New Bioactive Derivatives of Xanthorrhizol
María Isabel Aguilar,*1 Guillermo Delgado2 and María Luisa Villarreal3
1 Departamento de Farmacia, Conjunto "E" de la Facultad de Química. Universidad Nacional Autónoma de México. Ciudad Universitaria, Coyoacán, México 04510, D. F. E-mail: laurents@servidor.unam.mx
2 Instituto de Química, Universidad Nacional Autónoma de México. Circuito Exterior, Ciudad Universitaria, Coyoacán, México 04510, D.F.
3 Centro de Investigación Biomédica del Sur, IMSS, Argentina No. 1, Xochitepec, Morelos, México.
]]> Recibido el 6 de febrero del 2001.
Abstract
The chemical preparation and the antifungic and cytotoxic evaluations of several new derivatives of xanthorrhizol, a bioactive natural product isolated from certain plants used in traditional medicine, are described. Acylation of the phenol, bromination of the benzene ring, as well as reduction and oxidation of the olefin of the natural sesquiterpene, allowed obtaining a series of derivatives which displayed mild antifungic activities and did not show cytotoxic activities toward certain human tumor cell lines.
Key words: Xanthorrhizol, Iostephane heterophylla, antifungic agents, bisabolene derivatives.
Resumen
Se decribe la preparación química y las evaluaciones antifúngicas y citotóxicas de algunos derivados novedosos del xantorrhizol, un producto natural bioactivo aislado de ciertas plantas usadas en la medicina tradicional. La acilación del fenol, la bromación del anillo bencénico, y la oxidación y reducción de la olefina del sesquiterpeno natural permitieron la obtención de una serie de derivados, los cuales mostraron actividades fúngicas moderadas y no mostraron actividad citotóxica en ciertas líneas celulares.
Palabras clave: Xantorrhizol, Iostephane heterophylla, agentes antifúngicos, derivados del bisaboleno.
]]>Introduction
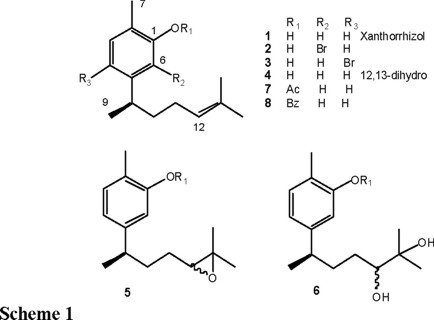
Xanthorrhizol (1), a sesquiterpene of the bisabolene type, has been isolated from Zingiberaceae and Asteraceae families of higher plants [1,2]. This compound constitutes the major secondary metabolite in the chloroform extract of the roots of Iostephane heterophylla (Asteraceae), a plant that has been medicinally used in Central Mexico to treat skin illnesses such as wounds and sores, among other diseases [3]. Previous evaluations on this compound, as antifungic and cytotoxic agent [3], for prolonging pentobarbital induced sleeping time [4], and as inhibitor of the tonic contraction of rat uterus [5], have shown positive results. Xanthorrhizol is a sesquiterpenic phenol, with an unsaturated prenylic chain at C-5. The antimicrobial activity of phenol derivatives is well documented in the literature, and phenol itself is employed as a standard of bactericidal activity in the USP Pharmacopoeia, reporting its antimicrobial action as the "phenol coefficient" [6]. Introduction of chlorine or bromine atoms in phenolic ring enhances the antiseptic activities, as well as the presence of alkyl chains, being more potent the lineal than the ramified ones. Acetylation of the phenolic group produces an ester that slowly liberates the phenol when it contacts the tissues. This property diminishes its irritant action and allows it to be employed as an antiinfective agent in nose, throat and ears [6].
Since chemical modifications of a bioactive natural product result in the modification of its biological properties [7-9], some new derivatives of xanthorrhizol (1) were prepared, in which the phenol was modified to acetate and benzoate esters. Other modifications of 1 were the bromination of the aromatic ring and the saturation and oxidation of the Δ12,13 double bond. In this paper, we report the preparation of the above mentioned derivatives and their evaluation in the anti-dermatophytic and cytotoxic bioassays.
Results and discussion
6-Bromo- (2), 4-bromo- (3), 12,13-dihydro- (4), 1-O-acetyl-(7), and 1-O-benzoyl- (8), 12, 13-epoxy- (5), and 12, 13-dihydro-dihydroxy-xanthorrhizol (6) were prepared from xanthorrhizol (1), obtained from the CHCl3 extract of the roots of I. heterophylla [3]. Halogenation of 1 with NBS afforded the sterically hindered 6-bromo-xanthorrhizol (2) as the main product, together with the bromination product at C-4 (3). Both compounds possessed similar spectroscopic characteristics and according to their MS, they showed to be isomers. The molecular ion appeared at m/z 298 [M++2], 297 [M++1] and 296 [M+], and other fragments were at m/z 217 M+-Br and 135 M+-C6H11O (base peak). The 1H NMR spectrum of 2, showed signals for two ortho benzenic hydrogens at δ 6.71 (H-5) and δ 7.03 (H-4), as a pair of doublets (J = 7.8), while in compound 3, the benzenic hydrogens appeared para to each other, as a pair of singlets at δ 6.65 (H-6) and δ 7.26 (H-3). The remaining signals corresponded to a benzylic hydrogen, and four methyls (1 H NMR data in the Experimental). Compound 4 was obtained quantitatively by catalytic hydrogenation of xanthorrhizol (10 % Pd / C), and its spectroscopic constants were similar to those of xanthorrhizol, being observed a difference of two additional units in the MS of 4. 4 was previously characterized [1]. Compounds 5 and 6 were obtained previously as minor natural products [3] and they were prepared as a C-12 epimeric mixture from 1: epoxidation with m-chloroperbenzoic acid, afforded 5, and diol 6 was obtained via trans-hydroxylation with performic acid [3]. Acetylation and benzoylation of xanthorrhizol (1) led to compounds 7 and 8. 1H (se Experimental) and 13C NMR (Table 1) data for compounds 2-8 were in agreement with the proposed structures, and the assignments were supported by comparison with the data in the literature [10].

Compounds 1-8 were tested for their antimicrobial activities toward Trichophyton mentagrophytes and Microsporum gypseum growing in vitro, and the results (Table 2) showed that the majority of the structural modifications to xanthorrhizol (derivatives 2, 3, 5-8) led to reduced but clear activities (MIC < 400 µg / mL), while the 12,13-dihydro derivative (4) displayed the same bioactivities of the standard antifungic reference (nystatin) and xanthorrhizol (1). These preliminary results suggested the structural requirements of xanthorrhizol (1) for the activity toward the tested microorganisms. Regarding the antiploriferative activities, although xanthorrhizol (1) showed activity against sarcoma 180 ascites in mice [11], the evaluation of compounds 1-8 in KB, UISO-SQC-1 and HCT-15 human tumor cell lines indicated that they did not display significant cytotoxicity (ED50 > 4 µg / ml).
]]> Experimental
IR spectra were taken on an 80A Nicolet FT. MS were measured on a 5985-B Hewlett Packard instrument. Optical rotation was determined on a 241 Perkin Elmer polarimeter. NMR spectra were recorded on a Varian VXR 300 spectrometer, operating at 300 MHz (1H) and 75 MHz (13C). CDCl3 was used as solvent. Chemical shifts are reported in parts per million (δ) relative to TMS, and coupling constants (J) are in Hertz. Carbon substitution degrees were established by DEPT multipulse sequence. Xanthorrhizol (1) was isolated from the roots of I. heterophylla, as described previously [2,3].
6-Bromo-xanthorrhizol (2) and 4-Bromo-xanthorrhizol (3). Xanthorrhizol (1) (225 mg, 1.02 mmol) and N-bromo-succinimide (178 mg, 1 mmol) were dissolved in CCl4 (2.5 mL). The reaction was initiated adding catalytic quantities of dibenzoyl peroxide, and refluxed for 2 h. The starting material was consumed, and the formation of two reaction products less polar than xanthorrhizol was observed by TLC. Evaporation of the solvent afforded a residue which was loaded to a Si gel column chromatography (70-230 mesh) packed with petroleum ether. The chromatography was developed using a gradient of petroleum ether-EtOAc. Two oily compounds were separated, and characterized as 2 (205 mg, 66 %), and 3 (66 mg, 21.6 %). 2: IR νmáx (film): 3507, 2961, 2856, 1607, 1489, 1450, 1120, 811 cm−1; 1H NMR (CDCl3, 300 MHz) δ: 1.19 (3H, d, J 7.0, H-9), 1.51 (3H, s, H-15), 1.58 (2H, m, H-11), 1.67 (3H, s, H-14), 1.93 (2H, m, H-10), 2.27 (3H, s, H-7), 3.12 (1H, q, J=7.0. H-8), 5.09 (1H, t, J=7.1, H-12), 6.71 (1H, d, J=7.8, H-5), 7.03 (1H, d, J=7.8, H-4); 13C NMR (75 MHz, CDCl3) in Table 1; EIMS m/z (rel. int.): 298 [M++2], 297 [M++1], 296 [M+, 2.0], 217 (M+-Br, 32.6), 214 (22.1), 201 (12.4), 135 (100), 134 (39.3), 115 (17.4), 105 (17.7), 91 (31.9), 79 (22.4), 77 (31.4). 4-Bromo-xanthorrhizol (3): IR νmax (film): 3500, 2950, 2860, 1610, 1450, 1115, 990, 815 cm−1; 1H NMR (CDCl3, 300 MHz) δ: 1.14 (3H, d, J=6.8, H-9), 1.53 (3H, s, H-15), 1.67 (3H, s, H-14), 1.4-1.7 (2H, m, H-11), 1.93 (2H, m, H-10), 2.18 (3H, s, H-7), 3.10 (1H, m, H-8), 4.72 (1H, br s, OH), 5.09 (1H, m, H-12), 6.65 (1H, s, H-6), 7.26 (1H, s, H-3); 13C NMR (CDCl3, 75 MHz) in Table 1.
12, 13-Dihydro-xanthorrhizol (4). A solution of xanthorrhizol (1) (80 mg) dissolved in ethyl acetate (10 mL) was hydrogenated using 10 % Pd / C (20 mg), as catalyst. The reaction was monitored by TLC (petroleum ether / ethyl acetate 85:15), and the reaction product was purified by preparative chromatography as a yellowish oil (100 % yield). [α]D-5.61° (c 1.3, MeOH); UV λmax (MeOH): 215, 273 nm (ε 17,982, 2324); IR νmax (film): 3400, 3372, 2955, 2867, 1620, 1587, 1458, 1365, 1160, 1116, 993, 811 cm−1; 1H NMR (CDCl3, 300 MHz) δ: 0.62 (3H, d, J=6.6, H-14), 0.63 (3H, d, J=6.6, H-15), 1.11-1.19 (2H, m, H-12, 13), 1.18 (3H, br d, J=7.2, H-9), 1.44-1.52 (2H, m, H-10), 2.21 (3H, s, H-7), 2.57 (1H, m, H-8), 4.85 (1H, br s, -OH), 6.61 (1H, d, J=1.6, H-6), 6.68 (1H, dd, J=7.8, 1.6, H-4), 7.03 (1H, d, J=7.8, H-3); 13C NMR (CDCl3, 75 MHz) in Table 2; EIMS m/z (rel. int.): 220 [M+, 8], 136 (27), 135 (100), 121 (15), 91 (15), 77 (5), 79 (5), 43 (7).
1-O-Acetyl-xanthorrhizol (7). Xanthorrhizol (1) (322 mg) was acetylated with acetic anhydride (3.3 mL) and pyridine (1 ml) overnight at room temperature. Usual work-up afforded 7 which was purified by column chromatography to obtain 280 mg as an oil (68.7 %). 1H NMR (CDCl3, 300 MHz) δ: 1.19 (3H, d, J=6.8, H-9), 1.50-1.63 (2H, m, H-10), 1.52 (3H, s, H-15), 1.67 (3H, s, H-14), 1.94-1.83 (2H, m, H-11), 2.13 (3H, s, H-7), 2.31 (3H, s, H-1'), 2.66 (1H, qt, H-8), 5.08 (1H, m, H-12), 6.82 (1H, d, J=1.8, H-6), 6.99 (1H, dd, J=7.7, 1.8, H-4), 7.14 (1H, d, J=7.7, H-3); 13C NMR (CDCl3, 75 MHz) in Table 1.
1-O-Benzoyl-xanthorrhizol (8). Xanthorrhizol (1) (250 mg), benzoyl chloride (0.5 mL) and pyridine (0.5 mL) were allowed to react overnight at room temperature. Usual work-up and purification of the product by column chromatography allowed to obtain 240 mg of 6 (62 %). IR νmax (film): 3061, 2960, 2865, 1601, 1579, 1451, 1176, 1120, 862, 821, 708 cm−1; 1H NMR (300 MHz, CDCl3) δ: 1.24 (2H, d, J 7.0, H-9), 1.51-1.69 (2H, m, H-10), 1.67 (3H, s, H-15), 1.54 (3H, s, H-14), 1.98-1.86 (2H, m, H-11), 2.19 (3H, s, H-7), 2.78-2.63 (1H, m, H-8), 5.09 (1H, m, H-12), 6.96 (1H, d, J=1.6, H-6), 7.01 (1H, dd, J=7.70, 1.6, H-4), 7.19 (1H, d, J=7.7, H-3), 7.69-7.48 (3H, m, H-3', 4', 5'), 8.23 (2H, m, H-2', 6'); 13C NMR (CDCl3, 75 MHz) in Table 1; EIMS m/z (rel. int.): 322 [M+, 6], 254 (35), 217 (20), 105 (100), 91 (18), 68 (5), 43 (7).
Biological Assays. Antimicrobial activity. Studies were performed with cultures of Trichophyton mentagrophytes (ATCC 10742) and Microsporum gypseum (ATCC 10435). The dermatophytes were maintained on Saboureaud's dextrose agar [12]. Cytotoxic activity. Cell cultures were maintained in Basal Medium Eagle (BME) with 10 % fetal bovine serum (FBS), and the assays were performed using standard protocols [12].
Acknowledgments
The authors thank María Isabel Chávez, Marisela Gutiérrez, Graciela Chávez, Luis Velasco and Javier Pérez-Flores for technical assistance.
]]>References
1. Rimpler, H.; Hänsel, R.; Kochendoerfer, L. Z. Naturforsch. 1970, 25b, 995-998. [ Links ]
2. Aguilar, M. I.; Delgado, G.; Bye, R.; Linares, E. Phytochemistry 1993, 33, 1161-1163. [ Links ]
3. Aguilar, M. I.; Delgado, G.; Hernández, M. L.; Villarreal, M. L. Nat. Prod. Lett. 2001, 15 (in the press). [ Links ]
4. Yamazaki, M.; Maebayashi, Y.; Iwase, N.; Kaneko, T. Chem. Pharm. Bull. 1988, 36, 2070-2074. [ Links ]
]]>5. Ponce-Monter, H.; Campos, M.; Aguilar, M.I.; Delgado, G. Phytotherapy Res. 1999, 13, 1-4. [ Links ]
6. Gisvold, O., Phenols and their Derivatives. In: Textbook of Organic Medicinal and Pharmaceutical Chemistry. 6th Ed. J. B Lippincott,. Philadelphia, 1971. [ Links ]
7. Silverman, R. B. The Organic Chemistry of Drug Design and Drug Action. Academic Press, New York, Chapt. 1, 1992. [ Links ]
8. Cannon, J. G. In: Burger's Medicinal Chemistry. Analog Design, M. E. Wolff, Ed. Wiley-Interscience, New York, 1995, 783-802. [ Links ]
9. Wermuth, C. Specific Substituent Effects. In: The Practice of Organic Chemistry, Academic Press, New York, Chapt. 17, 1996, 312-342. [ Links ]
]]>10. Joseph-Nathan, P.; Tovar-Miranda, R.; Martínez, E.; Santillán, R. L. J. Nat. Prod. 1988, 51, 1116-1128. [ Links ]
11. Itokawa, H.; Hirayama, F.; Funakoshi, K.; Takeya, K. Chem. Pharm. Bull. 1985, 33, 3488-3492. [ Links ]
12. Villarreal, M. L.; Alvarez, L.; Alonso, D.; Navarro, V.; García P.; Delgado, G. J. Ethnopharm. 1994, 42, 25-29. [ Links ]
]]>