
J. HERRERA MURILLO, J. F. ROJAS MARÍN
Laboratorio de Análisis Ambiental, Escuela de Ciencias Ambientales, Universidad Nacional,
Campus Omar Dengo, Heredia, Costa Rica. Corresponding author: J. Herrera Murillo; email: jherrer@una.ac.cr
S. RODRÍGUEZ ROMÁN
Escuela de Química, Universidad de Costa Rica, Ciudad Universitaria Rodrigo Facio San Pedro de Montes de Oca, Costa Rica
A. P. BAEZ PEDRAJO
Centro de Ciencias de la Atmósfera, Universidad Nacional Autónoma de México
Circuito Exterior, Ciudad Universitaria, México D. F. México
Received December 8, 2009; Accepted June 21, 2010
RESUMEN
Se realizaron mediciones de metales e iones inorgánicos presentes en muestras de PM10 colectadas en cinco sitios distribuidos en las ciudades de San José, Heredia y Belén. Las muestras fueron colectadas con muestreadores de alto volumen marca Thermo Andersen sobre filtros de fibra de vidrio y cuarzo. Los iones SO42–, NO3–, Cl–, F–, PO43–, NH4+, K+, Na+, Ca2+, Mg2+ fueron analizados por cromatografía de intercambio iónico, mientras que los metales traza (V, Cu, Cr, Pb, Ni, Mn y Fe) se cuantificaron por absorción atómica con horno de grafito. Los resultados obtenidos indican que el SO42– es el ion más abundante, mientras que el Al y el Fe son los metales que presentan mayores concentraciones. Al aplicar correlaciones de Spearman a los datos, se muestra una alta correlación entre SO42–, NO3– NH4+ indicando un origen antrópico común. De las especies metálicas analizadas solamente el Pb y el Cu presentan factores de enriquecimiento mayores a 10, indicando una posible contribución de origen antrópico. Las partículas colectadas en la ciudad de Belén muestran un mayor nivel de acidez y contenido de metales debido a la concentración de importantes actividades industriales en la zona.
ABSTRACT
Measurements of metals and inorganic ions were carried out on PM10. Sampling was made in five sites distributed in San José, Heredia and Belén cities. Samples were collected using Thermo Andersen PM10 high volume samplers with glass fiber and quartz filters. The ions SO42–, NO3–, Cl–, F–, PO43–, NH4+, K+, Na+, Ca2+, Mg2+ were analyzed by ion chromatography and in the case of metals V, Cu, Cr, Pb, Ni, Mn, Fe, Al using an atomic absorption spectrometer with a graphite furnace attachment. The results indicated that SO42– was the most abundant ion, and with respect to metals, Al and Fe had the highest concentration. Spearman correlation, applied to all data, showed a high correlation between SO42–, NO3– and NH4+ indicating a common anthropogenic origin. From the metal species that were assessed only Pb and Cu presented enrichment factors higher than 10, indicating an anthropogenic contribution. Particles collected in the industrial zone of Belén showed the highest level of acidity and metal content.
Keywords: Water soluble ions, particulate matter, metals, Costa Rica.
]]> 1. Introduction
The country of Costa Rica is crossed by a mountain range system running from NW to SE. The system includes many volcano cones with elevations up to 3600 m above sea level (asl), forming a central plateau known as the Gran Area Metropolitana (Metropolitan Area) at 1100 m asl with an area of about 3000 km2. This region contains 75% percent of the motor vehicles (734 200), 85% of the industry and 60% of the population (2,580,000) of the country, according to the latest census (INEC, 2000).
The Metropolitan Area is the highest–ranking center in the urban system in Costa Rica as a result of productive activity, economic and public services that are concentrated in this area. Four cities form the Metropolitan Area (Alajuela, Heredia, Cartago and San José) and many smaller population centers that, a few years ago, were in the middle of crop areas, but now, due to the region growth pattern, have been consolidated into a single urban center together with the main cities. Lack of urban planning has implicated a considerable ruin of air quality, as a result of growth experienced by the cities of the Metropolitan Area of Costa Rica during the past 20 years. Therefore, inhabitans have been exposed to annual average concentrations of PM10 close to 46 µg/m3 (Herrera and Rodríguez, 2007).
Atmospheric particles are made up of a mix of direct emissions generated from wide variety of sources and are formed by different mechanisms such as gas–particle conversion. Particles have an important influence in the transmission of radiation in the atmosphere and water cycle. For this reason, they can alter the energy exchange system between earth and the atmosphere by absorbing and scattering solar radiation, directly affecting the climate (Meinrat et al., 1997; Su and Chen, 1997; Qian et al., 1999; Dusek et al., 2006).
In addition, these particles can generate other effects to the environment, such as enrichment of coastal ecosystems due to air–water transfer and impact on health, worsening conditions such as asthma, chronic bronchitis and decreased lung function, among others (USEPA, 1997).
The assessment of chemical composition of particle determines not only their toxicological potential and behavior on the environment (Pacyna, 1998), but also it becomes a very valuable aid to assess the contribution of different sources, especially when developing strategies to control and reduce air pollution (Simpson et al., 2003; Bessagnet et al., 2004).
For the reasons mentioned above, this study determines concentrations of V, Cr, Cu, Ni, Pb, Mn, Fe, Al and inorganic ions SO42–, NO3–, Cl–, F–, PO43–, NH4+, K+, Na+, Ca2+, Mg2+ in PM10 collected in five sites of the Costa Rican Metropolitan Area during May to November, 2007.
2. Experimental
2.1 Sampling
]]> For the PM10 sampling, five monitoring sites were selected (Table I). The sites were representative of commercial, industrial and residential areas, all located in the municipalities of San José, Heredia and Belén. All of them belongs to Costa Rica metropolitan area. Figure 1 shows the spatial distribution of the sampling sites.
Sampling campaign was conducted between May and November 2007. Samples were collected three times a week: Mondays, Wednesdays and Fridays. To collect the samples, five high volume air samplers, Thermo Andersen with MFC (mass flow controller) were used during 24 ± 1 h, with a flow rate of 1.13 ± 10% m3 min–1. Once every three samplings, flow calibration of each sampler was performed comparing the readings of pressure drop generated by the flow passing through a calibrated critical orifice, Tisch Variflo model 454, with the sampler drop pressure reading.
For each sampling, ambient temperature and atmospheric pressure readings were taken using a Kestrel anemometer model 5000.
For sampling collection, Whatman Cat No. 1829–932 fiberglass filters were used twice a week. Whatman Cat No. 1851–865 quartz filters were used for the subsequent analysis of metals at least once a week. For the inorganic ions analysis, the samples collected in glass fiber filters and quartz were used for a total of three samples per week, while for analysis of metals only the filters of quartz (a weekly sample) were used. Those filters were conditioned at least 24 h in a desiccator under the following conditions: temperature of 15–30 °C and humidity less than 40%, before being weighted in an analytical balance. Plastic bags with hermetic seals were used to transport the filters to the field and back to the laboratory.
2.2 Chemical analysis
2.2.1 Inorganic ions
From each filter, a strip of 2.54 cm wide and 23.0 cm in length was cut. Then, it was placed in a 150 mL beaker and 75 mL of deionized water (DW) was added. After that, they were located in an ultrasonic bath tempered at 35 °C for 60 minutes. The obtained extract was transferred quantitatively to a 100 mL volumetric flask, then a portion was filtered for further analysis.
The analysis of ionic species was performed by microbore suppresed ion chromatography with suppression using a Dionex ICS–3000 equipment with a quaternary pump, according to the specifications listed in Table II.
]]>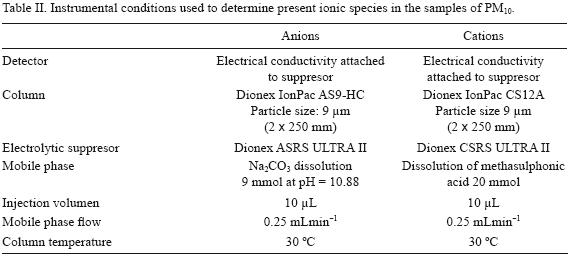
Ionic species were identified and quantified by interpolation in a calibration curve of seven standard solutions prepared in a working range of 0.25 to 10 mgL–1 for anions and 0.10 to 8 mgL–1 for cations, from 1000 mgL–1 stock solutions prepared from certified salts, Merck Suprapur. A fresh calibration curve was prepared for every 20 samples, together with a dissolution of quality control of 5 mgL–1 prepared from a certified Dionex synthetic sample. All the above steps were done in order to monitor the response and reproducibility of the peak shape and retention times.
In addition, the detection limit for each ion were determined. To achieve this, 10 sample blanks of different filters measured once at a time, using the same analysis procedure applied to collected samples, were analyzed. Detection limits were calculated by the concentration that generates a signal equal to the average of the signal of the blanks plus three times the blank's standard deviation. The results are shown in Table III.

2.2.2 Metals
The 2.54 by 23 cm strip for the quartz fiber filters were extracted in a 150 mL beaker by adding 5 ml of ultra–pure concentrated nitric acid and 25 ml DW were added, and heated on a hot plate until almost dryness. The solution remaining was poured into a 25 mL volumetric flask and DW was added up the mark.
Analysis of metals was made using atomic absorption spectrometry with an equipped Perkin Elmer Analyst 700 graphite furnace with autosampler. Metal species were identified and quantified by interpolation in a calibration curve of five standard solutions prepared in a working range from 5 to 50 µgL–1 for V, Cu, Cr, Ni, Pb, Fe and Al, and 5 to 30 µgL–1 for Mn, from the following 1000 mgL–1 certified commercial dissolutions, Perkin Elmer with traceability to NIST. Detection limits in ngm–3, using the IUPAC method, were obtained. The results are shown in Table III.
Blank filters were analyzed for metals and inorganic ions, obtaining lower concentrations of 5% of those found in samples.
]]>3. Results and discussion
3.1 Chemical composition
The average and the standard deviation of valid data concentration of PM10, ions and metals, for each of the sampling site, are presented in Table IV. According to the obtained results, only the sampling site REC–HE, has an annual average value greater than 50 µgm–3 (Reference value for PM10 in Decree 30221–S "Limits inmission of air pollutants in Costa Rica". The average concentration of PM10, were found to be higher for sites located in high traffic flow and industrial areas (41–58 µgm–3) than for those sites in commercial/residential mixed areas (30–33 µgm–3).

The predominants ions were found to be SO42– and Na+ followed by Cl–, PO43–, NH4+ and NO3– for those with high vehicular flow (CCLR–BE, CAT–SJ and REC–HE) while in the sites of the commercial/residential areas, the concentration of PO43– turned out to be greater than that of Cl–. Ionic constituents represent 22.0, 20.0, 14.6, 20.8 and 15.7% of total mass of PM10, collected in the sampling sites of CAT–SJ, JE–SJ, REC–HE, GOB–SJ and CCLR–BE, respectively. In the case of metals, Fe, Al, Mn and Cu concentrations were the most abundant in PM10, while Cr and Ni were the less abundant species with exception of the sites located in Heredia, where the Cr concentration was higher. This can be explained because the city of Heredia has an important concentration of metal–mechanic industries and high traffic flow.
To determine the degree of similarity or difference of the found PM10, ions and metals concentrations between the different sampling sites, the divergence ratio was calculated. It is defined as follows (Park and Kim, 2004):

where xij represents the average concentration of chemical species i in a given sampling site. Meanwhile, j and k represent two sampling sites and p the number of chemical components. If the value of CD is close to zero, data from both sites are considered to be similar. Otherwise, if the value tends to one, both are seen as different sites. Table V presents the calculated CD values from the PM10 and 16 different chemical species (ions and metals) for all the sampling sites. Based on the results, it can be noticed that the biggest differences are found between the site located in industrial zone (CCLR–BE) and those located in urban centers, especially with low vehicular flow such as JE–SJ. However, differences between coefficients are very small, so it can be inferred that large–scale transportation phenomena in the metropolitan area is more important that the contribution of local sources of emission.
]]>
In order to obtain a better explanation of the differences between the monitoring sites, the analyzed chemical species in the particles were divided into 5 classes: crustal origin, contribution of secondary components, sea salt, smoke and metals. The contribution of each category was estimated from the results of chemical analysis and the following guidelines: (1) crustal origin = concentration Al / 0.08, (2) contribution of secondary components = concentration SO42– + concentration NO3– + concentration NH4+, (3) sea salt = 2.54 (concentration Na – 0.3 concentration Al ), and here the expression (concentration Na – 0.3 concentration Al) represents the fraction of sodium that comes only from the marine aerosol (Chan et al., 1997), (4) smoke (calculated from the K fraction of non–crustal original) = concentration K – 0.25 concentration Al (Chan et al., 1997), (5) metals = the total of mass from all metals analized in the particles. The contribution of the five categories to PM10 is shown in Figure 2, where it can be seen that crustal origin particles are the main contributor in all the sites. In addition, it can be noted that contribution of marine aerosol is greater for sites located in Heredia city, due to the pattern of prevailing winds in the large metropolitan area, while the greatest contribution of metals is found in Belén, where there is a significant concentration of industrial activities.

3.2 Chemical forms of the main ionic species present in PM10
Chemical forms of the ions present in particles were determined from the calculation of Spearman correlations (Table VI). It can be seen that NH4+ is strongly correlated with SO42– and NO3–. According to the results showed in Table VI, it is suggested that the species that may be present in PM10 are: NaCl, KNO3, MgSO4, (NH4)2SO4, NaNO3, NH4NO3 and CaSO4.

An important portion of NO3– present in PM10 can reasonably be attributed to NH4NO3 presence. The main mechanism of secondary production of SO42– and NO3– includes ammonium according to the following reactions (Seinfeld, 1998):
]]>

Equations (2) to 5 indicate that each mole of SO42– can remove 2 moles of gaseous phase ammonia and, if there is an excess, it can react with HNO3(g) to produce NH4NO3. Molar concentrations of 2 [SO42–] + [NO3–] obtained for the five monitoring sites, are strongly correlated with molar concentrations of [NH4+], supporting the hypothesis that NH4NO3 and (NH4)2SO4 are the prevalent chemical forms of sulfate and nitrate in PM10 particles (Fig. 3).
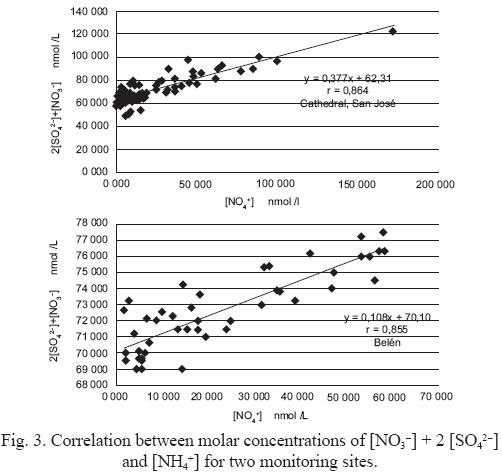
Using this same concept, Chu (2004) defined the index of availability of ammonium (J), as the relations between molar concentration of ammonia, present in PM10 and the concentration required for neutralize the concentration of sulfate and nitrate present in the sample according to the following equation:

If J > 100%, the atmosphere is rich in ammonia. In Figure 3, the slopes of the linear regression curves are <1 for all the sampling sites, indicating that the average values of J are >100%. Because of this, it can be concluded that sampling environments are rich in ammonia.
3.3 Fraction of ionic compounds in PM10 due to the contribution of marine aerosol
The pattern of prevailing winds in the large Metropolitan Area of Costa Rica supports the transport of air masses from the ocean to the central plateau; during the dry season, appears the persistent trade winds (NE), with high speeds of 30 kmh–1 between January, February and March. In the rainy season, the trade winds lessen the intensity in the morning, at approximately 10:00 a.m., bringing the breeze of the Pacific, heading West, Southwest, it enters to this area and forms a "front breeze", encouraging the formation of cumulus–kind clouds of vertical development enough to cause rains and thunderstorms. Because of this, it is important to assess the contribution of marine aerosol in composition of PM10 particles. To estimate this contribution, it is necessary to calculate the fraction of the various inorganic components influenced by marine aerosol. Ions like SO42–, K+, Mg2+ and Ca2+ have both marine and continental sources. Based on mass relation of these ions with respect to Na+, the component due to marine aerosol can be calculated using the following equation (Duce et al., 1983):
]]>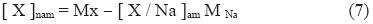
Where:
[X / Na]am= the mass relation of X ion with respect to Na+ in seawater
Mx = ion X concentration in PM10
MNa= Na+ concentration in PM10
Mass relation of ions SO42+, K+, Mg2+ and Ca2+ with respect to Na+ in seawater is 0.25, 0.037, 0.038 and 0.12, respectively (Berg, 1978). The difference between the ion concentration present in particles and component due to marine contribution corresponds to the fraction originated by different sources to the marine aerosol [X]nam. Figure 4 shows the distribution for each sampling site, and the fraction due to marine aerosol as the assigned to other sources. Examining the case of SO4–2, it appears that approximately 87% of mass concentration of this ion has a different origin to marine aerosols. In the case of K+ and Ca2+, this fraction represents at least 95 % of the different sampling sites. On the continent, fraction SO42– that do not come from marine aerosol is a tracer of anthropogenic activities and burning of fossil fuels, Knam mostly originates by burning biomass whereas Canam has a predominantly crustal origin.
3.4 Spatial variation of aerosol acidity
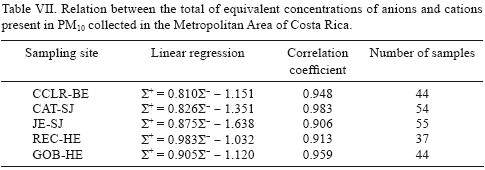
The relation between the total of the equivalent concentration (μeqm–3) of cations divided by the corresponding amount of anions in PM10 can be used to estimate the acidity of aerosols. The characteristics of the linear regression of the total of anions and cations, for the five monitoring sites, are shown in Table VII.
3.5 Enrichment factors
Elementary relation in particles samples can provide important information to understand its possible origin. In this way, enrichment factors of the elements present in aerosols, with respect to the concentration in the crustal material, are frequently calculated in order to identify possible sources and the contribution of anthropogenic emissions to metal levels PM10 (Zoller et al., 1974; Duce et al., 1975). If the value of the enrichment factor is close to the unity, the item has a predominantly crustal origin. However, due to differences between the crustal composition in different regions around the globe, values of enrichment factor between 1 and 10, are considered to be of natural origin. Elements such as Al, Si, Ti, Fe are commonly used as a point of reference for the calculation of factors, because they are very abundant in crustal material and are not significantly affected by pollution. In this study, to estimate those factors Fe was used as the reference, and the upper continental crustal concentration was taken from Taylor and McLennan (1985).
The enrichment factor of an E element in a sample of aerosol, is defined as:

where R is the reference element.
The values of enrichment factors calculated from data of metals concentration of metals present in the particles collected in the Metropolitan Area of Costa Rica, are shown in Table VIII. They have the following sequence: Pb> Cu> V> Mn> Ni> Fe> Al> Cr for the two sites in San Jose city, while for the remaining sites Cu has the higher enrichment factor followed by Pb.

Pb and Cu represent EF values in the range of 10–1000, indicating that concentration of these elements in particles is enriched by sources that are not of crustal origin such as combustion of fossil hydrocarbons, vehicular traffic, emissions of metalworking industries (electrical cable, piping and cabinet industries, for example). Smaller values were obtained for EF V, Mn and Ni showing that its main source is crustal with a very low, almost zero, input from anthropogenic sources.
]]> 3.6 Statistical analysisSpearman correlation matrix was created for each of the five sampling sites of PM10 (Table IX) for data on concentrations of inorganic ions and metals present in PM10. The following patterns can be shown:
–Sampling sites located in areas of high vehicular flow (REC–HE and CAT–SJ) have a well–defined pattern of correlation between PM10, SO42–, NO3– and NH4+, which can be associated with emissions, mainly from mobile sources, indicating that these species could have a common anthropogenic origin.
–For the site CCLR–BE, significant correlations are obtained between species such as metal Cr–Al, Pb–Mn, Ni–V and Pb–Cu showing the influence of both emissions from stationary sources (metalworking industry, combustion processes, etc.) and vehicle exhaust.
–The CCLR–BE, REC–HE and GOB–HE sites showed one important correlation between V and SO4–2 that indicates one possible contribution of emissions originated in industrial combustion of jet fuel. This sites have an important influence of industrial areas located near of them in comparation with San José City, that is mostly a comercial zone.
–It is important to emphasize that for REC–HE and GOB–HE, an important pattern of correlation is observed between: Na–Cl, PM10 –Ca, Ca–Mg and PM10–Mg that demonstrates the importance of crustal contribution and marine aerosol contribution for particle composition on these sites.
In addition, a principal component analysis was applied to particle data, and metal ions in each of the sites. This method is used as an aid in finding and interpreting complex and casual relations between variables of the same set of data. In this study, the most important contribution of analysis of main components is the elucidation of possible sources that could explain the chemical composition of PM10. The results are shown in Table X.
In the case of the sampling site CCLR–BE located in the industrial area of Belén, four factors were found. The first factor, which explains 44.3% of the total variance, includes SO42–, NH4+, V, Pb, Cr, Cu, Ni, Mn and Al, which indicates the important contribution of emission sources from combustion processes of oil and metal–mechanic industry located in the city. The second factor, with a 26.8%, includes the influence of vehicular traffic for having PM10, SO4–2, NO3–. The third factor explains the 16% and may be associated with the crustal origin, this included Mg2+, Fe, Al and Cr. Finally the fourth factor, with 11.5%, includes Na, K and Cl, evidencing the influence of marine aerosol in the composition of PM10.
For CAT–SJ, four factors were found too. The first factor explains 40.8% of total variance and includes PM10, NO3–, SO42–, NH4+, Cu and Cr, it can suggest the contribution of mobile combustion sources in the particles chemical composition. It is important to remember that this point is located in one high traffic flow zone. Comparing with other sites, the possible contribution of mobile combustion sources in this site is higher. The second factor with 14.4%, includes Na and Cl and can suggest the marine contribution. Third and fourth factor, with 22.2 and 14.3%, respectively, show the crustal contribution to PM10 composition and includes K, Fe, Ni and Al. Ni was present in two important minerals, (Ni, Fe)xSy and (Ni, Mg)SiO3•nH2O in soils of this region. The third factor includes K and Al and this can show a possible contribution of feldespats.
The principal components analysis applied to the data generated from collected samples in the transition zones (between commercial and residential areas), likes GOB–HE showed that the main factor that explains the data variance, PC1 with 30.3%, includes PM10, Na, K, Cl, Mg, Ca, leaving out the importance of marine aerosol in the chemical composition of the particles. In addition, a second factor was obtained, linked to emissions from vehicular traffic (SO4–2, NH4+, NO3–, V, Mn), explaining 23.9% of the variance. The third factor includes Mn, Ni, Mg, Ca and K that can shown the crustal contribution, with the 20.5%. It is important indicate that the fourth factor with 21.2%, shows the contribution of antropogenic sources, for example industrial source emisions.
]]>4. Conclusions
It can be concluded that, although the statistical tests indicate that there is no significant difference between particle composition in the sampling sites located in Metropolitan Area of Costa Rica, the patterns of correlation and principal component analysis indicate that the sources that affect the variability of the data differ between sites. That is why, while in CAT–SJ and REC–HE (commercial sites with high traffic flow), there is a strong influence of anthropogenic emissions from mobile sources as evidenced by the correlations between PM10–SO42–, PM10 – NO3–, NO3– – SO42– and NH4+ – SO42–, in the case of CCLR–BE, for being in an industrial area, there are significant correlations of V – SO42–, Pb – SO42–, Cr – Al, Pb – Mn, V – Ni and Pb – Cu. This characterizes the influence of emissions from industrial sources that use heavy fuels such as bunker and large–scale metallurgical processes.
The differences between sources that affect the variability of the chemical composition of the particles determine some properties as the acidity. The particles collected in Heredia have a lower acidity; due to a greater contribution of marine aerosol, San José and Belén have a higher degree of acidity caused in part by higher concentrations of secondary components such as NO3– and SO42–. This suggests that potential impacts in materials, ecosystems and human health can be greater in San José and Belén.
According to data obtained for metals concentrations in particles, only in the case of Pb and Cu enrichment factors are significant (over 10), however, it is necessary to pay attention to the fact that in the case of Belén, V has an important enrichment, that could be explained by the high concentration of industrial activities in the area, where heavy fuels (that contents this element) are consumed.
Due to the geomorphology of the Costa Rican territory, the influence of marine aerosol plays a key role in the chemical composition of PM10 samples, this being responsible for 13% and 5% of magnesium and sulfate, respectively, present in PM10.
Acknowledgments
The authors are grateful to the OVSICORI–UNA, Municipality of San José, Health Ministry and Meteorological Institute of Costa Rica for the collaboration provided to conduct this study.
]]> References
Andreae M. O. and P. J. Crutzen, 1997. Atmospheric aerosols: biogeochemical sources and role in atmospheric chemistry. Science 276, 1052–1058. [ Links ]
Arimoto R., R. A. Duce, D. L. Savoie, J. M. Prospero, R. Talbot, J. D. Cullen, U. Tomza, N. F. Lewis and B. J. Ray, 1996. Relationships among aerosol constituents from Asia and the North Pacific during Pem–West A. J. Geophys. Res. 101, 2011–2023. [ Links ]
Berg W. W. Jr. and J. W. Winchestor, 1978. Aerosol chemistry of marine atmosphere. In: Chemical Oceanography 7th ed. (J. P. Riley and R. Chester Eds.) Academic Press, London, 173–231. [ Links ]
Bessagnet B., A. Hodzic, R. Vautard, M. Beekmann, S. Cheinet, C. Honore, C. Liousse and L. Rouil, 2004. Aerosol modeling with CHIMERE–preliminary evaluation at the continental scale. Atmos. Environ. 38, 2803–2817. [ Links ]
Chan Y. C., R. W. Simpson, G. H. McTainsh, P. D. Vowles, D. D. Cohen and G. M. Bailey, 1997. Characterisation of chemical species in PM2.5 and PM10 aerosols in Brisbane, Australia. Atmos. Environ. 31, 3773–3785. [ Links ]
Chu S.–H., 2004. PM2.5 episodes as observed in the speciation trends network. Atmos. Environ. 38, 5237–5246. [ Links ]
Duce R. A., G. L. Hoffman and W. H. Zoller, 1975. Atmospheric trace metals at remote northern and southern hemisphere sites: Pollution or natural. Science 187, 59–61. [ Links ]
Duce R. A., R. Arimoto, B. J. Ray, C. K. Unni and P. J. Harder, 1983. Atmospheric trace elements at Enewetak Atoll 1, concentrations, sources and temporal variability. J. Geophys. Res. 88, 5321–5342. [ Links ]
Dusek U., G. P. Frank, L. Hildebrandt, J. Curtius, J. Schneider, S. Walter, D. Chand, F. Drewnick, S. Hings, D. Jung, S., Borrmann and M. O. Andreae, 2006. Size matters more than chemistry for cloud–nucleating ability of aerosol particles. Science 312, 1375–1378. [ Links ]
Herrera J. and S. Rodríguez, 2007. Cuarto Informe de Calidad del Aire de la ciudad de San José (2007). Informe técnico, Universidad Nacional, 15–16. [ Links ]
INEC, 2002. Compendio de Estadísticas del Cantón Central de San José. Informe Técnico, Instituto Nacional de Estadística y Censos, 23–26. [ Links ]
Meinrat O. A. and P. J. Crutzen, 1997. Atmospheric aerosols: biogeochemical sources and role in atmospheric chemistry. Science 276, 1052–1058. [ Links ]
Pacyna J. M., 1998. Source inventories for atmospheric trace metals. In: Atmospheric Particles (R. M. Harrison and R. E. van Grieken, Eds.). IUPAC Series on Analytical and Physical Chemistry of Environmental Systems, vol. 5. Wiley, Chichester, UK, 385–423. [ Links ]
Park S. S. and Y. J. Kim, 2004. PM2.5 particles and size–segregated ionic species measured during fall season in three urban sites in Korea. Atmos. Environ. 38, 1459–1471. [ Links ]
Qian Y., C. B. Fu and S. Y. Wang, 1999. Mineral dust and climate change. Advance in Earth Science 14, 391–394 (in Chinese with English abstract). [ Links ]
Seinfeld J. H. and S. N. Pandis, 1998. Atmospheric chemistry and physics from air pollution to climate change. Wiley, New York. 1345 pp. [ Links ]
Simpson D., H. Fagerli, J. E. Johnson, S. Tsyro, P. Wind and J.–P. Tuovinen, 2003. Transboundary acidification, eutrophication and ground level ozone in Europe–Part I. Unified EMEP Model description. EMEP Status Report 1/2003. Meteorological Synthesizing Centre–West, Oslo. 325 pp. [ Links ]
Su W. Y. and C. H. Chen, 1997. The effects of aerosol optical absorption characteristics on lower atmospheric heating rates. Plateau Meteorol. 16, 353–358. [ Links ]
Taylor S. R. and S. M. McLennan, 1985. The continental crust: Its composition and evolution. Blackwell, Oxford, UK. 317 pp. [ Links ]
USEPA, 1997. Technology Transfer Network OAR Policy and GuidanceWebsite. United States Environmental Protection Agency. http://www.epa.gov/ttn/oarpg/naaqsfin/pmhealth.html. (September, 23, 2009). [ Links ]
Xiao H. and C. Liu, 2004. Chemical characteristics of water soluble components in TSP over Guiyang, SW China, 2003. Atmos. Environ. 38, 6297–6306. [ Links ]
Yao X., C. K. Chan, M. Fang, S. Cadle, T. Chan, P. Mulawa, K. He and B. Ye, 2002. The water–soluble ionic composition of PM2.5 in Shanghai and Beijing, China. Atmos. Environ. 36, 4223–4234. [ Links ]
Zoller W. H., R. A. Gordon and R.A. Duce, 1974. Atmospheric concentrations and sources of trace metals at the South Pole. Science 183, 198–200. [ Links ]
]]>