Introduction
The distribution of tropical ecosystems is often correlated with soil fertility indicators (Russo et al., 2005; Turner et al., 2018). Among lowland tropical ecosystems, soil fertility may vary widely and thus might affect plant abundance and distribution. In many tropical regions of the world, the boundaries between forests and savannas are often contiguous (Hoffmann et al., 2012). However, the differences in plant species composition, abundance, and spatial distribution are enormous. Indeed, several factors explaining such differences are involved, but physical, chemical, and biological characteristics of soils likely play a significant role. Tropical forest soils usually have higher soil organic carbon and essential nutrients such as nitrogen, phosphorus, and potassium concentrations than savanna soils (van der Sande et al., 2018).
Soil fertility may affect the recovery of the vegetation following a disturbance in lowland tropical ecosystems (Turner et al., 2018; van der Sande et al., 2018) because it may have a substantial effect on plant communities during ecological succession (Powers and Marín-Spiotta, 2017). However, little is known about how soils from different ecosystems, such as savanna and forests, influence the successional trajectories of pioneer tropical trees (Estrada-Villegas et al., 2019). Consequently, analyzing the role of the soil in plant performance in tropical ecosystems is vital for restoration projects. Few studies have examined the effects of different soil types on the survival and growth of seedlings of pioneer tropical tree species.
A key but frequently neglected factor in the tropical plant species performance is the arbuscular mycorrhizal fungi (AMF). An increase in phosphorous and nitrogen soil concentrations can increase plant growth, survival, and diversity in tropical forests (Ayala-Orozco et al., 2017; Werden et al., 2018). AMF play an active role in the absorption of water and nutrients (mainly phosphorous and nitrogen in soils with low nutrient availability), improves survival, growth, resistance to pathogens, and abiotic stress (Veiga et al., 2013; Bender et al., 2014; Wagg et al., 2014; van der Heijden et al., 2015). Therefore, the effects of the soil type and plant-AMF symbiosis should be evaluated for improving the growth rates of tropical plant species on soils contrasting in fertility.
Ecological studies on AMF have focused on analyzing the role of AMF on the structure of plant communities, in ecological succession, and their interaction with different plant species (Janos, 1980a, b; Zangaro et al., 2012, 2013; Martínez-García et al., 2015). Studies have shown that plant-AMF symbiosis increases seedling survival and growth, and thus, plant inoculation with AMF could be a valuable procedure in ecological restoration projects (Janos, 1980a; Kardol and Wardle, 2010). However, plant-AMF compatibility and soil type could play a major role in the symbiosis with tropical tree seedlings under field conditions (Janos, 1980a, b; Guadarrama et al., 2004a, b; Zangaro et al., 2012, 2013, 2018; Martínez-García et al., 2015; Turner et al., 2018; van der Sande et al., 2018). Therefore, under field conditions, these aspects should be analyzed for a more appropriate evaluation of the AMF use in ecological restoration projects (Álvarez-Sánchez et al., 2009).
The AMF potential on plant growth of pioneer tree species (colonizing species of open spaces, mostly dispersed by wind and insects, growth at very fast rates, satisfying the need for a lot of nutrients, and a high affinity for AMF species) has been reported in several studies (Janos, 1980a, b; Guadarrama et al., 2004a; Zangaro et al., 2012, 2013; Martínez-García et al., 2015); however, its effectiveness in pioneer tree species under contrasting edaphic conditions is unknown. Humid tropical forests in Mexico are rapidly vanishing (Dirzo and García, 1992), and thus ecological restoration projects are urgently needed. Those that include the plant-AMF symbiosis are particularly interesting (Álvarez-Sánchez et al., 2009). Because the AMF affinity of tree seedling species can vary from one species to another, and it depends mostly on soil fertility levels, it is necessary to compare how the tropical rainforest and savanna soils influence the growth rates of pioneer tree species and how the presence of AMF modifies such effects. The objective of this study was to evaluate the impact of soil type (forest and savanna soils) and AMF inoculation on growth (height and stem diameter) of Entero-lobium cyclocarpum, Cassia grandis, Cochlospermum vitifolium, and Heliocarpus appendiculatus, which are pioneer tree species of humid tropical forests.
Materials and methods
Experimental Area
The experiment was carried out in La Trinidad Yaveo, Santiago Yaveo (Bajo Mixe region, Oaxaca, Mexico, 17° 27’ N, 95° 32’ W, 110 masl) in the Gulf of Mexico Coastal Plain. The climate is warm and humid, with 25 °C and 2300 mm of mean annual temperature and precipitation, with a rainy season from July to September (> 600 mm per month) and a dry season from January to April (< 60 mm per month) (Reyes-Carmona and Zamora-Serrano, 1974; Álvarez-Lopeztello et al., 2019b). The experiment was conducted from January to June 2019 in a tree nursery situated close to savanna and tropical rainforest fragments intermixed with induced grasslands, croplands, and forest plantations.
Soil sampling, sterilization, and analysis
The soils used in this study were collected from fragments of tropical rainforest and savanna located near the tree nursery where the experiment was conducted. In each ecosystem type, eight sites were randomly selected, and in each location, soil subsamples at rhizo-spheric levels (0-20 cm depth) were collected. Before collecting the soil, the litter layer was removed manually. The soil samples were sieved and mixed with sand (2mm in diameter) at 1:1 ratio (v/v) and sterilized with water-steam for 72 hours to eliminate all AMF propagules (Zangaro et al., 2018). Subsequently, the soil-sand mixture was transferred to black plastic bags (1000 cm3). To restore the original microbial community, except the AMF, 70 mL of a filtrate (with sterilized water) from the two soil types, were added to each plastic bag with sterilized soil, according to its source (forest or savanna soils), every 20 days, for four times. To produce the filtrate, 500 g of fresh soil from each ecosystem was suspended in 4.0 L of distilled water, and the supernatant was filtered through a filter (0.22 μm), to retain the AMF propagules (Zangaro et al., 2018).
We measured the following soil properties: soil texture (Bouyoucos), pH (1:2 soil: water ratio) and electrical conductivity (EC) with a digital potentiometer, soil organic carbon (SOC) and organic matter (SOM, Walkley-Black method), available phosphorus (P, Bray and Kurtz method), inorganic nitrogen (N, micro-Kjeldahl method), cation exchange capacity (CEC, Ag TU method) (SEMARNAT, 2002), and exchangeable cations (Ca and Mg, colorimetry method).
Seedling growth and AMF inoculation
Trap pots were established for the propagation of AMF spore species, with the soils of both vegetation types and sorghum (Sorghum vulgare L.) as a trap plant. They were kept under greenhouse conditions for 11 months to allow the propagation of most of the genera of AMF and the establishment of the consortium. The dominant AMF species in the inoculum (consortium) were: Acaulospora mellea Spain & N.C. Schenck, Acaulospora scrobiculata Trappe, Acaulospora spinosa C. Walker & Trappe, Claroideoglomus drummondii C. Walker & Schuessler, Diversispora spurca C. Walker & A. Schuessler, Funneliformis geosporus C. Walker & Schuessler and Septoglomus constrictum Sieverd., G. A. Silva & Oehl (Álvarez-Lopeztello et al., 2019a).
The plant species used in this experiment were all species that can become established on early successional plots typical of humid tropical forests: Enterolobium cyclocarpum (Jacq.) Griseb., Cassia grandis L.f., Cochlospermum vitifolium (Willd.) Spreng. and Heliocarpus appendiculatus Turcz. (Janzen, 1971; Pennington and Sarukhán, 2005), and previously recognized as AMF forming (Habte and Musoku, 1994; Siqueira et al., 1998; Guadarrama et al., 2004a; Allen et al., 2005). The collected seeds were disinfected with 2 % sodium hypochlorite for 20 minutes, washed four times with distilled water, and germinated in plastic trays with sterilized sand; before germination, the seeds of E. cyclocarpum and C. grandis were scarified with concentrated sulfuric acid for 8-10 minutes. The seedlings were transplanted into plastic bags 20 days after germination. Each seedling received 4-5 g of fresh inoculum soil containing approximately 586 ± 32.78 (mean ± standard deviation) AMF spores, colonized roots, and hyphae. The uninoculated seedlings received the same quantity of soil (4-5 g) from the AMF inoculum, which was previously steamsterilized with an autoclave.
The seedlings were grown under tree nursery conditions covered with a shade mesh of 50 %. The seedlings were watered with sterilized water when it was necessary. We conducted a 2 x 2 factorial experiment with two factors: soil type (rainforest [TS] or savanna [SS]) and AMF (inoculated with AMF [+AMF] or without AMF [-AMF]) and 30 replicates per treatment per species (except for C. grandis with 29 seedlings). Seeding growth was monitored by measuring the height and the basal width of the stem, every 20 days until the end of the experiment (147 days after the beginning of the experiment). Heigth (cm) and stem diameter (mm) were measured for each seedling with a flexometer (height) and a digital Vernier (stem diameter). The relative growth rate (height and stem diameter), defined as the difference between natural logarithms of the final and initial values in stem height or stem diameter over the elapsed time (days), was used for assessing plant growth.
Arbuscular mycorrhizal fungi colonization
At the end of the seedling growth in the experiment, root fungal colonization was assessed. Fine roots (approximately 2 mm in diameter) were extracted directly from 15 seedlings per species in the four treatments (for each seedling, 20 root segments of 2.5 cm in length were cut). Roots were cleared (10 % KOH and 5 % H2O2), acidified (1 % HCl) and stained with trypan blue (0.05 %) (Brundrett et al., 1996), and total AMF colonization was estimated using the magnified intersection method observing the presence of different AMF structures (arbuscules, hyphae, spores, and vesicles) at 40X magnification (McGonigle et al., 1990).
Data analysis
The soil properties were compared using the non-parametric Kruskal-Wallis test (P < 0.05). The interaction between soil type (rainforest and savanna soils) and AMF inoculation (inoculated with AMF and without AMF) using the Scheirer-Ray-Hare extension of the Kruskal-Wallis test (Sokal and Rohlf, 1995) was evaluated. To compare the differences in growth height (cm) and stem diameter (mm) rates, and mycorrhizal colonization (%) between the different treatments, Kruskal-Wallis tests were performed, as well. Dunnett tests (significance at P < 0.05) were used to perform pairwise differences between medians. All the analyses were made with the R software version 3.4.0 (R Development Core Team, 2019).
Results
The tropical rainforest soil was more acidic and had higher concentrations of soil organic matter, organic carbon, available phosphorous, inorganic nitrogen, electric conductivity, and cation exchange capacity, than the savanna soil (Table 1). No significant differences were detected in magnesium and calcium concentrations and clay, sand, and silt levels between both soil types.
Table 1 Soil properties (median ± standard deviation) of tropical rainforest and savanna soils from La Trinidad Yaveo (Gulf of Mexico), Oaxaca, Mexico
| Soil properties | Tropical rainforest | Savanna | x 2 | P-value |
| pH (water) | 4.95±0.19 | 5.68±0.62 | 10.28 | < 0.01 |
| Soil Organic Matter (%) | 9.61±34.54 | 3.76±30.23 | 11.29 | < 0.001 |
| Soil Organic Carbon (%) | 5.57±1.96 | 2.18±1.67 | 11.29 | < 0.001 |
| Available Phosphorus (mg Kg-1) | 20.0±4.99 | 2.81±4.95 | 4.01 | < 0.05 |
| Inorganic Nitrogen (mg Kg-1) | 14.49±3.51 | 7.11±3.62 | 11.28 | < 0.001 |
| Electric Conductivity (dS m-1) | 152.0±5.36 | 63.5±7.86 | 10.59 | < 0.01 |
| Cation Exchange Capacity (Cmol(+) Kg-1) | 12.1±8.34 | 9.2±8.01 | 7.20 | < 0.01 |
| Magnesium (Cmol(+) Kg-1) | 1.67±0.69 | 0.93±2.42 | 0.34 | > 0.5 |
| Calcium (Cmol(+) Kg-1) | 3.53±1.14 | 2.97±0.97 | 2.61 | > 0.5 |
| Clay (%) | 9.46±0.44 | 6.96±0.86 | 0.91 | > 0.5 |
| Silt (%) | 17.76±0.77 | 9.84±0.70 | 4.42 | > 0.5 |
| Sand (%) | 75.48±0.96 | 84.19±1.78 | 2.35 | > 0.5 |
P-values < 0.05 indicate significant differences between the two types of soil according to the Kruskal-Wallis test.
The soil type (rainforest and savanna soils) had significant effects in the height (Scheirer-Ray-Hare test H = 101.69, p < 0.001) and the stem diameter rates (Scheirer-Ray-Hare test H = 65.08, p < 0.001) on the four pioneer species; the highest growth rate (height and stem diameter) was detected in the tropical rainforest soil for the four species. The AMF inoculation showed significant differences in height (Scheirer-Ray-Hare test H = 22.08, p < 0.001) and stem diameter rates (Scheirer-Ray-Hare test H = 11.57, p < 0.001) of the studied species; the highest growth rate in the four pioneer species (height and stem diameter) was observed in the treatment that included the inoculation with AMF. However, the interaction soil type and inoculation was not significant in height (Scheirer-Ray-Hare test H = 0.52, p = 0.47) nor stem diameter rates (Scheirer-Ray-Hare test H = 0.25, p = 0.87).
The greatest growth in height in the four studied species was detected in the treatment that included the tropical rainforest soil and AMF inoculation (Figure 1). Heliocarpus appendiculatus did not show significant differences in height between treatments with and without AMF inoculation in tropical rainforest soil; the other three plant species displayed their highest growth in height in the treatment that included the tropical rainforest soil and AMF inoculation. The plants cultivated in the tropical rainforest soils without AMF inoculation exhibited a higher growth in height than those cultivated in savanna soil with or without AMF inoculation. The only exception to this trend was C. grandis plants, which showed similar growth rates in height in the tropical rainforest soil without AMF inoculum than the savanna soil inoculated with AMF.
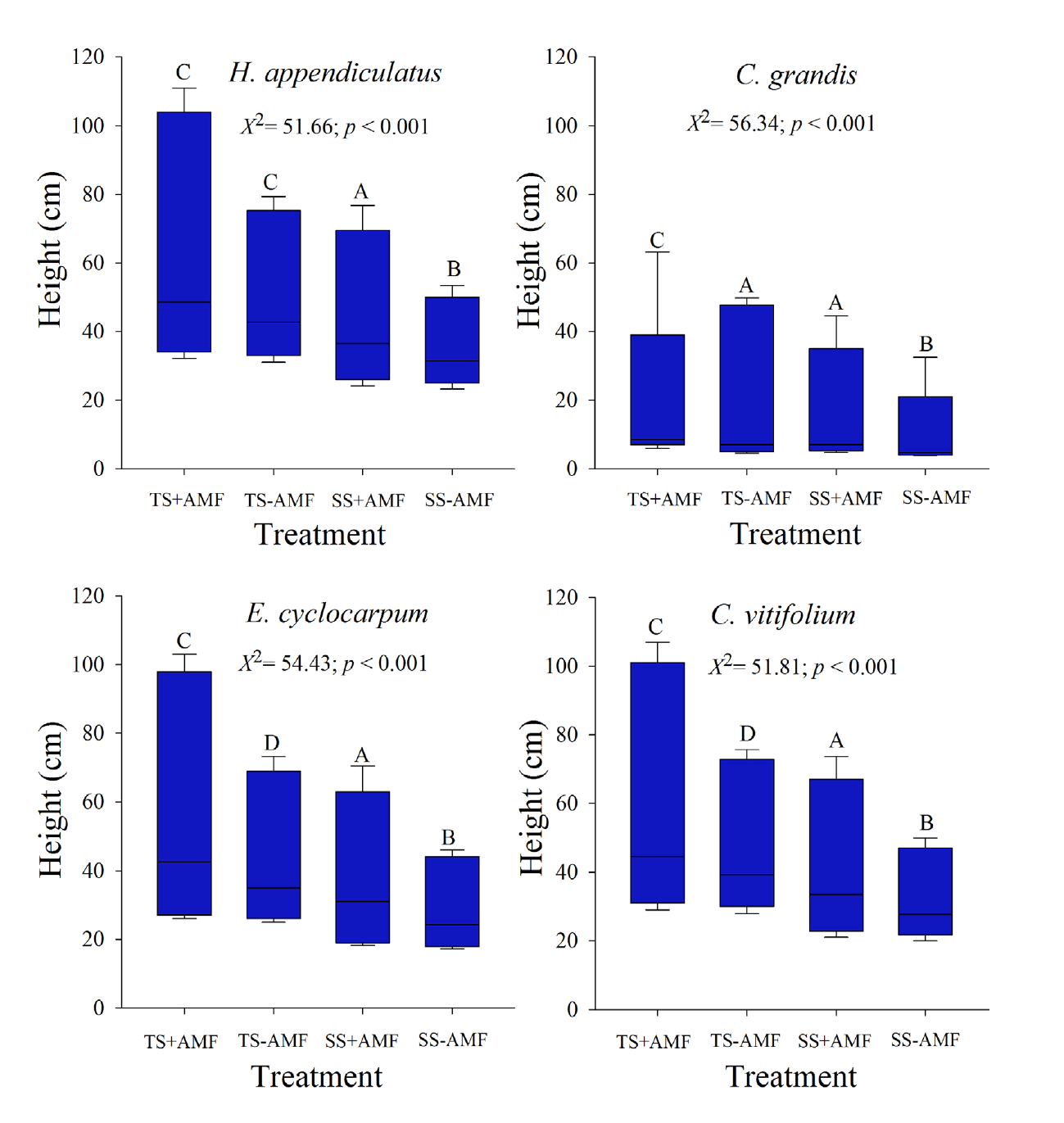
Figure 1 Changes in height (cm) of the four pioneer tree species evaluated associated with soil type and arbuscular mycorrhizal fungi (AMF) growing under nursery conditions. TS+AMF: tropical rainforest soils inoculated with AMF. TS-AMF: tropical rainforest soils without AMF inoculum. SS+AMF: savanna soils inoculated with AMF. SS-AMF: savanna soils without AMF inoculum. Median values followed by the same letter do not differ significantly between treatments according to the Dunnett test (P < 0.05).
The inoculation with AMF in tropical rainforest soils rendered stems with a significantly higher diameter in H. appendiculatus, E. cyclocarpum, and C. vitifolium than in tropical rainforest soils not inoculated with AMF (Figure 2). In none of the studied species, the growth in stem diameter was higher in the savanna soil without AMF inoculation than in the other treatments. No significant differences in stem diameter were detected between the two savanna soil treatments (with and without AMF inoculation); except for C. grandis, which showed higher stem diameter growth in the savanna soil inoculated with AMF.

Figure 2 Changes in stem diameter (mm) of the four pioneer tree species evaluated associated with soil type and arbuscular mycorrhizal fungi (AMF) growing under nursery conditions. TS+AMF: tropical rainforest soils inoculated with AMF. TS-AMF: tropical rainforest soils without AMF inoculum. SS+AMF: savanna soils inoculated with AMF. SS-AMF: savanna soils without AMF inoculum. Median values followed by the same letter do not differ significantly between treatments according to the Dunnett test (P < 0.05).
The soil type (Scheirer-Ray-Hare test H = 0.29, p = 0.58), the AMF inoculation (Scheirer-Ray-Hare test H=1.91, p = 0.16), and the interaction soil type and inoculation (Scheirer-Ray-Hare test H = 0.15, p = 0.69) did not show a significant effect in AMF colonization on the four studied species. AMF colonization in the four species showed consistent patterns among the different treatments (Table 2). No significant differences were observed in AMF colonization in the plant species cultivated in the tropical rainforest and savanna soils inoculated with AMF; in contrast to treatments without AMF inoculum which showed the lowest colonization rates and they were not statistically different from each other.
Table 2 Root colonization (%) (median ± standard deviation) of arbuscular mycorrhizal fungi (AMF) in tropical rainforest soils inoculated with different AMF treatments in the four pioneer tree seedling species evaluated
| Species | Treatment | χ2 | P-value | |||
| TS+AMF | TS-AMF | SS+AMF | SS-AMF | |||
| Heliocarpus appendiculatus | 51±5.07a | 4±2.01b | 50±5.13a | 5±2.30b | 44.83 | < 0.001 |
| Cassia grandis | 50±5.62a | 3±1.83b | 47±6.79a | 5±1.67b | 45.14 | < 0.001 |
| Enterolobium cyclocarpum | 52±4.45a | 5±1.84b | 50±6.35a | 4±1.88b | 44.75 | < 0.001 |
| Cochlospermum vitifolium | 50±6.06a | 3±1.88b | 42±7.80a | 2±2.23b | 45.10 | < 0.001 |
(TS+AMF): tropical rainforest soils inoculated with AMF. (TS-AMF): tropical rainforest soils without AMF inoculum. (SS+AMF): savanna soils inoculated with AMF. (SS-AMF): savanna soils without AMF inoculum. Values followed by the same letter do not differ significantly between treatments according to the Dunnett test (P < 0.05).
Discussion
We found evidence that the soil type plays a significant role in the studied plant growth rates. Overall, rainforest soil appears to improve tree growth more than that of savanna soil. This result may explain the observed selectivity of tropical rainforest species for tropical rain-forest ecosystems over savannas, even if both ecosystems are distributed at short distances (Hoffmann et al., 2012).
The detected effect of AMF inoculation in the studied tropical seedlings species could be reflected not only in the evaluated growth (height and stem diameter), but also in leaf area, and aboveground and root biomass (Janos, 1980a; Guadarrama et al., 2004a; Vandresen et al., 2007; Zangaro et al., 2012, 2013). Numerous studies show positive AMF-inoculated tropical tree plant responses on the growth and survival, but the magnitude of the effects greatly differ among tree species (Janos, 1980a; Guadarrama et al., 2004a; Vandresen et al., 2007; Zangaro et al., 2012, 2013). Therefore, the effect of AMF on tropical tree species is strongly influenced by plant identity, and also by the mycorrhizal fungi species, as well as many other environmental factors such as soil properties and management practices (Tran et al., 2019). Indeed, in soils with medium to low phosphorus availability, mycorrhizal colonization is generally associated with plant growth highest response (Janos, 1980a; Guadarrama et al., 2004a; Vandresen et al., 2007). However, there is also a scarcity of information on the relative importance and magnitude of AMF symbiotic features such as the type of AMF species, plant species, and soil properties on the growth of plants.
Phosphorus is one of the primary limiting nutrients in tropical ecosystems, and together with nitrogen, they are probably the most limiting nutrients of growth of different plant species (Hodge and Fitter, 2010; Smith and Smith, 2011). The highest growth in the treatment with tropical rainforest soil may be explained by the higher availability of soil nutrients observed in this ecosystem than those of savannas. In tropical ecosystems, it has been found that soil fertility (nitrogen and phosphorous) can have significant effects on the yield (growth and survival) of plants (Hall et al., 2004; Ayala-Orozco et al., 2017; Turner et al., 2018; Werden et al., 2018). The availability of phosphorus and nitrogen affects the leaves photosynthetic rates (Reich et al., 2009) and the growth of fine roots (Powers and Peréz-Aviles, 2013). The tropical rainforest and savanna are edaphically different ecosystems. Tropical rainforest ecosystems appear to have better soil conditions for plant growth than savanna ecosystems: soil organic matter (very high vs. high levels), inorganic nitrogen (medium vs. very low levels), and available phosphorus (medium vs. low concentrations) (Table 1). Despite being higher levels of soil fertility in the tropical rain-forest, the soil fertility is among low to intermediate levels, except for soil organic matter with a very high concentration, according to SEMARNAT (2002). The results obtained in this work concur with those of Bond (2010), Furley (1999), and those of Reyes-Carmona and Zamora-Serrano (1974); the latter was conducted in the same region. Such differences in soil fertility can be explained in part by microclimatic conditions (temperature and moisture), and by fire (persistent in the savanna).
With higher soil fertility levels, the benefits of mycorrhizal associations with the plant appear to decrease as the plant has less need for mycorrhizal to obtain its nutrients from the soil (Hodge and Fitter, 2010; Smith and Smith, 2011). The affinity of pioneer tree seedling species for AMF can vary from one species to another and this depends on soil fertility levels (e.g., available phosphorus, inorganic nitrogen, and soil organic matter), and climatic variations (moisture and temperature). Enterolobium cyclocarpum with a high percentage of AMF colonization could be an excellent example, this tropical tree species have been registered as not mycorrhizal (Gavito et al., 2008), but also as highly mycorrhizal (Habte and Musoku, 1994). Early-successional plant species appear to have a higher affinity and dependence on AMF than latesuccessional species, mainly due to their high growth rates and root production (Álvarez-Sánchez et al., 2009; Zangaro et al., 2012, 2013), demanding high nutritional contributions which are supplied by the AMF in tropical rainforest soils with intermediate levels of fertility; however, in savanna soils with low levels of soil organic carbon, available phosphorus, and inorganic nitrogen (Table 1), mycorrhizal symbiosis is not sufficient to supply the high demand of nutrients of earlysuccession tropical plant species.
AMF alter root architecture along with the formation of elaborate hyphal extensions permitting enhanced root conductance (Basyal and Emery, 2020) eventually leading to improved nutrients acquisition when compared to plants without symbiotic mycorrhizal fungi (Berruti et al., 2016), which also facilitates plant growth and photosynthesis (Barros et al., 2018). The extensive hyphal networks of mycorrhiza allow higher nutrients absorption such as phosphorus and nitrogen in soils with low to medium concentrations. However, it is important to note that a highly colonized plant does not necessarily translate to proportionally greater benefits or a higher response in the plant to AMF, or vice versa. A small amount of root colonization by AMF can still allow for the transport of phosphorus and other nutrients via the mycorrhizal pathway (Tran et al., 2019). Tree species with longer and finer roots are likely benefit less from forming AMF than those with shorter and wider diameter roots (Zangaro et al., 2007). Although we do not evaluate the length and biomass of the roots, some studies have found that root growth in mycorrhized plants tends to decrease with high levels of mycorrhizal colonization; that is, plants with a higher affinity for AMF invest less energy in the production of fine roots and, therefore, in soil exploration (Guadarrama et al., 2004a; Leuschner et al., 2009). Thus, we might expect that in conditions of higher soil fertility (contrary to our findings, see results section and Table 1), the studied species would display lower increases in root length with AMF inoculation. The benefits of symbiosis could be due to the extensive mycelial networks modulating nutrient absorption, water absorption and retention, and soil properties (Leifheit et al., 2014; van der Heijden et al., 2015).
An additional factor that can improve plant growth is the endophytic bacteria (added in the filtrates; see the section on materials and methods). Endophytic bacteria can promote growth, stress tolerance, nutrient up-take, and protection from soil-pathogens (Malfanova et al., 2011; Sessitsch et al., 2012). Bacteria might synthe-size biologically active compounds and provide nutrients for the host plant survival (Marschner et al., 2001). These positive interactions among plant-bacteria and AMF are known to have beneficial effects on plants by improving the availability of nutrients and, therefore, plant survival (Hashem et al., 2016).
Among the four evaluated species, no significant differences were detected in AMF colonization regardless of soil type, probably because (1) all the studied species are characteristic of early-successional stages, and (2) the soil fertility is medium to low, driving them to be more dependent on AMF, due to very fast growth and the need for a lot of nutrients. In species such as H. appendiculatus the inoculation with AMF improved survival and alleviated competition effects with other pioneer plant species in the nursery (Guadarrama et al., 2004b).
Two AMF non-inoculated treatments from all the tree species in this study presented a low AMF colonization rate (less than 6 %). We do not know to what extent the low rates of mycorrhizal colonization are by AMF contamination of sterile material, since to our knowledge, this problem has not been previously documented. However, the contamination of these treatments could be due to the proximity to probable sources of AMF (forests near the tree nursery) due to its natural dispersal by wind (Egan et al., 2014). However, the low colonization percentages of these treatments, 10 to 25 times lower than those inoculated, make those values non-significant.
Despite the vital role of AMF on plants, few studies have used AMF as a biotechnological tool in ecological restoration projects (Janos, 1980a, b; Álvarez-Sánchez et al., 2009; Zangaro et al., 2012, 2013; Bender et al., 2014). Although with few plant species, our study suggests the importance of AMF for tropical rainforest restoration as a key factor favoring the selectivity of tropical trees for tropical ecosystems instead of savanna areas.











 nueva página del texto (beta)
nueva página del texto (beta)



