
Optimization of acid hydrolysis of bagasse from Agave tequilana Weber
Optimización de hidrólisis ácida de bagaso de Agave tequilana Weber
J. Saucedo-Luna1, A. J. Castro-Montoya1*, J. L. Rico1 and J. Campos-García2
1 División de Estudios de Posgrado, Facultad de Ingeniería Química, Universidad Michoacana de San Nicolás de Hidalgo, Edif. V-1, Cd. Universitaria, 58060, Morelia, Michoacán, México. * Corresponding author. E-mail: ajcastro@umich.mx.
]]> 2 Instituto de Investigaciones Químico-Biológicas, Universidad Michoacana de San Nicolás de Hidalgo, Edif. S, Cd. Universitaria, 58060, Morelia, Michoacán, México.
Received 7 of October 2009;
Accepted 4 of March 2010
Abstract
The bagasse of Agave tequilana Weber is a lignocellulosic residue resulting from the production of tequila. At present, the bagasse is not industrialized, and poses a serious contamination problem. Thus, an ongoing research effort is to explore the possibility to obtain added value chemicals from it. The aim of the present work was to determine the effect of temperature, concentration of H2 SO4 and reaction time on the hydrolysis of agave bagasse to produce fermentable sugars. The hydrolysis was performed in two sequential batch stages: (a) in the first stage the optimal conditions were 151ºC, 2% of sulfuric acid and 10 min of reaction, yielding 236 g/Kg dry matter and concentration of 26.9g/L of fermentable sugars, and (b) in second stage the optimal experimental conditions for the depolimerization of the biomass resulting from the first stage were 175º C, 2% of sulfuric acid and 30 min of reaction, yielding 90 g/kg dry matter and concentration of 15 g/L of fermentable sugars. Total fermentable sugars yield from the sequential batch stages was 326 g/kg dry matter (48.50% of the theoretical value).
Keywords: agave bagasse, dilute-acid hydrolysis, optimization.
Resumen
]]> El bagazo de Agave tequilana Weber es un residuo lignocelulósico que se genera en el proceso de producción de tequila. Actualmente, este bagazo no tiene una aplicación industrial, sin embargo por su composición química puede ser utilizado para la producción de productos químicos con un valor agregado. El objetivo del presente trabajo consistió en determinar el efecto de la temperatura, la concentración de H2SO4 y del tiempo de reacción sobre la hidrólisis de bagazo de agave para la producción de azúcares fermentables. La hidrólisis se llevó a cabo en dos etapas en serie y las condiciones óptimas para cada una de ellas fueron las siguientes: para la primera etapa 151°C, 2 % de H2SO4 y 10 minutos de reacción con un rendimiento de 236 g/kg de materia seca y una concentración de 26.9 g/l; para la segunda etapa 175°C, 2 % de H2SO4 y 30 minutos de reacción con un rendimiento de 90 g/kg de materia seca y una concentración de 15 g/l. De manera global se obtuvo un rendimiento de 326 g/kg de materia seca equivalente a un 48.05 % del valor teórico.Palabras clave: bagazo de agave, hidrólisis ácida diluida, optimización.
1 Introduction
The blue agave (Agave tequilana Weber) is a plant highly cultivated in México and used as raw material in the production of tequila. The bagasse is a residual fiber obtained from the process and represents 40% of the processed agave (Iñiguez et al., 2001). The annual generation of bagasse in México is about 1.05x108 kg. From it, only small portion is used to prepare compost and the rest throw away. It is then clear that this particular situation is very attractive to look for better usage of agave bagasse. It is known that the ligno-cellulosic materials are considered as the only renewable source to produce biofuels and/or chemical products, with a great potential to supply energy (Bozell et al., 2000). It is worth mentioning that the bioprocess for the production of ethanol from lignocellulosic materials takes place mainly in three stages: hydrolysis of the cellulosic polymers to form monosaccharides, fermentation of sugars by yeast or bacteria to ethanol and finally, the purification of ethanol. Currently hydrolysis is carried out in either concentrated acid environment or in presence of enzymes. The latter requires pretreatment of the biomass in order to improve the enzymatic digestion. Several pretreatment processes have been investigated, for instance: Mechanical comminuting, pyrolysis, steam explosion, ammonia fiber explosion, CO2 explosion, ozonolysis, hydrolysis acid and alkaline, organosolv process and biological (Sun and Cheng, 2002). However, among all the pretreatment methods proposed, dilute-acid pretreatment has been widely studied because it is effective and inexpensive (Wyman, 1994).
The variables commonly studied in acid hydrolysis are the type and concentration of acid, reaction temperature, solid/liquid ratio and reaction time. The aim of such studies is frequently the determination of the total amount of fermentable sugars, or the concentration of monosac-charides such as xylose, glucose and arabinose. It was reported that for Bermuda grass and rye straw, dilute-acid hydrolysis is effective for the de-polimerization of hemicelluloses, to obtain a yield between 50 to 60% of xylose. The experimental conditions were 1.2% of sulfuric acid, 121 ºC and 60 min of reaction. Using similar experimental conditions, it was reported that hydrolysis of cellulose to glucose was 27-33% (Sun and Cheng, 2005). For raw Paja Brava, typical grass from Bolivia, the maximum yield to xylose was obtained using 0.5-1% sulfuric acid and 190 ºC, whereas for the hydrolysis of cellulose a higher temperature of 235ºC was needed (Sanchez et al., 2004). The barley straw, residue of brewery process was hydrolyzed by sulfuric acid to obtain 70.83 % of conversion to fermentable sugars for 110 °C, 1% of acid concentration and 210 minutes of reaction (Aguilar-Rivera and Canizalez-Leal, 2004). Acid hydrolysis was also applied to agricultural stubble of rice, wheat, sorghum and sugarcane bagasse using different acids, for instance, phosphoric acid (Gámez et al., 2004), sulfuric acid (Aguilar et al., 2002; Roberto et al., 2003), nitric acid (Rodriguez-Chong et al., 2004), and hydrochloric acid (Her-rera et al., 2004; Herrera et al., 2003). The concentration of acid ranges was from 1 to 6%, the temperature from 100 to 200 ºC, and reaction time up to 300 min. All of these reports agree that the depolymerization of hemicellulose requires lower severity for hydrolysis than that for cellulose. If high temperatures and long reaction times are applied, the monosaccharides resulted from hemicellulose degrade to form furan compounds and carboxylic acids, in agreement with our experimental observations, where furfural and hydroxymetylfurfural were qualitatively detected. For this reason, it is recommended that the hydrolysis process to be performed in two stages.
In that respect, no reports have been found in the open literature about the dilute-acid hydrolysis of agave bagasse to fermentable sugars. The aim of the present research project was to study the transformation of agave bagasse by dilute-acid hydrolysis, in order to evaluate the potential of the available biomass to produce fermentable sugars.
2 Materials and methods
2.1 Materials
]]> Bagasse from Agave tequilana Weber, was collected from different tequila distilleries (El Quiote, El Viejito, el Consuelo Jalisco-México), washed with water at room temperature until no sugars (monosaccharides) were detected in the liquid, dried, milled in a laboratory mill and sieved to particles of 0.5 mm diameter, homogenized in a single lot and stored until needed. The analysis of the main polymeric fractions was done by quantitative acid hydrolysis at standard conditions (Garrote et al., 1999). The reaction was followed by quantifying the glucose, xylose and arabinose (fermentable sugars) by liquid chromatography (HPLC) equipped with a refractive index detector and provided with a Metacarb 87ºC Varian column. Water was used as a mobile phase at 0.6 ml/min and the column temperature was 70ºC. Furfural was analyzed by spectrophotometry at 280 nm (Vázquez et al., 2007; Téllez-Luis et al., 2002).2.2 Acid hydrolysis
The experiments were performed using a Multiple Reactor System (Parr Instruments, Model 5000 Multiple Reactor System) provided with 6 reactors of 75 ml each. Each reactor has temperature and agitation control. For each of the operating conditions evaluated all the reactors were loaded simultaneously with 5 g of agave bagasse, 1 or 2% of dilute-sulfuric acid and various temperatures were applied, in the range of 100 to 200º C. A solid- liquid ratio 1/10 was used for all experiments. Every 10 minutes one of the reactors was stopped and cooled to room temperature to separate by filtration the liquid and solid phase. The solid phase was used to quantify the global yield (g of residual biomass/g of biomass treated) of hydrolysis and the liquid phase for the quantification of sugars and analysis of furfural by HPLC. The solid remain from 1st stage of acid hydrolysis was exposed to a second hydrolysis, using similar experimental conditions as first stage.
2.3 Design of experiments
The experiments were performed using a factorial design of experiments with multiple levels. The order of runs was random with 3 replicas each. The total concentration of fermentable sugars was fitted to Eq. (1), that is second order empirical model, which was then utilized to predict the optimal experimental conditions:

Where Y is the response variable, Xi and Xj are the independent variables, temperature, reaction time and concentration of catalyst, β0 is the intersection parameterβi are the lineal coefficients, βii are the square coefficients, βij are the interaction coefficients (i ≠ j) and ei is the random error.
3 Results and discussion
3.1 Composition of agave bagasse
]]> Composition of the agave bagasse was measured by quantitative acid hydrolysis (Garrote et al., 1999), with the follow results: cellulose (42±-2), hemicelluloses (20±1) and lignin (15± 1) %w dry basis (Table 1). These results are comparable to other biomass source used to the same objective, for instance, Canettieri et al., (2007), reported for eucalyptus grandis residue, 40%, 16% and 23 % for cellulose, hemicelluloses and insoluble lignin, respectively. In a different study, Rocha (2000), reported for sugarcane bagasse 47%, 27% and 20.5% for cellulose, hemicelluloses and insoluble lignin, respectively. The high cellulose and hemicelluloses content measured for agave bagasse indicates a high potential for production of a great variety of products, as ethanol, which may be then used as a fuel or raw material for synthesis of different chemicals, (Werpy and Petersen, 2004).
3.2 Hydrolysis in the first stage
Acid hydrolysis of agave bagasse was carried out using a multilevel factorial experiment design. The independent variables were temperature, residence time and catalyst concentration and fermentable sugars (glucose, xylose and arabinose) as variable dependent (Table 2). As example, in the figs. 1 - 2 show the concentration profiles of fermentable sugars as function of reaction time using 1% and 2 % of sulfuric acid as catalyst at various reaction temperatures. As expected, a low temperature fermentable sugars concentration is small with a maximum production of sugars about 2.5 g/L for 1% catalyst concentration and of 10 g/L for 2% catalyst concentrate of liquor; the hydrolysis rate becomes significant at greater temperatures with maximum sugar production of 25 g/L for 2% catalyst concentrate. In Fig. 3 are reported monosaccharides composition profiles for 1% concentration of catalyst and 200°C. It was observed that the glucose and xylose concentration reached a maximum value and then decreased with the reaction time. This suggests that decomposition reactions exist, conducting probably to furfural. They also show that there is a synergistic effect between temperature and the concentration of catalyst for both reactions: for the desired reaction that is the hydrolysis of cellulosic materials and for the undesirable reaction that is the decomposition of sugars. The optimal conditions for the hydrolysis of cellulosic materials were temperature 150ºC, 10 min of residence time and 2% of H2SO4 as catalyst.



Fermentable sugars yield was 200 g/ Kg of agave bagasse treated. The solid phase composition was quantified by quantitative acid hydrolysis. Results show solid remained were constituted in dry weight by cellulose (60%), insoluble lignin (25%), no hemicelluloses was found.
3.3 Hydrolysis in the second stage
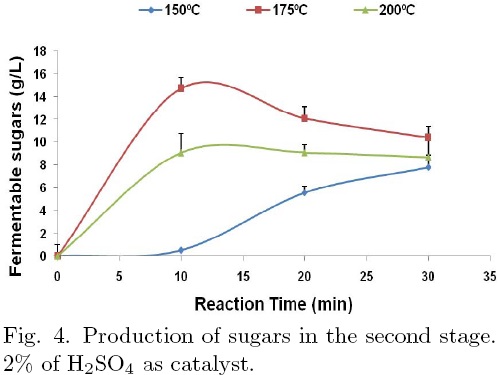
Solid remained from 1st stage of acid hydrolysis was exposed to 2nd stage of hydrolysis. Fig. 4 exhibits the results of the second hydrolysis stage, using again 2% de H2SO4 as catalyst. These results indicate that lower hydrolysis rates are observed, for hydrolysis of cellulose, which is more difficult to depolimerize than hemicellulose. However, the concentration profiles are similar to those observed during the first stage. The hydrolysis rate is greater as the temperature increases and a maximum sugars concentration of 14 g/L at 175ºC was observed, instead of that at 150ºC noticed in the first stage. Similarly, degradation of sugars is favored at higher temperatures and longer reaction times as it is show in the Fig. 5 for 2 % of catalyst and 175° C. Optimal conditions were 175ºC of temperature, 10 min and 2% of catalyst. Fermentable sugars yield was 84 g/Kg of agave bagasse. The Fig. 6 shows the furfural production profiles at optimal conditions for each step of hydrolysis: for high temperature and catalyst concentration the degradation of sugars is bigger.


Hydrolysate analysis show up that release of glucose and xylose were dependent on experimental operating conditions. The monosaccharides concentration (g/L) in first stage in optimal condition were of glucose (1.0), xylose (24.6) and arabinose (0.4) when reaction was carried out at 150ºC, reaction time 10 min and 2% of catalyst. Fermentable sugars yield was 200 g/Kg. Glucose concentration of 14 g/L was obtained with 2nd stage of acid hydrolysis when reaction time was 30 min, 175ºC, and 2 wt % sulfuric acid. Fermentable sugars yield was 84 g/Kg raw material. Total amount of fermentable sugars was 284 g/Kg of agave bagasse treated. Total yield was 40.98% over theoretical value.
3.5 Optimization
Mathematical models was developed to optimize the dilute- acid hydrolysis process of agave bagasse with sulfuric acid, an empirical model, Eq. (1), was used. The coefficient of this equation and the analysis of variance (ANOVA) were obtained using the software Statgraphics Centurion XV. The model found for Y was evaluated for the test-F with a significance level of 0.05. The analysis of variance demonstrates that the three studied variables have an individual and a synergetic effect on the hydrolysis. Eqs. (2)-(3) represent the empirical models found for the experimental conditions used in our study:


Where Y1 and Y2 are the total concentrations of sugars in the first and second stage, respectively; X1 is the reaction temperature, X2 is the reaction time and X3 is the concentration of H2SO4. Figs. 7-8 show the response surface for two hydrolysis stages. Fig. 5 indicates that the maximum concentration of fermentable sugars for the first stage was 26.8 g/L and 236 g/Kg dry matter, at 10 min, a reaction temperature of 151º C using 2wt % of sulfuric acid. In the other hand, Fig. 6 show a maximum of 15.0 g/L of fermentable sugars at 30 min, 180 ºC and 2 % of H2SO4 for the second stage and 90 g/Kg dry matter, was obtained. Total of fermentable sugars were 326 g/Kg and 48.5% respect of theoretical value. Maximum in the production of fermentable sugars was as expected since it is known that at high temperatures and long reaction times, the degradation of sugars to by-products, takes place.


Conclusions
The results from acid hydrolysis were obtained in two stages. The yield to fermentable sugars can be increased as high as 48.5% of theoretical value, obtaining a total concentration of 22.02 g/L and rendering 326 g/Kg of agave bagasse treated. In addition to hydrolysis, the decomposition of sugars, mainly glucose and xylose, was also observed and is related to the reaction temperature, acid concentration and reaction time. The optimization results indicate agave bagasse is a promising raw material for production of fermentable sugars.
References
Aguilar, R., Ramírez, J.A., Garrote, G. and Vazquez, M. (2002). Kinetic study of acid hydrolysis of sugar cane bagasse. Journal of Food Engineering 55, 309-318. [ Links ]
Aguilar-Rivera, N. y Canizalez-Leal, M.J. (2004). Cinética de la hidrolisis acida de la cascarilla de cebada. Revista Mexicana de Ingeniería Química 3, 257-263. [ Links ]
]]>Bozell, J.J., Moens, L., Elliott, D.C., Wang, Y., Neuenscwander, G.G., Fitzpatrick, S.W., Bilski, R.J. and Jarnefeld, J.L. (2000). Production of levulinic acid and use as a platform chemical for derived products. Resources Conservation and Recycling 28, 227-239. [ Links ]
Canettieri, E.V., de Moraes Rocha, G.J., de Carvalho Jr., J.A. and de Almeida and Silva, J.A. (2007). Optimization of acid hydrolysis from the hemicellulosic fraction of Eucalyptus grandis residue using response surface methodology. Bioresource Technology 98, 422-428. [ Links ]
Gámez, S., Ramírez, J.A., Garrote, G. and Vazquez, M. (2004. Manufacture of fermentable sugars solutions from sugar cane bagasse with phosphoric acid at atmospheric pressure. Journal of Agricultural and Food Chemistry 52, 4172-4177. [ Links ]
Garrote, G., Dominguez, G., and Parajo, J.C. (1999). Mild autohydrolysis: an environmentally friendly technology for xylooligosaccharide production from wood. Journal of Chemical Technology and Biotechnology 74, 1101-1109. [ Links ]
Herrera, A., Téllez-Luis, S.J., González-Cabriales, J.J., Ramirez, J.A. and Vazquez, M. (2004). Effect of the hydrochloric acid concentration on the hydrolysis of sorghum straw at atmospheric pressure. Journal of Food Engineering 63, 103-109. [ Links ]
]]>Herrera, A., Téllez-Luis, S.J., Ramorez, J.A., Voazquez, M. (2003). Production of xylose from sorghum straw using hydrochloric acid. Journal of Cereal Science 37, 267-274. [ Links ]
Iñiguez-Covarrubias, G. Lange, S.E. and Rowell, R.M. (2001). Utilization of byproducts from the tequila industry: part 1: agave bagasse as a raw material for animal feeding and fiberboard production. Bioresource Technology 77, 25-32. [ Links ]
Roberto, I.C., Mussato, S.I. and Rodriguez, R. (2003). Dilute-acid for optimization of xylose recovery from rice straw in a semi-pilot reactor. Industrial Crops and Products 17, 171-176. [ Links ]
Rocha, G.J.M. (2000). Deslignificao de bagaco de cana de acucar assistida por oxigenio. Sao Carlos, SP, Brazil: Instituto de Quimica de Sao Carlos-USP, PhD thesis, p. 136. [ Links ]
Rodriguez-Chong, A., Ramírez, J.A., Garrote, G. and Vazquez, M. (2004). Hydrolysis of sugar cane bagasse using nitric acid: a kinetic assessment. Journal of Food Engineering 61, 143-152. [ Links ]
]]>Sanchez, G., Pilcher, L., Rosalnder, C., Modig, T. Galbe, M., and Liden, G. (2004). Dilute-acid hydrolysis for fermentation of the Bolivian straw Paja Brava. Bioresource Technology 93, 249-256. [ Links ]
Sung, Y., and Cheng, J.J. (2005). Dilute acid pretreatment of rye straw and Bermuda grass for ethanol production. Bioresource Technology 96, 1599-1606. [ Links ]
Sung, Y., and Cheng, J. (2002). Hydrolysis of lignocellulosic materials for ethanol production: a review. Bioresource Technology 83, 1-11. [ Links ]
Téllez-Luis, S.J., Ramírez, J.A. and Vázquez, M. (2002). Modelling of the hydrolysis of sorghum straw at atmospheric pressure. Journal of the Science of Food and Agriculture 82, 505-512. [ Links ]
Vázquez, M., Oliva, M., Téllez-Luis, S.J. and Ramorez, J.A. (2007). Hydrolysis of sorghum straw using phosphoric acid: Evaluation of furfural production. Bioresource Technology 98, 3053-3060. [ Links ]
]]>Werpy, T. and Petersen, G. (2004). Top value added chemicals from biomass volume I: Results of screening for potential candidates from sugars and synthesis gas. Available at: http://www1.eere.energy.gov/bioenergy/pdfs/35523.pdf. Accessed: July 14, 2009. [ Links ]
Wyman, C.E. (1994). Ethanol from lignocellulosic biomass: technology, economics and opportunities. Bioresource Technology 50, 3-16. [ Links ]
]]>