Introduction
Anemia is associated with increased incidence of mortality and morbidity especially in elderly patients undergoing orthopedic surgery.1 In the majority of the cases, etiology for these anemic patients is iron deficiency or chronic inflammation.2 The effective treatment in these cases would be administration of iron and erythropoiesis stimulating agents (ESA).3,4
There is consensus regarding the use of oral or intravenous therapy for iron deficiency anemia as the first line of treatment but the use of ESA is limited to refractory cases.5
The published literature on the efficacy of iron therapy alone had focused on increased hemoglobin levels in the postoperative period; however, they are silent on avoidance of blood transfusion.6,7 Prior meta analyses and systematic reviews published on effectiveness of ESA in avoiding postoperative blood transfusion8,9 had limitations like 1) studies with preoperative blood transfusion were included which could had confounding effect on use of ESA 2) adverse events associated with use of ESA were not included. 3) Randomized controlled trials involving the effect of ESA on avoidance of red blood transfusion were not included.
Presently, role of ESA is limited in the management of preoperative anemia due to unanimity that iron monotherapy is adequate for management.10 There is also risk of possible side effects like stroke, thrombosis and mortality with use of ESA in high dose or for long term especially in patients with medical comorbidities.11,12
These potential concerns may be unfound because a) the dose of ESA administered in cases of preoperative anaemia is low and given for a shorter duration b) hemodilution and other hemodynamic responses subsequent to blood loss would reduce the risk of thrombosis.
We conducted this systematic review and meta-analysis to assess the safety and effectiveness of iron vis-a-vis combination therapy of iron and ESA in the management of anaemia prior to orthopedic surgery. Our hypothesis was that combined administration of iron and ESA would be more beneficial than iron therapy alone in avoiding post-surgical red blood cell transfusions. Secondary objectives were to assess change in hemoglobin level after treatment, postoperative change in level of hemoglobin, mortality, stroke, myocardial infarction, deep vein thrombosis, pulmonary embolism and renal dysfunction.
Material and methods
The current meta-analysis was performed according to the recommendations of the PRISMA statement.13 It was registered in PROSPERO under the number CRD42020216341.
Search strategy: we had searched for four databases for studies i.e. PubMed, Embase, Google Scholar and Cochrane database since inception to April 2021 to collect information on previous trials. In addition, we had also looked for any undergoing trials on clinicaltrials.gov. The keywords searched were «iron», «erythropoietin», «anemia» and «orthopedic». There was no restriction of our search for by language, date and publication status. The bibliographic details of all the included studies were searched manually for any additional citations. In case of duplication of publication, the study with the entire set of data was included. The complete search strategy has been listed in the electronic supplementary material (supplementary material [sm] 1).
Eligibility criteria. For inclusion in the meta-analysis the following criteria were met:
Study design: randomized controlled trial.
Subjects: adults undergoing orthopedic operations.
Intervention: administration of iron vis-a-vis combination of ESA and iron.
Outcome: reporting of either minimum of one primary or secondary outcome.
Studies were excluded in case of
Numerous confounding factors which were unable to assess the effectiveness of iron or ESA.
Inability to differentiate between population of interest from a larger population under consideration.
Patient administered ESA or iron only during operative procedure or in the postoperative period.
Patients received autologous blood transfusion in the perioperative period.
Primary and secondary outcome measures. The primary outcome of the study was to assess the proportion of patients required perioperative red blood cell transfusion. Secondary objectives were to assess change in hemoglobin level from baseline to time of surgery, postoperative hemoglobin levels and complications like mortality, stroke, myocardial infarction, deep vein thrombosis, pulmonary embolism and renal dysfunction.
Selection of study and data extraction. All the studies were independently screened for meeting the requirements of the study criteria using Rayyan web application.14 In case of any disagreement regarding the inclusion of study, the matter was resolved by the third author. The reviewers independently extracted the characteristics of the studies and outcome measures. The extraction form was developed as per Cochrane recommendations.15 The discrepancy between the data extracted twice was solved by analysis of full text by all the reviewers. The patient characteristics included were age, sex, site and indication for surgery. Other parameters collected were type of ESA and iron, route of administration, dosage, interval and timing of doses, type of comparator and use of any other interventions.
Assessment of bias risk. Cochrane collaboration’s tool was used to check for the quality of studies included in the meta-analysis.16 The areas checked in each study were: random generation of sequence; concealment of allocation; reporting of selective outcome; blinding of participants; incomplete outcome data; selective outcome reporting; potential sources of bias like conflict of interest. The performance of each study was checked for risk of bias and tabulated. Categorization of the risk of bias was carried out as low, medium and high risk. A study was labelled with low risk of bias if six out of seven domains were low risk.
Data synthesis and statistical analysis. The studies were combined to obtain data for assessment of relative risk and 95% confidence intervals for categorical data like mortality, myocardial infarction, renal dysfunction, stroke, deep vein thrombosis and pulmonary embolism. The outcome of red blood cell transfusion and subsequent improvement in hemoglobin levels were estimated using pooled weighted mean difference with 95% confidence interval (CI). Random effect model of DerSimonian and Laird approach17 was used to estimate pooled RRs and mean differences with inverse variance approach. In case of no event in a group, the RR was estimated by adding 0.5 to each cell. The testing of heterogeneity was carried out using χ2 test and quantified using I2. In case of I2 > 50%, substantial heterogeneity was considered. P value of < 0.10 was considered statistically significant to indicate heterogeneity using χ2 test. The statistical analyses were performed using Review Manager Software (Cochrane Collaboration, UK).
The criteria mentioned in Cochrane Handbook were used to convert medians, standard errors and 95% CI to means and standard deviations. Similar guidelines were followed in case a combination of multiple doses of ESA were required to assess its effect in comparison to iron therapy.
Subgroup and sensitivity analysis. A priori subgroup analysis was performed for two interventions i.e. 1) ESA and oral therapy of iron vis-a-vis oral iron only 2) combination of Intravenous iron and ESA vis-a-vis intravenous iron only. A significant degree of heterogeneity was anticipated hence sensitivity analyses were performed in studies with low vs high dose of ESA. The studies with high risk of bias were also excluded. The subgroup analysis was carried out as per the guidelines published by Richardson et al.18
Results
Literature search. In total of 1,273 articles were obtained in the initial search for studies involving iron and erythropoietin therapy. 49 duplicate articles were removed, leaving 1,224 for screening. After screening titles and abstracts, 36 full text articles remained for review. Following review of full text articles, 25 studies were excluded as ten among those did not had intervention of interest, eight studies involved administration of perioperative autologous blood transfusion and in seven studies, iron or ESA was administered either intraoperatively or postoperatively. A total of eleven studies19,20,21,22,23,24,25,26,27,28,29 (2,816 patients) were included in the meta-analysis (Figure 1).
Study characteristics. The studies included in the metanalysis were divided in two sub groups: a) ESA with oral iron vis-a-vis oral iron alone and b) ESA with intravenous iron vis-a-vis intravenous iron alone. Eight RCTs assessed oral iron therapy with combination of ESA and oral iron (Table 1). Three RCTs assessed ESA with intravenous iron or administration of intravenous iron alone (Table 2). The subgroup analysis was performed to assess number of RBC units transfused, hemoglobin after treatment, postoperative hemoglobin concentration, mortality, stroke, pulmonary embolism, deep vein thrombosis and other parameters.
Table 1: Characteristics of included randomized controlled trials involving ESA with iron vis-a-vis iron.
| Study | Total number of participants | Preoperative hemoglobin | Surgery | Intervention | Comparator | Pre or postoperative administration | Measured outcomes |
|---|---|---|---|---|---|---|---|
| Wu, 2016 | 62 | Male: 13-15 | Primary total bilateral hip replacement | EPO (70,000 IU) (n = 30) | Oral iron (n = 32) | Both pre and postoperative | 1. Hemoglobin concentration |
| Female: 12-15 | 2. Reticulocyte count | ||||||
| 3. Hematocrit | |||||||
| 4. RBC transfusion rate | |||||||
| 5. Deep vein thrombosis | |||||||
| 6. Pulmonary embolism | |||||||
| 7. Blood loss | |||||||
| Stowell, 2009 | 680 | 10 to 13 | Spine surgery | EPO (168,000 IU) (n = 340) | Oral iron (n = 340) | Preoperative | 1. Deep vein thrombosis and other thrombovascular events |
| Weber, 2005 | 695 | 10-13 | Elective orthopedic procedure (spine, hip and knee) | EPO (168,000 IU) (n = 460) | Oral iron (n = 235) | 1. Number of red blood cell transfusions | |
| 2. Hemoglobin | |||||||
| 3. Thrombotic events | |||||||
| Wurnig, 2001 | 1. Number of red blood cell transfusions | ||||||
| 2. Mortality | |||||||
| 3. Thrombovascular events | |||||||
| 4. Hemoglobin | |||||||
| 5. Iron studies (serum ferritin, serum iron) | |||||||
| Feagan, 2000 | 201 | 9.8 to 13.7 | Total hip joint replacement | EPO (80,000 IU) with oral iron (n = 44) | Placebo with oral iron (n = 78) | Preoperative | 1. Number of red blood cell transfusions |
| 2. Reticulocyte count | |||||||
| 3. Thrombovascular events | |||||||
| De Andrade, 1996 | 290 | < 15 | Major orthopedic surgery | EPO (105,000 IU) with oral iron (n = 101) | Placebo with oral iron (n = 103) | Pre and postoperative | 1. Blood loss |
| 2. Transfusions | |||||||
| EPO (315,000 IU) with oral iron (n = 112) | 3. Thrombovascular events | ||||||
| 4. Adverse events (nausea, vomiting, pyrexia) | |||||||
| Faris, 1996 | 185 | No haemoglobin criteria | Major orthopedic surgery | EPO (105,000 IU) with oral iron (n = 71) | Placebo with oral iron (n = 10) | Pre- and postoperative | 1. Transfusion |
| 2. Reticulocyte count | |||||||
| 3. Hemoglobin | |||||||
| EPO (315,000 IU) with oral iron (n = 69) | 4. Chest pain | ||||||
| 5. Depression | |||||||
| COPES, 1993 | 208 | 11-16 | Total hip replacement | EPO (189,000 IU) with oral iron (n = 53) | Oral iron (n = 78) | Pre and postoperative | 1. Transfusion intraoperatively and postoperatively |
| 2. Reticulocyte count | |||||||
| 3. Hemoglobin | |||||||
| EPO (294,000 IU) with oral iron (n = 77) | 4. Length of hospital stay | ||||||
| 5. Deep vein thrombosis |
Table 2: Characteristics of included randomized controlled trials involving ESA with intravenous iron vis-a-vis iron therapies.
| Study | Total number of participants | Preoperative haemoglobin (g/l) | Surgery | Intervention | Comparator | Pre or postoperative administration | Measured outcomes |
|---|---|---|---|---|---|---|---|
| Bernabeu-Wittel, 2016 | 306 | 9 to 12 | Osteoporotic hip fracture | EPO (40,000 IU) with IV iron (n = 100) | Placebo with IV iron (n = 103) | Pre and postoperative administration | 1. Number of red blood cell transfusions |
| Placebo with EPO (n = 100) | 2. Survival | ||||||
| EPO (40,000 IU) with intravenous iron (n = 100) | 3. Hemoglobin | ||||||
| 4. Health quality related measures | |||||||
| Kateros, 2010 | 79 | < 13 | Intertrochanteric fracture | EPO (20,000 IU) with IV iron (n = 38) | Placebo with IV iron (n = 41) | Pre and postoperative administration | 1. Blood loss |
| 2. Transfusions | |||||||
| 3. Hemoglobin | |||||||
| 4. Platelets | |||||||
| Olijhoek, 2001 | 110 | 10 to 13 | Elective orthopedic surgery | EPO (84,000 IU) with IV iron (n = 29) | EPO (20,000 IU) with oral iron (n = 29) | Preoperative only | 1. Hemoglobin |
| Placebo with IV iron (n = 25) | 2. Iron studies | ||||||
| Placebo with oral iron (n = 27) | 3. Thromboembolic events |
Risk of bias assessment. The risk of bias of the studies included was assessed and tabulated in supplementary material (sm 2, 3 and 4). The parameters to assess the risk of bias like allocation of participants and blinding was explicitly described in almost all the studies.
Publication bias. The funnel plot was studied for each comparison. Funnel asymmetry was noted for publication bias (sm 5). Egger’s linear regression analysis was used for evaluation of publication bias for three factors:
Proportion of patients who had received blood transfusion in the postoperative period (coefficient: -1.487; 95% CI, -4.587 to 1.612; p = 0.306).
The total number of blood transfusion units administered (coefficient: 0.70; 95% CI, -0.95 to 2.67; p = 0.345).
Length of stay in hospital (coefficient: -1.62; 95% CI, -3.45 to -0.34; p = 0.236).
Based on the analysis, it was noted that there was no publication bias for all the factors i.e. proportion of patients who had received blood transfusion in the postoperative period, units of blood transfused and length of stay.
Red blood transfusion rate with combination therapy of ESA and iron. Combination of ESA and iron decreased number of patients who required blood transfusion after surgical intervention in comparison to patients treated with iron therapy alone (RR, 0.73; 95% CI, 0.59 to 0.91, I2 = 65%; p = 0.005) (Figure 2). In subgroup analysis with oral and intravenous iron, the difference was not statistically significant (p = 0.24). With administration of erythropoietin either in high (≥ 80,000 IU) or low dose (≤ 80,000 IU) resulted in lower blood transfusion rates (p = 0.0007). But subgroup analysis, revealed there was no significant difference in transfusion rates with either high or low dose of erythropoietin (p = 0.08) (Figure 3) on blood transfusion rates.
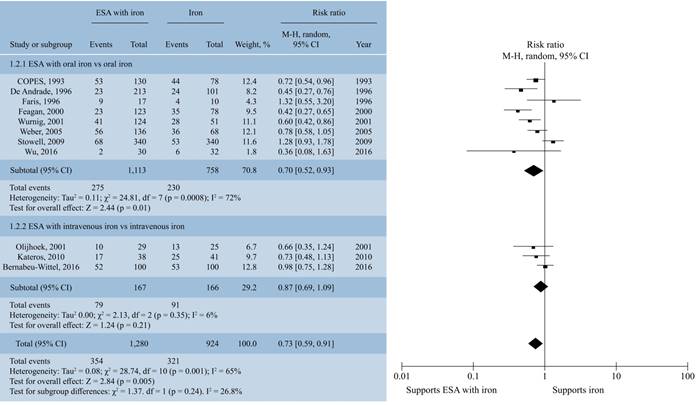
Figure 2: Forrest plot showing the effect of combination therapy with ESA and iron vis-a-vis iron on the proportion of patients requiring red blood cell transfusion. Subgroup analysis according to a) ESA with oral iron vs oral iron b) ESA with intravenous iron vs intravenous iron.
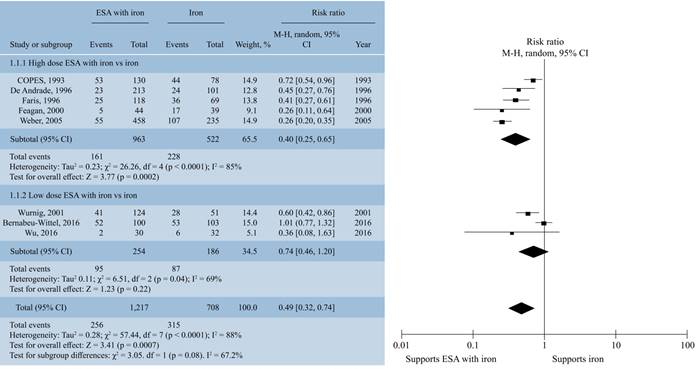
Figure 3: Forrest plot showing effect of high (≥ 80,000 IU) and low (≤ 80,000 IU) dose of erythropoietin stimulating agents in combination with iron on proportion of patients transfused with red blood cells.
The secondary parameters also supported the use of erythropoietin with iron vis-a-vis iron alone for decreasing the requirement of blood transfusion after operative procedure:
1. After treatment hemoglobin concentration (mean difference in hemoglobin from base line to time of surgery), 10.44 g/dl; 95% CI 4.35 to 16.52, I2 = 97%; p = 0.0008) (sm 6).
2. Postoperative hemoglobin concentration (mean difference in hemoglobin, 8.73 g/dl; 95% CI, 5.20 to 12.25, I2 = 99%; p < 0.00001) (sm 7).
There was no statistically significant difference in the subgroup analysis between oral or intravenous administration of iron on these above two parameters (after treatment, p = 0.51; postoperative hemoglobin, p = 0.81).
Side effects of combination of ESA and iron therapy. The combination of ESA and iron therapy in comparison to iron therapy did not significantly increase the risk of
Mortality (RR, 0.84; 95% CI, 0.32 to 2.18, I2 =0%; Z = 0.36; p = 0.72) (sm 8).
Myocardial infarction (RR, 1.45; 95% CI, 0.54 to 2.34, I2 = 0%; p = 0.68) (sm 9).
Stroke (RR, 1.42; 95% CI, 0.35 to 5.71, I2 = 0%; Z = 0.49; p = 0.62) (sm 10).
Deep vein thrombosis (RR, 1.56; 95% CI, 0.95 to 2.54, I2 = 0%; Z = 1.76; p = 0.08) (sm 11).
Pulmonary embolism (RR, 0.92; 95% CI, 0.15 to 5.83, I2 = 0%; Z = 0.08; p = 0.93) (sm 12).
Further subgroup analysis to study the effect of ESA dosage (high versus low) on deep vein thrombosis did not reveal any statistically significant difference (RR, 1.59; 95% CI, 0.98 to 2.59, I2 = 0%; Z = 1.89; p = 0.06).
Discussion
The meta-analysis supports our hypothesis that combined administration of ESA and iron vis-a-vis iron alone results in a lesser number of red blood cell transfusions in the postoperative period in orthopedic procedures. It was further supported by outcomes measures like 1) increase in hemoglobin levels after operation, 2) decrease in number of red blood cell units transfused, 3) the proportion of the patients who had received blood transfusion following surgery.
The current meta-analysis adds to the literature as previous systematic reviews were limited in the way that a) they did not include all the RCTs to assess decrease in rate of blood transfusion, b) a large number of studies included preoperative autologous transfusion, c) in addition, they were silent on adverse events associated with ESA therapy.
ESA is extensively used for the treatment of anaemia in cases of renal failure, malignancy and surgical patients. It is also used in patients to increase the hematocrit levels prior to surgery and reduce the possibility of allogeneic blood transfusion. In a review by Cherian et al30 on blood conservation methods for patients undergoing total hip replacement had affirmed that no single methodology is superior to other in preventing allogenic blood transfusion. Deutsch et al31 in their randomized control trail on knee arthroplasty patients had administered erythropoietin in one group and followed preoperative blood donation protocol in another group. They concluded that the blood parameters like hemoglobin or reticulocyte counts were higher on the day of surgery in the erythropoietin group in comparison to those with preoperative blood donation. However, there was no major difference in the rate of blood transfusion in the postoperative period as observed in the present study.
In contrast, a systematic review conducted by Alsaleh et al32 on arthroplasty patients had found a significant decrease in the requirement of postoperative blood transfusion after administration of erythropoietin in the preoperative period. The blood parameters including hemoglobin levels were maintained in the immediate postoperative period. Zhao et al33 in their meta-analysis on arthroplasty patients had also concluded that preoperative erythropoietin administration is associated with higher postoperative hemoglobin and lower requirement of autologous blood transfusion.
The studies had shown marked decrease in mortality in ICU patients.34,35,36 Some RCTs had shown that when the target Hb to be achieved was 13 g/dl there are chance of marked increase in stroke, thrombosis, mortality in patients suffering from comorbidities.11,28,29
There are a lot of concerns regarding the safety of ESA like stroke, myocardial infarction, mortality, stroke, renal failure, deep vein thrombosis and pulmonary embolism. This had restricted its use in patients undergoing operative procedure. Our analysis had shown that there is no evidence to support the fear prohibiting its use in orthopedic patients.
The studies were stratified in two groups, one in which a high dose of ESA (> 80,000 IU) was administered and another one in which a low dose of ESA (> 80,000 IU) was given. The division in low and high dose of erythropoietin was carried out as described by Kei T et al.37 There was a trend towards increased risk of deep vein thrombosis in cases where a high dose of erythropoietin was given. Stowell et al38 in their RCT on spine cases had reported an DVT in 4.7% cases in which a high dose of ESA (600 IU/kg/day) was administered for 12 days. Furthermore, it would be pertinent to mention here that they had not used DVT prophylaxis measures, so the results had to be interpreted with caution. De Andrade et al39 had reported increased risk of DVT in the group receiving high doses of ESA while no significant increase in risk of DVT was noted in patients receiving low doses of ESA in patients undergoing orthopedic surgeries.
The strength of the current systematic review was that an extensive literature search was carried out for a large population undergoing orthopedic surgery and important clinical outcomes. Hence, we were able to identify a large number of randomized controlled trials that involve comparison of ESA and iron with iron therapy alone. The analysis of these studies has revealed that addition of ESA to iron therapy can lead to reduction in the requirement of blood transfusion after surgery.
There were limitations to our review. The broad inclusion criteria of all orthopedic surgeries lead to a high degree of heterogeneity among studies with use of blood transfusion in the perioperative period. Factors like inclusion of both anemic and non-anemic patients at the baseline, different types of surgeries and standard of care, duration of treatment, use of different doses of erythropoietin further added to this heterogeneity. Moreover, the earlier studies mainly used oral iron whereas the newer ones had mainly used intravenous iron which might have affected the outcomes. The sensitivity analysis was performed to address the issue of heterogeneity and studies with low risk of bias were included.
Low heterogeneity was observed in case of assessment of adverse effects like mortality, deep vein thrombosis, etc. with ESA therapy.
The requirement of blood transfusion after orthopedic surgery was affected by the combined administration of iron and ESA vis-a-vis iron alone, however the finding is weakened by the heterogeneity between the studies and possibility of publication bias. The study is not adequately powered to address the question of adverse events with the combination of ESA and iron. Large multicentric trials are needed to assess the adverse events with combination therapy of ESA and iron.
Conclusion
The combination of ESA and iron is more effective than iron therapy alone in decreasing the requirement of blood transfusion and increasing the hemoglobin in the postoperative period. The incidence of adverse events like stroke, deep vein thrombosis, mortality, etc. was not increased with the combined therapy. However, larger trials are needed to support or refute the same.











 nueva página del texto (beta)
nueva página del texto (beta)



