
Radiation Grafting of 4-Vinylpyridine and N, N-Dimethylaminoethylmetha-Crylate from Binary Mixtures onto Polypropylene
Juan Carlos Ruiz, Emilio Bucio, Roustam Aliev and Guillermina Burillo*
Instituto de Ciencias Nucleares, Universidad Nacional Autónoma de México, Circuito Exterior, Ciudad Universitaria, 04510, México D.F. E-mail: burillo@nuclecu.unam.mx
Recibido el 18 de junio del 2004.
Aceptado el 2 de septiembre del 2004.
Abstract
Radiation grafting of 4-vinylpyridine (4-VP) and N,N-dimethylaminoethylmethacrylate (DMAEMA) from binary mixtures in toluene solutions was carried out onto polypropylene (PP) films. The simultaneous γ-iradiation of PP and monomers solutions (direct method) at dose rate of 6 kGy/h and doses from 5 to 35 kGy was used. Dependences of grafting yield on dose and ratio of monomers concentration in solution were found. The grafted films were characterized with the FTIR-ATR spectroscopy. The grafted and ungrafted (in the presence or in the absence of PP) copolymers compositions were determined, and their dependence on compositions of the initial monomers nixtures were shown. Monomers reactivity ratios were calculated. These data indicate a random 4-VP and DMAEMA copolymerization.
Key words: 4-Vinylpyridine, N,N-Dimethylaminoethylmethacrylate, polypropylene, radiation grafting, copolymerization.
Resumen
Se realizó el injerto de 4-vinilpiridina (4-VP) y dimetilaminoetilmetacrilato (DMAEMA) en películas de polipropileno, mediante radiación gamma, utilizándose el método de radiación simultánea del PP y la solución de monómeros en tolueno, a una razón de dosis de 8 kGy/h y dosis de radiación de 5 a 35 kGy. Se estudió la dependencia del rendimiento de injerto con la dosis y la concentración de ambos monómeros en la solución. Las películas injertadas se caracterizaron mediante espectroscopia infrarroja (FTIR-ATR). Se determinó la composición de los copolímeros 4-VP-co-DMAE-MA en presencia y ausencia de las películas de PP y en el copolímero injertado, así como su dependencia con la composición inicial de los monómeros utilizados y su reactividad, los resultados obtenidos nos indican una copolimerización al azar de 4-VP y DMAEMA.
Palabras clave: 4-Vinilpiridina, N,N-Dimetilaminoetilmetacrilato, polipropileno, injerto por radiación, copolimerización.
Introduction
]]> It is known that homopolymers containing tertiary amino groups, such as N,N-dimethylaminoethylmethacrylate (DMAE-MA) or 4-vinylpyridine (4-VP) form salts with several benzoic acids derivatives, some of them with liquid crystal properties, but these compounds are often hygroscopic and have poor mechanical properties [1,2]. Therefore preliminary graft polymerization of these monomers onto hydrophobic and mechanically durable polymer matrices is of interest. Radiation grafting of DMAEMA onto polyethylene (PE) as well as salts of poly(DMAEMA-g-PE) with benzoic acids formation were reported earlier [3,4]. Radiation grafting of 4-VP or DMAE-MA onto PP has been investigated in some works [5-11]. Recently, we reported the 4-VP and DMAEMA radiation grafting onto PP by two step method, where the monomers were grafted one after another [12]. But grafting of these monomers from their binary mixtures onto polymers has never been investigated before. Nevertheless, there is an essential interest in polymers (PE, PP, nylon, teflon a.o.) modification by this method using some other monomers (styrene, acrylic acid, acrylamide, acrylonitrile, N-isopropylacrylamide, 4-VP a.o.) [13-20]. Kinetics regularities of graft copolymerization were determined, and monomers reactivity ratios were calculated in some cases.In the present study, we report conditions and regularities of the 4-VP/DMAEMA binary mixtures radiation grafting onto PP as well as some characteristics of the grafted copolymers.
Experimental
PP isotactic films (PEMEX, México) with a thickness of 70 ìm were cut in 1 5 cm pieces. 4-VP and DMAEMA (Aldrich Chemical Co) was purified by distillation under a reduced pressure. Toluene, dimethylformamide (DMF), hexane and methanol from Baker were used as received.
PP films were placed in glass ampoules which contained 4-VP and DMAEMA of different monomers ratios in 50% toluene solution. The reaction mixtures were deaerated under vacuum by repeated freezing and thawing method. Then the ampoules were vacuum sealed and irradiated with a Co60 ã-source (Gammabeam 651 PT, Nordion International Inc.) at a dose rate of 6 kGy/h and doses from 5 to 35 kGy. The residual monomers and ungrafted copolymers formed were removed from the grafted samples by extraction with toluene for 24 h, and then with dimethylformamide and methanol. The grafted samples were dried under vacuum to a constant weight.
The grafting yield was calculated by the equation: Grafting % = (W-W0)100/W0, where W and W0 are weights of the grafted and initial films, respectively.
The ungrafted copolymers formed at 15 kGy in the presence or in the absence of the PP films were precipitated in hexane, then sediments were dried under vacuum to a constant weight.
FTIR-ATR spectra of the starting and modified polymer films were analyzed with the Perkin-Elmer (model Paragon) spectrometer. SeZn glass was used for contact with the samples surfaces, and the IR radiation penetrated into samples to the depth of 8-10 ìm.
Compositions of the grafted and ungrafted 4-VP/DMAEMA copolymers were determined by elemental analysis in Desert Analytics (Arizona, USA). Monomers reactivity ratios were calculated by the Fineman-Ross method.
]]>Results and discussion
Figure 1 shows the grafting percent as a function of dose, obtained at different concentrations of 4-VP/DMAEMA in 50% toluene solution. The grafting values increased with the dose, but they decreased with increase in the 4-VP concentration from 10 to 25 %. Nevertheless, the further increase in the 4-VP concentration to 30 and 40% resulted in the grafting increase. The same tendency was found for dependence of the grafting percent on the 4-VP/DMAEMA molar ratios (Fig.2). At first, an increase in the 4-VP concentration led to decrease in the grafting percent to some minimum but then with higher amount of 4-VP (more than two times), the grafting percent increased again. This minimum grafting percent with its following increase at the 4-VP higher concentration was caused, probably, by a different diffusion of the monomer inside the polymer.


The IR-spectra analysis confirmed the polyDMAEMA and poly4-VP grafting onto PP: the spectra of samples contained both modifying polymers. The carbonyl group of DMAEMA is responsible for the strong absorption at 1717 cm-1, and the three peaks at 1594, 1555 and 1413 cm-1 attributed to the pyridine ring stretching vibration [21]. These spectra are similar to that one observed earlier in the case of two-step method of these monomers grafting [12].
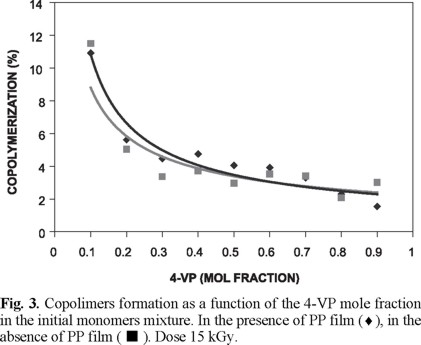
Copolymerization percent of two monomers in presence and absence of PP films as a function of the 4-VP mole fraction is shown in Fig. 3. Practically the same decrease in amounts of copolymers formed in both cases was observed.
]]>
Monomers reactivity ratios were calculated from the experimental data shown in Fig.4, and they are presented in Table 1. It is seen an essential difference in the r4-VP and rDMAE-MA values for the grafted and ungrafted copolymers but in all three cases these values indicate a random 4-VP and DMAEMA copolymerization.


Conclusions
An efficient radiation grafting of 4-VP and DMAEMA from binary mixtures in toluene was performed under mild irradiation conditions. It has been found that grafting percent depended both the dose and monomers ratio in the initial mixture. A comparative analysis of grafted and ungrafted copolymers showed an essential difference in dependence of their compositions on compositions of the initial monomers mixtures. These dependences and calculated values of the monomers reactivity ratios indicate a random 4-VP and DMAEMA copolymerization.
]]> Acknowledgements
The authors thank José Rangel, Susana Castillo and Francisco García from ICN UNAM, and Araceli Ordoñez from IIM UNAM for their technical assistance.
References
1. Bazuin, C.G.; Brandys, F.A.; Eve, T.M.; Plante, M. Makromol. Chem., Macromol. Symp. 1994, 84,183-196. [ Links ]
2. Bazuin, C.G.; Tork, A. Macromolecules 1995, 28, 8877-8880. [ Links ]
3. Bucio, E.; Burillo, G. Polym. Bull. 1997, 38, 587-594. [ Links ]
]]>4. Burillo, G.; Bucio, E.; Cervera, E.; Ogawa, T. J. Appl. Polym. Sci. 2000, 78, 972-978. [ Links ]
5. Hartley, F.R.; McCaffrey, D.J.A.; Murray, S.G.; Nicholson, F.N. J. Organometal. Chem. 1981, 206, 347-359. [ Links ]
6. Ellinghorst, G.; Niemöller, A.; Vierkotten, D. Radiat. Phys. Chem. 1983, 22, 635-642. [ Links ]
7. Kaur, I.; Barsola, R. J. Appl. Polym. Sci. 1990, 41, 2067-2073. [ Links ]
8. Gawish, S.W.; Kantouch, A.; El Naggar, A.M.; Mosleh, S. J. Appl. Polym. Sci. 1992, 4, 1671-1676. [ Links ]
]]>9. Kaur, I.; Misra, B.N.; Barsola, R. J. Appl. Polym. Sci. 1993, 48, 575-581. [ Links ]
10. Bucio, E.; Aliev, R.; Burillo, G. Radiat. Phys. Chem. 1998, 52, 193-196. [ Links ]
11. Tan, S.; Li, G.; Shen, J. J Appl. Polym. Sci. 2000, 77, 1861-1868. [ Links ]
12. Bucio, E.; Aliev, R.; Burillo, G. Polym. Bull. 2002, 47, 571-577. [ Links ]
13. Kubota, H. J. Appl. Polym. Sci. 1992, 46, 383-388. [ Links ]
]]>14. El-Salmawi, K.M.; El-Naggar, A.M.; Said, H.M.; Zahran, A.H. Polym. Int. 1997, 42, 225-234. [ Links ]
17. Kondo, T.; Koyama, M.; Kubota, H.; Katakai, R. J. Appl. Polym. Sci. 1998, 67, 2057-2064. [ Links ]
18. Hegazy, El-S.A.; Kamal, H.; Maziad, N.; Dessouki, A.M. Nucl. Instr. Meth. B. 1999, 151, 386-392. [ Links ]
19. Choi, S-H; Nho, Y.C. Radiat. Phys. Chem. 2000, 58, 157-168. [ Links ]
19. El-Naggar, A.M.; Zohdy, M.H.; Sahar, S.M.; Allam, E..A. Polym. Int. 2001, 50, 1082-1088. [ Links ]
]]>20. Palacios, O.; Aliev, R.; Burillo, G. Polym. Bull. 2003, 51, 191-197. [ Links ]
21. Socrates, G. Infrared Characteristics Group frecuencies, Tables and Charts. John Wiley and Sons, Chichester, 1994, 34, 95,130. [ Links ]
]]>