
Citroflavonoides como posible alternativa en el tratamiento de la diabetes y sus complicaciones
Citroflavonoids as an alternative for the treatment of diabetes and its complications
González-Sánchez Avel,1 Cabañas-Wuan Ángel, 1 Arana-Argáez Víctor,1 Hernández-Núñez Emanuel,2 Ortiz-Andrade Rolffy1
1 Laboratorio de Farmacología, Facultad de Química, Universidad Autónoma de Yucatán.
2 Laboratorio de Espectrometría de Masas, Facultad de Química, Universidad Autónoma de Yucatán.
]]>Correspondencia
Dr. Rolffy Rubén Ortiz Andrade
Laboratorio de Farmacología, Facultad de Química,
Universidad Autónoma de Yucatán
Calle 41 No. 421 x 26 y 28, Col. Industrial,
CP 97150, Mérida, Yucatán, México
Tel: (99) 99225711, Ext. 119
e-mail: rolffy.ortiz@uady.mx
]]> Fecha de recepción: 31 de mayo de 2011.
Resumen
Los flavonoides constituyen una de las subfamilias de polifenoles naturales, a las que la comunidad científica ha dedicado más atención en los últimos años, principalmente por sus efectos antioxidantes. Debido a su gran diversidad estructural, abundancia en la naturaleza, su demostrada ubicuidad y sus múltiples propiedades farmacológicas observadas experimentalmente, junto con su amplia presencia en numerosos remedios de la medicina tradicional, representan una importante alternativa terapéutica para el descubrimiento de nuevos agentes farmacológicos para el tratamiento de enfermedades crónicas de elevada prevalencia como es el caso de la diabetes mellitus.
Palabras clave: diabetes mellitus, flavonoides, antioxidantes.
Abstract
Flavonoids represent natural polyphenols subfamilies, which the scientific community has devoted more attention in recent years, principally for its antioxidant properties. Because of their structural diversity, abundance in nature, demonstrated ubiquity and pharmacological properties observed in experimentation, besides their wide presence in traditional medicine, represent an important therapeutic alternative in the discovery of new pharmacological agents, for the treatment of chronic diseases, as in the case of diabetes mellitus.
]]> Key words: diabetes mellitus, flavonoids, antioxidants.
Introducción
En 1930, Rusznyak y Szent-György aislaron un nuevo compuesto a partir de la naranja dulce (Citrus sinensis), al cual denominaron Vitamina P.1 Actualmente, dicha sustancia se conoce como flavonoide. Desde aquel entonces, estos compuestos han llamado la atención de muchos investigadores, sobre todo, con el descubrimiento de la llamada "Paradoja Francesa",2 fenómeno asociado al consumo de vino tinto, fuente importante de flavonoides.3
En cuanto a su estructura química, los flavonoides representan un grupo de compuestos polifenólicos que poseen un núcleo básico de flavano con dos anillos aromáticos (A y B), los cuales se encuentran interconectados por un anillo heterocíclico de tres átomos de carbono (anillo C), el cual a su vez, puede estar unido al anillo B en C-2 (flavona), en C-3 (isoflavona) o en C-4 (neoflavona). Debido a la diversidad de modificaciones que pueden presentarse en los tres anillos, principalmente el C-2, los flavonoides representan uno de los grupos más amplios de compuestos, así como también la clase más diversa de metabolitos secundarios en plantas.4 Prueba de ello es que a la fecha se han identificado más de 4,000 flavonoides en frutas, vegetales, granos, cortezas, raíces, tallos y flores, así como en el té verde y en el vino (figura 1).5,6

El creciente interés en los flavonoides se debe principalmente a su amplia actividad farmacológica y a sus importantes efectos antioxidantes. Además, presentan una gran variedad de propiedades entre las que se destacan el impacto sobre la regulación del crecimiento celular y la inducción de enzimas de destoxificación como las monooxigenasas dependientes del citocromo P-450. Asimismo, se ha comprobado su potente capacidad de inhibición in vitro de la oxidación de las lipoproteínas de baja densidad (LDL) por los macrófagos y reducir la citotoxicidad de las LDL oxidasas. Por otra parte, pueden unirse a los polímeros biológicos como: enzimas, transportadores de hormonas y ADN; formar quelatos con iones metálicos transitorios tales como Fe2+, Cu2+, Zn2+; catalizar el transporte de electrones y depurar radicales libres. Es por ello que, debido a este hecho, se han descrito efectos protectores en patologías tales como DM, cáncer, cardiopatías, infecciones víricas, úlcera estomacal y duodenal, e inflamaciones.7-9
Presencia de los flavonoides en la naturaleza
Los flavonoides se encuentran ampliamente distribuidos en el reino vegetal, encontrándose en frutas, verduras, semillas y flores, así como en productos como la cerveza, vino, té verde, té negro y soya.10,11
]]> En nuestra cultura nutrimental contamos con alimentos ricos en flavonoides tales como: la fresa, naranja ácida, uvas negras, toronjas, espinacas, remolacha, pimientos, cebolla, aguacate, berenjena, plátano, brócoli y coliflor, entre una lista bastante extensa.10 Los principales flavonoides presentes en la dieta humana se presentan en la tabla 1.1,2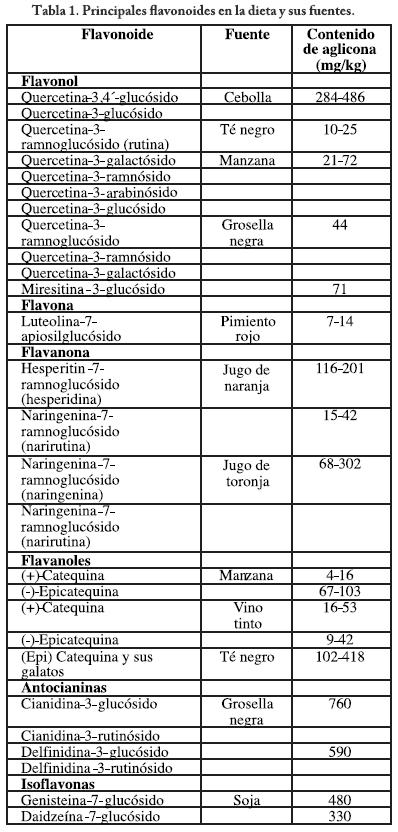
Características estructurales de los citroflavonoides
Los flavonoides comprenden un grupo de compuestos polifenólicos ampliamente distribuidos en la naturaleza. Existen 13 subclases de flavonoides con más de 5,000 compuestos, todos presentando un esqueleto hidrocarbonado del tipo C6-C3-C6 derivado del ácido shikímico y de tres residuos de acetato.13-15
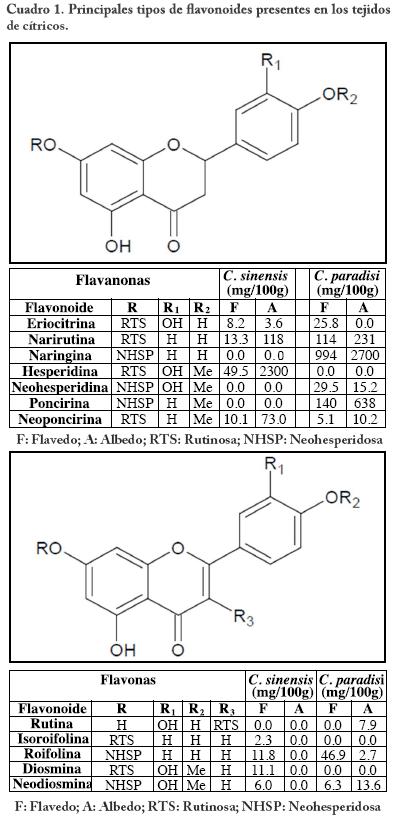
Las plantas del género Citrus se caracterizan por producir un amplio rango de constituyentes flavonoides, principalmente los del tipo flavanona, flavona y del tipo flavona polimetoxiladas tanto en la porción del flavedo (epicarpio) como en el albedo (tejido blanco esponjoso) (Cuadro 1).16
Actividades biológicas y farmacológicas de los citroflavonoides
Los flavonoides han presentado diversas actividades biológicas y farmacológicas a nivel experimental, entre las que se destacan: antioxidante,16 antitumoral,17,18 antiviral anti-H1N1,19 tripanomicida y lehismanicida,20,21 en cuanto a las actividades farmacológicas se han reportado: antidiabética, hipoglucemiante y antihiperglucemiante,22-24 antiinflamatoria,25 antitrombótica,25 hepatoprotectora,26-29 ansiolítica y anticonvulsivante,30,31 antiamiloidogénica,32 neuroprotectora,33 antilipidémica,34,35 y antihipertensiva.36,37 Por otra parte, han demostrado experimentalmente la inhibición de enzimas como la topoisomerasa IV,38 xantina oxidasa,39 aldosa reductasa,40 α-glucosidasa y amilasa24 transcriptasa inversa del virus de inmunodeficiencia humana (VIH),41 proteína quinasa C,42 tirosina quinasa,43 calmodulina,44 hexoquinasa,45 glucosa-6-fosfatasa,46 fosfolipasa A247 y 11β-hidroxiesteroide deshidrogenasa tipo I,48 sólo por mencionar algunas.
]]> Diabetes mellitusLa Asociación Americana de Diabetes (ADA, por sus siglas en inglés), define a la Diabetes Mellitus (DM) como un grupo de enfermedades, caracterizada por hiperglucemia, la cual es resultado de un defecto en la secreción y/o acción de la insulina. Esta hiperglucemia crónica se asocia al daño pancreático a largo plazo, defunción y/o fallo de diferentes órganos, especialmente ojos, riñones, nervios, corazón así como de venas y arterias.49
La fisiopatología de la DM se relaciona con dos eventos perfectamente identificables: 1) la destrucción autoinmune de las células β-pancreáticas con la consecuente deficiencia en la secreción de insulina, y 2) la deficiente acción de la misma sobre los tejidos periféricos, o bien, un efecto combinado de estas dos características. La base de las anormalidades en el metabolismo de carbohidratos, lípidos y proteínas en la Diabetes Mellitus tipo 2 (DM2) es aceptada como evento primario en el desarrollo de la resistencia a la insulina en los tejidos periféricos y como evento secundario a los defectos asociados a una carencia relativa de secreción de dicha hormona. La secreción inadecuada de insulina, así como, los defectos en la acción de la misma, comúnmente coexisten en el mismo paciente, y frecuentemente no está claro, si una o ambas, son la causa primaria de la hiperglicemia (Figura 2).49,50-5
Terapia farmacológica de la DM
En individuos con DM2, una terapia común comienza con un esquema dietético con consumo bajo en carbohidratos y ejercicio, seguido de agentes antidiabéticos orales (ADO's), estos últimos se usan cuando no es posible alcanzar un adecuado control tras un periodo razonable de intervención en los hábitos y estilo de vida. Estos fármacos están representados por cinco familias distribuidos en tres grupos farmacológicamente bien definidos, y ejercen su efecto sobre diferentes factores fisiopatológicos que contribuyen al desarrollo de la enfermedad. Por otro lado, la terapia con insulina ha sido considerada como la última opción terapéutica cuando la dieta, el ejercicio y los ADO's orales han fallado en el control glucémico (Figura 3).55-66
a) Secretagogos de insulina
Son agentes que incrementan la secreción de insulina en las células β-pancreáticas mediante bloqueo de los canales de potasio sensibles a ATP (KATP), a través de la unión a receptores específicos a estos canales. Se encuentran disponibles agentes tanto de acción rápida como acción lenta. Los dos principales grupos de secretagogos de insulina empleados son:55-60
• Sulfonilureas (SU, por ejemplo glibenclamida)
• Meglitinidas (MG, por ejemplo repaglinida)
b) Antihiperglucemiantes
]]> Son fármacos que impiden el aumento de los niveles plasmáticos de glucosa, inhibiendo competitivamente a enzimas que se encuentran en el borde en cepillo del intestino delgado, las cuales son responsables de la hidrólisis de oligosacáridos y disacáridos a monosacáridos, más fáciles de absorber, por lo que se retrasa la entrada de la glucosa a la circulación sistémica y disminuye los niveles postprandiales de glucosa.55-60• Inhibidores de las α-glucosidasas intestinales (IAG, por ejemplo acarbosa y miglitol)
c) Sensibilizadores de insulina
Estos agentes incrementan la sensibilidad del músculo e hígado hacia la insulina. Dependiendo de la clase de fármaco que se trate, incrementará la sensibili dad a la insulina por un mecanismo diferente y tendrá su efecto primario, igualmente, en diferentes tejidos. 55-60
• Biguanidas (por ejemplo metformina). Actúan disminuyendo la producción hepática de glucosa e incrementando la captación de la misma por el músculo esquelético, mediante la activación de la Adenosina Monofosfato Protein Kinasa Activada (AMPK) hepática y muscular, que a su vez inhibe la lipogénesis y activa la β-oxidación.55-60
• Tiazolidinedionas (TZD, por ejemplo rosiglitazona). Son fármacos agonistas de los Receptores Activadores de Proliferación de Peroxisomas Gamma (PPAR-γ, por sus siglas en inglés), que al activarse se unen a elementos de respuesta de ADN, alterando la transcripción de genes que regulan el metabolismo de carbohidratos y lípidos. Su efecto más importante es la estimulación e incremento en la captación de insulina por células del músculo esquelético. Estos agentes disminuyen la resistencia a insulina en los tejidos periféricos, así como la producción hepática de glucosa. 55-60
Una nueva clase de agentes farmacológicos con efecto antidiabético que se han desarrollado para el tratamiento de la diabetes mellitus, son:
1. Miméticos de incretinas
Son un grupo de agentes farmacológicos que imitan algunos efectos endógenos de hormonas incretínicas, incluyendo la mejor secreción de insulina glucosa-dependiente; aunque exhiben efectos glucorregulatorios similares a los del péptido similar al Glucagón-1 (GLP-1, por sus siglas en inglés), sus acciones no son directamente mediadas solamente por el receptor GLP-1.61-63
• Análogos del GLP-1, por ejemplo exenatida y liraglutida
]]> • Análogos de la amilina, por ejemplo pramlintide2. Inhibidores de la dipeptidil-peptidasa IV (DPP-IV)
Inhiben la acción de esta enzima e impiden la degradación de una variedad de péptidos, incluyendo al GLP-1, y por lo tanto aumentando su concentración plasmática así como su periodo de acción.62-64
• Vildagliptina, sitagliptina y saxagliptina
3.Inhibidores del cotransportador dependiente de sodio y glucosa tipo 2 (SGLT-2)
Inhiben este cotransportador, impidiendo la reabsorción de glucosa produciendo glucosuria, disminuyendo así los niveles plasmáticos de glucosa. Estos fármacos aún se encuentran en fase de investigación clínica III.65-72
• Dapagliflozina, remogliflozina y sergliflozina
Flavonoides como una alternativa terapéutica de enfermedades crónicas
Numerosos grupos de investigación han demostrado que los flavonoides presentan diversas actividades sobre la maquinaria metabólica de diferentes células humanas, dichos experimentos han evidenciado efectos tanto in vitro como in vivo que pudiesen hacer sospechar que estos metabolitos secundarios representen una alternativa terapéutica para la regulación de padecimientos que comprometan procesos metabólicos en las células, tal es el caso de los padecimientos de tipo crónico degenerativos cómo la diabetes, la hipertensión y el cáncer.
Debido a la gran diversidad estructural de estos compuestos, su abundancia en la naturaleza y demostrada ubicuidad, los flavonoides representan una importante alternativa para el descubrimiento de nuevos agentes antidiabéticos. Es por ello que en la actualidad diversos grupos de investigación a nivel mundial intensifican la búsqueda de fuentes alternativas para la obtención de compuestos de tipo flavonoide. Una de estas importantes fuentes hasta ahora olvidada, son los flavonoides derivados de los desechos de plantas cítricas denominados citroflavonoides.73
]]> Citroflavonoides y diabetes mellitusLos citroflavonoides han sido ampliamente estudiados desde diferentes puntos de vista, entre ellos destacan los que se encuentran de manera más abundante en los cítricos como hesperidina, naringina, rutina y diosmina. Estos citroflavonoides presentan evidencias de efectos hipoglucemiantes, antihiperglicémicos y/antidiabéticos tanto en ensayos in vivo como in vitro. Los flavonoides más estudiados son: hesperidina, diosmina, naringenina y rutina, los cuales se describen a continuación según sus evaluaciones biológicas y farmacológicas relacionadas con sus efectos antidiabéticos.
Hesperidina
La hesperidina es el flavonoide que por su abundancia en el género Citrus representa la molécula con mayor importancia dentro de este grupo. En cuanto a los efectos sobre la homeostasis de glucosa. Lee y colaboradores (2004) sugieren que la hesperidina juega un papel importante en la prevención de la hiperglicemia, evidenciado por el aumento de la glucólisis y la concentración de glucógeno hepático semejante a lo ocasionado por fármacos antihiperglucémicos cómo la metformina y fenformina.23
Por otro lado, Uehara y colaboradores (2010) reportan que una dieta de esta flavona (10 mg/kg) reduce los niveles de glucosa sanguínea, mediante la alteración de las enzimas reguladoras de glucosa (glucosa cinasa y glucosa-6-fosfatasa), en modelos de DM1 y DM2. EKakadiya y Shah (2010) demostraron que la hesperidina, redujo significativamente los niveles de glucosa sanguínea, HbA1c y glucógeno.74,75
Estos resultados demuestran los efectos hipoglucemiantes de la hesperidina, sin embargo, es necesario el esclarecimiento del modo de acción por el cual se ejercen estos efectos benéficos sobre la homeostasis de glucosa.
Diosmina
La diosmina es una flavanona presente en los cítricos en proporción 9:1 con respecto a la hesperidina. En un estudio realizado por pari y srinivasan (2010) en un modelo de DM2, se demostró su actividad antihiperglucémica al observar la reducción de los niveles de glucosa plasmática dosis dependiente. Adicionalmente, se evidenció su efecto sobre el metabolismo hepático al incrementar la actividad de la hexoquinasa y la glucosa-6-fosfato deshidrogenasa, y disminuir la actividad de la glucosa-6-fosfatasa y la fructosa-1,6-bifosfatasa. Finalmente, en otro estudio se demostró que la administración oral de diosmina en dosis de 100 mg/kg redujo los niveles de HbA1c e incremento la insulina en plasma. Estos resultados confirman los efectos antihiperglicémicos e hipoglucemiantes de la diosmina.76
Naringenina
La naringenina es la flavona que al glicosilarse da origen a la quercetina. Se ha reportado que dicha aglicona incrementa la captación de glucosa por las células del músculo esquelético de forma dependiente de AMPC según los hallazgos de Zygmunt y 22 cols. (2010). Estos efectos hacen sospechar que la naringenina presenta un comportamiento similar a la insulina, al no incrementar la captación de glucosa en los mioblastos, indicando que en el mecanismo de acción de este flavonoide, los transportadores de glucosa dependientes de insulina (GLUT4) podrían estar involucrados.77
]]> Por otro lado, Purushotham, Tian y Belury, evaluaron en un bioensayo in vitro con células FaO (productoras de glucosa a partir de precursores no glucosídicos), observando que la presencia de naringenina en el medio de cultivo ocasionó una potente inhibición de la producción de glucosa hepática, efecto similar al que ejercen los antidiabéticos orales del tipo biguanida.78En cuanto a los efectos in vivo, Ortiz-Andrade y cols. demostraron que la naringenina induce una supresión en el incremento de glucosa plasmática en ratas normoglucémicas y diabéticas, en periodos agudos y subcrónicos de administración. Estos resultados sugieren que el efecto antidiabético ocasionado por la naringenina es llevado a cabo por vía extrapancreática, particularmente por una posible supresión en la absorción de carbohidratos a nivel intestinal, lo que reduce el incremento de los niveles postprandiales de glucosa sanguínea.79 Esta aseveración es reforzada con lo descrito por Li y cols., donde se demuestra que la naringenina inhibe la captación de glucosa en el borde del cepillo intestinal y renal de ratas diabéticas, en un modelo in vitro. Esta inhibición, explica al menos en parte, la actividad antihiperglucemiante de la naringenina en experimentos in vivo, relacionándolo principalmente con una inhibición de los cotransportadores sodio-glucosa (SGLT).80
Otros grupos de investigación también han demostrado que la naringenina está implicada en la modulación del metabolismo de carbohidratos al inhibir la actividad enzimática de la fructosa-1,6-bifosfatasa y la glucosa-6-fosfatasa, proteínas importantes en la gluconeogénesis. Además, Rayidi y cois, sugieren que este efecto es llevado a cabo a través de la regulación del AMPc o cualquier otro metabolito regulador de la gluconeogénesis. De la misma forma, naringenina produjo un aumento de la actividad de la hexoquinasa, lo que también indica un incremento en la absorción de glucosa por los hepatocitos y un aumento de la glicólisis ocasionado por ésta flavona.81
Los flavonoides presentes en las especies vegetales se encuentran principalmente como glicósidos. Sin embargo, existen reportes en la literatura científica que describen que dichos compuestos son los menos bioactivos. Esto indica que el flavonoide en forma libre (aglicona) son la forma bioactiva de este grupo de metabolitos secundarios, los cuales son principalmente absorbidos por difusión pasiva.82
Por otro lado, se plantea que los glicósidos podrían ser modificados (desglicosilados) al ingresar por vía oral al organismo mediante las enzimas del tracto gastrointestinal, o bien, por la flora normal que ahí habita. Por lo que, la actividad farmacológica desencadenada en el organismo, será ocasionada por la correspondiente aglicona. Posteriormente, los flavonoides al ser absorbidos pueden metabolizados en el enterocito y en el hígado para ser finalmente excretados. Es por ello, que en el presente artículo se hace mención de dos glucósidos de flavonoide que dan origen a agliconas que han sido objeto de amplios estudios farmacológicos encaminados a la búsqueda y esclarecimiento de sus efectos beneficios sobre la homeostasis de glucosa en el organismo, tal es el caso de la naringina y la rutina, glucósidos de naringenina y quercetina respectivamente.82
Naringina
Lee y colaboradores (2004) han descrito que la flavanona naringina presenta actividad antihiperglucemiante al ser avaluada en ratones diabéticos C57BL/KsJ-db/db, reduciendo los niveles de glucosa en sangre, a través de la disminución de la actividad de las enzimas hepáticas glucosa-6-fosfatasa, fosfoenolpiruvato carboxicinasa y glucocinasa hepática (precursores de la gluconeogénesis). Así, como un aumento en la concentración de glucógeno, insulina, péptico C y lectina. Sin embargo, el mecanismo por el cual este compuesto ocasiona tales regulaciones aún no está esclarecido. Por otro lado, estudios realizados por Pari y Suman (2010) demostraron la actividad antihiperglucemiante de esta flavanona, al ser evaluada en ratas diabéticas observándose la capacidad de disminuir el estrés oxidativo al aumentar las concentraciones de vitamina C, vitamina E y glutatión. De esta manera, la naringina disminuye las hidroxiperoxidasas y otras sustancias reactivas, que son producidas durante el curso patológico de la inducción de la diabetes experimental. Estos resultados indican que los efectos benéficos de la naringina están relacionados con su propiedad antioxidante (ó atrapar radicales libres). Sin embargo, el mecanismo por el cuál se llevan a cabo los efectos antidiabéticos aún no están del todo esclarecidos.83 Estas aseveraciones son reforzadas con los resultados de, Ali y colaboradores (2004) quienes proponen que el mecanismo de acción de la naringenina, está relacionado con su actividad antioxidante.84
Rutina
Rutina es el glucósido de quercetina más común en los cítiricos, y es reconocido por su capacidad de disminuir la permeabildiad capilar. 85 Por otro lado, en cuanto a sus efectos farmacológicos, Rauter (2010) logró demostrar que administraciones subagudas de este flavonoide mejoran la tolerancia a la glucosa en ratas diabéticas.86 G Finalmente Kamalakkannan, Mainzen y Prince demostraron que la administración subaguda de rutina en ratas diabetizadas con STZ, ejerció una disminución en los niveles de glucosa plasmática, hemoglobina glucosilada, y las hidroperoxidasas lipídicas, así como un aumento en las concentraciones séricas de insulina, péptido C y hemoglobina total Estos resultados si bien no esclarecen el mecanismo de acción hipoglucemiante y antihiperglucémico de la rutina, si evidencian sus efectos benéficos sobre la regulación del metabolismo de carbohidratos.87
Quercetina
]]> La quercetina está principalmente presente en la naturaleza como O-glucósido en donde uno o más grupos azúcar se encuentran unidos a los grupos fenólicos por enlaces glucosídicos.85Este compuesto presenta una gran variedad de estudios biológicos y farmacológicos relacionados con sus efectos sobre sitios clave de la regulación de la secreción de insulina, tal es el caso de lo descrito por Torres-Piedra y cols. (2010) como un potente inhibidor in vitro de la enzima 11β-hidroxiesteroide deshidrogenasa tipo 1 (11 β-HSD1), la cual es estimuladora de la acción de la hormona glucocorticoide, antagonista de la insulina y estimuladora de la gluconeogénesis.48
De la misma forma se ha demostrado su actividad antidiabética en diferentes modeles de diabetes experimental, evidenciando que, la quercetina protege a las células β pancreáticas del estrés oxidativo producido por la administración de STZ en ratas, debido a una potente actividad antioxidante, según lo descrito por Coskun y cols. (2004).88
Por otro lado, Vessal y cols. (2003) describen que la quercetina no presenta efecto a nivel de glucosa sanguínea en ratas normoglucémicas, sin embargo, en ratas diabéticas inducidas con STZ, disminuyó considerablemente los niveles de glucosa en una prueba de tolerancia a la glucosa. De la misma forma, en este mismo estudio, Vessal y cols. describen un potente efecto estimulador de la enzima glucoquinasa hepática. Esto permite suponer que los efectos antioxidantes de la quercetina juegan un papel importante en la, regeneración de los islotes pancreáticos, lo que explicaría el incremento de la secreción de insulina.89
De la misma forma Abdelmoaty y cols.90 no encontraron efectos hipoglucemiantes en ratas normoglucémicas, sin embargo, en ratas diabéticas inducidas con STZ, quercetina previno la hiperglicemia característica de la patología, lo cual sugiere que su efecto antidiabético podría ser un producto secundario a su capacidad antioxidante. Sin embargo, Coldiron, Sanders y Watkins (2002), no encontraron modificaciones considerables en los marcadores del estrés oxidativo, en un tratamiento con quercetina. Por lo que para elucidar el mecanismo de acción aplicable al tratamiento de la diabetes y sus potenciales efectos terapéuticos, es necesario elucidar los mecanismos moleculares implicados.91
Conclusiones
Los citroflavonoides más estudiados se dividen en dos grupos: a) glicosilados (hesperidina, naringina, rutina y diosmina) y agliconas (naringingenina y quercetina). Las agliconas se forman por la hidrolisis enzimática de los flavonoides glicosilados. Estos compuestos se encuentran presentes en una gran variedad de especies vegetales y son recomendados por sus propiedades antioxidantes. El presente trabajo representa una recopilación de la literatura científica enfocada a explicar los hallazgos sobre los efectos benéficos en la homeostasia de glucosa por los flavonoides aislados de los cítricos. Estos resultados permiten proponer a los citroflavonoides como una alternativa terapéutica para el tratamiento de la diabetes debido a sus importantes efectos antioxidantes. Dichos efectos, representan un mecanismo de acción novedoso que a la fecha ha despertado gran interés por su relación con una gran variedad de patologías del tipo crónico degenerativas, las cuales, parecen estar relacionadas entre sí. Es por ello, que moléculas con potentes efectos antioxidante como los flavonoides, con fuentes de obtenciones sustentables y asequibles, incrementan el interés de su estudio.
Los citroflavonoides aquí descritos, demuestran además la capacidad de interferir en algunos de los mecanismos fisiopatológicos de la diabetes o bien, un efecto regulador en las consecuencias metabólicas de dicho padecimiento. Esto incrementa el potencial terapéutico de estos metabolitos secundarios. Sin embargo, es necesario esclarecer los mecanismos moleculares implicados en la regulación de los efectos farmacológicos observados, los cuales permitan establecer parámetros de dosificación, potencia y eficiencia que haga de éstos compuestos líderes en el desarrollo de nuevas alternativas terapéuticas de la diabetes y/o sus complicaciones.
]]> Agradecimientos
González-Sánchez, Avel y Cabañas Wuan, Ángel agradecen la beca otorgada por el Programa Institucional de Orientación a la Investigación de la Universidad Autónoma de Yucatán para realizar una estancia de investigación en el Laboratorio de Farmacología, FQ-UADY.
Referencias
1. Rusznyak S, Szent-Gyorgyi A. Vitamin P: Flavonols as Vitamins. Nature. 1936; 138:27. [ Links ]
2. Ranaud S, de Lorgeril M. Wine, alcohol, platelets, and the French Paradox for coronary heart disease. Lancet. 1992; 339 (8808): 1523-1526. [ Links ]
3. Rosenkranz S, Knirel D, Dietrich H. Inhibition of the PDGF receptor by red wine flavonoids provides a molecular explanation for the "French Paradox". The FASEB Journal. 2002; 16:1958-1960. [ Links ]
]]>4. Harborne JB, Williams CA. Advances in flavonoids research since 1992. Phytochemistry. 2000; 55:481-504. [ Links ]
5. Van Hoorn DEC, Van NK, Boelens PG. Biological activities of flavonoids. Science & Medicine. 2003; 9(3): 152-161. [ Links ]
6. De Groot H, Raven U. Tissue injury by reactive oxygen species and the protective effects of flavonoids. Fundam Clin Pharmacol. 1998; 12:249-255. [ Links ]
7. Martínez S, González J, Culebras J, Tuñón M. Los flavonoides: propiedades y acciones antioxidantes. Rev Nutrición Hosp. 2002; 17(6):271-278. [ Links ]
8. Bors W, Heller W, Quista M. Flavonoids as antioxidants: determination of radical-scavenging efficiencies. Methods Enzymol. 1990; 186: 343-355. [ Links ]
]]>9. Pérez G. Los flavonoides: Oxidantes o Prooxidantes. Rev Cub Inv Biomed. 2003; 22(1): 48-57 [ Links ]
10. Aherne SA, O'Brien NM. Dietary flavonols: chemistry, food content, and metabolism. Nutrition. 2002; 18(1): 75-81. [ Links ]
11. Russo RO, Speranza M. Los flavonoides en la terapia cardiovascular. Rev. Costarr. Cardio. 2006; 7(6) 1-6. [ Links ]
12. Erlund I. Chemical analysis and pharmacokinetics of the flavonoids quercetin, hesperitin and naringenin in humans. Tesis Doctoral. 2002. Facultad de Agricultura y Silvicultura de la Universidad de Helsinki. [ Links ]
13. Kochhar A, Nagi M. Effect of supplementation of traditional medicinal plants on blood glucose in non-insulin-dependent diabetics: A Pilot Study. J Med Food. 2005; 8(4):545-549. [ Links ]
14. Zibula SMX, Ojewole JAO. Hypoglycemic effects of Hypoxis hemerocallidea corn 'African potato' methanolic extract in rats. Med J Islam Acad Sci. 2000; 13(2):75-78. [ Links ]
15. Akerele O. WHO's traditional Medicine Programme: Progress and Perspectives. WHO chronide. 1984; 38(2):76-81. [ Links ]
16. Van Acquire SA, Van den Berg DJ, Tromp MN. 1996. Structural aspects of antioxidant activity of flavonoids. Free Radie. Biol. Med. 20:331-342. [ Links ]
17. Groot H, Rauen U. Tissue injury by reactive oxygen species and the protective effects of flavonoids. Fundam Clin Pharmacol. 1998; 3:249-255. [ Links ]
18. Vinson J. A. Flavonoids in food as in vitro and in vivo antioxidants. Adv Exp Med Biol. 1998; 439:151-164. [ Links ]
19. Roschek B, Fink RC, Matthew D. Elderberry flavonoids bind to and prevent H1N1 infection in vitro. Phytochemistry. 2009; 70:1255-1261. [ Links ]
20. Tasdemir D, Kaiser M, Brun R. Antitrypanosomal and antileishmanial activities of flavonoids and their analogues: In vitro, in vivo, structure-activity relationship, and quantitative structure-activity relationship studies. Antimicrob Agents Chemother. 2006; 40(4): 1352-1364. [ Links ]
21. SülsenVP, Cazorla SI, Frank FM. Trypanocidal and leishmanicidal activities of flavonoids from argentine medicinal plants. Am. J. Trop. Med. Hyg. 2007; 77(4):654-659. [ Links ]
22. Mahmood V, Mina H, Mohammad V. Antidiabetic effects of quercetin in Streptozocin-induced diabetic rats. Comp Biochem Physiol. 2003; 135:357-364. [ Links ]
23. Jung U, Lee M, Jeong K, Choi M. The Hypoglycemic effects of hesperidin and naringin are partly mediated by hepatic glucose-regulating enzymes in C57BL/KsJ-db/db Mice. J Nutr. 2004; 22:2499. [ Links ]
24. Jong SK, Chong SK, Kun HS. Inhibition of a-glucosidase and amylase by luteolin, a flavonoid. Biosci. Biotechnol. Biochem. 2000; 64(11):2458-2461. [ Links ]
25. Bohm H, Boeing H, Hempel J. Flavonols, Flavone and anthocyanins as natural antioxidants of foods and their possible role in the prevention of chronic diseases. Ernahrungswiss. 1998; 2:147-163. [ Links ]
26. Mankanil KL, Krishna V, Manjunatha BK, Vidya SM, Jagadeesh SSD, Manohara YD, Raheman AU, Avinash KR. Evaluation of hepatoprotective activity of stem bark of Pterocarpus marsupium Roxb. Indian J Pharmacol. 2005; 37(3):165-168. [ Links ]
27. Wul Y, Wang F, Zheng Q, Lu L, Yao H, Zhoul C, Wul X, Zhaol Y. Hepatoprotective effect of total flavonoids from Laggera alata against carbon tetrachloride-induced injury in primary cultured neonatal rat hepatocytes and in rats with hepatic damage. J Biomed Sci. 2006. 13:569-578. [ Links ]
28. Qureshil MN, Kuchekar BS, Logade NA., Haleem MA. In vitro antioxidant and in vivo hepatoprotective activity of Leucas ciliata leaves Rec. Nat. Prod. 2010; 4(2): 124-130. [ Links ]
29. Chandrasekhar KS, Prasanna KS. Hepatoprotective Activity of Leucas lavandulaefolia against carbon tetrachloride-Induced hepatic damage in rats. IJPSR. 2010; 1(2):101-103. [ Links ]
30. Raines T, Jones P, Moe M. Investigation of anxiolytic effects of luteolin, a lemon balm flavonoid in the male Sprague-Dawley rat. AANA Journal. 2009; 77(1):33-36. [ Links ]
31. Rodrigues AE, Oliveira RFR, Lindoso CGB. Anxiolytic and anticonvulsant effects on mice of flavonoids, linalool, and a-tocopherol presents in the extract of leaves of Cissus sicyoides L. (Vitaceae). J Biomed Biotech. 2008; 2009:1-5. [ Links ]
32. Choi Y, Donghyun KT, Paik SR. Molecular simulations for anti-amyloidogenic effect of flavonoid myricetin exerted against Alzheimer's ß-amyloid fibrils formation. Bull Korean Chem Soc. 2008; 29(8):1505-1509. [ Links ]
33. Vauzour D, Vafeiadou K, Rodríguez MA. The neuroprotective potential of flavonoids: A multiplicity of effects. Genes Nutr. 2008; 3:115-126. [ Links ]
34. Miyake Y, Suzuki E, Ohya S. Lipid-lowering effect of eriocitrin, the main flavonoid in lemon fruit, in rats on a high-fat and high-cholesterol diet. J Food Scie. 2006; 71(9):S633-S636. [ Links ]
35. Chen J, Li X. Hypolipidemic effect of flavonoids from mulberry leaves in triton WR-1339 induced hyperlipidemic mice. Asia Pac J Clin Nutr. 2007; 16(1):290-294. [ Links ]
36. Perez VF, Duarte J, Jimenez R. Antihypertensive effects of the flavonoid quercetin. Pharmacological Reports. 2009; 61:67-75. [ Links ]
37. Larson AJ, Symons D, Jalili T. Quercetin: A treatment for hypertension? A review of efficacy and mechanisms. Pharmaceuticals. 2010; 3:237-250. [ Links ]
38. Xavier BF, Serge S, Cameron B. Glycosylated Flavones as Selective Inhibitors of Topoisomerase IV. Antimicrob Agents Chemother. 1997; 41(5):992-998. [ Links ]
39. Van HDE, Nijveldt RJ, Van LPA. Accurate prediction of xanthine oxidase inhibition based on the structure of flavonoids. European J. Pharmacol. 2002; 451:111-118. [ Links ]
40. Goodarzi MT, Zal F, Malakooti M. Inhibitory activity of flavonoids on the lens aldose reductase of healthy and diabetic rats. Acta Médica Iranica. 2005; 44(1):42-44. [ Links ]
41. Moore PS, Pizza C. Observations on the inhibition of HIV-1 reverse transcriptase by catechins. Biochemical J. 1992. 288:717-719. [ Links ]
42. Ferriola PC, Cody V, Middleton E. Protein kinase C inhibition by plant flavonoids: Kinetic mechanisms and structure-activity relationships. 1989; Biochem Pharmacol 38:1617-1624. [ Links ]
43. Nakashima S, Koike T, Nozawa Y. Genistein, a protein tyrosine kinase inhibitor, inhibits thromboxane A2-mediated human platelet responses. Mol Pharmacol. 1991; 39:475-480. [ Links ]
44. Nishino H, Naitoh E, Iwashima A, Umezawa K. Quercetin interacts with calmodulin, a calcium regulatory protein. Experientia. 1984; 40:84-85. [ Links ]
45. Babujanarthanam R, Kavitha P, Pandian MR. Quercitrin, a bioflavonoid improves glucose homeostasis in streptozotocin-induced diabetic tissues by altering glycolytic and gluconeogenic enzymes. Fundam Clin Pharmacol. 2009; 24(3):357-364. [ Links ]
46. Guigas B, Naboulsi R, Villanueva GR, Taleux N, Lopéz-Novoa JM, Leverve XM, El-Mir MY. The flavonoid silibinin decreases glucose-6-phosphate hydrolysis in perifused rat hepatocytes by an inhibitory effect on glucose-6-phosphatase. Cell Physiol Biochem. 2007; 20:925-934. [ Links ]
47. Lindahl M, Tagesson C. Flavonoids as phospholipase A2 inhibitors: importance of their structure for selective inhibition of group II phospholipase A2. Inflammation. 1997.21:347-356. [ Links ]
48. Torres-Piedra M, Ortiz-Andrade RR, Villalobos-Molina R, Singh N, Medina-Franco J, Webster S, Binnie M, Navarrete-Vázquez G, Estrada-Soto S. A comparative study of flavonoid analogues on Streptozotocin/Nicotinamide induced diabetic rats: Quercetin as a potential antidiabetic agent acting via 11ß-hydroxysteroid dehydrogenase type 1 inhibition. Eur J Med Chem. 2010; 45(6):2606-2612. [ Links ]
49. American Diabetes Association. Diagnosis and classification of Diabetes mellitus. Diabetes Care. 2010; 33(1):S62-S69. [ Links ]
50. Lebovitz HE. Type 2 Diabetes: An overview. Clinical Chemistry. 1999. 45(8B):1339-1345. [ Links ]
51. American Diabetes Association. Diagnosis and classification of Diabetes mellitus. Diabetes Care. 2004; 27(1):S5-S10. [ Links ]
52. Unger RH. Reinventing type 2 diabetes. Pathogenesis, treatment and prevention. JAMA. 2008; 299:1185-1187. [ Links ]
53. Youngren JF. Regulation of insulin receptor function. Cell Mol Life Sci. 2007; 64:873-891. [ Links ]
54. Stumvoll M, Goldstein BJ, Van Haeften TW. Pathogenesis of type 2 diabetes. Endocrine Research. 2007; 32:19-37. [ Links ]
55. Cheng AYY, Fantus IG. Oral antihyperglycemic therapy for type 2 diabetes mellitus. CMAJ. 2005; 172(2):213-226. [ Links ]
56. Krentz AJ, Bailey CJ. Oral Antidiabetic Agents. Current role in type 2 Diabetes mellitus. Drugs. 2005; 65(3):385-409. [ Links ]
57. DeFronzo AF. Pharmacologic therapy for type 2 Diabetes mellitus. Ann Intern Med. 1999; 131(4):281-300. [ Links ]
58. Inzucchi SE. Oral antihyperglycemic therapy for type 2 Diabetes. JAMA. 2002; 287(3):360-369. [ Links ]
59. Lebovitz HE. Treating hyperglycemia in type 2 Diabetes: New goals and strategies. CCJM. 2002; 69(10):809-819. [ Links ]
60. Koski RR. Practical review of oral antihyperglycemic agents for Type 2 diabetes mellitus. The Diabetes Educator. 2006; 32(6):869-876. [ Links ]
61. Hoogwerf BJ. Exenatide and Pramlintide: New glucose-lowering agents for treating Diabetes mellitus. CCJM. 2006; 73(5):477-484. [ Links ]
62. Hinnen D, Nielsen LL, Waninger A. Incretin mimetics and DPP-IV inhibitors: New paradigms for the treatment of type 2 Diabetes. J Am Board Fam Med. 2006; 19(6):612-618. [ Links ]
63. Bloomgarden ZT. Incretin concepts. Diabetes Care. 2010; 33(2):20-24. [ Links ]
64. Tahara A, Yokono AM, Nakano R, Someya Y, Hayawaka M, Shibasaki M. Evaluation of the antidiabetic effects of dipeptidyl peptidase-TV inhibitor ASP8497 in streptozotocin-nicotinamide-induced mildly diabetic mice. Pharmacology. 2008; 83:177-187. [ Links ]
65. Castaneda F, Burse A, Boland W, Kinne RKH. Thioglycosides as inhibitors of HSGLT-1 and HSGLT-2: Potential therapeutic agents for the control of hyperglycemia in diabetes. Int J Med Scis. 2007; 4(3):131-139. [ Links ]
66. Jabbour SA, Goldstein BJ. Sodium-glucose co-transporter 2 inhibitors: Blocking renal tubular reabsorption of glucose to improve glycaemic control in patients with Diabetes. Int J Clin Pract. 2008; 62(8): 1279-1284. [ Links ]
67. Pérez G, González O, Cano M. Type 2 sodium-glucose cotransporter (SGLT2) inhibitors: from familial renal glucosuria to the reatment of type 2 diabetes mellitus. Nefrologia. 2010;30(6):618-625. [ Links ]
68. Idris I, Donnelly R. Sodium-glucose co-transporter 2 inhibitors: an emerging new class of oral antidiabetic drug. Diabetes Obes Metab. 2009;11:79-88. [ Links ]
69. Jabbour SA. The importance of reducing hyperglycemia while preserving insulin secretion: The rationale for sodium-coupled glucose co-transporter 2 inhibition in Diabetes. U. S. Endocrinology. 2009;5(1):75-78. [ Links ]
70. Katsuno K, Fujimori Y, Takemura Y, Hiratochi M, Itoh F, Komatsu Y, Fujikura H, Isaji M. Sergliflozin, a novel selective inhibitor of low-affinity sodium glucose cotransporter (SGLT-2), validates the critical role of SGLT2 in renal glucose reabsorption and modulates plasma glucose level. J Pharmacol and Exp Ther. 2007; 320(1):323-330. [ Links ]
71. Fujimori Y, Katsuno K, Nakashima I, Ishikawa TY, Fujikura H, Isaji M. Remogliflozin etabonate, in a novel category of selective low-affinity sodium glucose cotransporter (SGLT2) inhibitors, exhibits antidiabetic efficacy in rodent models. J Pharmacol and Exp Ther. 2008; 327(1):268-276. [ Links ]
72. List JF, Woo V, Morales E, Tang W, Fiedorek FT. Sodium-glucose cotransport inhibition with dapagliflozin in type 2 Diabetes. Diabetes Care. 2009; 32(4):650-657. [ Links ]
73. Gattuso G, Becerra D, Gargiulli C, Leuzzi U, Caristi C. Flavonoids composition of Citrus juice: Review. Molecules. 2007; 12:1641-1663. [ Links ]
74. Akiyama S, Katsumata S, Suzuki K, Ishimi Y, Wu J, Uehara M. Dietary hesperidin exerts hypoglycemic and hypolipidemic effects in streptozotocin-induced marginal type 1 diabetic rats. J Clin biochem nutr. 2010; 46: 87-92. [ Links ]
75. Kakadiya J; Shah N. Effect of hesperidin on cardiovascular complication in streptozotocin-nicotinamide induced type 2 diabetic rats. Int J Pharmacy Pharm Sci. 2010; 2(3): 165-169. [ Links ]
76. Pari L; Srinivasan S. Antihyperglycemic effect of diosmin on hepatic keys enzymes of carbohydrate metabolism in streptozotocin-nicotinamide-induced diabetic rats. Biomed Pharmacother. 2010; 64(7):477-481. [ Links ]
77. Zygmunt K, Faubert B, MacNeil J, Tsiani E. Naringenin, a citrus flavonoid, increases muscle cell glucose uptake via AMPK. Biochem Biophys Res Commun. 2010; 398(2): 178-183. [ Links ]
78. Purushotham A, Tian M, Belury A. The citrus flavonoid naringenin suppresses hepatic glucose production from FaO hepatoma cells. Mol Nutr Food Res. 2009; 53(2): 300-307. [ Links ]
79. Oriz-Andrade R, Sánchez-Salgado J, Navarrete-Vázquez G, Webster S, Binnie M, Garcia-Jimenez S, León-Rivera R, Villalobos-Molina R, Estrada-Soto S. Antidiabetic and toxicological evaluations of naringenin in normoglycaemic and NTDDM rat models and its implications on extra-pancreatic glucose regulation. Diabetes Obes Metab. 2008; 10(11): 1097-1104. [ Links ]
80. Li JM, Che CT, Lau CB, Leung PS, Cheg CH: Inhibition of intestinal and renal NA+-glucose cotransporter by naringenin. Int J Biochem Cell Biol. 2006; 38(5-6): 985-995. [ Links ]
81. Rayidi S, Pari L. Effect of naringenin on carbohydrate metabolism in streptozotocin-nicotinamide induced diabetic rats. Biomirror. 2010; 2:1-8. [ Links ]
82. Pinent M, Castell A, Baiges I, Montagut G, Aróla L, Ardévol A. Bioactivity of flavonoids on insulin-secreting cells. Compr Rev Food Sci Food Saf. 2008; 7:299-308. [ Links ]
83. Pari L, Suman S. Antihyperglycemic and antilipidperoxidative effects of flavonoid naringin in streptozotocin-nicotinamide induced diabetic rats. Int J Biol Med Res. 2010; 1(4): 206-210. [ Links ]
84. Ali M, Abd El Kader M. The influence of naringin on the oxidative state of rats with streptozotocin-induced acte hyperglicaemia. Z. Naturforsch. 2004; 59c:726-733. [ Links ]
85. Shanno, R. Rutin: a new drug for the treatment of increased capillary fragility. Am. J. Med. Sci. 1946; 211(5): 539-543. [ Links ]
86. Rauter P, Martins A, Borges C, Mota-Filipe H, Pinto R, Sepodes B, Justino J. Antihyperglycaemic and protective effects of flavonoids on streptozotocin-induced diabetic rats. Phytother. Res. 2010; (24):S133-S138. [ Links ]
87. Kamalakkannan N, Prince PS. Antihyperglycaemic and antioxidant effect of rutin, a polyphenolic flavonoid, in streptozotocin-induced diabetic wistar rats. Basic Clin Pharmacol Toxicol. 2006; 98(1): 97-103 [ Links ]
88. Coskun O, Kanter M, Korkmaz A, Oter S. Quercetin, a flavonoid antioxidant, prevents and protects streptozotocin-induced oxidative stress and ß-cell damage in rat pancreas. Pharmacol Res. 2004; 51(2):117-123. [ Links ]
89. Vessal, M, Hemmati, M, Vasei M. Antidiabetic effects of quercetin in streptozotocin induced diabetic rats. Comp Biochem Physiol. 2003; 135(3): 357-364. [ Links ]
]]>90. Abdelmoaty MA, Ibrahim MA, Ahmed NS, Abdelaziz MA. Confirmatory studies on the antioxidant and antidiabetic effect of quercetin in rats. Indian J Clin Biochem. 2010; 25(2): 188-192. [ Links ]
91. Coldiron A, Sanders R, Watkins J. Effects of combined and coenzyme Q10 treatment on oxidative stress in normal and diabetic rats. 2002; 16(4): 197-202.
]]>