a gran escala de la preparación de licopeno.
Palabras clave: anti-solvente, licopeno, precipitación.
Lycopene (CAS Number: 502-65-8) is one of over 600 carotenoids found in nature. It accumulates in relatively few tissues and can most easily be observed in ripe tomato, red pepper, watermelon, and red grapefruit, giving them a characteristic red pigmentation. Lycopersicon esculentum is considered one of the best resources of lycopene production. Lycopene, which has an intense red color, is the most abundant carotenoid in L. esculentum, accounting for approximately 85% of the total carotenoids present. Concentrations of lycopene in L. esculentum vary from 30 to 200mg/kg in fresh fruit and from 430 to 2,950 mg/kg on a dry basis (Leoni, 1993).
Lycopene is important, not only because of its use as natural food colorant but also because of its recognized health benefits. Although it has no provitamin A activity, lycopene functions as an antioxidant and quenches singlet oxygen in vitro. The quenching constant of lycopene is more than double that of β-carotene and 10 times greater than that of α-tocopherol (Shi et al., 1999). Epidemiological studies have shown that serum levels of lycopene and dietary intake of lycopene from L. esculentum are inversely related to the risk of certain types of cancer, such as prostate cancer, digestivetract cancers, and lung cancer (Le Marchand et al., 1989).
Many different techniques, such as conventional solvent extraction (Sadler et al., 1990) and supercritical fluid extraction (SFE) (Cadoni et al., 2000; Vasapollo et al., 2004; Shi et al., 2009), have been used for the extraction of lycopene from L. esculentum. For further isolation and purification, thin-layer chromatography (TLC; Liu et al., 2008), column chromatography (Hakala and Heinonen, 1994), and high-performance liquid chromatography (HPLC) techniques (Cadoni et al., 2000; Miguel et al., 2006) have been adapted. However, these isolation techniques require sensitive and complex manipulations and are time consuming.
In the anti-solvent precipitation process, solid crystals are formed and precipitate from a solution. Anti-solvent precipitation is also a chemical solid-liquid separation technique in which mass transfer of a solute from the liquid solution to a pure solid crystalline phase occurs. Crystallization is therefore, an aspect of precipitation, obtained through a variation in the solubility conditions of the solute in the solvent, as compared to precipitation resulting from a chemical reaction (O'Grady et al., 2007; O'Ciardhá et al., 2011). The anti-solvent approach for crystallization is considered a powerful isolation and purification technique (O'Grady et al., 2007). It has been reported that indomethacin (Lee et al., 2011) and paracetamol (Ó'Ciardhá et al., 2011) can be prepared by an anti-solvent method. The production of fine lycopene particles using a supercritical fluid as an anti-solvent has also been reported (Miguel et al., 2006).
We now know the anti-solvent precipitation can accelerate the isolation process and this in turn may improve lycopene recovery. To the best of our knowledge, few studies have examined lycopene isolation using organic anti-solvent precipitation. Therefore, in this study, we developed a simple and effective extraction and isolation protocol of lycopene from Lycopersicum esculentum by conventional solvent extraction and organic anti-solvent precipitation. This protocol has application potential for the large-scale preparative isolation of lycopene.
]]>Materials and methods
Sample preparation and materials. Lycopersicon esculentum was provided by Chal-Tomato Co., Ltd. (Daejeo, Busan, Korea). Fresh L. esculentum fruits were thoroughly washed with tap water and the seeds were removed. The fruits were then chopped into cubes, which were freeze-dried for 3 days until a moisture content of approximately 0.8% was obtained. The freeze-dried L. esculentum were ground in a mill and passed through a 500 mesh sieve. The sieved material was stored at -20°C until use. Lycopene (Sigma L9879) for quantitative analysis was purchased from Sigma-Aldrich. Solvents and other reagents were analytical grade.
Soxhlet extraction. Soxhlet extraction of freeze-dried Lycopersicum esculentum was performed to determine the initial amount of lycopene present in the L. esculentum. A total of 5 g of powdered freeze-dried L. esculentum was extracted for 12 h using 150 ml of hexane. After extraction, the solvents were removed under a vacuum evaporator and the material was weighed. The dried extract was dissolved in 20 ml of methanol:hexane (1:2, v/v). A 20 μl aliquot of methanol: hexane solution was used for HPLC.
Recovery procedure. The recovery procedure of natural lycopene from Lycopersicum esculentum was composed of two steps. First, all carotenoids were extracted from L. esculentum by conventional solvent extraction using organic solvents that possess different polarities. A 100 g sample was weighed into a 3 liter glass tube with a glass filter bottom (50 mm x 1500 mm), and crude lycopene was extracted for 1 h with 1 liter of different organic solvents: hexane, ethanol, and ethyl acetate. The resulting crude lycopene extract was evaporated at reduced pressure to approximately 1% of the initial volume. We selected the extraction solvent showing the highest extraction efficiency for the next isolation procedure. Second, the crystallization of lycopene was carried out by adding suitable anti-solvent to the crude lycopene extract. After adding 100 ml of methanol as an anti-solvent, precipitation of lycopene from the mixture occurred within several minutes. The isolated lycopene was stored at -20 °C until further analysis. All of the lycopene recovery procedures were conducted within 2 h.
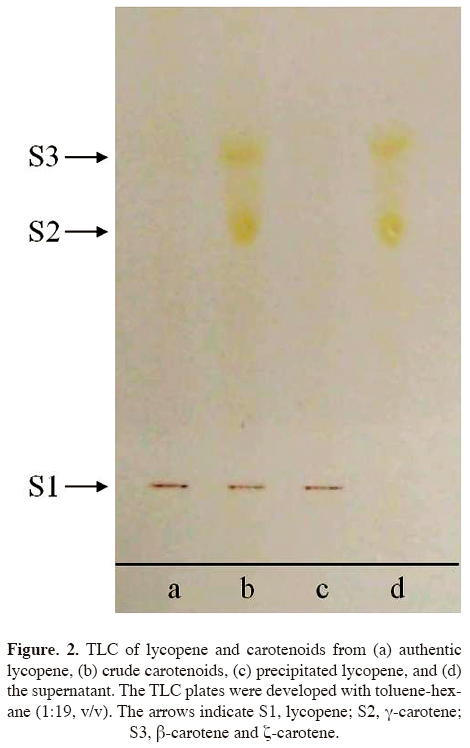
Thin-layer chromatography (TLC). To confirm the purity of the precipitated lycopene, TLC was conducted according to the method of Britton (2008) with slight modification. Briefly, Silica Gel 60 F254 (0.25mm, Merck, Darmstadt, Germany) activated at 110 °C for 10 min was used for the TLC plates. The crude carotenoids shown in figure 1A and the precipitated lycopene and supernatant shown in figure 1B were developed with toluene-hexane (1:19, v/v) on the TLC plates. Authentic lycopene (Sigma L9879) was also developed.
Analysis by high-performance liquid chromatography for the quantification of lycopene. The quantification analysis of the lycopene of the Soxhlet extract, solvent extract, and anti-solvent precipitate was performed using a Waters 600E HPLC system (USA) equipped with Waters 486 UV detector, as described by Vasapollo et al. (2004) with the minor modifications. The chromatographic analysis was conducted using a reverse-phase ZORBAX Eclipse plus C18 column (4.6 mm x 250 mm, Agilent) with 5 μm particles. The characterization of the lycopene extracts was performed in isocratic mode and the mobile phase used was methanol:THF (90:10; v/v). Lycopene samples were dissolved in 20 μl of a methanol:hexane (1:2, v/v) solution. Chromatographic separation of extracts was performed at a constant flow rate of 1 ml/min. Lycopene was detected at 472 nm. For quantitative analysis, standard lycopene (Sigma L9879) was also analyzed using the HPLC system under the same conditions.
To avoid exposure to light and oxygen, all experiments were conducted using dark amber glassware or transparent glassware wrapped with aluminum foil and filled with nitrogen gas. The nitrogen gas was obtained from a N2 generator (G4010E, Domnick Hunter, England; less than 100 ppm oxygen content). All isolation procedures were performed at least three times independently.
Results
]]> Isolation of lycopene from Lycopersicon esculentum. To isolate lycopene from Lycopersicon esculentum, conventional solvent extraction and anti-solvent precipitation were performed. Soxhlet extraction was conducted to determine the initial amount of lycopene in freeze-dried L. esculentum, which was 4.52 (± 0.18) mg/g. To determine the optimal solvent for the extraction of lycopene from L. esculentum, different solvents were tested, including hexane, ethyl acetate, and ethanol. The amounts of crude lycopene extracted from L. esculentum were 3.58, 4.39, and 1.25 mg/g, respectively. The most effective extraction solvent was ethyl acetate (Table 1). The extraction yields (X0) were 79.20%, 97.12%, and 27.65% by hexane, ethyl acetate, and ethanol, respectively, as determined from the initial amount of lycopene.
For further isolation of lycopene, we employed the anti-solvent precipitation method. Ethyl acetate (PI; 4.4) as an extraction solvent has a dissolving capacity for lycopene and for carotenoids and glycerides too. Anti-solvent extraction can selectively isolate carotenoids and glycerides from a mixture of lycopene, carotenoids, and glycerides. With regard to lycopene solubility, we selected methanol (PI; 5.1) as an anti-solvent. Lycopene not dissolved in methanol was precipitated and recovered as a quasi-crystalline solid form (3.50 mg) (Figure 1).
Purity of the precipitated lycopene. As figure 2 shows, on the TLC plate, the crude carotenoid fractions gave red (Rf = 0.14), orange (Rf = 0.6), and yellow (Rf = 0.72) spots, while the supernatant gave orange and yellow spots and, the lycopene precipitated with methanol, as an anti-solvent, gave a single red spot that was free of other carotenes as impurities. The Rf value of the red spot was the same of that of authentic lycopene (Figure 2). The orange and yellow spots represent ϒ-carotene and β- and ζ-carotene, respectively (Britton, 2008).
Chromatograms of lycopene. We used HPLC to confirm the lycopene recovery. Lycopene was identified by comparing the retention times of the pigment in the extraction mixture with those of their respective standard compounds (Sigma products). The HPLC chromatograms of extracts and anti-solvent fractionates are shown in figure 3. The UV spectra demonstrated maximum adsorption bands at 502, 472, and 448 nm. These results are similar to those reported by Davis et al. (2003). In our study, comparison of the chromatograms showed that the relative peak area of lycopene significantly increased after isolation by the anti-solvent fractionation. When the area of the lycopene peak was calculated and compared with standard compounds, the recovery rate of precipitated lycopene from crude ethyl acetate extract was 79.73%.
Discussion
]]> The most important source of lycopene is Lycopersicum esculentum and associated processed food products, in which lycopene constitutes more than 60% of the carotenoids present. Conventional methods for the extraction of carotenoids from many various sources have used pure solvents such as dichloromethane or a mixture of polar and non-polar solvents (e.g., hexane, acetone, and ethanol). Supercritical fluid extraction with the CO2 method has recently been proposed (Vasapollo et al., 2004). This extraction method provides an added advantage of using non-organic solvents, large-scale facilities and higher costs are associated with this procedure. Therefore, we adapted conventional solvent extraction techniques to develop a method that is low cost and does not require large-scale facilities for the preparative separation and purification of lycopene.Preliminary extractions from the raw material (hot wind-dried tomato powder) were performed for selection of effective solvents to extract lycopene. In these experiments, the conventional organic solvent extracted low levels of lycopene because the hardness of the dried tomato power prevented lycopene from dissolving in the organic solvent. Therefore, the freeze-drying process was adapted under vacuum in order to analyze dried soft tomato tissue. The final isolation of pure lycopene from the crude carotenoids mixture is generally achieved by chromatographic methods (HPLC, TLC, column chromatography; Choksi and Joshi, 2007). However, these protocols also require large-scale facilities and/or are time consuming processes for the large-scale preparative isolation of lycopene.
In the last decade, salting-out as a means to induce super saturation has been drawing greater attention. In this method, a secondary solvent known as an anti-solvent or precipitant is added to the solution resulting in the reduction of the solubility of the solute in the original solvent and consequently, the generation of a super-saturation driving force (Nowee et al., 2008). To our knowledge, no previous study had attempted to adapt the conventional anti-solvent precipitation technique for the isolation of lycopene. This was the objective of the current study.
In this study, the total lycopene content in freeze-dried tomato was found to be 4.52 ± 0.18 mg/g of dry matter (n = 3), as determined by Soxhlet extraction with hexane. The lycopene extraction rates for dried tomato powder by solvent extraction using different solvents are shown in Table 1. Extractions performed with ethyl acetate resulted in higher yields of lycopene (4.39 ± 0.27 mg/g; 97.12%).
We contemplated the adaption of anti-solvent precipitation to effectively isolate lycopene from crude extract. An anti-solvent used to isolate lycopene was selected in consideration of the distribution coefficient ratio of the concentration of solute dissolved in the anti-solvent versus the concentration of solute dissolved in the extraction solvent. The equation of the correlation coefficient of distribution is as follows:
Kow = C octanol / C water ..... (1)
where Kow is the partition coefficient, C octanol, is the concentration of solute dissolved in octanol, and C water is the concentration of solute dissolved in water.
The anti-solvent derived from the equation of the partition coefficient was methanol, which can mix with ethyl acetate (0.66 < Kow < 0.73) to remove residue solvent in tomato concentrate and methanol (Kow < 0.77). Methanol has a high partition coefficient value to dissolve carotenoids and glycerides. Methanol showed a low partition coefficient value against lycopene in organic solvents (3.17 < Kow), which in turn can precipitate lycopene dissolved in the solvent extraction.
As seen figure 1, methanol as an anti-solvent is added to the crude lycopene solution. This results in a reduction in the solubility of the lycopene in the original solvent, consequently, generating a super-saturation driving force. The percentage of the lycopene yield was 79.73% for anti-solvent precipitation from a lycopene ethyl acetate mixture. These results implied that anti-solvent precipitation using organic solvent as an anti-solvent is a far more efficient extraction method than chromatographic purification.
To confirm the purity of the precipitated lycopene, TLC was performed (Figure 2). Judging from a previous report (Britton, 2008), the single red spot with no other carotenoids impurities on TLC can be identified as lycopene, as the precipitated and authentic lycopene have the same Rf value.
]]> These results indicate that our simple protocol is very effective for obtaining high-purity lycopene. The analysis of lycopene in food products may be carried out by HPLC or by direct spectrophotometry, which is the standard method for fast quantification of lycopene (Baysal et al., 2000). In this paper, the crude extracts and pure lycopene of Lycopersicon esculentum were used for pigment analysis by HPLC. The spectra for the dried tomato samples using this method, for which lycopene constitutes the predominant carotenoid content, exhibited apparent absorption maxima at 502, 472, and 448 nm. These maxima are almost corresponded with the maxima at 505, 475, and 445 observed for lycopene in organic solvents such as hexane. The maxima are also located at or near the same wavelengths observed for lycopene when it is extracted into aqueous solutions of dilute detergents (Davis et al., 2003). The previous reported lycopene recovery rates of Baysal et al. (2000), Cadoni etal. (2000), Shi et al. (2009) and Vasapollo et al. (2004) using the supercritical CO2 extraction method were 53.93%, 87%, 56.8%, and 60%, respectively. In our results, the recovery rate of lycopene was 77.43% by conventional solvent extraction and anti-solvent precipitation using organic solvent as an anti-solvent.In conclusion, the conventional solvent extraction and anti-solvent processes are feasible methods to rapidly (2 h) purify lycopene from L. esculentum. These results demonstrate that this lycopene isolation method is suitable for the large-scale industrial preparative separation and purification of lycopene.
Literature cited
Baysal T., Ersus S. and Starmans D.A.J. 2000. Supercritical Co2 extraction of β-carotene and lycopene from tomato paste waste. Journal of Agricultural and Food Chemistry 48:5507-5511. [ Links ]
Britton G. 2008. TLC of carotenoids. In: Waksmundzka-Hajnos M., Sherma J. and Kowalska T. Eds. Thin Layer Chromatography in Phytochemistry, pp. 543-573, CRC Press, New York. [ Links ]
Cadoni E., De Giorgi M. R., Medda E. and Poma G. 2000. Supercritical Co2 extraction of lycopene and β-carotene from ripe tomatoes. Dyes and Pigments 44:27-32. [ Links ]
]]>Choksi P.M. and Joshi V.Y. 2007. A review on lycopene - Extraction, purification, stability and applications. International Journal of Food Properties 10:289-298. [ Links ]
Davis A.R., Fish W.W. and Perkins-Veazie P. 2003. A rapid spectrometric method for analyzing lycopene content in tomato and tomato products. Postharvest Biology and Technology 28:425-430. [ Links ]
Hakala S.H. and Heinonen I.M. 1994. Chromatographic purification of natural lycopene. Journal of Agricultural and Food Chemistry 42:1314-1316. [ Links ]
Le Marchand L., Yoshizawa C.N., Kolonel L.N., Hankin J.H. and Goodman M.T. 1989. Vegetable consumption and lung cancer risk: a population-based case-control study in Hawaii. Journal of National Cancer Institute 81:1158-1164. [ Links ]
Lee H.E., Lee M.J., Kim W.S., Jeong M.Y., Cho Y.S. and Choi G.J. 2011. In-line monitoring and interpretation of an indomethacin anti-solvent crystallization process by near-infrared spectroscopy (NIRS). International Journal of Pharmaceutics 420:274-281. [ Links ]
]]>Leoni C. 1993. I derivati industriali del pomodoro. In: Stazione sperimentale per l'industria delle conserve alimentary. Ed. Collana di Monografie Tecnologiche. Vol. 7. Parma. [ Links ]
Liu B., Huang X.Y., Di D.L. and Zhang B.C. 2008. Study on quality standard for lycopene soft gels. Food Science and Technology 34:243-245. [ Links ]
Miguel F., Martín A., Gamse T. and Cocero M.J. 2006. Supercritical anti solvent precipitation of lycopene: Effect of the operating parameters. Journal of Supercritical Fluids 36:225-235. [ Links ]
Nowee S.M., Abbas A. and Romagnoli J.A. 2008. Antisolvent crystallization: Model identification, experimental validation and dynamic simulation. Chemical Engineering Science 63:5457-5467. [ Links ]
Ó'Ciardhá C.T., Frawley P.J. and Mitchell N.A. 2011. Estimation of the nucleation kinetics for the anti-solvent crystallisation of paracetamol in methanol/water solutions. Journal of Crystal Growth 328:50-57. [ Links ]
]]>O'Grady D., Barrett M., Casey E. and Glennon B. 2007. The effect of mixing on the metastable zone width and nucleation kinetics in the anti-solvent crystallization of benzoic acid. Chemical Engineering Research and Design 85:945-952. [ Links ]
Sadler G., Davis J. and Dezman D. 1990. Rapid extraction of lycopene and β-carotene from reconstituted tomato paste and pink grapefruit homogenates. Journal of Food Science 55:1460-1461. [ Links ]
Shi J., Maguer M.L., Kakuda Y., Liptay A. and Niekamp F. 1999. Lycopene degradation and isomerization in tomato dehydration. Food Research International 32:15-21. [ Links ]
Shi J., Yi C., Xue S.J., Jiang Y., Ma Y. and Li D. 2009. Effects of modifiers on the profile of lycopene extracted from tomato skins by supercritical Co2. Journal of Food Engineering 93:431-436. [ Links ]
Vasapollo G., Longo L., Rescio L. and Ciurlia L. 2004. Innovative supercritical Co2 extraction of lycopene from tomato in the presence of vegetable oil as co-solvent. Journal of Supercritical Fluids 29: 87-96. [ Links ]
]]>