Uso de tacrolimus en pediatría
Use of tacrolimus in pediatrics
M. en C. Herlinda Reyes–Pérez1, Dra. Mara Medeiros–Domingo2
1 Laboratorio de Farmacología
2 Departamento de Nefrología, Hospital lnfantil de México Federico Gómez, México, D.F., México,
]]>Resumen
El tacrolimus es un fármaco inmunosupresor inhibidor de calcineurina utilizado principalmente para prevenir el rechazo de órganos sólidos. También se emplea como inmunosupresor de segunda línea en otras enfermedades de origen inmunológico entre las que destacan: síndrome nefrótico, lupus eritematoso sistémico, dermatitis atópica, vitíligo y psoriasis entre otras.
En este artículo se revisa la farmacocinética, monitoreo terapéutico, efectos adversos y toxicidad del tacrolimus en pacientes pediátricos, haciendo énfasis en los padecimientos en los que ha demostrado utilidad.
Palabras clave. Tacrolimus; farmacocinética; monitoreo terapéutico; trasplante.
Abstract
Tacrolimus is a calcineurin inhibitor immunosuppressive agent used to prevent rejection in solid organ transplantation. It has also been used in several immunological diseases including nephrotic syndrome, systemic lupus eritematosus, atopic dermatitis, vitiligo and psoriasis.
This article is a review of tacrolimus pharmacokinetics, therapeutic monitoring, adverse effects, toxicity and clinical use in children.
Key words. Tacrolimus; pharmacokinetics; drug monitoring; transplant.
]]>Apoyado por Fondo Sectorial Salud–2004–c01–193
Solicitud de sobretiros:
Dra. Mara Medeiros Domingo,
Departamento de Nefrología, Hospital Infantil de México Federico Gómez,
Calle Dr. Márquez # I 62, Col. Doctores,
C.P. 06720, México, D.F.,
México.
]]> Fecha de recepción: 29–03–2006.
Información General sobre el tacrolimus
El tacrolimus (FK506) es un compuesto macrólido, altamente lipofílico y con peso molecular de 804. Es insoluble en agua pero altamente soluble en solventes orgánicos, tiene una intensa actividad inmunosupresora. Se emplea principalmente para prevenir y tratar el rechazo de órganos sólidos, pero también en otras enfermedades de origen auto inmune.1,2
Presentaciones farmacéuticas
Formulación intravenosa: contiene tacrolimus 5 mg/ mL, alcohol y un surfactante. Debe diluirse en dextrosa o solución salina y se recomienda administrar en infusión continua para reducir su nefrotoxicidad.
Formulación oral: cápsulas de 1 y 5 mg de tacrolimus en hidroxipropilmetilcelulosa.
Ungüento: 1 g de pomada al 0.1% y 0.03% contienen 1 mg y 0.3 mg de tacrolimus monohidratado respectivamente, en aceite mineral, parafina, propilencarbonato, petrolato y cera blanca.
]]>Mecanismo de acción
El tacrolimus inhibe la activación de los linfocitos–T al unirse a la proteína intracelular FKBP12 (proteína fijadora de FK 12). Se forma un complejo tacrolimus–FKBP12, calcio, calmodulina y calcineurina que subsecuentemente inhibe la actividad fosfatasa de la calcineurina y previene la fosforilación y translocación del factor nuclear de células T activadas (NFAT), un componente nuclear que inicia la transcripción de genes de linfocinas tales como interleucina 2 (IL–2) e interferón gama
Métodos de determinación
Los parámetros farmacocinéticos del tacrolimus dependen de la matriz que se emplee para su determinación, ya que existe diferencia en la concentración sangre/plasma. Por esto se prefiere la determinación en sangre total tomada en tubo con EDTA. El tacrolimus es estable en sangre total aproximadamente un año a –70° C, por al menos dos semanas a 4° C y 22° C y al menos dos a tres días a 37° C.5
El método que con mayor frecuencia se utiliza en la práctica clínica cotidiana es el MEIA (inmunoensayo de micropartículas ligado a enzima). La especificidad analítica depende del anticuerpo y de las condiciones preanalíticas (modo de extracción, volumen y tipo de precipitante, pH). Ninguno de los anticuerpos empleados en MEIA es específico para el compuesto original y todos tienen reacción cruzada con diversos metabolitos. También se ha empleado el inmunoensayo enzimático multiplicado cuantitativo (EMIT), ambos métodos tienen un valor diagnóstico similar.6
Recientemente se dio a conocer otro método, el de cromatografía líquida de alta presión (HPLC) acoplado a masas que podría ser intercambiable con el MEIA, es exacto y muy práctico, ya que puede leer muestras de sangre tomada de la punta del dedo de la mano; sin embargo su costo es aún demasiado alto.7
Farmacocinética
]]> Absorción. El tacrolimus se absorbe rápidamente, alcanzando el pico máximo en 0.5–1 hora, sin embargo algunos pacientes con trasplante hepático pueden tener el pico a las dos horas.8 En la absorción influye la baja solubilidad del medicamento y la motilidad gastrointestinal. La biodisponibilidad es pobre y muy variable, de 4–89% (=25%) y se ve reducida en presencia de alimentos, por esto se recomienda administrar en forma consistente ya sea en ayunas o con el mismo tipo de alimentos. Esta variabilidad se ha visto en diversas poblaciones de pacientes sin importar el tipo de órgano trasplantado.4Distribución. Se distribuye en la mayoría de los tejidos. Atraviesa la placenta con concentraciones en plasma del cordón umbilical de un tercio de las encontradas en el plasma materno y se encuentra presente en la leche materna en niveles similares a los reportados en plasma.9
Se sabe que la distribución de tacrolimus depende parcialmente de la unión reversible a proteínas del plasma (albúmina, α1– glicoproteína) y al hematócrito. Las concentraciones plasmáticas de albúmina y α1glicoproteína así como el conteo de eritrocitos se incrementan levemente durante los primeros meses post–trasplante y pueden contribuir a alteraciones en la fracción libre de tacrolimus en plasma y por lo tanto a la distribución y eliminación.
Metabolismo. Se lleva a cabo vía el citocromo CYP 3 A4 intestinal y hepático siendo la hidroxilación y la desmetilación las principales rutas metabólicas. Se han detectado por lo menos 15 metabolitos, siendo los principales el 13–O–desmetil y 15–O–desmetil tacrolimus.4 Ninguno de ellos presenta efecto inmunosupresor apreciable en humanos.
El tacrolimus también es sustrato de la glicoproteína P (P–gp), producto del gen de resistencia a fármacos (MDR1). La P–gp es una bomba de salida dependiente de adenosintrifosfato (ATP) que contribuye a la protección del cuerpo de toxinas ambientales, limitando su absorción a partir del lumen gastrointestinal o incrementando su excreción biliar y urinaria. Varios estudios han reportado que la farmacocinética de tacrolimus está inversamente relacionada con la cantidad de MDR1 en el intestino.10,12 Existe una biodisponibilidad reducida en pacientes con actividad anormalmente alta de P–gp intestinal10 y se ha reportado una biodisponibilidad incrementada en pacientes pediátricos con trasplante hepático, que cursaron con episodios diarreicos relacionados con supresión de CYP3A y P–gp debida a citocinas liberadas por virus o bacterias lo que incrementó la absorción.13 Estudios recientes sugieren que gran parte de la variabilidad asociada con tacrolimus es debida a diferencias genéticas en la expresión de CYP3A4 y P–gp,14 esto incluye las diferencias étnicas, ya que existe evidencia de que afroamericanos y latinos tienen menor biodisponibilidad de tacrolimus que los caucásicos.15
Excreción. La vida media de eliminación varía de 12–19 horas.16 La principal vía de eliminación es la biliar (más de 90% de la dosis de tacrolimus se elimina en la bilis) y menos de 1% de la dosis es excretada inalterada en la orina.4,17
Farmacocinética en pacientes pediátricos
Dado que la población pediátrica se distingue por numerosas diferencias fisiológicas relacionadas con la composición corporal (relación agua/grasa), proteínas plasmáticas y actividad metabólica, el comportamiento farmacocinético del tacrolimus es diferente en esta población.
Las rutas oxidativas catalizadas por el sistema enzimático del citocromo P 450 están inmaduras durante los primeros meses de vida y necesitan de 6 a 12 meses para alcanzar una actividad metabólica importante, la cual a esa edad podría ser más alta que el promedio de actividad en los adultos. Esta maduración gradual de la actividad de numerosos sistemas enzimáticos durante los primeros meses de vida contribuye significativamente a la gran variabilidad interindividual en la velocidad de eliminación en pacientes pediátricos de diferentes edades.18
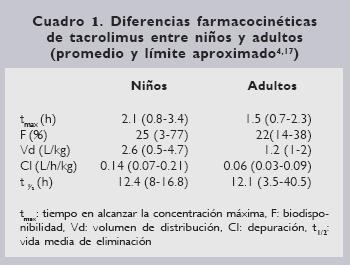
]]> Algunos estudios han reportado que los niños requieren de dosis hasta cinco veces más altas para alcanzar concentraciones similares a las de los adultos.19 Las diferencias en las dosis administradas en pacientes pediátricos y adultos para alcanzar concentraciones mínimas comparables en sangre, se explica por las diferencias en depuración y distribución,20 ya que la depuración del tacrolimus en niños es más rápida y tienen un mayor volumen de distribución (Cuadro 1).
Efectos adversos y toxicidad
Las reacciones adversas más frecuentes del tacrolimus vía oral e intravenoso son temblor, cefalea, diarrea, náusea, parestesias, dispepsia e hipertensión. Cuando se administra en forma tópica puede presentarse sensación de quemazón y picor, enrojecimiento, dolor, sensación de calor, aumento de la sensibilidad de la piel (calor y frío), hormigueo, exantema, foliculitis e infecciones virales, acné, dolor de cabeza y dermatitis tipo rosácea.
Los efectos adversos y toxicidad se detallan en el cuadro 2, generalmente mejoran al disminuir la dosis, también puede ayudar cambiar a un intervalo de dosificación de cada ocho horas en vez de cada 12 horas, manteniendo la misma dosis en mg/kg/día para evitar los picos elevados de concentración que son los que se han asociado con toxicidad,21–22 cuando los efectos adversos comprometen la rehabilitación del paciente como en el caso de diabetes mellitus, se ha recomendado el cambio a otro esquema de inmunosupresión –por ejemplo ciclosporina.
En pacientes con trasplante renal cobra particular interés la nefrotoxicidad, que puede manifestarse como trastornos tubulares incluyendo acidosis tubular renal23 hasta la falla renal aguda con disminución de la velocidad de filtración glomerular y retención de azoados, generalmente la nefrotoxicidad mejora al disminuir o suspender el medicamento. Se ha propuesto el uso de teofilina en los casos de insuficiencia renal aguda oligúrica que no responde al uso de diuréticos. También se ha propuesto el uso de fenobarbital, como inductor enzimático en casos de intoxicación por tacrolimus.24
Interacciones medicamentosas
]]> El tacrolimus puede interactuar con otros fármacos inductores e inhibidores del CYP3 A424,39 y así disminuir o aumentar su biodisponibilidad, algunos medicamentos pueden incrementar el riesgo de nefrotoxicidad (Cuadro 3).
Influencia del tiempo post'trasplante
Varios estudios en adultos han reportado una disminución en la dosis de tacrolimus requerida para mantener concentraciones mínimas similares al incrementarse el tiempo post–trasplante. Entre ellos se encontró en 50 adultos trasplantados reducción de las proporciones dosis/concentración de tacrolimus después del primero y tercer mes de tratamiento. A los dos años la disminución de la dosis de tacrolimus fue reducida hasta 50%, las razones postuladas para esto son biodisponibilidad incrementada, reducción en la dosis de corticosteroides y concentraciones incrementadas de hematócrito y albúmina.40–42 En el caso de niños esta influencia no está del todo clara ya que en un estudio retrospectivo con pacientes con trasplante de hígado, la dosis de tacrolimus en el primer mes de trasplante fue mayor que la requerida tres a cinco años después; mientras que otros investigadores no han observado cambios significativos en el primer año.19
Monitoreo terapéutico
Actualmente el monitoreo terapéutico del tacrolimus se lleva a cabo midiendo las concentraciones mínimas también conocidas como niveles en valle; sin embargo, esto ha sido cuestionado al presentarse algunos casos de toxicidad y rechazo aún cuando las concentraciones mínimas estaban dentro del límite considerado como aceptable. Diversos estudios43–47 han demostrado que existe buena correlación entre la concentración en sangre y el área bajo la curva de concentración plasmática vs tiempo (ABC), y que dicha correlación se puede mejorar mediante el uso de diferentes tiempos de muestreo. Puede darse el caso de que el régimen de muestreo óptimo variara de acuerdo al tipo de trasplante, tiempo post–trasplante u otros factores.
En pacientes con trasplante renal después de una dosis promedio de tacrolimus de 0.16 mg/kg/ día, el ABC fue de 104 mg*h/L, mientras que en trasplante hepático, con una dosis promedio de 0.3 mg/kg/día, el ABC fue de 252 mg*h/L. Se ha encontrado correlación entre ABC y la concentración mínima de tacrolimus en sangre total (Cmin) después de la primera dosis oral (r =0.90) y el estado estacionario (r =0.83), lo que sugiere que la Cmin de tacrolimus es un buen indicador de la exposición sistémica. En pacientes con trasplante hepático la depuración parece incremen' tarse con el tiempo: para un niño de 5 kg la depuración se incrementa de 0.163 (en el día 0) a 0.223 L/h/kg (en el día 60), lo cual significa que podría ser necesario en ciertos pacientes pediátricos incrementar gradualmente la dosis de tacrolimus después del trasplante. En tanto que puede disminuir cuando se presenta infección por hepatitis C.17 En trasplante de pulmón se ha propuesto cuantificar al menos dos puntos entre cero y cuatro horas, para una estimación adecuada de ABC.48
]]> Los niveles en valle objetivo de tacrolimus varían de acuerdo al tipo de trasplante, tiempo post–trasplante y si se emplean otros inmunosupresores (Cuadro 4).49 En pacientes con trasplante renal también se emplea como terapia de rescate para el rechazo resistente a esteroides.En el caso de pacientes con trasplante de corazón se ha reportado que la incidencia de rechazo agudo es menor cuando los niveles en valle de tacrolimus el primer mes post–trasplante son mayores a 8 ng/mL.50
Modelos de muestreo limitado en pacientes pediátricos Actualmente no existen reportes de modelos de muestreo limitado de tacrolimus en pacientes pediátricos tal y como lo existe para ciclosporina.51 Varios estudios realizados en adultos han propuesto algoritmos de muestreo limitado para predecir el área bajo la curva a las 12 horas (ABC12). Entre ellos un estudio de 12 pacientes con trasplante de hígado en donde se calculó el ABC con cuatro puntos (1, 2.5, 6 y 9 horas) encontrándose una r2 de 0.99 o una con tres puntos (1, 4 y 12 horas) con una r2 de 0.92 que predijo mejor el ABC12.52 En tanto que en un estudio con pacientes con trasplante renal el ABC12 fue mejor predicha usando algoritmos que incluyeron tiempos de muestreo de cinco horas (C5) con r2de 0.915, una combinación de 5 (C5) y dos horas (C2) con r2 de 0.983 y una combinación de cinco (C5), dos (C2) y una horas (C1 con r2 de 0.997,53 también se ha propuesto el monitoreo del ABC 0 h del tacrolimus durante el período de inducción después del trasplante renal.54 No se ha encontrado correlación entre la dosis por superficie de área corporal y el ABC (r2 =0.098) en niños bajo tratamiento con tacrolimus, pero sí hay una buena correlación entre el ABC y la concentración a las dos horas C2 (r =0.8573), cuatro horas C4 (r =0.9120), y a las seis horas (r =0.9034).55
Uso en padecimientos diferentes del trasplante de órganos sólidos
Artritis reumatoide. Recientemente se llevó a cabo un estudio multicéntrico, doble ciego fase IV donde se evaluó el tacrolimus como monoterapia versus placebo durante seis meses, obteniendo mejor respuesta en los pacientes que habían mostrado intolerancia a los fármacos antirreumáticos que los que habían mostrado resistencia a éstos.56
Síndrome nefrótico corticorresistente. El tacrolimus se ha empleado con éxito en series pequeñas de niños y adultos con síndrome nefrótico resistente al tratamiento con esteroides. Loeffler y col.57 estudiaron 16 niños que recibieron tacrolimus a dosis de 0.1 mg/kg/día dividido en dos dosis, obteniendo 81% de remisión completa en los primeros dos meses post–tratamiento y 13.4% de remisión parcial. Se requieren más estudios para determinar si en realidad es menos tóxico que la ciclosporina y cuál es la evolución a largo plazo de los pacientes tratados con esta terapia.58–60
Lupus eritematoso. Recientemente se publicó un estudio piloto en nueve pacientes con nefropatía lúpica clase IV que recibieron tratamiento con tacrolimus y prednisolona por seis meses, con remisión completa de la proteinuria en 67% y parcial en 13%, sin embargo aún queda por definir el tiempo de tratamiento y si los resultados son comparables a la terapia estándar que consiste en ciclofosfamida y esteroides,61 se considera una buena alternativa terapéutica cuando los pacientes no pueden recibir el tratamiento convencional.62
Padecimientos dermatológicos. El tacrolimus tópico ha resultado útil en el manejo de una gran variedad de enfermedades de la piel entre las que destaca la dermatitis atópica;63,64 Koo y col.65 reportaron recientemente los resultados obtenidos en 8 000 pacientes con esta enfermedad tratados con tacrolimus al 0.3 y 0.1% dos veces al día, encontrando una disminución de 52% del área afectada al mes de tratamiento y de 91% a los 18 meses, ofreciendo una alternativa terapéutica en pacientes pediátricos.66
También se ha empleado para tratamiento de vitíligo en niños con respuesta que varía de 63 a 89% dependiendo del sitio afectado, la concentración de tacrolimus empleada y el tiempo de tratamiento.67,68
]]> Existen reportes de su utilidad en el tratamiento de psoriasis cuando se combina el tacrolimus al 0.1% con gel de ácido salicílico al 6%.69En niños existe un estudio de casos retrospectivo en donde se evaluó la eficacia de tacrolimus 0.1% en psoriasis inversa, en donde 12 de 13 pacientes tuvieron una limpieza completa de sus lesiones dos semanas después de iniciar el tratamiento.70También se ha empleado en pacientes con dermatitis de contacto,71 rosácea,72,73 dermatitis seborreica74,75 y liquen plano.76
Conclusiones
El tacrolimus es un potente inmunosupresor que ha demostrado su utilidad en diversos padecimientos pediátricos entre los que destacan los trasplantes de órganos sólidos y la dermatitis atópica. Por su estrecho margen de seguridad se recomienda el monitoreo terapéutico con la determinación de los niveles en valle en sangre total. Los niños tienen mayor variabilidad farmacocinética que los adultos, probablemente relacionada a las diferencias en depuración hepática según el grupo de edad, y en general requieren mayor dosis en mg/ kg para obtener valores sanguíneos similares a los del adulto. Se requieren más estudios farmacocinéticos en este grupo de edad.
Referencias
1. Pollock–Barziv SM, Dipchand Al, McCrindle BW, Nalli N, West LJ. Randomized clinical trial of tacrolimus vs cyclosporine–based immunosuppression in pediatric heart transplantation: preliminary results at 15–month follow–up. J Heart Lung Transplant. 2005; 24: 190–4. [ Links ]
2. Peura JL, Zile MR, Feldman DS, VanBakel AB, McClure C, Uber W, et al. Effects of conversion from cyclosporine to tacrolimus on left ventricular structure in cardiac allograft recipients. J Heart Lung Transplant. 2005; 24: 1969–72. [ Links ]
3. Fung JJ. Tacrolimus and transplantation: a decade in review. Transplantation. 2004; 77: S41–3. [ Links ]
4. Venkataramanan R, Swaminathan A, Prasad T, Jain A, Zuckerman S, Warty V, et al. Clinical pharmacokinetics of tacrolimus. Clin Pharmacokinet. 1995; 29: 404–30. [ Links ]
5. Ingels SC, Koenig J, Scott MG. Stability of FK506 (tacrolimus) in whole–blood specimens. Clin Chem. 1995; 41: 1320–1. [ Links ]
6. Bartlomiejczyk I, Zochowska D, Sanko–Resmer J, Matuszewicz D, Paczek L. Therapeutic monitoring of tacrolimus concentrations in blood of renal and liver transplant recipients: comparison of microparticle enzyme immunoassay and enzyme multiplied immunoassay methods. Transplant Proc. 2006; 38: 94–6. [ Links ]
7. Webb NJ, Roberts D, Preziosi R, Keevil BG. Fingerprick blood samples can be used to accurately measure tacrolimus levels by tandem mass spectrometry. Pediatr Transplant. 2005; 9: 729–33. [ Links ]
8. Jusko WJ, Piekoszewski W, Klintmalm GB, Shaefer MS, Hebert MF, Piergies AA, et al. Pharmacokinetics of tacrolimus in liver transplant patients. Clin Pharmacol Ther. 1995; 57: 281–90. [ Links ]
9. French AE, Soldin SJ, Soldin OP, Koren G. Milk transfer and neonatal safety of tacrolimus. Ann Pharmacother. 2003; 37: 815–8. [ Links ]
10. Kaplan B, Lown K, Craig R, Abecassis M, Kaufman D, Leventhal J, et al. Low bioavailability of cyclosporine microemulsion and tacrolimus in a small bowel transplant recipient: possible relationship to intestinal P–glycoprotein activity. Transplantation. 1999; 67: 333–5. [ Links ]
11. Masuda S, Uemoto S, Goto M, Fujimoto Y, Tanaka K, Inui K. Tacrolimus therapy according to mucosal MDRI levels in small–bowel transplant recipients. Clin Pharmacol Ther. 2004; 75: 352–61. [ Links ]
12. Hashida T, Masuda S, Uemoto S, Saito H, Tanaka K, Inui K. Pharmacokinetic and prognostic significance of intestinal MDRI expression in recipients of living–donor liver transplantation. Clin Pharmacol Ther. 2001; 69: 308–16. [ Links ]
13. Maezono S, Sugimoto K, Sakamoto K, Ohmori M, Hishikawa S, Mizuta K, et al. Elevated blood concentrations of calcineurin inhibitors during diarrheal episode in pediatric liver transplant recipients: involvement of the suppression of intestinal cytochrome P450 3A and P–glyco–protein. Pediatr Transplant. 2005; 9: 315–23. [ Links ]
14. Staatz CE, Tett SE. Clinical pharmacokinetics and pharmacodynamics of tacrolimus in solid organ transplantation. Clin Pharmacokinet. 2004; 43: 623–53. [ Links ]
15. Mancinelli LM, Frassetto L, Floren LC, Dressier D, Carrier S, Bekersky I, et al. The pharmacokinetics and metabolic disposition of tacrolimus: a comparison across ethnic groups. Clin Pharmacol Ther. 2001; 69: 24–31. [ Links ]
16. Plosker GL, Foster RH. Tacrolimus: a further update of its pharmacology and therapeutic use in the management of organ transplantation. Drugs. 2000; 59: 323–89. [ Links ]
17. Wallemacq PE, Verbeeck RK. Comparative clinical pharmacokinetics of tacrolimus in paediatric and adult patients. Clin Pharmacokinet. 2001; 40: 283–95. [ Links ]
18. Kearns GL, Abdel–Rahman SM, Alander SW, Blowey DL, Leeder JS, Kauffman RE. Developmental pharmacology—drug disposition, action, and therapy in infants and children. N Engl J Med. 2003; 349: 1157–67. [ Links ]
19. McDiarmid SV, Colonna JO 2nd, Shaked A, Vargas J, Ament ME, Busuttil RW. Differences in oral FK506 dose requirements between adult and pediatric liver transplant patients. Transplantation. 1993; 55: 1328–32. [ Links ]
20. Filler G, Grygas R, Mai I, Stolpe HJ, Greiner C, Bauer S, et al. Pharmacokinetics of tacrolimus (FK 506) in children and adolescents with renal transplants. Nephrol Dial Transplant. 1997; 12: 1668–71. [ Links ]
21. KhalafH, Al–Asseri A, Bhuiyan J, Nafea O, Al–Sebayel M. Tacrolimus (FK 506) given three times daily after liver transplantation for minimizing nephrotoxicity and neurotoxicity. Transplant Proc. 2003; 35: 2787–8. [ Links ]
22. Mollison KW, Fey TA, Krause RA, Andrews JM, Bretheim PT, Cusick PK, et al. Nephrotoxicity studies of the immunosuppressant tacrolimus (FK506) and ascomycin in rat models. Toxicology. 1998; 125: 169–81. [ Links ]
23. Ogita K. Renal tubular acidosis secondary to FK506 in living donor liver transplantation: A case report. Asian J Surg. 2003; 26: 218–20. [ Links ]
24. Quiros–Tejeira RE, Chang IF, Bristow LJ, Karpen SJ, Goss JA. Treatment of acute tacrolimus whole–blood elevation with phenobarbital in the pediatric liver transplant recipient. Pediatr Transplant. 2005; 9: 792–6. [ Links ]
25. Dresser GK, Spence JD, Bailey DG. Pharmacokinetic–pharmacodynamic consequences and clinical relevance of cytochrome P450 3A4 inhibition. Clin Pharmacokinet. 2000; 38: 41–57. [ Links ]
26. Banerjee R, Leaver N, Lyster H, Banner NR Coadministration of itraconazole and tacrolimus after thoracic organ transplantation. Transplant Proc. 2001; 33: 1600–2. [ Links ]
27. Christians U, Jacobsen W, Benet LZ, Lampen A. Mechanisms of clinically relevant drug interactions associated with tacrolimus. Clin Pharmacokinet. 2002; 41:813–51. [ Links ]
28. Tada H, Yanagiwara S, Ito K, Suzuki T. Role of diltiazem on tacrolimus pharmacokinetics in tacrolimus–induced nephrotoxic rats. Pharmacol Toxicol. 1999; 84: 241–6. [ Links ]
29. Toda F, Tanabe K, Ito S, Shinmura H, Tokumoto T, Ishida H, et al. Tacrolimus trough level adjustment after administration of fluconazoleto kidney recipients. Transplant Proc. 2002; 34: 1733–5. [ Links ]
30. van Gelder T. Drug interactions with tacrolimus. Drug Saf. 2002; 25: 707–12. [ Links ]
31. Buck ML. Pharmacokinetic changes during extracorporeal membrane oxygenation: implications for drug therapy of neonates. Clin Pharmacokinet. 2003; 42: 403–17. [ Links ]
32. Page RL 2nd, Klem PM, Rogers C. Potential elevation of tacrolimus trough concentrations with concomitant metronidazole therapy. Ann Pharmacother. 2005; 39: 1109–13. [ Links ]
33. Shitrit D, Ollech JE, Ollech A, Bakal I, Saute M, Sahar G, et al. Itraconazole prophylaxis in lung transplant recipients receiving tacrolimus (FK 506): efficacy and drug interaction. J Heart Lung Transplant. 2005; 24: 2148–52. [ Links ]
34. Floren LC, Bekersky I, Benet LZ, Mekki Q, Dressier D, Lee JW, et al. Tacrolimus oral bioavailability doubles with coadministration of ketoconazole. Clin Pharmacol Ther. 1997; 62: 41–9. [ Links ]
35. Hebert MF, Fisher RM, Marsh CL, Dressier D, Bekersky I. Effects of rifampin on tacrolimus pharmacokinetics in healthy volunteers. J Clin Pharmacol. 1999; 39: 91–6. [ Links ]
36. Hebert MF, Park JM, Chen YL, Akhtar S, Larson AM. Effects of St. John's wort (Hypericum perforation) on tacrolimus pharmacokinetics in healthy volunteers. J Clin Pharmacol. 2004; 44: 89–94. [ Links ]
37. Jones TE, Morris RG. Pharmacokinetic interaction between tacrolimus and diltiazem: dose–response relationship in kidney and liver transplant recipients. Clin Pharmacokinet. 2002; 41: 381–8. [ Links ]
38. Ocran KW, Plauth M, Mai I, Lochs H. Tacrolimus toxicity due to drug interaction with mibefradil in a patient after liver transplantation. Z Gastroenterol. 1999; 37: 1025–8. [ Links ]
39. Moreau C, Taburet AM, Furlan V, Debray D, Loriot MA. Interaction between tacrolimus and omeprazole in a pediatric liver transplant recipient. Transplantation. 2006; 81: 487–8. [ Links ]
40. Hu RH, Lee PH, Tsai MK. Clinical influencing factors for daily dose, trough level, and relative clearance of tacrolimus in renal transplant recipients. Transplant Proc. 2000; 32: 1689–92. [ Links ]
41. Undre NA, Schafer A. Factors affecting the pharmacokinetics of tacrolimus in the first year after renal transplantation. European Tacrolimus Multicentre Renal Study Group. Transplant Proc. 1998; 30: 1261–3. [ Links ]
42. Christiaans M, van Duijnhoven E, Beysens T, Undre N, Schafer A, van Hooff J. Effect of breakfast on the oral bioavailability of tacrolimus and changes in pharmacokinetics at different times posttransplant in renal transplant recipients. Transplant Proc. 1998; 30: 1271–3. [ Links ]
43. Braun F, Schutz E, Peters B, Talaulicar R, Grupp C, Undre N, et al. Pharmacokinetics of tacrolimus primary immunosuppression in kidney transplant recipients. Transplant Proc. 2001; 33: 2127–8. [ Links ]
44. Cantarovich M, Fridell J, Barkun J, Metrakos P, Besner JG, Deschenes M, et al. Optimal time points for the prediction ofthe area–under–the–curve in liver transplant patients receiving tacrolimus. Transplant Proc. 1998; 30: 1460–1. [ Links ]
45. Jorgensen KA, Povlsen JV, Madsen S, Madsen M, Hansen HE, Pedersen AR, et al. Two–hour blood tacrolimus levels are not superior to trough levels as estimates of the area underthe curve in tacrolimus–treated renal transplant patients. Transplant Proc. 2002; 34: 1721–2. [ Links ]
46. Jorgensen K, Povlsen J, Madsen S, Madsen M, Hansen H, Pedersen A, et al. C2 (2–h) levels are not superior to trough levels as estimates of the area under the curve in tacrolimus–treated renal–transplant patients. Nephrol Dial Transplant. 2002; 17: 1487–90. [ Links ]
47. Pisitkun T, Eiam–Ong S, Chusil S, Praditpornsilpa K, Pansin P, Tungsanga K. The roles of C4 and AUCO–4 in monitoring of tacrolimus in stable kidney transplant patients. Transplant Proc. 2002; 34: 3 173–5. [ Links ]
48. Ragette R, Kamler M, Weinreich G, Teschler H, Jakob H. Tacrolimus pharmacokinetics in lung transplantation: new strategies for monitoring. J Heart Lung Transplant. 2005; 24: 1315–9. [ Links ]
49. Di Filippo S, Zeevi A, Me Dade K, Bastien O, Webber S. Impact of TGFb1 gene polymorphisms on acute and chronic rejection in pediatric heart transplant allografts. Transplantation. 2006; 81: 934–9. [ Links ]
50. Albornoz–Lopez R, Aumente–Rubio MD, Arizon–del Prado JM, Cardenas– Aranzana M, Lopez–Malo de Molina MD, Gago–Sanchez Al. [Tacrolimus blood levels and incidence of graft rejection in heart transplantation]. Farm Hosp. 2005; 29: 158–63. [ Links ]
51. Medeiros M, Pérez–Urizar J, Muñoz R, Castañeda–Hernández G. Limited sampling model for area–under–the–curve monitoring in pediatric patients receiving either Sandimmune or Neoral cyclosporin A oral formulations. Pediatr Transplant. 1999; 3: 225–30. [ Links ]
52. Ku YM, Min DI. An abbreviated area–under–the–curve monitoring for tacrolimus in patients with liver transplants. Ther Drug Monit. 1998; 20: 2 19–23. [ Links ]
53. Stolk LM, van Duijnhoven EM, Christiaans MH, van Hooff JP. Trough levels of tacrolimus. Ther Drug Monit. 2002; 24: 573; author reply 573–4. [ Links ]
54. Uchida K, Tominaga Y, Haba T, Katayama T, Matsuoka S, Sato T, et al. Usefulness of monitoring of AUC(0–4h) during the induction period of immunosuppressive therapy with tacrolimus after renal transplantation. Transplant Proc. 2002; 34: 1736–7. [ Links ]
55. Filler G, Feber J, Lepage N, Weiler G, Mai I. Universal approach to pharmacokinetic monitoring of immunosuppressive agents in children. Pediatr Transplant. 2002; 6: 411–8. [ Links ]
56. Yocum DE, Furst DE, Bensen WG, Burch FX, Borton MA, Mengle–Gaw LJ, et al. Safety of tacrolimus in patients with rheumatoid arthritis: long–term experience. Rheumatology (Oxford). 2004; 43: 992–9. [ Links ]
57. Loeffler K, Gowrishankar M, Yiu V. Tacrolimus therapy in pediatric patients with treatment–resistant nephrotic syndrome. Pediatr Nephrol. 2004; 19: 281–7. [ Links ]
58. Meyrier A. Treatment of focal segmental glomerulosclerosis. Expert Opin Pharmacother. 2005; 6: 1539–49. [ Links ]
59. Tang S, Tang AW, Tarn MK, Ho YW. Use of tacrolimus in steroid– and cyclophosphamide–resistant minimal change nephrotic syndrome. Am J Kidney Dis. 2003; 42: E13–5. [ Links ]
60. Duncan N, Dhaygude A, OwenJ, Cairns TD, Griffith M, McLean AG, et al. Treatment of focal and segmental glomerulosclerosis in adults with tacrolimus monotherapy. Nephrol Dial Transplant. 2004; 19: 3062–7. [ Links ]
61. Mok CC, Tong KH, To CH, Siu YP, Au TC. Tacrolimus for induction therapy of diffuse proliferative lupus nephritis: an open–labeled pilot study. Kidney Int. 2005; 68: 813–7. [ Links ]
62. Politt D, Heintz B, FloegeJ, Mertens PR. Tacrolimus– (FK 506) based immunosuppression in severe systemic lupus erythematosus. Clin Nephrol. 2004; 62: 49–53. [ Links ]
63. Simpson D, Noble S. Tacrolimus ointment: a review of its use in atopic dermatitis and its clinical potential in other inflammatory skin conditions. Drugs. 2005; 65: 827–58. [ Links ]
64. Woo DK, James WD. Topical tacrolimus: a review of its uses in dermatology. Dermatitis. 2005; 16: 6–21. [ Links ]
65. Koo JY, Fleischer AB Jr., Abramovits W, Pariser DM, McCall CO, Horn TD, et al. Tacrolimus ointment is safe and effective in the treatment of atopic dermatitis: results in 8 000 patients. J Am Acad Dermatol. 2005; 53: S195–205. [ Links ]
66. Frohna JG. Efficacy and tolerability of topical pimecrolimus and tacrolimus in the treatment of atopic dermatitis: meta–analysis of randomised controlled trials. J Pediatr. 2005; 147: 126. [ Links ]
67. Kanwar AJ, Dogra S, Parsad D. Topical tacrolimus for treatment of childhood vitiligo in Asians. Clin Exp Dermatol. 2004; 29: 589–92. [ Links ]
68. Silverberg NB, Lin P, Travis L, Farley–Li J, Mancini AJ, Wagner AM, et al. Tacrolimus ointment promotes repigmentation of vitiligo in children: a review of 57 cases. J Am Acad Dermatol. 2004; 51: 760–6. [ Links ]
69. Carroll CL, Clarke J, Camacho F, Balkrishnan R, Feldman SR Topical tacrolimus ointment combined with 6% salicylic acid gel for plaque psoriasis treatment. Arch Dermatol. 2005; 141: 43–6. [ Links ]
70. Steele JA, Choi C, Kwong PC. Topical tacrolimus in the treatment of inverse psoriasis in children. J Am Acad Dermatol. 2005; 53: 713–6. [ Links ]
71. Saripalli YV, Gadzia JE, Belsito DV. Tacrolimus ointment 0.1% in the treatment of nickel–induced allergic contact dermatitis. J Am Acad Dermatol. 2003; 49: 477–82. [ Links ]
72. Goldman D. Tacrolimus ointment forthe treatment of steroid–induced rosacea: a preliminary report. J Am Acad Dermatol. 2001; 44: 995–8. [ Links ]
73. Bamford JT, Elliott BA, Haller IV. Tacrolimus effect on rosacea. J Am Acad Dermatol. 2004; 50: 107–8. [ Links ]
74. Braza TJ, DiCarlo JB, Soon SL, McCall CO. Tacrolimus 0.1 % ointment for seborrhoeic dermatitis: an open–label pilot study. Br J Dermatol. 2003; 148: 1242–4. [ Links ]
75. Meshkinpour A, Sun J, Weinstein G. An open pilot study using tacrolimus ointment in the treatment of seborrheic dermatitis. J Am Acad Dermatol. 2003; 49: 145–7. [ Links ]
76. Eisman S, Orteu CH. Recalcitrant erosive flexural lichen planus: successful treatment with a combination of thalidomide and 0.1 % tacrolimus ointment. Clin Exp Dermatol. 2004; 29: 268–70. [ Links ]
]]>