Convenient Reductive Amination of Aldehydes by NaBH4/Cation Exchange Resin
Davood Setamdideh* and Farhad Sepehraddin
Department of Chemistry, College of Sciences, Mahabad Branch, Islamic Azad University, Mahabad, Iran. d.setamdideh@iau-mahabad.ac.ir; davood.setamdideh@gmail.com
Received June 10, 2013. ]]> Accepted September 18, 2013.
Abstract
Different secondary amines have been synthesized by reductive amination a variety of aldehydes and anilines with NaBH4/DOWEX(R)50WX8 as reducing system in THF at room temperature in high to excellent yields of products (85-93%).
Keywords: NaBH4, DOWEX(R)50WX8, Reductive amination, Carbonyl compounds.
Resumen
Se sintetizaron diversas aminas secundarias por aminacion reductiva de una variedad de aldehidos y anilinas empleando como sistema reductor NaBH4/DOWEX(R)50Wx8 en THF a temperatura ambiente con rendimientos de altos a excelentes.
Palabras clave: NaBH4, DOWEX(R)50WX8, aminacion reductiva, aminas secundarias.
]]> Introduction
Amines are important in drugs and in active pharmaceutical intermediates. Amines can be achieved by reduction of nitro, cyano, azide, carboxamide derivatives or alkylation of amines (using alkyl halides or sulfonates). On the other hand, direct reductive amination (DRA) is other approach which offers significant advantages. Reductive amination reaction in a single operation can be performed by some non-borohydride reducing system [1]. Also, a variety of borohydrides reducing systems and reducing agents have been used for DRA such as Lewis acid /THF then NH3BH3 [2], NaBH4/cellulose sulfuric Acid/EtOH [3], NaBH4-silica chloride [4], NaBH4-silica-gel-supported sulfuric acid [5], [Zr(BH4)2(Ph3P)2] [6], 2-picoline-BH3 [7], 5-ethyl-2-methylpyridine borane/ AcOH [8], sodium tetrakis [3,5-di (trifluoromethyl)phenyl]borate (NaBArF4)/ hydrioiridium(III) complex/H2O [9], NaBH4-H3PW12O40 [10], NaBH4/guanidine hydrochloride/H2O [11], Cu(PPh3)2BH4, MeOH/NH2SO3H [12], NaBH4/ Brønsted acidic ionic liquid (1-butyl-3-methyl imidazolium tetrafluoroborate [(BMIm)BF4]) [13], NaBH4/ LiClO4/diethyl ether [14], zinc borohydride V-methyl piperidine [15], dibutylchlorotin hydride complex [16], NaBH4-PhCO2H, NABH4/ρ-TsOH [17], Zr[(BH4)2(Cl)2(dabco)2] [18], benzylamine-BH3 [19], NaBH4-NiCl2 [20], (t-Bu)(Me)(i-Pr)N-BH3 [21], silicagel-Zn(BH4)2 [22], Ti(O-i-Pr)4-NaBH4 [23], NaBH4-wet-clay-microwave [24], NaBH4-H2SO4 [25], NaBH4/Mg(ClO4)2 [26], 2,6-dibo-rane-methanol [27], NaBH3CN [28] and NaBH4/X(OH)3 (X: B, Al, Ga) [29]. These methods have many advantages and some disadvantages such as toxic byproducts, excess amount of reagents, using expensive reagents, prolonged reaction time, higher reaction temperature, acidic conditions and so on. Therefore, there is interest in developing synthesis of secondary amines under new systems. So, in continuing our efforts for the development of new reducing systems [30], we have carried out extensive re-examination of the DRA reaction to develop a convenient system by NaBH4/DOWEX(R)50WX8 as reducing system in THF at room temperature.
Results and Discussion
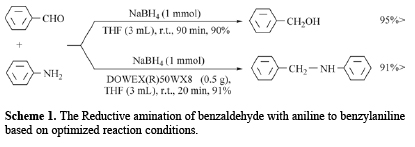
Recently, we have reported that the DOWEX(R)50WX4 (low price cation exchange resin, strong acid) can be used as recyclable catalyst for the regioselective synthesis of Oximes by NH2OH.HCl/DOWEX(R)50WX4 system [31], reduction of a variety of carbonyl compounds such as aldehydes, ketones, α-diketones, acyloins and α,β-unsaturated carbonyl compounds to their corresponding alcohols by NaBH4/DOWEX(R)50WX4 system [32] and the synthesis of cyanohydrins by NaCN/ DOWEX(R)50WX4 [33]. In this context, we now wish to report an efficient, facile one-pot reductive mono-V-alkylation of anilines using aldehydes by NaBH4/DOWEX(R)50WX8 as reducing system in THF at room temperature. The model reaction has been selected by reductive amination of benzaldehyde with aniline. This reaction was carried out in different solvents, different amounts of the NaBH4 and DOWEX(R)50WX8 for the selection of appropriate conditions at room temperature. Among the tested different solvents, the reaction was most facile and proceeded to give the highest yield in THF. The optimization reaction conditions showed that using 1 molar equivalents of NaBH4 and 0.5 g of DOWEX(R)50WX8 in THF were the best conditions to complete the reductive amination of benzaldehye (1 mmol) and aniline (1 mmol) to Ν-benzylaniline (Table 1, Entry 1). Our observation reveals that reductive ami-nation completes within 20 min with 91% yields of product as shown in scheme 1.

The efficiency of this protocol was further examined by using various structurally different aldehydes and anilines. In this approach, the corresponding secondary amines were obtained in excellent yields (88-93%) and within appropriate times (2045 min) as shown in Table 2.
Imine formation is usually the rate-determining step for reductive aminations therefore addition of co-reactants is desirable. It is notable, in the absence of DOWEX(R)50WX8, imine formation does not occur and the aldehyde is reduced to benzyl alcohol as shown in scheme 1. Since the insolubility of DOWEX(R)50WX8 in THF, the reaction takes place under heterogeneous conditions. The mechanism for the influence of DOWEX(R)50WX8 is not clear, but as shown in scheme 2, we think that with the addition of DOWEX(R)50WX8 (as cation exchange resin, strong acid) to the reaction mixture (substrate & NaBH4 in THF), Na+ with H+ slowly being changed and hydrogen ion concentration increase (scheme 2, step II). Therefore, carbonyl group can be protonated, thus it is more readily to attack the nitrogen of aniline for the imine formation (scheme 2, step III). Also, this process causes to protonate nitrogen of the imine as intermediate (scheme 2, step V). Consequently the imine intermediate become more reducible by reducing agents (scheme 2, step VI). On the other hands, we observed sodium borohydride is decomposed thus, THF-BH3 complex is formed and hydrogen gas slowly is liberated in situ (scheme 2, step IV) [21]. Hydrogen gas generation seems to be directly related to concentration of H+ (scheme 2, step II & Table 1, entries 7) and the solubility of the NaBH4 (scheme 2, step I) in the reaction solvent.
]]>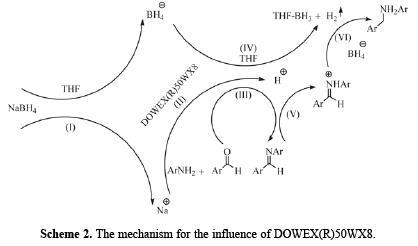
The synergistically generated molecular hydrogen combines with more easily hydride attack to protonated imine intermediate, thus accelerates the rate of reduction reaction (scheme 2, step VI).
The reductive amination of cinnamaldehyde with 1 molar equivalents of aniline and 4-methyl aniline by 1 molar equiva lents of NaBH4 in the presence of 0.5 g of DOWEX(R)50WX8 was carried out exclusively in 1,2-reduction manner within 20 minutes at room temperature as shown in scheme 3. In these reactions the corresponding cinnamylanilines were obtained in 90-91% yields (entries 22-23).

The reductive amination of aldehyde in the presence of ketones was 100%. So, this is a general trend for the reductive amination of various aldehydes in the presence of ketones (scheme 4).
]]>
We have also checked the reusability of the catalyst using the recovered DOWEX(R)50WX8 from the reductive amination reaction. It was observed that recovered catalyst could be satisfactorily used for the second run without regeneration. Whereas, third run of the recovered catalyst leads to poor yields and longer reaction times. After regeneration of DOWEX(R)50WX8 (It was stirred in HCl 10-5% for 30-60 minutes, then washed with distillated water), the reduction reaction has been carried out like the first run in the presence of the regenerated DOWEX(R)50WX8.
The products were determined from the 1H-chemical shift of the CH2 group which appeared around 4.22-4.68 ppm as a singlet. Also the NH stretching frequency in FT-IR spectrum appeared around 3380-3427 cm 'as shown in table 2.
Experimental
General
All substrates and reagents were purchased from commercially sources with the best quality and used without further purification. IR and 1H NMR spectra were recorded on PerkinElmer FT-IR RXI, 100 and 400 MHz Bruker spectrometers, respectively. The products were characterized by their 1H NMR or IR spectra and comparison with authentic samples (melting or boiling points). Organic layers were dried over anhydrous sodium sulfate. All yields referred to isolated pure products. TLC was applied for the purity determination of substrates, products and reaction monitoring over silica gel 60 F254 aluminum sheet.
Reductive amination of benzaldehyde and aniline with NaBH4/DOWEX(R)50WX8, A typical procedure
In a round-bottomed flask (10 mL) equipped with a magnetic stirrer, a solution of benzaldehyde (0.106 g, 1 mmol) , aniline (0.093 g, 1 mmol) and DOWEX(R)50WX8 (0.5 g) in THF (3 mL) was prepared. The resulting mixture was stirred for 5 min at room temperature. Then the NaBH4 (0.036 g, 1 mmol) was added to the reaction mixture and stirred at room temperature. TLC monitored the progress of the reaction (eluent; CCl4/Ether: 5/2). The reaction was filtered after completion within 20 min. Evaporation of the solvent and short column chromatography of the resulting crude material over silica gel (eluent; CCl4/ Ether: 5/2) afforded the A-benzylaniline (0.l66 g, 91% yield, Table 2, entry 1).
]]>Conclusions
In this investigation, we have shown that the NaBH4/ DOWEX(R)50WX8 is suitable for the reductive amination of a variety of aldehydes and anilines to their corresponding secondary amines in high to excellent yields. Reduction reactions were carried out with 1 molar equivalents of NaBH4 in the presence of 0.5 g DOWEX(R)50WX8 in THF at room temperature. High efficiency of the reductions, shorter reaction times and easy work-up procedure makes as an attractive new protocol for reductive amination of aldehydes and it could be a useful addition to the present methodologies.
Acknowledgments
The authors gratefully appreciated the financial support of this work by the research council of Islamic Azad University branch of Mahabad.
References
1. a) Nador, F.; Moglie, Y.; Ciolino, A.; Pierini, A.; Dorn, V.; Yus, M. Tetrahedron Lett. 2012, 53, 3156-3160. [ Links ] b)Cano, R.; Yus, M.; Ramon, D. J. Tetrahedron 2011, 67, 8079-8085. [ Links ] c) Connor, D.; Lauria, A.; Bondi, S. P.; Saba, S. Tetrahedron Lett. 2011, 52, 129-132. [ Links ] d) Azizi, N.; Khajeh Amiri, A. R.; Ghafuri, H.; Saidi, M. R.; Bolourtchian, M. J. Iran. Chem. Soc. 2010, 7, 428-431. [ Links ] e) Patel, J. P.; Li, A. H.; Dong, H.; Korlipara, V. L.; Mulvihill. M. J. Tetrahedron Lett. 2009, 50, 5975-5977. [ Links ] f) Bhor, M. D.; Bhanushali, M. J.; Nandurkar, N. S.; Bhanage, B. M. Tetrahedron Lett. 2008, 49, 965-969. [ Links ] g) Byun, E.; Hong, B.; De Castro, K. A.; Lim, M.; Rhee, H. J. Org. Chem. 2007, 72, 9815-9817. [ Links ] h) Gnanamgari, D.; Moores, A.; Rajaseelan, E.; Crabtree, R. H. Organometallics 2007, 26, 1226-1230. [ Links ] i) Robichaud, A.; Ajjou, A. N. Tetrahedron Lett. 2006, 47, 3633-3636. [ Links ] j) Nugent, T. C.; Ghosh, A. K.; Wakchaure, V. N.; Mohanty, R. R. Adv. Synth. Catal. 2006, 348, 1289-1299. [ Links ] k) Matsushita, T. I. K.; Shinozawa, J.; Yada, S.; Takagi, Y. Tetrahedron 2005, 61, 2105-2109. [ Links ] l) Cho, B. T.; Kang, S. K. Tetrahedron 2005, 61, 5725-5734. [ Links ] m) Itoh, T.; Nagata, K.; Miyazaki, M.; Ishikawa, H.; Kurihara, A.; Ohsawa, A. Tetrahedron 2004, 60, 6649-6655. [ Links ] n) Itoh, T.; Nagata, K.; Kurihara, A.; Miyazaki, M.; Ohsawa, A. Tetrahedron Lett. 2002, 43, 3105-3108. [ Links ] o) Chen, B. C.; Sundeen, J. E.; Guo, P.; Bednarz, M. S.; Zhao, R. Tetrahedron Lett. 2001, 42, 1245-1246. [ Links ] p) Apodaca, R.; Xiao, W. Org. Lett. 2001, 3, 1745-1748. [ Links ] q) Chandrasekhar, S.; Raji Reddy, C.; Ahmed, M. Synlett 2000, 1655-1657. [ Links ] r) Suwa, T.; Sugiyama, E.; Shibata, I.; Baba, A. Synlett 2000, 556-558. [ Links ] s) Suwa, T.; Shibata, I.; Nishino, K.; Baba, A. Org. Lett. 1999, 1579-1581. [ Links ] t) Pienemann, T.; Schafer, H. J. Synthesis 1987, 11, 1005-1007. [ Links ]
2. Ramachandran, V.; Gagare, P. D.; Sakavuyi, K.; Clark, P. Tetrahedron Lett. 2010, 51, 3167-3169. [ Links ]
3. Heshmatollah, A.; Zakieh, T. Bull. Korean Chem. Soc. 2010, 31, 1927-1930. [ Links ]
4. Alinezhad, H.; Tajbakhsh, M.; Hamidi, N. Turk. J. Chem. 2010, 34, 307-312. [ Links ]
5. Alinezhad, H.; Tajbakhsh, M.; Zare, M. Synth. Commun. 2009, 39, 2907-2916. [ Links ]
6. Firouzabadia, H.; Iranpoora, N.; Alinezhadb, H. J. Iran. Chem. Soc. 2009, 6, 177-186. [ Links ]
7. Sato, S.; Sakamoto, T.; Miyazawa, E.; Kikugawa, Y. Tetrahedron 2004, 60, 7899-7906. [ Links ]
8. Burkhardt, E. R.; Coleridge, B. M. Tetrahedron Lett. 2008 49, 5152-5155. [ Links ]
9. Lai, R. Y.; Lee, C. I.; Liu, S. T. Tetrahedron 2008, 64, 1213-1217. [ Links ]
10. Heydari, A.; Khaksar, S.; Akbari, J.; Esfandyari M.; Pourayoubia, M.; Tajbakhsh, M. Tetrahedron Lett. 2007, 48, 1135-1138. [ Links ]
11. Heydari, A.; Arefi, A.; Esfandyari, M. J. Mol. Catal. A: Chem. 2007, 169-172. [ Links ]
12. Bhanushali, M. J.; Nandurkar, N. S.; Bhor, M D.; Bhanage, B. M. Tetrahedron Lett. 2007, 48, 1273-1278. [ Links ]
13. Reddy, P. S.; Kanjilal, S.; Sunitha, S.; Prasad, R. B. N. Tetrahedron Lett. 2007, 48, 8807-8810. [ Links ]
14. Saidia, M. R.; Brownb, R. S.; Ziyaei-Halimjani, A. J. Iran. Chem. Soc. 2007, 4, 194-198. [ Links ]
15. Alinezhad, H.; Tajbakhsh, M.; Zamani, R. Synlett 2006, 431-434. [ Links ]
16. Kato, H.; Shibata, I.; Yasaka, Y.; Tsunoi, S.; Yasuda, M.; Baba, A. Chem. Commun. 2006, 4189-4191. [ Links ]
17. Cho, B. T.; Kang, S. K. Tetrahedron 2005, 61, 5725-5734. [ Links ]
18. Firouzabadi, H.; Iranpoor, N.; Alinezhad, H. Bull. Chem. Soc. Jpn. 2003, 76, 143-151. [ Links ]
19. Peterson, M. A.; Bowman, A.; Morgan, S. Synth. Commun. 2002, 32, 443-448. [ Links ]
20. Saxena, I.; Borah, R.; Sarma, J. C. J. Chem. Soc. Perkin Trans. 1 2000, 503-504. [ Links ]
21. Brown, H. C.; Kanth, J. V. B.; Dalvi, P. V.; Zaidlewicz, M. J. Org. Chem. 1999, 64, 6263-6274. [ Links ]
22. Ranu, B. C.; Majee, A.; Sarkar, A. J. Org. Chem. 1998, 63, 370-373. [ Links ]
23. Neidigh, K. A.; Avery, M. A.; Williamson, J. C.; Bhattacharyya, S. J. Chem. Soc. Perkin Trans. 1 1998, 2527-2532. [ Links ]
24. Varma, R. S.; Dahiya, R. Tetrahedron 1998, 54, 6293-6298. [ Links ]
25. Abdel-Magid, A. F.; Carson, K. G.; Harris, B. D.; Maryanoff, C. A.; Shah, R. D. J. Org. Chem. 1996, 61, 3849-3869. [ Links ]
26. Brussee, J.; van Benthem, R. A. T. M.; Kruse, C. G.; van der Gen, A. Tetrahedron Asymmetry 1990, 1, 163-166. [ Links ]
27. Nose, A.; Kudo, T. Chem. Pharm. Bull. 1980, 34, 4817-4820. [ Links ]
28. Lane, C. F. Synthesis 1975, 135-146. [ Links ]
29. a) Setamdideh, D.; Hasani, S.; Noori, S. J. Chin. Chem. Soc. 2013, 60, 1267-1271. [ Links ] b) Pourhanafi, S.; Setamdideh, D.; Khezri, B. Orient. J. Chem. 2013, 29, 709-712. [ Links ]
30. a) Setamdideh, D.; Rafigh, M. E-J. Chem. 2012, 4, 2338-2345. [ Links ] b) Setamdideh, D.; Rahmatollahzadeh, M. J. Mex. Chem. Soc. 2012, 56, 169-175. [ Links ] c) Setamdideh, D.; Ghahremani, S. S. Afr. J. Chem. 2012, 65, 91-97. [ Links ] d) Setamdideh, D.; Khezri, B.; Rahmatollahzadeh, M. J. Serb. Chem. Soc. 2013, 78, 1-13 e) Mohamadi, [ Links ] M.; Setamdideh, D.; Khezri, B. Org. Chem. Inter. 2013, 2, doi:10.1155/2013/127585. [ Links ] f) Setamdideh, D.; Khaledi, L. S. Afr. J. Chem. 2013, 66, 150-157. [ Links ] g) Kamari, R.; Setamdideh, D. Orient. J. Chem. 2013, 29, 497-499. [ Links ] h) Latifi Mamaghani, E.; Setamdideh, D. Orient. J. Chem. 2013, 29, 953-955. [ Links ]
31. Setamdideh, D.; Khezri, B.; Esmaeilzadeh, S. J. Chin. Chem. Soc. 2012, 59, 1119-1124. [ Links ]
32. Setamdideh, D.; Karimi, Z.; Alipouramjad, A. J. Chin. Chem. Soc. 2013, 60, 590-596. [ Links ]
33. Sophighaderi, S.; Setamdideh, D. Orient. J. Chem. 2013, 29, 1135-1137. [ Links ]
]]>