Introduction
The ecological features of forest fringe communities (edge or saum communities) include a gradient from pre-forest or forest edges to non-forest biotopes (normally grasslands) in which hemicryptophytes and chamaephytes often dominate and form special communities (Capelo, 2007; Čarni, 2003; Dengler & Boch, 2008). This medium-sized, non-nitrophilous, dense perennial vegetation is found in contact with well-conserved forest or pre-forest environments that often show high floristic richness due to their growth in situations that are ecologically transitory (in terms of light conditions and nutrient status) (Loidi, Herrera, García-Mijangos, Biurrun, & Mucina, 2004; Taffetani, Rismondo, & Lancioni, 2011). The corresponding environment in these communities is of the transitional type between the access of light and, in addition, the shelter effect of the ecoclimate and pedosphere of the forest margin, which increases the plant biodiversity (Biondi, Čarni, Vagge, Taffetani, & Ballelli, 2001; Taffetani et al., 2011).
The forest fringe communities of the southwestern Iberian Peninsula are included in the Trifolio medii-Geranietea sanguinei class (Costa et al., 2012; Dengler & Boch, 2008; Loidi et al., 2004; Rivas-Martínez, 2011). The Origanetalia vulgaris is the order of the Trifolio medii-Geranietea sanguinei class found in the Iberian Peninsula and is further divided into six alliances. The vegetation of the forest fringe of the West Iberian Mediterranean is classified within the alliance Origanion virentis (Costa et al., 2012). In the southwestern Iberian Peninsula, a large set of endemic fringe species enable Capelo (1996) to recognize two suballiances within the Origanion virentis alliance: Origanenion virentis and Stachyo lusitanicae-Cheirolophenion sempervirentis.
The silicicolous, acidophilous to neutrophilous fringe communities of the mesomediterranean and supramediterranean belts of the West Iberian Mediterranean territories are classified within the Origanenion virentis suballiance, whereas the more thermophilous fringe communities of the southwestern Iberian Peninsula, which grow on calcicolous (calco-dolomitic) to acidophilous substrata, are classified within the Stachyo lusitanicae-Cheirolophenion sempervirentis suballiance. In southern Portugal, the Origanion virentis alliance encompasses the less thermophilous communities of the subcontinental areas in the northern and eastern Luso-Extremadurese territories. Furthermore, the thermophilous and oceanic territories of the West Iberian Mediterranean reflect the transition of the Origanenion virentis to Stachyo lusitanicae-Cheirolophenion sempervirentis communities. The latter comprises thermophilous forest-edge communities: thermo or mesomediterreanean, euoceanic to subhyperoceanic, dry to humid bioclimate, within well-defined biogeographical units - Coastal Lusitanian-Andalusian Province and western and southwestern margins of the Luso-Extremadurese Subprovince. Stachyo lusitanicae-Cheirolophenion sempervirentis suballiance are strongly influenced by the different thermophilous forests of Quercion broteroi alliance.
The present paper aims to provide a phytosociological and syntaxonomical analysis of the forest fringe communities included in the Stachyo lusitanicae-Cheirolophenion sempervirentis suballiance, from the southwestern Iberian Peninsula. Of particular interest is the forest fringe community dominated by Cynara algarbiensis Coss. ex Mariz, an acidophilic species, typically found on siliceous substrata (Franco, 1971). This hemicryptophyte is an artichoke exclusive to the southwestern Iberian Peninsula (Coutinho, 1939; Valdés, Talavera, & Fernández-Galiano, 1987), exclusively found in the oceanic areas, which are mainly situated on mountainous territories of the Monchique Sierran District and thermophilous areas of the lower Guadiana Valley in the Alentejan and Andevalese Districts.
Material and methods
Field research was carried out from 2009 to 2012. Phytosociological relevés have been collected according to the sigmatist school of the Zürich-Montpellier approach (Braun-Blanquet, 1965; Géhu & Rivas-Martínez, 1981; Biondi, 2011). Taxonomic nomenclature follows the work of Coutinho (1939), Franco (1971), Valdés et al. (1987), Franco and Rocha-Afonso (1994), and Castroviejo (1986-2010). This biogeographical and bioclimatological analysis was carried out according to Rivas-Martínez (2007), Monteiro-Henriques (2010) and Rivas-Martínez et al. (2014). Syntaxonomical typologies were checked according to Rivas-Martínez (2011) and Costa et al. (2012). The data set of the samples, which included 52 relevés and 195 species, was submitted to the Un-Weighted Pair-Group Method using Arithmetic Averages (UPGMA), with Bray-Curtis distance, to produce the dissimilarity measure, using PRIMER 6 software version 6.1.5 (Clarke, 1993; Clarke & Gorley, 2006; PRIMER-E Ltd., 2006). The Braun-Blanquet‘s abundance-dominance indexes were transformed according to van der Maabel (1979). In order to establish a solid synthetic table and submit the data matrix to multivariate analysis procedures, some relevés were taken from the literature (Capelo, 1996; Costa, Capelo, Santo, & Lousã, 2002; Costa et al., 1993; Costa et al., 2012; Pereira, 2009; Pinto-Gomes & Paiva-Ferreira, 2005).
The field research area covered the south and southwest mainland of Portugal, whose altitudes ranged from 90 to 395 m. The main feature is the domain of turbiditic material (especially schists, siltstones and greywackes) that form the Baixo Alentejo flysh group. These formations include two distinct geomorphological units: the rolling topography with low, gentle slopes of Alentejo in the north and east, and the tectonic sub-coastal reliefs of the Caldeirão, Monchique, Cercal and Grândola mountains in the south and west. According to biogeographic typology units of Quinto-Canas (2014) and Rivas-Martínez et al. (2017), the study area corresponds to the Monchique Sierran and Alentejan Districts. The bioclimatic analysis reveals that the researched area presents a mediterranean pluviseasonal oceanic, euoceanic to subhyperoceanic bioclimate, with thermo-mesomediterranean thermotype and dry to humid ombrotype (Monteiro-Henriques, 2010; Rivas-Martínez, 2007).
Results and discussion
The cluster analysis dendrogram (Figure 1) and synoptic table (Appendix 1) reveal a clear separation between all phytosociological associations of the Stachyo lusitanicae-Cheirolophenion sempervirentis woodland fringe communities, based on diagnostic species as a response to different ecological and biogeographical attributes. The dendrogram of relevés classification distinguished at the first level two main groups (group A and group B), which represent seven community types: (A) Picrido spiniferae-Cynarietum algarbiensis (clusters 1-7); (B1) Bartsio asperae-Origanetum virentis (clusters 30-36); Picrido algarbiensis-Cheirolophetum sempervientis (clusters 12-19); (B2) Stachyo lusitanicae-Origanetum virentis (clusters 21-29); Origano virentis-Leucanthemetum sylvatici (clusters 37-52); Senecio lopezii-Cheirolophetum sempervirentis (cluster 20) and Leucanthemo sylvatici-Cheirolophetum sempervirentis (clusters 8-11).
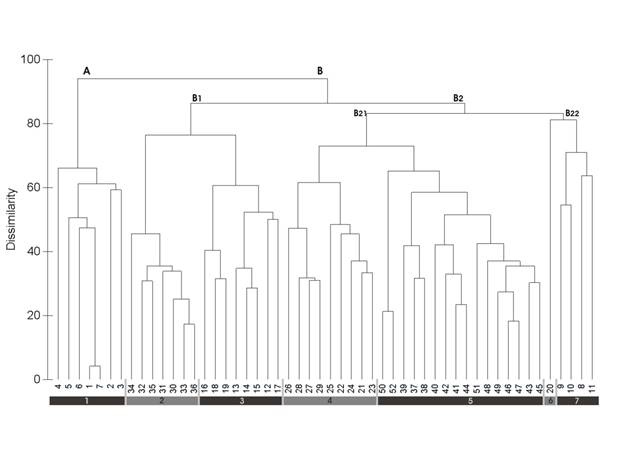
Figure 1 UPGMA hierarchical clustering, based on Bray-Curtis distance: 1) Picrido spiniferae-Cynarietum algarbiensis (1-7); 2) Bartsio asperae-Origanetum virentis(30-36) ; 3) Picrido algarbiensis-Cheirolophetum sempervientis (12-19); 4) Stachyo lusitanicae-Origanetum virentis (21-29); 5) Origano virentis-Leucanthemetum sylvatici (37-52); 6) Senecio lopezii-Cheirolophetum sempervirentis (20); 7) Leucanthemo sylvatici-Cheirolophetum sempervirentis (8-11).
Group A has a high dissimilarity in relation to the other associations, and including relevés dominated by C. algarbiensis, which did not have any phytosociological correspondence. According to the ecological and floristic profile, we propose the new Picrido spiniferae-Cynarietum algarbiensis association. Group B comprises two subgroups. The subgroup B1 encompasses samples ascribed to the calcicolous substrata associations: Bartsio asperae-Origanetum virentis, dominated by Bartsia aspera (Brot.) Lange, which spreads across the Portuguese Divisorian Sector and represents the fringe of Arisaro simorrhini-Quercetum broteroi or Lonicero implexae-Quercetum rotundifoliae; and, Picrido algarbiensis-Cheirolophetum sempervientis, described in the limestones of Algarvese District (Algarvese-Monchiquese Sector), which constitutes the fringe of gall-oak and portuguese-oak groves belonging to Quercetum alpestris-broteroi. The subgroup B2 comprises two block communities of the external forests fringe growing on calcicolous to silicicolous substrata. The first block (B21) belongs to silicicolous, acidophilous to neutrophilous syntaxa included in: Stachyo lusitanicae-Origanetum virentis, a fringe community of the cork oak of Asparago aphylly-Quercetum suberis, suggested by Costa et al. (2002) for the Portuguese Divisorian and Ribataganian territories, being scarce in the Sadese District and half-western Luso-Extremadurese areas; Origano virentis-Leucanthemetum sylvatici, described in the Monfurado mountain in the Alentejan District, which corresponds to the forest fringe of Sanguisorbo hybridae-Quercetum broteroi. Samples of the second block (B22) have been included in the associations dominated by Cheirolophus sempervirens (L.) Pomel: Senecio lopezii-Cheirolophetum sempervirentis for silicicolous substrate, linked to upper sub-humid to humid ombrotypes of the Monchique Sierran (Figure 2) and Vicentinan Coastal Districts (Capelo, 2007), which mainly represents the forest fringe of the Avenello strictae-Quercetum marianicae; and Leucanthemo sylvatici-Cheirolophetum sempervirentis, given by Costa et al. (1993) and Capelo (2007) for the Portuguese Divisorian and Arrabida Sierran territories, that constitutes the calcicolous fringe of Arisaro simorrhini-Quercetum broteroi and Viburno tini-Quercetum rivasmartinezii woodlands (Capelo, 2007). The consistency of the UPGMA classification dendrogram and detailed analysis of the floristic composition of each group defined in the dendrogram allowed us to propose a new association which is described below.

Figure 2 Simplified scheme of the external forest fringe associations present in the Monchique Sierran District: A) upper sub-humid to humid ombrotypes; B) lower sub-humid ombrotype. Associations: 1) Lavandulo viridis-Quercetum suberis, 2) Picrido spiniferae-Cynarietum algarbiensis, 3) Avenello strictae-Quercetum marianicae, 4) Senecio lopezii-Cheirolophetum sempervirentis, 5) Ulici argentei-Quercetum rotundifoliae, 6) Trifolio subterranei-Poetum bulbosae, 7) Salicetum atrocinereo-australis.
The relevés ascribed to Picrido spiniferae-Cynarietum algarbiensis (holotypus relevé: 6, Appendix 2; Figure 2) appear clearly defined in group A (samples 1-7, Figure 1). These are relatively dense forests fringes whose features are easily distinguishable from surrounding syntaxa. Cynara algarbiensis is the dominant hemicryptophyte and the main diagnostic species in this association. This relevé occurs on siliceous substrate, mainly over deep humid soils, derived from schists and greywackes, in the thermo to mesomediterranean, euoceanic to subhyperoceanic (Figure 3), lower sub-humid bioclimate. The assosiation is distributed on potential areas of the climatic domain of Lavandulo viridis-Querco suberis sigmetum, in the southwestern areas of Portugal, especially in the Monchique Sierran District (Espírito-Santo et al., 2017). The new C. algarbiensis forest fringe community proposed here is constantly accompanied by Picris spinifera Franco and other characteristic species from the Origanion virentis alliance, such as Origanum virens Hoffmanns. & Link, Agrimonia eupatoria L. and Clinopodium vulgare L. subsp. arundanum (Boiss.) Nyman (Appendix 2, relevés 1-7). This vegetation unit accompanies forests of Lavandulo viridis-Quercetum suberis, as well as their mantel shrub formation, especially Cisto populifolii-Arbutetum unedonis and Lavandulo viridis-Cytisetum striati. Due to the ecological position (transitional habitat) of these external fringes, the floristic composition also contains taxa from other communities and types of habitats that are in contact with the C. algarbiensis community. In fact, this forest fringe community is enriched with woodland, pre-woodland and dense scrubland elements from Quercetea ilicis such as Arbutus unedo L., Myrtus communis L., Phlomis purpurea L., and Rubia peregrina L., among others, and Cytisetea scopario-striati such as Cytisus striatus (Hill) Rothm. and Pteridium aquilinum (L.) Kuhn. On the other hand, Picrido spiniferae-Cynarietum algarbiensis are also often in contact with Stipo giganteae-Agrostietea castellanae grasslands of Brachypodium phoenicoides (L.) Roem. & Schult. and Dactylis hispanica Roth subsp. lusitanica (Stebbins & Zohary) Rivas Mart. & Izco communities. In more disturbed areas the presence of Cisto-Lavanduletea species in the companion group, such as Cistus ladanifer L., Cistus crispus L. and Ulex argenteus Welw. ex Webb., suggests that this new association does not show high requirements for nutrients or soil moisture. Furthermore, the influence of human activities increases in the abundance of nitrophilous or semi-nitrophilous species in this forest fringe. This group includes semi-nitrophilous species from Stellarietea mediae, such as Galactites tomentosus Moench, Stachys arvensis (L.) L., Coleostephus myconis (L.) Rchb. f., Malva hispanica L., among others, and nitrophilous species from Artemisietea vulgaris, such as Dittrichia viscosa (L.) Greuter subsp. revoluta (Hoffmanns & Link) Pinto da Silva & Tutin, Carlina corymbosa L., Piptatherum miliaceum (L.) Cosson subsp. thomasii (Duby) Freitag, Scabiosa atropurpurea L., Daucus carota L., Eryngium campestre L., Cynara humilis L. The increasing abundance of many nitrophilous species reveals the secondary habitat in which C. algarbiensis appears and co-dominates, due to its ecological capacity. This invasion of C. algarbiensis into another habitat formerly occupied by Stellarietea mediae and Artemisietea vulgaris emphasizes the concept of versaumung (Mucina & Kolbek, 1993), which characterizes some typical forest edge vegetation from Trifolio medii-Geranietea sanguinei. The versaumung process is when the species from the forest fringe communities expand and colonize the neighboring grasslands (secondary habitat), mainly in agroecosystem areas that are no longer devoted to agricultural or pastoral activities (Loidi et al., 2004; Willner, 2011).
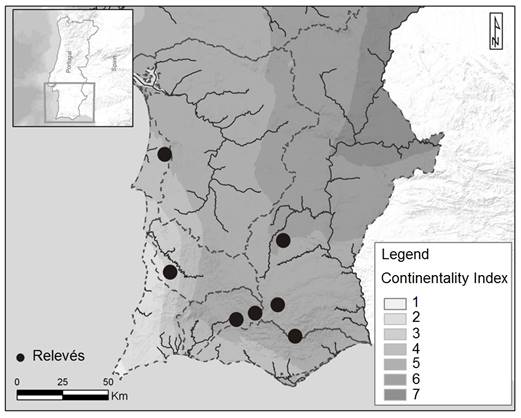
Figure 3 Simple continentality map (Monteiro-Henriques 2010) following Rivas-Martínez (2007) and relevés locations of Picrido spiniferae-Cynarietum algarbiensis. Corresponding legend numbers: 1) attenuated euhyperoceanic, 2) evident subhyperoceanic, 3) attenuated subhyperoceanic, 4) evident semihyperoceanic, 5) attenuated semihyperoceanic, 6) evident euoceanic, 7) attenuated euoceanic.
The original floristic composition, ecological patterns and characteristic biogeographical territory (Table 1) allow us to distinguish a new association, the Picrido spiniferae-Cynarietum algarbiensis. We place this new silicicolous community, at the syntaxonomic level, in the thermophilous forest fringe of Stachyo lusitanicae-Cheirolophenion sempervirentis. Because of the substrata nature given to the descriptions of the thermophilous fringe vegetation classified within the Stachyo lusitanicae-Cheirolophenion sempervirentis suballiance, we propose changing its diagnosis to encompass both calcicolous and silicicolous communities. Originally, these thermophilous communities had been placed in the Origanion virentis alliance. Later, Capelo (1996) showed in detail that this alliance isn’t able to encompass the large variety of different sub-mesophytic, basophilous to neutro-acidophilous communities in the southwestern Iberian Peninsula. Consequently, this same author recognised two suballiances within the Origanion virentis alliance: Origanenion virentis and Stachyo lusitanicae-Cheirolophenion sempervirentis. Rivas-Martínez (2011) then suggested the suballiance Origanenion virentis for the silicicolous substrata (acidophilous and neutro-acidophilous) in the Iberian Atlantic Mediterranean territories and the Stachyo lusitanicae-Cheirolophenion sempervirentis suballiance for the Portuguese Divisorian with Luso-Extremadurese disjunctions, in which they included calcicolous and calco-dolomitics, neutrophilous and neutro-acidophilous associations. Costa et al. (2012) also recognize two suballiances: first, Origanenion virentis, which is ascribed to the silicicolous to neutrophilous communities of the West Iberian Mediterranean territories; and second, Stachyo lusitanicae-Cheirolophenion sempervirentis in which they include six associations encompassing the forest fringe communities growing in the Coastal Lusitanian-Andalusian calcicolous (calco-dolomitic) and neutrophilous territories. Nevertheless, this ecological concept of subdivisions proposed by Costa et al. (2012) for the Stachyo lusitanicae-Cheirolophenion sempervirentis does not comprise the silicicolous communities. The results reported in Table 1 clearly reveal the presence of calcicolous to silicicolous communities classified within the Stachyo lusitanicae-Cheirolophenion sempervirentis, which support and confirm later descriptions of Capelo (2007), who considered this suballiance as edaphically indifferent.
Picrido spiniferae-Cynarietum algarbiensis does not have any correspondence to the Natura 2000 network natural habitats. Nevertheless, part of the character of these forests fringes comes from the shelter effect and the ecological dependence on forests. Therefore, it is to be noted that management objectives of the forest habitat under the Natura 2000 code: 9330 Quercus suber forests from Annex I of Council Directive 92/43/EEC, includes fringe communities within its definition (ALFA, 2006).
Table 1 Biogeographical, substratum and floristic comparision between forest fringe associations of the Stachyo lusitanicae-Cheirolophenion sempervirentis suballiance. The dominant taxa habit of the forest association (E = hard-leaved evergreen oaks; D = late-deciduous taxa) was identified for each forest fringe studied.
| Forest fringe association | Woodland association | Dominant taxa | Characteristics and differentials | Biogeography (sector or district) | Substratum affinity |
|---|---|---|---|---|---|
| Picrido algarbiensis-Cheirolophetum sempervientis | Quercetum alpestris-broteroiPinto Gomes & Paiva Ferreira 2005 | D | Cheirolophus sempervirens (L.) Pomel; Picris algarbiensis Franco; Cephalaria leucantha (L.) Roem. & Schult. | Algarvese District | Calcicolous |
| Origano virentis-Leucanthemetum sylvatici | Sanguisorbo hybridae-Quercetum broteroiPereira 2009 | D | Origanum virens Hoffmanns. & Link; Leucanthemum sylvaticum Nyman; Conopodium marianum Lange | Alentejan District | Silicicolous |
| Senecio lopezii-Cheirolophetum sempervirentis | Avenello strictae-Quercetum marianicae Vila-Viçosa, Vázquez, Mendes, Del Rio, Musarella, Cano-Ortiz & Meireles 2015 | D | C. sempervirens; Senecio lopezii Boiss.; Klasea algarbiensis (Cantó) Cantó; Euphorbia monchiquensis Franco & P. Silva; Centaurea prolongoi Boiss. ex DC. | Monchique Sierran and Vicentinan Coastal Districts | Silicicolous |
| Leucanthemo sylvatici-Cheirolophetum sempervirentis | Arisaro simorrhini-Quercetum broteroi Br.-Bl., P. Silva & Rozeira 1955 corr. Rivas-Martínez 1975; Viburno tini-Quercetum rivasmartinezii Rivas-Martínez, Lousã, T. E. Díaz, Fernández-González & J. C. Costa 1990 corr. Capelo & J. C. Costa 2001 | D-E | C. sempervirens; L. sylvaticum; Calamintha nepeta (L.) Savi; Silene vulgaris (Moench) Garcke | Portuguese Divisorian Sector and Arrabida Sierran District | Calcicolous |
| Bartsio asperae-Origanetum virentis | Arisaro simorrhini-Quercetum broteroi Br.-Bl., P. Silva & Rozeira 1955 corr. Rivas-Martínez 1975; Lonicero implexae-Quercetum rotundifoliae Lousã, Espírito Santo & Costa 1994 | D-E | Origanum virens; Bartsia aspera (Brot.) Lange; Prunella xintermedia Link; Lathyrus sylvestris L.; Bupleurum paniculatum Brot.; Anthyllis vulneraria L. subsp. maura (Beck) Maire | Portuguese Divisorian Sector | Calcicolous |
| Picrido spiniferae-Cynarietum algarbiensis | Lavandulo viridis-Quercetum suberis Quinto-Canas, Vila-Viçosa, Meireles, Ferreira, Martínez-Lombardo, Cano & Pinto-Gomes 2010 | E | Cynara algarbiensis Coss. ex Mariz; Picris spinifera Franco | Monchique Sierran and Alentejan Districts | Silicicolous |
| Stachyo lusitanicae-Origanetum virentis | Asparago aphylly-Quercetum suberis Costa, Capelo, Lousã & Espírito Santo 1996 | E | Origanum virens Hoffmanns. & Link; Stachys germanica L. subsp. lusitanica (Hoffmanns & Link) Cout.; Stachys officinalis (L.) Trevisan subsp. algeriensis (De Noé) Franco; Galium scabrum L.; Heleborus foetidus L.; Geranium sanguineum L. | Portuguese Divisorian Sector and Ribataganian District | silicicolous |
Syntaxonomical scheme
A. Artemisietea vulgaris Lohmeyer, Preising & Tüxen in Tüxen 1950 ex von Rochow 1951.
B. Stellarietea mediae Tüxen, Lohmeyer & Preising ex von Rochow 1951.
C. Trifolio medii-Geranietea sanguinei Müller 1962; where + is the order, * is the alliance, and ** is the suballiance of class.
+ Origanetalia vulgaris Müller 1962.
* Origanion virentisRivas-Martínez & Bolòs in Rivas-Martínez, Díaz, Prieto, Loidi & Penas 1984.
** Stachyo lusitanicae-Cheirolophenion sempervirentisCapelo 1996.
1 - Bartsio asperae-Origanetum virentis Lopes, Lousã & Ladero in Costa et al. 2012.
2 - Leucanthemo sylvatici-Cheirolophetum sempervirentisCosta, Ladero, Díaz, Lousã, Espírito Santo, Vasconcellos, Monteiro & Amor 1993.
3 - Origano virentis-Leucanthemetum sylvaticiPereira 2009.
4 - Picrido algarbiensis-Cheirolophetum sempervientisPinto-Gomes & Paiva-Ferreira 2005.
5 - Senecio lopezii-Cheirolophetum sempervirentisCapelo 1996.
6 - Stachyo lusitanicae-Origanetum virentis (Capelo 1996) Capelo & Costa in Costa, Capelo, Espírito Santo & Lousã 2002.
7 - Picrido spiniferae-Cynarietum algarbiensis Quinto-Canas, Mendes, Vila-Viçosa, Cano Ortiz & Pinto-Gomes ass. nova hoc loco.
D. Poetea bulbosae Rivas Goday & Rivas-Martínez in Rivas-Martínez 1978.
+ Poetalia bulbosae Rivas Goday & Rivas-Martínez in Rivas Goday & Ladero 1970.
* Molineriello minutae-Trifolion subterranei Rivas Goday 1964 nom. inv. Et nom. mut.
8 - Trifolio subterranei-Poetum bulbosae Rivas Goday 1964.
E. Stipo giganteae-Agrostietea castellanae Rivas-Martínez, Fernández-González & Loidi 1999.
F. Cytisetea scopario-striati Rivas-Martínez 1974.
+ Cytisetalia scopario-striati Rivas-Martínez 1974.
* Ulici europaei-Cytision striati Rivas-Martínez, Báscones, Díaz, Fernandez-González & Loidi 1991.
9 - Lavandulo viridis-Cytisetum striati Pinto-Gomes, Cano-Ortiz, Quinto-Canas, Vila-Viçosa & Martínez-Lombardo 2012.
G. Salici purpureae-Populetea nigrae (Rivas-Martínez & Cantó ex Rivas-Martínez, Báscones, T.E. Díaz, Fernández-González & Loidi) Rivas-Martínez & Cantó 2002.
+ Salicetalia purpureae Moor 1958.
* Salicion salviifoliaeRivas-Martínez, T.E. Díaz, F. Prieto, Loidi & Penas 1984.
10 - Salicetum atrocinereo-australis J.C. Costa & Lousã in J.C. Costa, Lousã & Pães 1998.
H. Quercetea ilicis Br.-Bl. ex A. & O. Bolòs 1950.
+ Quercetalia ilicis Br.-Bl. ex Molinier 1934 em. Rivas-Martínez.
* Quercion broteroi Br.-Bl., P. Silva & Rozeira 1956 corr. Rivas-Martínez 1972
** Quercenion broteroi Rivas-Martínez 1987.
11 - Arisaro simorrhini-Quercetum broteroi Br.-Bl., P. Silva & Rozeira 1955 corr. Rivas-Martínez 1975.
12 - Asparago aphylly-Quercetum suberis Costa, Capelo, Lousã & Espírito Santo 1996.
13 - Lavandulo viridis-Quercetum suberis Quinto-Canas, Vila-Viçosa, Meireles, Ferreira, Martínez-Lombardo, Cano & Pinto-Gomes 2010.
14 - Quercetum alpestris-broteroiPinto Gomes & Paiva Ferreira 2005.
15 - Sanguisorbo hybridae-Quercetum broteroiPereira 2009.
** Paeonio broteroi-Quercenion rotundifoliae Rivas-Martínez in Rivas-Martínez, Costa & Izco 1986.
16 - Lonicero implexae-Quercetum rotundifoliae Lousã, Espírito Santo & Costa 1994.
** Quercenion rivasmartinezii-suberis Capelo in J. C. Costa, C. Neto, C. Aguiar, J. Capelo, M. D. Espiríto Santo, J. Honrado, C. Pinto-Gomes, T. Monteiro-Henriques, M. Sequeira, M. Lousã 2012.
17 - Avenello strictae-Quercetum marianicae Vila-Viçosa, Vázquez, Mendes, Del Rio, Musarella, Cano-Ortiz & Meireles 2015.
18 - Viburno tini-Quercetum rivasmartinezii Rivas-Martínez, Lousã, T. E. Díaz, Fernández-González & J. C. Costa 1990 corr. Capelo & J.C. Costa 2001.
* Querco rotundifoliae-Oleion sylvestris Barbéro, Quézel & Rivas-Martínez in Rivas-Martínez, Costa & Izco 1986.
** Querco rotundifoliae-Oleenion sylvestris J. C. Costa, C. Neto, C. Aguiar, J. Capelo, M. D. Espiríto Santo, J. Honrado, C. Pinto-Gomes, T. Monteiro- Henriques, M. Sequeira, M. Lousã 2012.
19 - Ulici argentei-Quercetum rotundifoliae Quinto-Canas, Vila-Viçosa, P. Mendes, E. Cano & Pinto-Gomes inéd.
+ Pistacio lentisci-Rhamnetalia alaterni Rivas-Martínez 1975.
* Ericion arboreae Rivas-Martínez 1987.
** Ericenion arboreae Rivas-Martínez 1975.
20 - Cisto populifolii-Arbutetum unedonis Br.-Bl., P. Silva & Rozeira 1964 nom inv.
Conclusions
In accordance with the phytosociological and numerical analysis, in this document we propose a new forest fringe association, named Picrido spiniferae-Cynarietum algarbiensis, for the thermomediterranean to mesomediterranean, euoceanic to subhyperoceanic, lower sub-humid bioclimatic belt areas of the Monchique Sierran District. Accordingly, natural regeneration and preservation of the higher layers and stages of plant dynamics seems to be a valuable management scheme for successful dispersal and long-term conservation of small and often spatially isolated Cynara algarbiensis communities.











 texto en
texto en 


