Costing of scaling up HIV/AIDS treatment in Mexico
Costos del tratamiento de VIH/SIDA en México
Sergio Bautista-Arredondo, MScI; Tania Dmytraczenko, PhDII; Gilbert Kombe, MD, MPHII; Stefano M Bertozzi, MD, PhDI, III, IV
IHealth Economics Division at the National Institute of Public Health (INSP), Cuernavaca, Mexico
IIPHRplus. Abt Associates Inc., Washington, DC, USA ]]>
IIICIDE (Centro de Investigación y Docencia Económicas), Mexico City
IVUniversity of California, Berkeley, USA
ABSTRACT
OBJECTIVE: To determine the net effect of introducing highly active antiretroviral treatment (HAART) in Mexico on total annual per-patient costs for HIV/AIDS care, taking into account potential savings from treatment of opportunistic infections and hospitalizations.
MATERIAL AND METHODS: A multi-center, retrospective patient chart review and collection of unit cost data were performed to describe the utilization of services and estimate costs of care for 1003 adult HIV+ patients in the public sector.
RESULTS: HAART is not cost-saving and the average annual cost per patient increases after initiation of HAART due to antiretrovirals, accounting for 90% of total costs. Hospitalizations do decrease post-HAART, but not enough to offset the increased cost.
CONCLUSIONS: Scaling up access to HAART is feasible in middle income settings. Since antiretrovirals are so costly, optimizing efficiency in procurement and prescribing is paramount. The observed adherence was low, suggesting that a proportion of these high drug costs translated into limited health benefits.
Key words: acquired immunodeficiency syndrome; antiretroviral therapy, highly active; costs and cost analysis; health care costs; Mexico
OBJETIVO: Determinar el efecto neto de la introducción de la terapia antirretroviral altamente activa (TARAA) en México sobre los costos anuales totales por paciente en el tratamiento de VIH/SIDA, tomando en cuenta el posible ahorro en el tratamiento de infecciones oportunistas y hospitalización.
MATERIAL Y MÉTODOS: Se hizo un estudio retrospectivo, multicéntrico, mediante la revisión de los expedientes de los pacientes y la recolección de datos de costos unitarios para describir la utilización de los servicios y calcular los costos de la atención de 1 003 pacientes adultos VIH positivos en el sector público.
RESULTADOS: La TARAA no ahorra costos y el costo promedio anual por paciente aumenta después de su inicio debido a los antirretrovirales, que representan 90% del costo total. Las hospitalizaciones disminuyen después de iniciada la TARAA, pero no lo suficiente como para compensar el aumento en costos.
CONCLUSIONES: Incrementar el acceso a la TARAA es factible en países con ingresos medios. Debido al alto costo de los antirretrovirales resulta esencial que se optimice la eficiencia en la compra y prescripción. El apego al tratamiento observado fue bajo, lo que sugiere que una proporción de estos altos costos en medicamentos no se traducen en beneficios a la salud significativos.
Palabras clave: síndrome de inmunodeficiencia adquirida; terapia antirretroviral altamente activa; costos y análisis de costos; costos de la atención a la salud; México
Mexico ranks third in the Americas in the total number of HIV cases reported.1 While AIDS is the 16th leading cause of death in Mexico, it jumps to as high as fourth when only men aged 25 to 34 are considered.2 The disease has been reported in all 32 states of Mexico. From 1983 to 2004, 93979 cumulative cases of AIDS were recorded in Mexico3. Due to delays and underestimation in reporting, the government estimates that 116000 to 177000 people are currently infected with HIV.4
Antiretroviral treatment (ART), when used in combinations of two or more drugs, has dramatically improved the health and lives of people living with HIV/AIDS (PLHA) around the world.5,6,7 However, the high cost and substantial clinical requirements of providing these cocktails, known as Highly Active Antiretroviral Therapy (HAART), have until recently kept them out of the reach of the vast majority of PLHA in low- and middle-income countries. This situation started to change recently in some countries, reflecting the confluence of two factors: First, the price reduction of antiretroviral drugs, which had a substantial impact on the affordability of therapy. In Latin American and Caribbean countries prices dropped as much as 54% from 2001 to 2002 and in some countries they continue to drop as a result of negotiations between ministries of health and pharmaceutical companies.8 Generic production and/or purchasing from unlicensed producers (Brazil, South Africa) also contributed to the reduction in drug prices. Secondly, the international community has intensified its efforts to combat AIDS. In recent years, a number of international initiatives have improved access to ART in poor countries by making financial resources available for the purchase of pharmaceutical products.9,10,11
]]> This study was conducted at the time when Mexico started to significantly increase access to HAART, which happened around 2002. Before that, only patients eligible for care in one of Mexico's five social health insurance institutions had access to free ART. The uninsured population who received care at Ministry of Health (SSA) facilities had more difficulty accessing ART treatment, having no access to the drugs at all or having to pay for it largely out-of-pocket. In 2001 the Ministry of Health committed to providing, by 2006, ART to everyone who needs it, regardless of their insurance status.12 According to official sources, this goal was met by the end of 2003.1 Currently, every patient who approaches the Mexican Health System has access to antiretrovirals either through the traditional Social Security System or through the Popular Insurance System, introduced by the Government as a pilot project in 2001, and significantly expanded over the entire period until 2006.1There are previous studies documenting the economic burden of HIV/AIDS in Mexico,2,13-16 however, this is the first study to our knowledge describing and analyzing the changes in costs associated with characteristics of the provision of HIV/AIDS care, in the context of the rapid scaling up of HAART in Mexico. The authors were especially interested in contrasting the Mexican experience with that of Brazil, which reported overall cost savings due to the decrease in opportunistic infections (OI) and hospitalization. The present analysis compared experiences in facilities of the three major Mexican health subsystems providing HIV/AIDS care -the SSA (Mexican Ministry of Health), IMSS/ISSSTE- the Mexican Social Security Institute and the Mexican Social Security and Services Institute for State Workers which are the two largest social health insurance entities in Mexico; and the National Institutes of Health (INS). The SSA and IMSS together provide health care for more than 90% of the Mexican population. Annual total costs per patient of HIV/AIDS treatment were estimated, as well as the costs of specific treatment categories (including ART, OI drugs and procedures, hospitalizations, outpatient visits, and laboratory analyses) before and after the introduction of HAART.
Material and Methods
Study sites and sample of patients
A multicenter, retrospective patient chart review was conducted and complementary unit cost data were collected to describe the utilization of services and estimate care costs for adult (>18 years of age) HIV+ patients in the public sector in Mexico. A total of 11 health facilities were selected for the study. The analysis was conducted from the perspective of the Mexican Public Health System and considers direct costs exclusively. Sites were chosen to reflect several criteria, including health subsystem, geographic location, and level of care. To ensure representation of the three health subsystems providing care for PLHA in the public sector in Mexico, five sites were selected from the SSA, four from the IMSS/ISSSTE, and two from the INS. Of these 11 health facilities, three are highly specialized tertiary care facilities; seven provide secondary care; and one is a specialized HIV outpatient clinic. For more detail, the general methodology of the study has been described elsewhere.17
As costs and patterns of care are likely to differ considerably between Mexico's largest urban center, Mexico City, and other areas of the country, facilities from three urban centers were selected: Mexico City (six sites), Guadalajara (three sites) and Cuernavaca (two sites). These centers are located in states with varying prevalence of the disease.2 Since HIV cases as well as patient care are concentrated in urban areas, the cities selected not only provide most of the care for HIV patients in these states, but for a significant number of HIV patients from neighboring states as well.
A sample of patients was randomly selected from each study site. The study was approved by the Ethics Committee of the Mexican National Institute of Public Health, and by all health facilities from which information was extracted. Patients meeting three criteria were eligible for inclusion in the study: diagnosis of HIV infection confirmed by ELISA, Western Blot or laboratory culture, or symptomatic AIDS; being 18 years of age or older at the time of the first consultation, and having at least one documented medical visit to a study site between January 1, 2000, and December 31, 2001. Subjects initiating treatment during the study period were deliberately over-sampled to see how cost patterns changed as HAART was initiated. Those who died during the study period were also over-sampled to see how costs changed in the last year of life. For those included in the study, data were captured retrospectively for a period of up to three years as calculated from the last consultation in the study period or until the beginning of the patient chart. A total of 1003 HIV+ patients were selected.3
Study instruments, costing methodology, and data management
Two study instruments were developed. A utilization questionnaire applied at the patient level collected comprehensive data on patient sociodemographic characteristics; clinical events; use of outpatient, inpatient, laboratory services, and prescribed medications. The data were entered from medical charts, into a structured computer-based interface.4 A costing questionnaire was applied at the facility level and data were collected on unit costs of ambulatory, inpatient, and laboratory services and medications.5
]]> In this study, two different approaches were used to generate unit costs. For the most relevant cost categories, namely drug costs and the cost of ART-associated monitoring tests, researchers performed microcosting,18 reviewed the primary purchasing data in the case of drugs, and conducted a detailed analysis of the relevant laboratory procedures.19 For the remainder of the unit costs (cost per bed day, cost per visit, procedures costs, etc.), the facility-specific unit cost estimates provided by the facility's administrative office were used. Treatment costs were estimated multiplying utilization data from patient charts by unit costs. For all years, US dollar 2002 unit costs (including antiretroviral costs) were applied to utilization data. The authors' interest is to describe changes in cost patterns determined by changes in service utilization. Therefore prices are kept as constant. In any event, antiretroviral drug prices have not changed significantly in Mexico since 2002. For the pre- post-HAART initiation analyses, patient-months were aligned according to the date of initiation of HAART. Thus, Year 1 (Y1) refers to the 12-month period beginning with the month when treatment was initiated and Y-1 is the 12-month period prior to initiation; henceforth The five-year period covered by the study (1997-2001) generates data up to five years on either side of the initiation date of HAART (Y-5 to Y5). However, due to the small sample size at the endpoints of the distribution, the reporting of findings was restricted to analysis for Y-3 to Y3.
Results
Characteristics of the sample of patients
As shown in Table I, the patient sample is broadly similar to the national PLHA population in terms of percentage of men, modes of transmission, and sexual preference. The one exception is the low reported level of men who have sex with men (MSM) in the IMSS/ISSSTE, which is likely to be the result of reporting bias due to stigma or fear of discrimination since social security coverage is employment-based. The IMSS/ISSSTE population's educational level is higher, as expected, given that IMSS/ISSSTE insures formal sector employees.
Rapid scaling up of HAART in Mexico
There was a progressive and rapid move toward HAART in the patient sample, as shown in Table II. In 1997, 59% of patients receiving ART were on double therapy and only 11% were on triple therapy. By 2001 the share of patients on HAART had more than tripled (40%), while the share of double therapy recipients had dropped to 35%. The number of patients on monotherapy dropped steadily from 30% in 1997 to 25% in 2001. This relative decline obscures the growth in the absolute number of patients in the sample that receive a single medication, despite treatment guidelines recommending HAART as the norm and double therapy in exceptional cases.
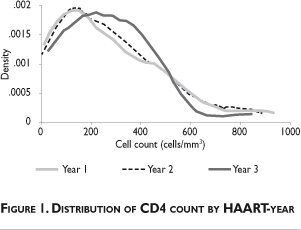
Data from the study appears to confirm the commonly held belief that patients only initiate HAART in Mexico in advanced stages of the disease. The median CD4 count was 150 cells/mm3 at Y-1 (figure 1). In this year there is a high concentration of patients around the median, indicating that a large number of patients who likely qualify for triple therapy are instead receiving either double, mono, or no ART.
]]>
Total costs are substantially higher under triple therapy
There is a marked increase in the average annual cost per patient after initiation of HAART (see Table 3). This is overwhelmingly due to the cost of antiretrovirals. These drugs are the single largest cost component throughout the study period. ART costs are adjusted by estimated adherence, that is, by the estimated number of months on treatment. A detailed analysis of adherence is hindered by two factors: lack of reporting patients' prescription renewals in the clinical files; and the fact that patients were only required to visit a doctor every three months. In between visits, patients were allowed to fill their prescriptions directly at the pharmacy. Patient records rarely reflect whether the prescription was actually filled. In these analyses, ART utilization was assumed to continue for up to three months. If no outpatient visit occurred after three months, then consumption of ART was assumed to cease until the following visit.
A significant drop was observed in cost per patient in the second year after initiation of HAART. Total patient costs declined from an average of $5585 in Y1 to $3575 in Y2. A drop in ART costs accounts for the majority (92.5%) of this decrease. This decrease in ART costs is explained by two factors: reduced adherence in Y2 compared to Y1 and switching to less costly combinations -mono- or double therapy or in some cases lower-cost triple therapy. Outpatient visits, OI drugs and procedures and lab tests also decrease in Y2 suggesting an overall reduction in adherence, not just to ART.
A surprising result, given what has been reported about costs of AIDS care in other countries (Guinness et al. 2002), is that hospitalization costs are not a major determinant of total costs. In this sample, hospitalization represented less than 6% of total costs in any given year. While our use of institutional costs may have somewhat underestimated the cost per bed-day, the hospitalization rate is so low that even a significant underestimation would not change the observation that hospitalization costs are a minor component of total costs.
Study results show a decline in hospital days after patients begin triple therapy (Y-1toY1)). However, this decline is not large enough to offset the increase in costs due to ART. Hospitalization costs would have to be on average more than 25 times higher in Y-1 in order to offset the change in ART drug costs in Y1. Said differently, even a 90% reduction in antiretroviral costs would not be enough to render ART cost-savings in the first year (not even considering the net present value of the cost of future years of treatment).
Costs vary across subsystems
The total cost patterns discussed above are replicated uniformly across the three Mexican subsystems. There is a marked increase in cost after initiation of HAART, and antiretrovirals are the major contributing factor to this increase. Costs are higher in IMSS/ISSSTE and the INS subsystems as compared to the SSA. However, total cost per patient masks some important differences across subsystems. For instance, the INS spends more on lab tests -both in absolute terms and as a share of total spending (6.2% on average across years)- than the SSA (2.7%) and IMSS/ISSSTE (3.3%). Conversely, a significantly larger share of IMSS/ISSSTE's total costs is associated with the provision of outpatient services (7.6% on average compared to 0.7% and 2.4% at the SSA and INS respectively). This, along with the fact that utilization of OI drugs does not vary significantly over the study period, reflects that IMSS/ISSSTE patients received more monitoring visits than their SSA and INS counterparts.
Treatment costs are higher for patients in advanced stages of illness and in their last year of life
]]> As the health of the patient worsens -measured by CD4 count- associated treatment costs increase. Costs of antiretrovirals, hospitalizations, and OI drugs and procedures increase steadily as CD4 falls. There is one exception to that trend, and that is patients with CD4 count greater than 500 receiving ART. This patient population had significantly higher levels of adherence with resultant higher ART costs. To the extent that this group selects for patients who have increased their CD4 on therapy to >500, it is not surprising that they would have higher adherence/utilization.The hypothesis that treatment costs are higher at more advanced stages of illness is further reinforced by the finding that costs are significantly higher for patients in the last year of life. Average costs in the year preceding death are greater across all treatment components, considering either stage of illness (table 4) or HAART-year (table III).
Discussion
Mexico's experience has shown that providing HAART to PLHA is both operationally feasible and affordable for a middle income country with a concentrated epidemic. The costs of the antiretrovirals and the associated monitoring are in the range of one to two times per capita GDP at current prices, depending on the setting in which care is delivered. While this annual expense is probably not competitive with the most cost-effective health interventions supported by the public sector in Mexico, it is not among the least cost-effective either.
Antiretrovirals represent the largest proportion of costs for HIV/AIDS care. Our conservative approach to estimating ART utilization suggests that true costs are likely to be even higher than those estimated here. If the sector is successful at improving the low levels of adherence observed during this period of initial roll-out of triple therapy, drug costs will increase proportionately.20 The Mexican government, like all governments in developing countries facing large costs for the treatment of HIV/AIDS, is concerned about the cost of treatment and is exploring a number of responses. Given that ART costs are the single largest determinant of treatment cost, negotiating lower drug prices and ensuring the use of the most cost-effective combinations of drugs are central to containing costs and ensuring sustainability of the universal coverage program.
This study found evidence that antiretrovirals were not cost saving when they were initially rolled-out and are unlikely to become cost saving even under optimistic assumptions of reductions in drug costs or increases in effectiveness. Although study results show a decline in hospital days after patients begin triple therapy, this decline is not nearly large enough to offset the increase in costs attributable to ART. This is consistent with the findings of a study of HIV/AIDS care costs in Thailand21 yet contrary to data from a Brazilian study which suggested that ART actually has a cost-saving effect due to the sharp decline in annual AIDS-related admissions per patient following introduction of HAART.22 Furthermore, the effect of ART is to prolong life rather than cure HIV, such that on average the burden of hospitalization costs will be postponed, not eliminated. Even though the intention to show that HAART is cost-saving might seem laudable for advocate reasons at fist sight, it might also be dangerous when decision makers realize that this is not the case. HAART represents an enormous improvement in quality of life and life expectancy for those receiving it, and has changed the prognosis of PLHA in developed countries. It does not have to be cost-saving to be socially desirable to continuing scaling up treatment in middle income countries, but it is absolutely necessary that the budgetary implications of this decision be taken into account.
This study also highlights several important issues of quality of care when rapidly scaling up HAART. Enormous variability in patterns of care was found and the sporadic adherence to official guidelines and norms suggest that there is much room for improvement in the effectiveness of HIV/AIDS care. Further investigation is necessary to understand which are the most important determinants of this variability, and should address training and management needs, and especially provider knowledge, practices and incentives.
Adherence to ART treatment emerges as a central problem. While it is difficult to distinguish between poor medical recordkeeping and poor adherence, the authors were conservative in the coding of adherence failure and the problem may be worse than reported. Reasons for patients suspending treatment may be structural, such as stock-outs at the hospital pharmacy, or related to patient behavior. Given the high levels of adherence necessary for effective treatment,23 further investigation into its determinants and strategies for improving patient adherence is warranted.
The highest rate of utilization of inpatient services occurs in the 12 months before patients start triple therapy. This suggests that many patients begin therapy when they already have the advanced stages of the disease. The median CD4 count during this period is 150 cells/mm3 with a mean 223 cells/mm3. The point recommended for the initiation of ART therapy in the latest guidelines is between 200-350 cells/mm3 yet half of the patients are initiating with CD4 count lower than 150. Late initiation of ART therapy also contributes to suboptimal response to treatment.24-26
]]> Study limitationsThe study was not designed to be statistically representative of ART treatment in Mexico; even though within facilities patients were randomly selected, convenience was considered rather than taking a random sample of facilities. Patient records across facilities are not linked. Thus, it is not possible to follow patients across subsystems of the Mexican healthcare system, whether public or private. Hence, costs estimated using this sample do not include the cost of care received in other facilities and therefore likely underestimate actual treatment costs.
Further, the quality of patient records is poor. Symptoms are more often recorded than diagnoses. This precluded the estimation of OI-specific costs. Furthermore, inadequate recording suggests another source of downward bias in the estimation of utilization and thus costs.
For admission, outpatient visits, procedures and some diagnostic tests, unit costs were estimated by facilities for accounting purposes and may not reflect economic costs. However, this is not likely to significantly bias the results, given that antiretrovirals account for 77-98% of total treatment costs.
Acknowledgements
This study was conducted in collaboration with many organizations in Mexico, in addition to the National Institute of Public Health (INSP). The authors would like to thank the National Center for the Prevention and Control of HIV/AIDS (Censida); the Federal Ministry of Health (SSA); the State Ministries of Health of Morelos, Jalisco, and the Federal District; the Mexican Social Security Institute (IMSS); the Mexican Social Security and Services Institute for Workers of the State of Jalisco; the Condesa Clinic in Mexico City; the National Cancer Institute; and the National Institute of Nutrition and Medical Sciences.
References
1. CENSIDA. El SIDA en cifras, 2005. [Accessed on October 2007]. Available from: http://www.salud.gob.mx/conasida/estadis/2005/El%20SIDA%20en%20cifras.pdf [ Links ]
2. Uribe-Zúñiga P, Magis-Rodríguez C, Bravo-García E. AIDS in Mexico. J Int Assoc Physicians AIDS Care 1998; 4:29-33. [ Links ]
3. CENSIDA. Panorama epidemiológico del VIH/SIDA e ITS en México. Mexico City: CENSIDA, 2004. [ Links ]
4. CONASIDA. Panorama epidemiológico del VIH/SIDA e ITS en México. Mexico City: CONASIDA, 2002. [ Links ]
5. Wood E, Braitstein P, Montaner JSG, Schechter MT, Tyndall MW, O'Shaughnessy MV, et al. Extent to Which Low-Level Use of Antiretroviral Treatment Could Curb the AIDS Epidemic in Sub-Saharan Africa. Lancet 2000; 355:2095-2100. [ Links ]
6. Wilkinson D, Floyd K, Gilks CF. Antiretroviral drugs as a public health intervention for pregnant HIV-infected women in rural South Africa: an issue of cost-effectiveness and capacity. AIDS 1998; 12:1675-1682. [ Links ]
7. Marins JR, Jamal LF, Chen SY, Barros MB, Hudes ES, Barbosa AA, et al. Dramatic improvement in survival among adult Brazilian AIDS patients. AIDS 2003; 17:1675-1682. [ Links ]
8. Fitzgerald J, Gomez B. On Open Competition Model for Regional Price Negotiations Yields Lowest ART Prices in the Americas. Presentation at the 8th STI/AIDS Congress, December 2-5, 2003. Puntal del Este, Uruguay. [ Links ]
9. The Global Fund to Fight AIDS, Tuberculosis and Malaria. A force for change: the Global Fund at 30 months. Geneva: The Global Fund to Fight AIDS, Tuberculosis and Malaria, 2004. [ Links ]
10. The White House. Fact sheet: the President's Emergency Plan for AIDS Relief. Washington: The White House, 2003. [ Links ]
11. World Health Organization. Treating 3 million by 2005: making it happen: the WHO strategy. The WHO and UNAIDS global initiative to provide antiretroviral therapy to 3 million people with HIV/AIDS in developing countries by the end of 2005. Geneva: WHO, 2004. [ Links ]
12. Ministry of Health, Mexico. Programa nacional de salud 2001-2006. Ciudad de México: SSA, 2001. [ Links ]
13. Magis-Rodríguez C, Rivera-Reyes MP, Gasca-Pineda R, Gutiérrez JP. El gasto en la atención y la prevención del VIH/SIDA en México: tendencias y estimaciones 1997-2002. Salud Publica Mex 2005;47(5):361-368. [ Links ]
14. Izazola-Licea JA, Ávila-Figueroa C, Arán D, Piola S, Perdomo R, Hernández P, et al. Country response to HIV/AIDS: National Health Accounts on HIV/AIDS in Brazil, Guatemala, Honduras, Mexico and Uruguay. AIDS 2002, 16 (suppl 3):S66-S75. [ Links ]
15. Aracena B, Gutiérrez JP, Bertozzi SM, Gertter P. Cost of AIDS Care in Mexico: What Are its Main Individual Predictors? Arch Med Res 2005;36:560-566. [ Links ]
16. Bautista-Arredondo S, Mane A, Bertozzi SM. Economic impact of antiretroviral therapy prescription decisions in the context of rapid scaling-up of access to treatment: lessons from Mexico. AIDS 2006, 20:101-109. [ Links ]
17. USAID, Partners for Health Reform plus. Costing of HIV/AIDS Treatment in Mexico. [Accessed on march 2008]. Available from: http://www.phrplus.org/Pubs/Tech020_fin.pdf. [ Links ]
18. Drummond MF, O'Brian BJ, Stoddart GL, Torrance GW. Methods for the Economic Evaluation of Health Care Programmes. 2nd edition. Oxford University Press, 1997. [ Links ]
19. Gasca R, Bertozzi S, Mowatt R. Estimación del Costo de la Cuantificación de la Carga Viral en Pacientes con VIH/SIDA: Un Modelo para Guiar la Toma de Decisiones de Inversión en Servicios de Salud. Cuernavaca, México: Instituto Nacional de Salud Pública, 2004. [ Links ]
20. Bautista-Arredondo S, Mane A, Bertozzi S. Economic impact of antiretroviral therapy prescription decisions in the context of rapid scaling-up of access to treatment: lessons from México. AIDS 2006, 20:101-109. [ Links ]
21. Kitajima T, Kobayashi Y, Chaipah W, Sato H, Chadbunchachai W, Thuennadee R. Costs for medical services for patients with HIV/AIDS in Khon Kaen, Thailand. AIDS 2003, 17:2375-2381. [ Links ]
22. Teixeira PR, Vitoria MA, Barcarolo J. Antiretroviral treatment in resource-poor settings: the Brazilian experience. AIDS 2004, 18:S5-S7. [ Links ]
23. Chesney M. Adherence to HAART regimens. AIDS Patient Care STDS 2003, 17:169-177. [ Links ]
24. Dybul M, Fauci AS, Bartlett JG, Kaplan JE, Pau AK. Panel on Clinical Practices for Treatment of HIV. Guidelines for using antiretroviral agents among HIV-infected adults and adolescents. Ann Intern Med 2002; 137:381-433. [ Links ]
25. Antiretroviral treatment. HIV infection in adults: better-defined first-line treatment. Prescrire Int 2004; 13:144-150. [ Links ]
26. World Health Organization. Scaling up antiretroviral therapy in resource-limited settings: guidelines for a public health approach. Geneva, Switzerland: WHO, 2001. [ [ Links ]Accessed on October 2007]. Available at: http://www.who.int/hiv/pub/prev_care/en/ScalingUp_E.pdf
Received on: August 01, 2007
Accepted on: August 25, 2008 ]]>
Sources of financial support: The authors would like to thank the Office of HIV/AIDS of the Bureau for Global Health and the Latin America and Caribbean Health Sector Reform Initiative at the United States Agency for International Development for jointly funding this study.
Address reprint requests to: Stefano M. Bertozzi. Av. Universidad 655, col. Santa María Ahuacatitlán. 62508 Cuernavaca, Mor, México. E-mail: sbertozzi@insp.mx.
1 By the end of 2006, the Popular Insurance System covered 5.1 million families in Mexico.
2 Two of them, explain the trend of HIV/AIDS total expenditures in Mexico using the National Health Accounts on HIV/AIDS;13,14 one15 analyzes the individual socio-demographic characteristics of patients that explain the variability of treatment costs; and one analyzing the inefficiencies associated with patterns of prescription of ARVs in Mexico.16
3 A convenience framework was used and the sample size was designed to provide representativeness of the sites included in the study.
4 The interface was programmed in Microsoft Visual Basic 6 and the data stored in Microsoft Access 2000.
5 The data were gathered using a structure computer-based interface programmed in Microsoft Excel XP 2002.