Characterization of Bone Cements Prepared with Either Hydroxyapatite, α-TCP or Bovine Bone
Caracterización de cementos óseos preparados con hidroxiapatita, α-TCP o hueso bovino
R.F. Vargas-Coronado*, L.H. Chan-Chan*, J.M. Cervantes-Uc*, J.V. Cauich-Rodríguez*, M.C. Piña Barba**
* Centro de Investigación Científica de Yucatán, A.C. Calle 43 # 130, Col. Chuburná de Hidalgo CP 97200, Mérida, Yucatán.
]]> ** Instituto de Investigaciones en Materiales-UNAM Circuito Exterior s/n Cd. Universitaria Delegación Coyoacán, C.P. 04510 México, D.F.
Correspondencia:
Juan V. Cauich Rodríguez
Correo electrónico: jvcr@cicy.mx
Fecha de recepción: 5 de Febrero de 2013.
Fecha de aceptación: 18 de Abril de 2013.
ABSTRACT
]]> In this work, we report the preparation of bone cements by using methyl methacrylate (MMA) as a base monomer and either hydroxyapatite (HA), alpha tricalcium phosphate (α-TCP) or bovine bone particles as bioactive fillers.In general, it was observed that curing times increased by the addition of any of these fillers (from 4 to 6.7 min). Maximum temperatures decrease slightly by the addition of 20 wt.% of either α-TCP or bovine bone (80.3°C and 73.2°C respectively) but it did not change by the addition of HA (84.3°C) with respect to PMMA only bone cement used as control. Residual monomer content was lower than 4% in the bioactive bone cements.
By using α-TCP or bovine bone compressive strength increased with respect to the unfilled bone cement but it was reduced when HA was used. However, all these formulations fulfill the 70 MPa required for bone cement use. Flexural strength was increased by using either a-TCP o bovine bone but the addition of HA decreased this properties compared to the base bone cement. However, the minimum flexural strength (50 MPa) was fulfilled only in those experimental formulations containing low amounts of α-TCP. The minimum tensile strength (30 MPa) was satisfied by all formulations but it was always lower than the exhibited by the unfilled bone cement.
Keywords: bone cements, hydroxyapatite, α-tricalcium phosphate, bovine bone.
RESUMEN
Este trabajo reporta la preparación de cementos óseos utilizando metacrilato de metilo (MMA) como monómero base y rellenos bioactivos tales como hidroxiapatita (HA), fosfato tricálcico alfa (α-TCP) o hueso bovino.
En general, los tiempos de curado aumentaron con la inclusión de estos refuerzos (de 4 hasta 6.7 min). La temperatura máxima alcanzada durante la polimerización del cemento disminuyó ligeramente al adicionar 20% de α-TCP o hueso bovino (80.3°C y 73.2°C respectivamente) y se mantuvo sin cambio en las formulaciones con HA (84.3°C) con respecto al control de solo PMMA. El contenido de monómero residual en los cementos bioactivos fue menor al 4%.
La presencia de α-TCP o hueso bovino aumentó la resistencia a la compresión del cemento base y la adición de HA la disminuyó, cumpliendo en todos los casos con la resistencia mínima a la compresión (70 MPa) sugerida para su uso como cemento óseo. La adición de α-TCP o hueso bovino aumentó la resistencia a la flexión del cemento base pero la adición de HA la redujo aunque el requerimiento mínimo de resistencia a la flexión (50 MPa) fue cumplido solamente al usar concentraciones bajas de α-TCP. La resistencia tensil mínima (30 MPa) fue satisfecha por todas las formulaciones aunque siempre fue menor que la exhibida por el cemento base.
Palabras clave: cementos óseos, hidroxiapatita, fosfato tricálcico alfa, hueso bovino.
]]>INTRODUCTION
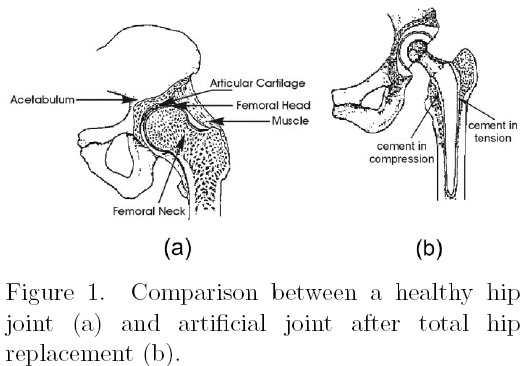
Bone cements are polymeric biomaterials used with the aim of fixing articular prosthesis as it is shown in Figure 1. These materials are used not only as fillers between the metallic prosthesis and the bone but also with the purpose to transfer complex mechanical loads. Conventional bone cements are prepared with a liquid component, mainly methyl methacrylate monomer, and a solid part, mainly composed of poly(methyl methacrylate). In spite of their great used in the modern orthopedics they suffer from high residual monomer content, high polymerization exotherms, high shrinkage and poor bioactivity. These properties in turn lead to chemical and thermal necrosis of the surrounding tissue, which along with their poor mechanical performance, cause the aseptic loosening of the implant. Due to these disadvantages, new bone cement formulations include low toxicity activators, low heat of polymerization monomers and the use of bioactive ceramics in order to improve their biocompatibility [1-5].

It has been reported that between 200,000and 300,000 total hip and knee replacements are conducted annually in U.S.A only, costing the health system approximately $13.8 billion US [6-7]. Worldwide it is estimated that 800,000 total hip replacements are performed annually [8]. Mexican state hospitals, IMSS and ISSSTE, reported in 2008 more than 10,000 hip replacements where 11,000 bone cement kits were used with a $10 million pesos associated cost [9].
Therefore, considering the current limitation of modern bone cements and their importance in the Mexican and worldwide orthopedics, this work is aimed to improve the biocompatibility of bone cements formulations by using either hydroxyapatite (HA), alpha-tricalcium phosphate (α-TCP) or bovine hydroxyapatite. HA is the mineral component of bone and teeth, and is considered as biocompatible, non resorbable and osteoconductive ceramic whereas α-TCP can be soluble at physiological pH and therefore, capable of generating a porous substrate with osteointegration potential. On the other hand, bovine bone is also osteoconductive and has been used as a filler in many orthopedic and dent al applicati ons , fo r (example, Bio-Oss®, Osteograft®, Nukbone®, etc.
The addition of these bioactive fillers is expected to improve osteoblast adhes1 on [10], reducing at the same time, the aseptic loosening of implant, without sacrificing other properties. Therefore, these bone cements were studied in terms of residual monomer content (RMC), curing properties such as curing time and exotherm of reaction in addition of their tensile, compressive and bending properties. The assessment of the mechanical properties on bone cements are of prime importance as the external loads applied during several physical activities can exceed bone cement capability to sustain these loads. Tensile stresses can be found in the lateral portion of the implant due to its bending while compressive stresses can be found in the medial region when a person is standing (see Figure 1b). Finally, it has been proposed that a combination of tension, compression and shear is occurring during the in vivo loading in an artificial joint [11].
MATERIALS AND METHODS
]]> a) Bone cement preparationExperimental bone cements were prepared by hand mixing a liquid (L) and a solid (P) component at P/L=2 (g/ml). The liquid component was composed of methyl methacrylate (with 100 ppm of hydroquinone) and N,N-dimethyl-p-toluidine (DMPT) as activator. The solid component was formulated with a commercial acrylic (Nictone®) and benzoyl peroxide (PBO) (reference bone cement, PMMA).
Hydroxyapatite, from Plasma Biotal (average particle size of 9.3 μm) or α-TCP (average particle size of 7.6 μm) was added to the solid component at 5, 10, 15 and 20 wt.% (A1-A4 and B1-B4, respectively). Bovine bone particles (average particle size of 245 μm) were incorporated only at 20 wt.% (C4).
b) Determination of physical and thermal properties
The content of residual monomer (RMC) was calculated by 1H NMR on a Varian Gemini 200 (0.050 g in 0.6 ml CDCl3). The monomer was quantified on at least two bone cements plates (3x1x0.1 cm) after 7 days of preparation. The areas for the CH2=C- at δ 5.6 ppm and 3.75 ppm for OCH3 group of the methyl methacrylate monomer were used to determine the percentage of monomer present in the total sample.
Peak temperature and curing time were determined according to ISO 5833 (annex C) at 20°C using a teflon mould [12]. The change in temperature with time was recorded immediately after mixing the powder and the liquid. Curing time (tc) is defined as:
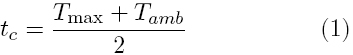
where Tamb is the temperature in the mould at the beginning of the experiment and Tmax is the maximum temperature reached during the polymerization. An average of at least three measurements for each condition is reported.
The glass transition temperature, Tg, of experimental bone cements was determined by means of a DMA-7 (Perkin-Elmer) in the extension mode. Bone cements machined as 20x 3x 0.1 mm strips were deformed under a static force of 60 mN and a dynamic force of 40 mN. Experiments were conducted from -50°C to 150°C at a heating rate of 2°C/min, 1 Hz and under nitrogen flow. The Tg was determined from the peak of the Tan δ vs temperature curve.
]]> Porosity was determined in both filled and unfilled composites from density measurements as the complement of the ratio of the observed and theoretical densities. The density was determined by the Archimedes' principle using a density kit attached to an Ohaus Voyager V12130 balance. Water at 20°C was used as the standard of known density. Theoretical densities were calculated following the rule of mixtures with ρHA=3.156 g/cm3, ρPMMA=1.2 g/cm3.
c) Determination of mechanical properties
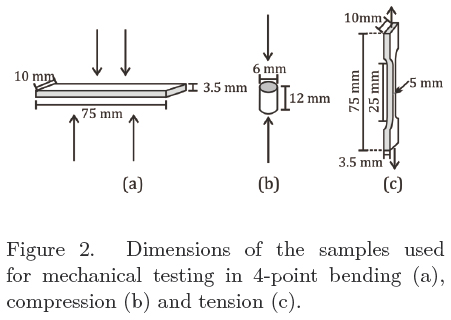
Filled and unfilled bone cements were tested on tension, compression and bending. Tensile tests were conducted using ISO 527 dumbbell specimens. A crosshead speed of 5 mm/min was used and the final deformation was obtained by means of a contact extensometer. From the load vs. displacement curve Young's modulus (ET), tensile strength (σT) and strain (ε) were obtained. Compression tests were carried out according to ISO 5833 (annex E) at 20 mm/min with cylinders of dimensions as indicated in Figure 2b where compressive strength (σC) and modulus (EC) were obtained. Bending modulus (EB) and bending strength (σB) were obtained at 5 mm/min by means of a 4-point bending experiments also according to ISO 5833 (annex F). All samples were tested using an Instron 1125 after storing the specimens at 25°C during 1 week. At least 5 specimens for each type of testing were used.
For comparison purposes Simplex P (Howmedica), CMW 3 (DePuy), Cemfix (Teknimed) and Osteobond (Zimmer) were prepared according to manufacturer instructions and mechanically tested. Fracture surfaces of the bioactive bone cement° were obéerved after the tensile experiments by using a JEOL JSM 5900-LV SEM.
RESULTS AND DISCUSSION
a) Curing properties of bioactive bone cements
]]> Table I summarizes the properties of cements prepared with methyl methacrylate and bioactive fillers. From this table it can be seen that curing times were in the 4.4-6-7 min. range, being the longest time observed with 5 or 10 wt.% of HA, 15 wt.% of α-TCP and 20 wt.% bovine bone. The increase in curing time can be explained as the L/P ratio changed from 0.5 (unfilled bone cement) up to 0.62 (bone cements with 20 wt.% of the filler) i.e. the acrylic was substituted by the ceramic particles. Under this condition, there are less initiator (BPO present in the acrylic) and more monomer (more inhibitor) leading to an incomplete monomer conversion. These curing times although different to those exhibited by commercial bone cements (approximately 10 min.) can be extended by an adequate control of the PBO/DMPT ratio. A suitable curing time is required in order to provide enough time for the cement to be placed by the surgeon in the femoral cavity, either by hand or by using an injection gun.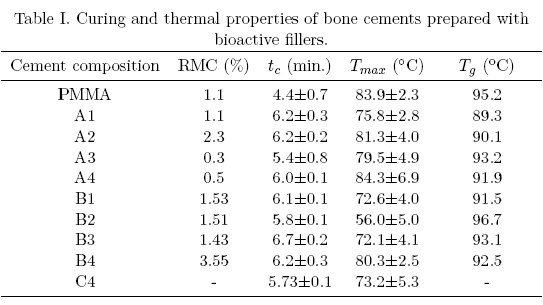
The maximum temperature reached during the polymerization were between 56°C and 84°C being the latest similar to those exhibited by CMW-3 (84.7 C), Osteobond (83.9 C) and Simplex P (89.7°C) [13]. The addition of α-TCP and bovine bone tended to decrease the maximum temperature when compared to the control bone cement (PMMA only) although HA incorporation did not affect this parameter.
Table I also shows that the amount of residual monomer increased when the filler concentration increased, being as high as 3.5% in the bone cement prepared with 20 wt.% α-TCP (B4). Residual monomer arises from the incomplete polymerization of methyl methacrylate, and is present even in those formulations without the filler when it reached 1%. If we consider and initial monomer concentration of 33%, and taking into account the residual monomer, we can obtain a degree of conversion close to 97%. Although RMC in the 2-6% range has been reported [13], this parameter is relevant to its clinical performance as it can lead to pulmonary embolisms and patient death. In this regard, it has also being reported that as the amount of residual monomer increase, the glass transition temperature is simultaneously lowered due to its plasticizing effect. However, in our study this was not observed as the Tg was located between 89°C and 96°C.
b) Mechanical properties of bioactive bone cements
Table III shows the mechanical properties of commercial bone cements while Tables III to V (table IV)shows the mechanical behavior of the experimental bioactive bone cements. From these tables it can be seen that commercial bone cements perform better as the fulfill the minimum compressive strength (70 MPa) and bending strength (50 MPa) required for bone cement use as recommended by the ISO 5833 standard in addition to the minimum tensile strength (30 MPa) generally accepted for this application [11,14-17]. Most of the experimental bioactive bone cements developed during the course of this study, fulfill the minimum compressive strength and the minimum tensile strength but not the minimum bending strength required for bone cement use. Only those formulations containing 5 wt.% and 10wt.% of α-TCP fulfill all the requirements.
]]>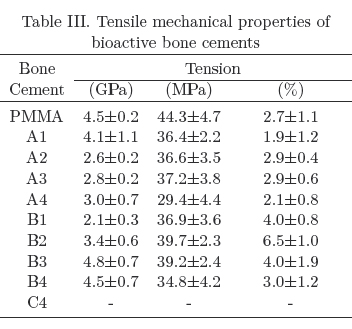
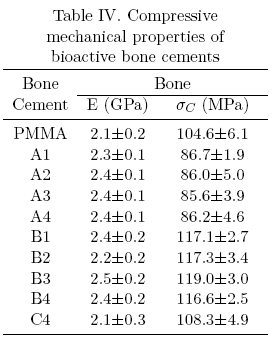
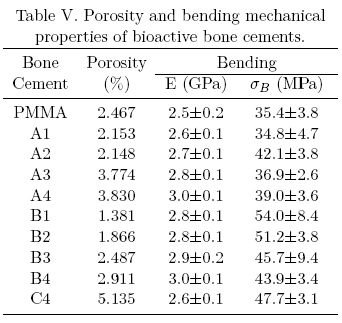
The mechanical behavior exhibited by the experimental bone cements can be explained in several manners including Tg, porosity and polymer-ceramic interface (adhesion, size and size distribution of the filler), etc. [18]. In the first case, the glass transition temperature does not explain the mechanical behavior as all bone cements showed a similar Tg. On the other hand, porosity in bone cements containing HA and α-TCP was similar to unfilled bone cements and only those formulations with bovine bone exhibited a higher porosity. The later behavior can be explained by the higher particle size (250 /xm for bovine bone vs. less than 10 μm for HA or α-TCP). Low porosity bone cements exhibited higher compressive and bending; properties but lower tensile properties (see B1 formulation) when compared with the unfilled bone cement. It is known that materials that are strong in compression are weak in tension but this does not explain the observed properties as the porosity was not controlled. Furthermore, the control bone cement exhibited a. high porosity but performed well in tension. Taking all these results together, we can say that α-TCP and bovine bone liad a reinforcing effect in compression and bending when compared to the unfilled bone cement.
Finally, the observed mechanical behavior can be explained in terms of the poor adhesion between the polymeric matrix and the ceramic filler in addition to a poor dispersion of the ceramic as shown in Figure 3.
]]>
Previous studies have demonstrated that osteoblast adhesion is improved in bone cement formulations containing α-TCP over those containing HA [19] 20% HA. However, it isexpected that bone cements prepared with bovine bone particles should exhibit a similar biocompatibility, a behavior that has to be proved experimentally.
CONCLUSIONS
Bioactive bone cements with good thermal and mechanical properties were prepared through the addition of HA, α-TCP and bovine bone particles to a PMMA matrix. It was observed that in all formulations the amount of residual monomer content never exceeded 4% and that maximum temperature reached was never higher than 90°C. Curing times were shorter that those exhibited by commercial bone cements (6 min. vs. 10 min.) although this parameter can be adjusted by varying the PBO and DMPT concentrations. Mechanical properties of the experimental bone cements were comparable to commercial formulations, mainly in compression when α-TCP was used. However, these new formulations have the advantage of providing an osteoconductive support for bone growth and improved mechanical performance. It can be concluded that these bioactive bone cements are a good alternative to current commercial formulations as they offer good physicochemical and mechanical properties. However, the cost associated with HA and α-TCP production and bovine bone cleaning has to be considered. Currently, we are working in the addition of antibiotics to improve their performance.
Acknowledgements
The authors wish to thank CONACYT (Project J27664U) for financial support.
REFERENCES
]]>1. Liso P.A., Vazquez B., Rebuelta M., Hernández A.L., Rotger R., San Román J. "Analysis of the leaching and toxicity of new amine activators for the curing of acrylic bone cements and composites". Biomaterials 1997, 18: 15-20. [ Links ]
2. Tanzi M.C., Sket I., Gatti A.M., Monari E. "Physical characterization of bone cement cured with new accelerator systems". Clinical Mater. 1991, 8: 131-136. [ Links ]
3. Saito M., Maruoka A., Muri T., Sugano N. and Hino K. "Experimental studies on a new bioactive bone cement: hydroxyapatite composite resin". Biomaterials 1994, 15: 156-160. [ Links ]
4. Hamizah A.S., Mariatti M., Othman R., Kawashita M., Noor Hayati A.R. "Mechanical and thermal properties of polymethyl methacrylate bone cement composites incorporated with hydroxyapatite and glass-ceramic fillers". J Appl Polym Sci. 2012, 125: E661-669. [ Links ]
5. Lopez-Heredia M.A., Sa, Y., Salmon P., de Wijn J.R., Wolke J.G.C. Jansen J.A. "Bulk properties and bioactivity assessment of porous polymethylmethacrylate cement loaded with calcium phosphates under simulated physiological conditions". Acta Biomaterialia 2012, 8: 3120-3127. [ Links ]
]]>6. Goswami T., Alhassan S. "Wear rate model for UHMWPE in total hip and knee arthroplasty". Mater. Design 2008, 29:289-296. [ Links ]
7. Tsukimura N., Yamada M., Aita H., Hori N., Yashino F., Chang-Il Lee M., Kimoto K., Jewett A., Ogawa T. "N-acetyl cysteine (NAC)-mediated detoxification and functionalization of poly(methyl methacrylate) bone cement". Biomaterials 2009, 30: 3378-3389. [ Links ]
8. Li C., Gragner C., Del Schutte Jr H., Biggers Jr S.B., Kennedy J.M., Latour Jr R.A. "Progressive failure analysis of laminated composite femoral prostheses for total hip arthroplasty". Biomaterials 2002, 23: 4249-4262. [ Links ]
9. Pacheco-Salazar O. "Preparación y caracterización de cementos óseos acrílicos con partículas núcleo-coraza de PBA- P(MMA-co-St) como fase sólida". Thesis MSc. Centro de Investigación Científica de Yucatán, A.C. 2012. [ Links ]
10. Anselme K. "Osteoblast adhesion on biomaterials". Biomaterials 2000, 21: 667-681. [ Links ]
]]>11. Lewis G. "Properties of acrylic bone cement: State of the art review". J. Biomed. Mater. Res. Appl. Biomater. 1997, 38: 155-182. [ Links ]
12. SO 5833:1992 (E), Implants for surgery - Acrylic resin cement, International Standard Organization, 1992. [ Links ]
13. Kühn K.D. "Bone cements: Up to date comparison of physical and chemical properties of commercial materials". Springer (Berlin), 2000. [ Links ]
14. Kusy R.P., "Characterization of self-curing acrylic bone cements". J. Biomed. Mater. Res. 1978, 12: 271-305. [ Links ]
15. Wright T.M. and Li S. Biomaterials. In Orthopedic Basic Science: Biology and Biomechanics of the musculoskeletal system, 2nd Ed. Ed.: Buckwalter J.A., Einhorn T.A. and Simon S. R. American Academy of Orthopedic Surgeons (Illinois) 2000: 181-215. [ Links ]
]]>16. Harper E.J., Behiri J.C., Bonfield W., "Flexural and fatigue properties of a bone cement based upon polyethylmethacrylate and hydroxyapatite". J. Mater. Sci., Mater. Med. 1995, 6: 799-803. [ Links ]
17. Dunne N. "Mechanical properties of bone cements in Orthopedic bone cement"s. Woodhead Publishing (Cambridge) 2008. [ Links ]
18. Canul-Chuil A., Vargas-Coronado R., Cauich-Rodríguez J.V., Martínez-Richa A., Fernández E., Nazhat S. "Comparative study of bone cements prepared with either HA or α-TCP and functionalized methacrylates". J. Biomed. Mater. Res. Appl. Biomat., 2003, 64B: 27-37. [ Links ]
19. Salih V., Mordan N., Abou Neel E.A., Armitage D.A., Jones F.H., Knowles J.C., et al. "Surface characterization of various bone cements prepared with functionalized methacrylates/bioactive ceramics in relation to HOB behavior". Acta Biomaterialia, 2006, 2: 143-157. [ Links ]
]]>