Enzymatic oxidation of volatile malodorous organosulfur compounds in a two-phase reactor
Oxidación enzimática de compuestos organoazufrados volátiles y de mal olor, utilizando un reactor de dos fases
Julio C. Cruz, Jesús García y Marcela Ayala*
Departamento de Biocatálisis e Ingeniería Celular. Instituto de Biotecnología, UNAM. Av. Universidad #2001, Col. Chamilpa, C.P. 62210, Cuernavaca, Morelos, México. E-mail: *maa@ibt.unam.mx
]]>Artículo recibido el 20 de enero de 2015.
Aceptado el 06 de mayo de 2015.
Abstract
In this work we report the oxidation of volatile organosulfur compounds (VOC) catalyzed by the enzyme chloroperoxidase from Caldariomyces fumago. VOC are regarded as atmospheric pollutants due to their unpleasant odor and low detection threshold. Diethyl sulfide, dimethyl disulfide, propanethiol, butanethiol and hexanethiol were found to be substrates for the enzyme in a liquid medium reaction at pH 6, under peroxidatic conditions. Product analysis showed that sulfides were oxidized to their respective sulfoxides whereas thiols were oxidized to the corresponding disulfides. The identified products showed significantly lower vapor pressure than the parental compounds; thus, the products are not considered atmospheric pollutants. A 70-mL two-phase reactor was assembled in order to determine the efficiency of the enzymatic treatment. The liquid phase, consisting of 15% organic solvent and 85% buffer, was contacted with the gaseous phase, consisting of a substrate-enriched air stream. Using dimethyl disulfide as model substrate, we found that only enzymatic oxidation occurred in this system; by controlling the enzyme and peroxide concentration, we found that the substrate is transferred to the aqueous phase where 1 mol of enzyme converted approximately 12,400 mol of substrate, thus highlighting the potential of enzymatic treatment of malodorous gaseous streams.
Key Words: Environmental biocatalysis, organosulfur compounds, chloroperoxidase, peroxidation, volatile substrates.
Resumen
En este trabajo reportamos la oxidación de una serie de compuestos organoazufrados volátiles (COV) catalizada por la enzima cloroperoxidasa obtenida del hongo Caldariomyces fumago. Los COV se consideran contaminantes atmosféricos debido a su olor desagradable y a su bajo umbral de detección. El sulfuro de etilo, disulfuro de dimetilo, propanotiol, butanotiol y hexanotiol fueron transformados por la enzima en un medio de reacción acuoso a pH 6 y en presencia de peróxido de hidrógeno. El análisis de los productos demostró que los sulfuros fueron oxidados a sus respectivos sulfóxidos, mientras que los tioles fueron oxidados a sus correspondientes disulfuros. Los productos identificados tienen una presión de vapor significativamente menor que los compuestos originales, por lo que son mucho menos volátiles y por tanto no se consideran contaminantes atmosféricos. Se ensambló un reactor de dos fases de 70 mL de volumen con el fin de determinar la eficiencia del tratamiento enzimático. La fase líquida, compuesta por 85% de amortiguador y 15% de solvente orgánico, se puso en contacto con la fase gaseosa, compuesta por aire enriquecido con el sustrato. Usando disulfuro de metilo como sustrato modelo, encontramos únicamente reacción enzimática en este sistema; al controlar la concentración de enzima y de peróxido en la fase líquida se logró transferir el sustrato a la fase acuosa en donde 1 mol de enzima convirtió aproximadamente 12,400 moles de sustrato, resaltando el potencial de los tratamientos enzimáticos para las corrientes gaseosas con mal olor por COV.
]]> Palabras Clave: Biocatálisis ambiental, compuestos organoazufrados, cloroperoxidasa, peroxidación, sustratos volátiles.
Introduction
Chloroperoxidase (CPO, EC 1.11.1.10) from Caldariomyces fumago is an attractive enzyme for application in several and diverse fields such as the fine chemical and pharmaceutical industry[1], diagnosis[2] as well as the oil-related industry[3] and environment remediation[4,5]. The versatility of CPO is determined by its ability to catalyze different reactions under controlled conditions, including halogenation (chlorination, bromination and iodination), dehydrogenation, oxygen insertion and peroxide dismutation. In addition, low substrate specificity allows CPO to catalyze the transformation of a wide range of compounds of different chemical nature[6]. One of the most interesting catalytic modality of CPO is the peroxygenation activity, which catalyzes the oxidation of olefins and sulfur compounds to generate epoxides and sulfoxides, respectively[1,6].
The broad specificity displayed by CPO towards organosulfur compounds is well established. The enantioselective generation of (R)-sulfoxides catalyzed by CPO has been thoroughly studied. The enzyme is able to catalyze the oxidation of β-carbonyl sulfides, cycloalkyl methyl sulfides, alkyl aryl sulfides, dialkyl sulfides and cyclic sulfides to their respective (R)-sulfoxides[7-9]. The enantioselectivity is generally favored in the absence of halogen ions and pH near neutrality (pH 6). On the other hand, under acidic conditions (pH 3) and in the presence of chloride, the enzyme catalyzes the oxidation of sulfides to the racemic mixture of sulfoxides[10]. Under these conditions, the sulfoxidation of aromatic and heterocyclic compounds has been reported[11]. Furthermore, significant enzymatic transformation of the sulfur-containing fraction in straight diesel fuel has been demonstrated, thus highlighting the ability of CPO to catalyze the oxidation of chemically diverse organosulfur compounds[12].
Short-chain, volatile organosulfur compounds (VOC) have low odor threshold and very unpleasant odor, characteristic of the putrid odor of rotten egg and vegetables[13]. Exposure to even low concentrations of these compounds leads to dizziness, vomiting, headaches and eye irritation[14]. Thus, these compounds are classified as atmospheric pollutants particularly affecting the area surrounding sources of emission, such as waste water treatment plants, composting sites, paper pulping process and thermal-sludge treatment plants[15].
Considering the low specificity of CPO for sulfur compounds and the environmental problem that VOC represent, it was interesting to study the ability of the enzyme to catalyze the oxidation of short-chain, volatile sulfides and thiols. The results are focused on the kinetic behavior of the enzyme, the identification of products and the operation of a two-phase reactor for the removal of VOC from a gaseous stream.
Materials and methods
Chemicals and enzyme. CPO was produced from Caldariomyces fumago UAMH 89362 and purified as reported elsewhere[16] obtaining a preparation with a Rz (A398nm/A280nm) of at least 1.2. Hydrogen peroxide, diethyl sulfide, dimethyl disulfide, propanethiol, butanethiol, hexanethiol and phenyl sulfide were purchased from Sigma Co. Organic solvents and salts were obtained from J.T. Baker.
]]> Kinetic characterization of CPO-catalyzed oxidation of VOC. Biocatalytic oxidation of organosulfur compounds was performed at 25°C in closed vials, to prevent volatilization of the substrates. Kinetic characterization was performed using substrate concentrations of up to 5 mM, due to insolubility of the compounds. The reaction mixture contained 10% acetonitrile, 60 mM phosphate buffer pH 6 and 1 mM H2O2. The reaction was started by addition of the enzyme (0.05-0.1 nmol). Substrate conversion was monitored by UV absorbance (typically 220-250 nm in a Perkin Elmer DAD) through reverse phase HPLC (Hypersil ODS 2.1 X 100 μm column from Agilent), using a 30% acetonitrile/70% water isocratic phase. Reduction in the area of the UV signal was used to calculate substrate conversion. Reaction rate was calculated as the ratio of substrate conversion to reaction time. Controls with peroxide and without enzyme were also performed, in order to calculate the non-enzymatic conversion. Kinetic characterization was performed using substrate concentrations of up to 5 mM, due to insolubility of the compounds. Given that saturation could not be achieved in all cases, the value of kcat/Km was calculated from the slope of the linear section in the initial rate vs substrate concentration plot. For the control reactions, in the absence of the enzyme and in the presence of peroxide, the rate constant of non-enzymatic conversion (knon) was also calculated from the slope of rate vs substrate concentration plot. Results shown are the average of at least three independent reactions.Product identification. For product identification, 10-mL reactions were monitored until at least 70% substrate conversion was achieved. The reaction mixture was extracted with methylene chloride, dried through an anhydride Na2SO4 bead and analyzed through gas chromatography (Agilent 6890N) coupled to a mass selective detector (Agilent 5973). A nonpolar HP-5ms column (30m x 0.25 mm x 0.25 μm) operating at low temperature (typically 40°C) was used to separate and identify the products.
Reactor operation. A two-phase reactor was assembled as follows. An air stream was passed through a reservoir containing 2 mL of 226.1 mM of dimethyl disulfide (DMDS) in acetonitrile and the resulting DMDS-enriched stream was bubbled at the bottom of a glass vessel containing 70 mL of the liquid reaction medium (15% tert-butanol and 85% 60 mM phosphate buffer pH 6). Gas flow was maintained between 40-60 mL/min, total duration of each experiment (i.e. until the reservoir was exhausted) was around 80-90 min. Enzyme (1 μmol CPO = 40,000 enzyme units) and peroxide were successively added and samples were taken from the liquid phase to be analyzed by HPLC. The reaction was monitored using 230 nm for product detection, as substrate concentration in the liquid medium was extremely low and could not be accurately determined.
Conversion calculations of the enzymatic reaction in the reactor. Product concentration in the system was calculated form a calibration curve. The ratio of total product accumulated in the liquid phase to the total amount of enzyme added to the liquid phase was calculated for each condition tested in this study. Substrate conversion was referred to the theoretically limiting substrate, H2O2 in most cases. Experiments were performed at least in duplicate.
Results and discussion
All compounds tested in this work were transformed in the presence of CPO at pH 6 in a reaction medium containing 10% organic solvent. Under these conditions, non-enzymatic oxidation of the substrates also occurred. The rate of the enzyme-catalyzed reaction was calculated as the difference between the total rate in the presence of the enzyme and peroxide minus the control rate in the presence of only peroxide. In some cases, enzyme saturation could not be achieved due to low substrate solubility. However, the pseudo-first order rate constants of the catalyzed (kcat/Km) and non-enzymatic (knon) reactions were calculated from the linear section of the rate vs substrate concentration plot, as described in the experimental section, and are shown in Table I. The catalytic proficiency, also shown in Table I, is the ratio of these rate constants and has been interpreted by other authors as the ability of enzymes to lower the activation barrier of the reaction[17]. In units of concentration, the catalytic proficiency relates to the affinity between the enzyme and the chemically distorted substrate in the transition state. According to our data, the dissociation constant of VOCs is in the range of 100 nM or less, suggesting a mild interaction within the active center of the enzyme.
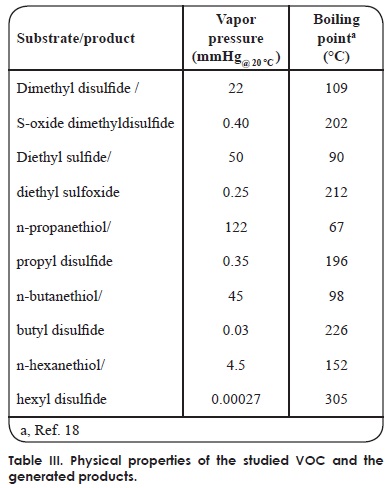
The reaction products, identified by mass spectrometry, are shown in Table II and Figure 1; no other products could be detected. Physical properties of substrates and products are shown in Table III. The oxidation generates compounds with higher boiling points, reduced vapor pressures and thus decreased volatility. Consequently, the oxidation decreases the atmospheric pollutant character of the studied sulfur compounds. We observed that the oxidation of sulfides generated the corresponding sulfoxides, as expected. It is well known that the CPO catalytic mechanism for the oxidation of sulfides involves a direct oxygen transfer within the active site of the enzyme, as evidenced by almost quantitative incorporation of labeled oxygen from H2O2[18] into the sulfoxide[19]. The formation of a short-lived sulfur-based free radical has also been invoked, but has not been demonstrated yet[20]. On the other hand, we observed that thiols were oxidized to the corresponding disulfides. This reaction has not been described before for CPO. The non-enzymatic formation of a disulfide has been reported as a consequence of CPO-catalyzed halogenation of thiols at acidic pH[21], thus ruling out a similar mechanism under the peroxidatic conditions (i.e pH 6 and absence of KCl) used in the present study. In order to further confirm this type of reaction being catalyzed by CPO, we also identified diphenyl disulfide (see Table II) as the product of the oxidation of an aromatic thiol, benzenethiol. This reaction has been reported for other heme proteins such as myeloperoxidase (MPO) and horseradish peroxidase (HRP) using aliphatic thiols as substrates; it has been noted that oxidation rate is widely variable for these enzymes. For example, HRP-catalyzed oxidation of cysteine and cysteine derivatives showed rate constants in the range of 24-660 M-1 s-1; only cysteine methyl ester and 2,3-dimercaptopropanol showed higher oxidation rate constants, in the order of 103 and 104 M-1 s-1, respectively[22]. On the other hand, MPO seemed less sensitive to the nature and charge of the substrate, which were oxidized with rate constants in the range of 103-104 M-1 s-1[23]. In the case of the substrates analyzed in this work, the rate constants of the CPO-catalyzed reaction are in the range of 103-104 M-1 s-1 (Table I), thus highlighting the potential of this versatile enzyme. Regarding the possible mechanism, the reaction could involve the formation of free radicals; it has been suggested that the first enzymatic intermediary of the catalytic cycle, an oxo-ferryl radical cation called Compound I, may be able to subtract an electron from the thiol group; the free radical, after losing a proton, combines with a second free thyl radical to form the disulfide (Figure 2).

Conclusions
In conclusion, CPO from C. fumago is able to catalyze the oxidation of short-chain sulfides and mercaptans to generate the corresponding sulfoxides and disulfides. The products are less volatile compounds, and its ability to pollute the atmosphere is thus reduced by oxidation. Based on limiting concentrations of peroxide, a ca. 40% conversion of the volatile compound contained in a gaseous stream was achieved, by controlling the concentration of enzyme in the reactor.
This example of a peroxidase able to catalyze the oxidation of volatile sulfur hydrocarbons highlights the versatility of low specificity enzymes in the environmental field; this reaction could be of practical application in the remediation of gaseous effluents. Further improvement of the system is necessary, through reactor engineering, in order to improve substrate transfer to the liquid medium.
Acknowledgments
The results presented in this work are part of the master thesis of Julio C. Cruz (UNAM) and bachelor thesis of Jesús García (Universidad Politécnica del Estado de Morelos). This work was supported by PAPIIT UNAM IN212510, BIOCATEM Network (Grant 245413) and Conacyt 179241. The authors thank Rosa Román from Institute of Biotechnology, UNAM and René Guardián Tapia from CIICAp-UAEM for technical support.
References
]]>1. Casella, L., Monzani, E. & Nicolis, S. Potential applications of peroxidases in the fine chemical industries, in Biocatalysis based on heme peroxidases (eds. Torres, E. & Ayala, M.) 111-153 (Springer-Verlag; 2010). [ Links ]
2. Márquez-Rocha, F.J., Pica-Granados, Y., Sandoval-Villasana, A.M. & Vázquez-Duhalt, R. Determination of genotoxicity using a chloroperoxidase-mediated model of PAH-DNA adduct formation. Bull. Environ. Contam. Toxicol. 59, 788-795 (1997). [ Links ]
3. Ayala, M. & Vázquez-Duhalt, R. Enzymatic catalysis on petroleum products in Petroleum biotechnology (eds. Vázquez-Duhalt, R. & Quintero-Ramírez, R.) 67-111 (Amsterdam, Elsevier Science, 2004). [ Links ]
4. Torres, R., Bustos-Jaimes, I. & Le Borgne, S. Potential use of oxidative enzymes for the detoxification of organic pollutants. Appl. Catal. Environ. 46, 1-15 (2003). [ Links ]
5. Torres, C. & Vázquez-Duhalt, R. Applications and prospective of peroxidase biocatalysis in the environmental field in Biocatalysis based on heme peroxidases (eds. Torres, E. & Ayala, M.) 179-206 (Springer-Verlag, 2010). [ Links ]
]]>6. Hofrichter, M. & Ullrich, R. Heme-thiolate haloperoxidases: versatile biocatalysts with biotechnological and environmental significance. Appl. Microbiol. Biotechnol. 71, 276-288 (2006). [ Links ]
7. Allenmark, G.A. & Andersson, M.A. Chloroperoxidase-induced asymmetric sulfoxidation of some conformationally restricted sulfides. Chirality 10, 246-252 (1998). [ Links ]
8. Colonna, S., Gaggero, N., Carrea, G. & Pasta, P. A new enzymatic enantioselective synthesis of dialkyl sulfoxides catalysed by monooxygenases. Chem. Commun. 439-440 (1997). [ Links ]
9. Vargas, R.R., Bechara, E.J.H., Marzorati, L. & Wladislaw, B. Asymmetric sulfoxidation of a [beta]-carbonyl sulfide series by chloroperoxidase. Tetrahedron: Asymmetry 10, 3219-3227 (1999). [ Links ]
10. Pasta, P., Carrea, G., Colonna, S. & Gaggero, N. Effects of chloride on the kinetics and stereochemistry of chloroperoxidase catalyzed oxidation of sulfides. Biochim. Biophys. Acta Protein Struct. Mol. Enzymol. 1209, 203-208 (1994). [ Links ]
]]>11. Ayala, M., Robledo, N.R., López-Munguía, A. & Vázquez-Duhalt, R. Substrate specificity and ionization potential in chloroperoxidase-catalyzed oxidation of diesel fuel. Environ. Sci. Technol. 34, 2804-2809 (2000). [ Links ]
12. Ayala, M., Tinoco, R., Hernández, V., Bremauntz, P. & Vázquez-Duhalt, R. Biocatalytic oxidation of fuel as an alternative to biodesulfurization. Fuel Process. Technol. 57, 101-111 (1998). [ Links ]
13. Schiffman, S.S. & Williams, C.M. Science of odor as a potential health issue. J. Environ. Qual. 34, 129-138 (2005). [ Links ]
14. Andersson, F., Annika, T.S., Karlsson, A., Svensson, B.H. & Ejlertsson, J.R. Occurrence and abatement of volatile sulfur compounds during biogas production. J. Air Waste Manage. Assoc. 54, 855-861 (2004). [ Links ]
15. Smet, E., Lens, P. & Van Langenhove, H. Treatment of waste gases contaminated with odorous sulfur compounds. Crit. Rev. Environ. Sci. Technol. 28, 89-117 (1998). [ Links ]
]]>16. Pickard, M.A., Kadima, T.A. & Carmichael, R.D. Chloroperoxidase, a peroxidase with potential. J. Ind. Microbiol. Biotechnol. 7, 235-241 (1991). [ Links ]
17. Radzicka, A. & Wolfenden, R. A proficient enzyme. Science 267, 90-93 (1995). [ Links ]
18. Brown, R.L. & Stein, S.E. Boiling point data in NIST Chemistry WebBook, NIST Standard Reference Database Number 69 (eds. Linstrom, P.J. & Mallards, W.G.) National Institute of Standards and Technology http://webbook.nist.gov (last seen on 2012). [ Links ]
19. Goto, Y., Matsui, T., Ozaki, S.I., Watanabe, Y. & Fukuzumi, S. Mechanisms of sulfoxidation catalyzed by high-valent intermediates of heme enzymes: electron-transfer vs oxygen-transfer mechanism. J. Am. Chem. Soc. 121, 9497-9502 (1999). [ Links ]
20. Kobayashi, S., Nakano, M., Kimura, T. & Schaap, A.P. On the mechanism of the peroxidase-catalyzed oxygen-transfer reaction. Biochemistry 26, 5019-5022 (1987). [ Links ]
]]>21. Silverstein, R.M.& Hager, L.P. Chloroperoxidase-catalyzed oxidation of thiols and disulfides to sulfenyl chlorides. Biochemistry 13, 5069-5073 (1974). [ Links ]
22. Burner, U. & Obinger, C. Transient-state and steady-state kinetics of the oxidation of aliphatic and aromatic thiols by horseradish peroxidase. FEBS Lett. 411, 269-274 (1997). [ Links ]
23. Burner, U., Jantschko, W. & Obinger, C. Kinetics of oxidation of aliphatic and aromatic thiols by myeloperoxidase compounds I and II. FEBS Lett. 443, 290-296 (1994). [ Links ]
24. Valderrama, B., Ayala, M. & Vázquez-Duhalt, R. Suicide inactivation of peroxidases and the challenge of engineering more robust enzymes. Chem. Biol. 9, 555-565 (2000). [ Links ]
25. Ayala, M., Batista, C.V. & Vázquez-Duhalt, R. Heme destruction, the main molecular event during the peroxide-mediated inactivation of chloroperoxidase from Caldariomyces fumago. J. Biol. Inorg. Chem. 16, 63-68 (2011). [ Links ]
]]>Información sobre los autores
Julio César Cruz
Julio César Cruz es Ingeniero bioquímico, con especialidad en biotecnología, egresado en 2007 del Instituto Tecnológico de Villahermosa, Tabasco, donde se tituló con la tesis basada en la caracterización bioquímica de una enzima tipo peroxidasa. Al término de la licenciatura realizó sus estudios de maestría en el Posgrado en Ciencias Bioquímicas del Instituto de Biotecnología de la Universidad Nacional Autónoma de México, y obtuvo el grado de Maestro en Ciencias en 2010. Durante ese periodo desarrolló la tesis sobre la oxidación de compuestos azufrados volátiles en fase sólido-gas, usando como biocatalizador la enzima cloroperoxidasa. Posteriormente, en 2011 ingresó a la Escuela de Salud Pública de México del Instituto Nacional de Salud Pública, donde a la fecha está por concluir el doctorado en Ciencias en Salud Pública y Epidemiología, el tema de su tesis es el estudio de la interacción entre contaminación ambiental (medida a través de la exposición a partículas finas suspendidas en el aire menores a 2.5 μm), obesidad y el polimorfismo genético del gen SERPINE 1 ( -675 4g/5g), así como su efecto en la inflamación de vías aéreas y función pulmonar en población infantil/adolescente. Las áreas de su interés son la investigación sobre temas ambientales, como la contaminación atmosférica, biorremediación y salud pública.
Jesús García
Jesús García estudió Ingeniería en Tecnología Ambiental en la Universidad Politécnica del Estado de Morelos. Realizó su tesis en el laboratorio de Biocatálisis Ambiental que dirigía el Dr. Rafael Vázquez Duhalt y bajo la tutoría de la Dra. Marcela AyalaAceves se tituló con la tesis "Oxidación de compuestos azufrados volátiles en un sistema en fase gas-líquido catalizada por la cloroperoxidasa de Caldariomyces fumago", en marzo de 2012.
Marcela Ayala
Marcela Ayala realizó sus estudios profesionales en la Facultad de Química de la Universidad Nacional Autónoma de México, en donde obtuvo el título de Ingeniera Química. Al terminar su licenciatura, ingresó al Instituto de Biotecnología de la misma universidad para realizar sus estudios de doctorado, obteniendo el grado de Doctora en Ciencias. Ingresó como investigadora al Instituto Mexicano del Petróleo, en donde trabajó hasta el 2005, año en que se incorporó como Investigadora Asociada en el Instituto de Biotecnología de la UNAM y desde 2009 es Investigadora Titular "A" en esta institución.
Actualmente es Investigadora de Tiempo Completo en la UNAM. Su especialidad es la obtención y caracterización de biocatalizadores basados en oxidoreductasas. Este tipo de enzimas tienen una diversidad de aplicaciones, desde campos como el ambiental hasta industrias de alto valor agregado como la farmacéutica. Dentro de las líneas de investigación de su interés están el estudio de la transferencia intramolecular de electrones en las peroxidasas y lacasas, catálisis de reacciones enzimáticas redox utilizando peroxidasas y lacasas y biocatálisis oxidativa en solventes orgánicos. Miembro del SNI, Nivel 1, con 20 publicaciones en revistas internacionales indizadas, ha escrito varios capítulos de libros y es editora de un libro sobre aplicaciones de peroxidasas.
Ha sido distinguida con la mención honorífica en el Certamen Juvenil de Ciencia y Tecnología 2002, por el Instituto Mexicano de la Juventud (SEP), con el Premio Alfredo Sánchez Marroquín 2003 otorgado a la mejor tesis doctoral en el área de biotecnología, por la Sociedad Mexicana de Biotecnología y Bioingeniería y con la Distinción Universidad Nacional para Jóvenes Académicos 2011, en el área de Innovación Tecnológica (DUNJA2011).
]]>