Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Revista mexicana de ciencias forestales
versão impressa ISSN 2007-1132
Rev. mex. de cienc. forestales vol.9 no.48 México Jul./Ago. 2018
https://doi.org/10.29298/rmcf.v8i48.129
Articles
Structure, composition and diversity of the urban forest of Linares, Nuevo León
1Facultad de Ciencias Forestales, Universidad Autónoma de Nuevo León. México.
2Escuela Técnica Superior de Ingenieros en Montes, Universidad Politécnica de Madrid. España.
3Instituto de Ecología Aplicada, Universidad Autónoma de Tamaulipas, México.
4Campo Experimental Valle de México, Instituto Nacional de Investigaciones Forestales, Agrícolas y Pecuarias. México.
The urban forests and green areas of the cities provide direct benefits to the population such as improving the temperature and air quality, protect the soil and allow the permeability of water to the subsoil. The aim of this study was to determine the structure, composition and diversity of the urban trees of Linares, Nuevo León. Dasometric information was collected from urban green areas and public parks, counting 6 ridges, 14 parks and 25 public spaces with a total area of 273 904 m2. The variables of diameter (d1.30), total height (h) and diameter of the crown (dcrown) were recorded. The density of plant species by urban green area and the total number of species was calculated. Diversity was assessed using the Shannon-Weiner and Margalef indices. The total number of trees registered was 2 066 belonging to 41 species, 34 genera and 25 families. The most representative was Fabaceae with six species; Fraxinus americana (ash) is outstanding from the ecological point of view, with 25.67 N ha-1, which represents 34.03 % of total abundance and 1 225.38 m2 ha-1 which is equivalent to 46.93 % of dominance and 30.91 % of IVI, followed by Quercus virginiana with 22.46 % and Washingtonia robusta with 11.56 %. The Shannon-Weiner Index recorded H'= 1.99 and H'max = 3.17, and the Margalef Index DMg = 5.24. It is concluded that the urban trees described here are mainly composed of introduced species, the most important of which is ash.
Key words: Abundance; green areas; urban forest; dominance; Margalef index; Shannon index
Los bosques urbanos y áreas verdes de las ciudades proveen beneficios directos a la población, como mejorar la temperatura y la calidad del aire, protegen el suelo y permiten la permeabilidad de agua al subsuelo. El objetivo del trabajo fue determinar la estructura, composición y diversidad del arbolado urbano de Linares, Nuevo León. Se recabó información dasométrica de áreas verdes urbanas y parques públicos; se trabajó con seis camellones, 14 parques y 25 plazas con una superficie de 273 904 m2. Se registró el diámetro normal (d1.30), la altura total (h) y el diámetro de copa (dcopa). Se calculó la densidad de especies vegetales por área verde urbana y su número total. La diversidad fue evaluada mediante los índices de Shannon-Weiner y de Margalef. El total de individuos fue de 2 066, pertenecientes a 41 especies, 34 géneros y 25 familias. La más representativa fue Fabaceae con seis taxones; Fraxinus americana (fresno) es sobresaliente desde el punto de vista ecológico, con 25.67 N ha-1, que representa 34.03 % de la abundancia total, 1 225.38 m2 ha-1 que equivale a 46.93 % de dominancia y 30.91 % de IVI; seguida de Quercus virginiana con 22.46 % y Washingtonia robusta con 11.56 %. El índice de Shannon-Weiner registró un valor de H’ = 1.99 y H'max = 3.17, y el índice de Margalef DMg = 5.24. Se concluye que el arbolado urbano estudiado está compuesto, principalmente, por especies introducidas, de las cuales la más importante es el fresno.
Palabras clave: Abundancia; áreas verdes; bosque urbano; dominancia; índice de Margalef; índice de Shannon
Introduction
Cities are home to a growing proportion of the world's population, and in the face of urban sprawl, green areas play a key role in maintaining social, economic and ecological well-being (McDonnell and Hahs, 2009; Acharya, 2011; Ramalho and Hobbs, 2012). Urban parks are green areas managed mainly for the purpose of coexistence and recreation, and unlike forests or protected parks, they are the most accessible green spaces for the population residing in urbanized areas (Oleyar et al., 2008; Nagendra and Gopal, 2011; Niemelä et al., 2011). Urban parks also provide multiple environmental services: mitigate air pollution, reduce noise, provide scenic beauty, recreation, opportunities for contact with nature (Dobbs et al., 2011; Escobedo et al., 2011; Zhang and Jim, 2014); and they favor the conservation of biological diversity by giving habitat and food to the local fauna (Chace and Walsh, 2006; Carbó-Ramírez and Zuria, 2011).
There is considerable information describing the composition and diversity of trees in urban parks in various parts of the world, and in the United States, Europe and Asia, where they have developed most (Lososová et al., 2011; Nagendra and Gopal, 2011; Jim and Zhang, 2013; Pesola et al., 2017; Riley et al., 2017). In Mexico, there is not much information about the green areas within the urban areas of several cities. The best assessed areas are large metropolis such as Mexico City, where general studies have been conducted covering the entire urban area (Benavides, 1992; Checa-Artasu, 2016) or on a particular area such as Chapultepec Forest (Benavides and Young, 2012).
In the northeastern part of Mexico, there are few records on the subject of green areas and urban forests. Alanís (2005) and Jiménez et al. (2013) evaluated the green areas of the Monterrey municipality, Nuevo León, through geographic information systems and in situ forest inventories. In Tamaulipas, Mora and Martínez (2012) elaborated a work on the wild plants of an urban forest located in Ciudad Victoria. In the case of the municipality of Linares, Nuevo León, information is scarce and limited to studies carried out on the main square of the city on the trees on the sidewalks (Zamudio, 2001) and on a student campus (Alanís et al., 2014). Based on the above, the objective of this study was to determine the structure, composition and diversity of urban trees in the different squares, parks and ridges of the urban area of Linares, Nuevo León.
Materials and Methods
Study area
The Linares municipality (Figure 1) is located southeast of the state of Nuevo León. Geographically it limits towards the north with the Hualahuises and Montemorelos municipalities; both to the south and to the east with the state of Tamaulipas and to the west with Galeana and Iturbide municipalities. Its geographic coordinates correspond to 24°51' N and 99°24' W (Municipios Mx, 2018).

Upper left image = NE of Mexico and SE United States of America; Lower left image = Linares municipality, Nuevo León; Right image = Linares urban area with its green areas; COA = Coahuila; NL = Nuevo León; ZAC = Zacatecas; SLP = San Luis Potosí; TAM = Tamaulipas.
Figure 1 Location of the study area.
Floristic inventory
A census was made of all the plant species present in public areas such as plazas, parks and ridges of the city of Linares. Thus, it was considered that the squares and parks are clearly delimited public spaces, dominated by vegetation and intended as public green areas for the realization of recreational or outdoor sports activities. This information was collected during the months of March and April 2016.
The dendrometric variables of normal diameter (d1.30 m) were measured with a Haglöf Mantax Blue 1270 mm caliper; total height (h), with a Suunto PM-5 hypsometer; and the diameter of the crown (dcrown), with a 100 m metric cross fiber glass TruperTM, tape according to the four cardinal points north-south and east-west. The coordinates of each square, park and ridge were recorded with an eTrex 20x Garmin® mountain crossing GPS.
Data analysis
For each species, their abundance was determined, according to the number of trees, their coverage, depending on the crown area, and their frequency based on their presence in the sampling sites. The relativized variables were used to obtain a weighted value at the taxon level called the Importance Value Index (IVI), which acquires percentage values on a scale of 0 to 100 (Mostacedo and Fredericksen, 2000).
For the estimation of relative abundance the following equation was used:
Where:
ARi = Relative abundance of the i species in regard to total abundance
Ai = Absolute abundance of the i species (N ha-1)
Dominance was calculated through the equation:
Where:
DRi = Relative dominance of the i species in regard to total dominance
D = Absolute dominance of the i species (m2 ha-1)
The absolute and relative frequencies were obtained with the equations:
Where:
Fi= Absolute frequency (percentage of presence in the sampling sites)
fi = Number of sites in which de i species is present
N = Number of sampling sites
FRi= Relative frequency of the i species in regard to total frequency
The Importance Value Index (IVI) is defined through the equation:
To estimate the alpha diversity, we used the Margalef index (DMg) and the Shannon-Weiner index (H') [18], using the following equations:
Where:
S = Number of present species
N = Total number of individuals
ni = Number of individuals of the i species
By means of a correlation analysis, the relationship between the parameters of wealth and diversity and the distance of ages from urban parks was explored.
Results
There were 41 species belonging to 34 genera and 25 families of vascular plants (Table 1). The most representative family was Fabaceae with six species, Fagaceae with four species, later Moraceae and Arecaceae with three each, and finally, Bignoniaceae, Oleaceae, Pinaceae and Rutaceae with two. Of the 41 species recorded in the study, 14 were native and 27 exotic (Table 2).
Table 1 Distribution of species and number of individuals per assessed area.
| Areas | Species | Individuals | |
|---|---|---|---|
| Squares | 21 | 37 | 1 071 |
| Parks | 14 | 21 | 835 |
| Ridges | 6 | 9 | 160 |
| Total | 41 | 41 | 2 066 |
Table 2 Scientific and common name, family and origin of the tree species recorded in the study area.
| Scientific name | Common name | Family | Origin |
|---|---|---|---|
| Azadirachta indica A.Juss. | Neem | Meliaceae | Exotic |
| Bauhinia variegata L. | Pata de vaca | Fabaceae | Exotic |
| Caesalpinia mexicana A.Gray | Árbol del potro | Caesalpinaceae | Native |
| Carya illinoinensis (Wangenh.) K.Koch | Nogal | Yuglandaceae | Native |
| Celtis laevigata Willd. | Palo blanco | Fabaceae | Native |
| Citrus sinensis (L.) Osbeck | Naranjo | Rutaceae | Exotic |
| Cordia boissieri A.DC. | Anacahuita | Boraginaceae | Native |
| Delonix regia (Bojer) Raf. | Flamboyán | Fabaceae | Exotic |
| Diospyros texana Scheele | Chapote prieto | Ebenaceae | Native |
| Ebenopsis ebano (Berland.) Barneby & J.W.Grimes. | Ébano | Fabaceae | Native |
| Eucalyptus globulus Labill. | Eucalipto | Myrtaceae | Exotic |
| Ficus benjamina L. | Ficus | Moraceae | Exotic |
| Ficus microcarpa L.f. | Laurel de la India | Moraceae | Exotic |
| Fraxinus americana L. | Fresno | Oleaceae | Exotic |
| Helietta parvifolia (A. Gray ex Hemsl.) Benth. | Barreta | Rutaceae | Native |
| Jacaranda mimosifolia D.Don | Jacaranda | Bignoniaceae | Exotic |
| Leucaena leucocephala (Lam.) de Wit | Leucaena | Fabaceae | Exotic |
| Ligustrum japonicum Thunb. | Trueno | Oleaceae | Exotic |
| Melia azedarach L. | Canelón | Meliaceae | Exotic |
| Phoenix dactylifera L. | Palma datilera | Arecaceae | Exotic |
| Pinus halepensis Mill. | Pino halapensis | Pinaceae | Exotic |
| Pinus pseudostrobus Lindl. | Pino real | Pinaceae | Native |
| Platanus rzedowskii Nixon & J.M.Poole | Sicomoro | Moraceae | Native |
| Prosopis laevigata (Humb. & Bonpl. ex Willd.) M.C. Johnst. | Mezquite | Fabaceae | Native |
| Prunus persica (L.) Batsch | Durazno | Rosaceae | Exotic |
| Punica granatum L. | Granado | Lythraceae | Exotic |
| Quercus macrocarpa Michx. | Encino macrocarpa | Fagaceae | Exotic |
| Quercus rubra L. | Encino Rojo | Fagaceae | Exotic |
| Quercus texana Buckley | Encino Cartamus | Fagaceae | Exotic |
| Quercus virginiana Mill. | Encino siempre verde | Fagaceae | Native |
| Salix humboldtiana Willd. | Sauce | Salicaceae | Exotic |
| Sapindus saponaria L. | Jaboncillo | Sapindaceae | Native |
| Sapium sebiferum (L.) Roxb. | Chainis | Euphorbiaceae | Exotic |
| Schinus molle L. | Pirul | Anacardiaceae | Exotic |
| Syagrus romanzoffiana (Cham.) Glassman | Palma coco plumoso | Arecaceae | Exotic |
| Tabebuia rosea (Bertol.) DC. | Palo de rosa | Bignoniaceae | Exotic |
| Taxodium mucronatum Ten. | Sabino | Taxodiaceae | Native |
| Thuja occidentalis L. | Tuya | Cupressaceae | Exotic |
| Vitex agnus-castus L. | Árbol casto | Lamiaceae | Exotic |
| Washingtonia robusta var. gracilis (Parish) Parish ex Becc. | Palma Washingtonia | Arecaceae | Exotic |
| Yucca filifera Chabaud | Palma pita | Asparagaceae | Native |
A total abundance of 75.43 N ha-1 was recorded. The most outstanding species was Fraxinus americana L. with 25.67 N ha-1, which represents 34.03 % of the total. Quercus virginiana Mill. with 28.61 % and Washingtonia robusta var. gracilis (Parish) Parish ex Becc. with 15.15 % follow; the remaining species together sum 22.21 %.
The crown coverage of the urban green areas (parks, squares and ridges) was 2 611.31 m2 ha-1. Fraxinus americana registered greater dominance with 1 225.38 m2 ha-1, followed by Quercus virginiana with 552.04 m2 ha-1 and Carya illinoinensis (Wangenh.) K.Koch with 255.63 m2 ha-1; the remaining species represented 22.14 %.
Quercus virginiana stands out as the main species in urban green areas in Linares, as it was recognized 33 of the 41 green areas analyzed (17.65 %). Washingtonia robusta var. gracilis was in 25 of them, which is equivalent to 13.37 %, followed by Fraxinus americana in 22 (11.76 %).
The highest calculated value for the Importance Value Index was for Fraxinus americana with 30.91 %, followed by Quercus virginiana with 22.46 % and for Washingtonia robusta with 11.56 %, which together add up to 64.93 % (Table 3). In contrast, Citrus sinensis (L.) Osbeck, Punica granatum L., Prunus persica (L.) Batsch and Tabebuia rosea (Bertol.) DC.recorded 0.20 % each, which is the lowest value.
Table 3 Abundance (N ha-1), dominance (m2 ha-1), frequency, Importance value Index of the species registered in urban green areas in Linares, N L.
| Scientific name* | Abundance | Dominance | Frequency | Importance values | ||||
|---|---|---|---|---|---|---|---|---|
| N ha-1 | % | m2 ha-1 | % | N/Site | % | IVI | IVI rel | |
| Fraxinus americana L. | 25.67 | 34.03 | 1 225.38 | 46.93 | 22.00 | 11.76 | 92.72 | 30.91 |
| Quercus virginiana Mill. | 21.58 | 28.61 | 552.04 | 21.14 | 33.00 | 17.65 | 67.39 | 22.46 |
| Washingtonia robusta var. gracilis (Parish) Parish ex Becc. | 11.43 | 15.15 | 161.23 | 6.17 | 25.00 | 13.37 | 34.69 | 11.56 |
| Carya illionensis (Wangenh.) K.Koch | 2.15 | 2.86 | 255.63 | 9.79 | 10.00 | 5.35 | 17.99 | 6.00 |
| Ligustrum japonicum Thunb. | 3.50 | 4.65 | 75.96 | 2.91 | 5.00 | 2.67 | 10.23 | 3.41 |
| Quercus rubra L. | 1.50 | 1.98 | 32.97 | 1.26 | 8.00 | 4.28 | 7.53 | 2.51 |
| Cordia boissieri A.DC. | 0.51 | 0.68 | 12.64 | 0.48 | 8.00 | 4.28 | 5.44 | 1.81 |
| Syagrus romanzoffiana (Cham.) Glassman | 0.69 | 0.92 | 10.41 | 0.40 | 6.00 | 3.21 | 4.53 | 1.51 |
| Quercus macrocarpa Michx. | 0.47 | 0.63 | 11.32 | 0.43 | 6.00 | 3.21 | 4.27 | 1.42 |
| Thuja occidentalis L. | 1.02 | 1.36 | 21.77 | 0.83 | 3.00 | 1.60 | 3.79 | 1.26 |
| Ebenopsis ebano (Berland.) Barneby & J.W.Grimes. | 0.22 | 0.29 | 13.98 | 0.54 | 5.00 | 2.67 | 3.50 | 1.17 |
| Azadirachta indica A.Juss. | 0.47 | 0.63 | 13.11 | 0.50 | 4.00 | 2.14 | 3.27 | 1.09 |
| Phoenix dactylifera L. | 0.91 | 1.21 | 38.38 | 1.47 | 1.00 | 0.53 | 3.21 | 1.07 |
| Pinus pseudostrobus Lindl. | 0.80 | 1.06 | 23.45 | 0.90 | 2.00 | 1.07 | 3.03 | 1.01 |
| Jacaranda mimosifolia D.Don. | 0.44 | 0.58 | 21.13 | 0.81 | 3.00 | 1.60 | 2.99 | 1.00 |
| Melia azedarach L. | 0.26 | 0.34 | 10.99 | 0.42 | 4.00 | 2.14 | 2.90 | 0.97 |
| Taxodium mucronatum Ten. | 0.18 | 0.24 | 13.44 | 0.51 | 4.00 | 2.14 | 2.90 | 0.97 |
| Leucaena leucocephala (Lam.) de Wit | 0.18 | 0.24 | 9.53 | 0.36 | 4.00 | 2.14 | 2.75 | 0.92 |
| Sapium sebiferum (L.) Roxb. | 0.69 | 0.92 | 18.11 | 0.69 | 2.00 | 1.07 | 2.68 | 0.89 |
| Bauhinia variegata L. | 0.40 | 0.53 | 13.60 | 0.52 | 3.00 | 1.60 | 2.66 | 0.89 |
| Ficus benjamina L. | 0.29 | 0.39 | 12.64 | 0.48 | 3.00 | 1.60 | 2.48 | 0.83 |
| Salix humboldtiana Willd. | 0.15 | 0.19 | 2.15 | 0.08 | 3.00 | 1.60 | 1.88 | 0.63 |
| Prosopis laevigata (Humb. & Bonpl. ex Willd.) M.C. Johnst. | 0.11 | 0.15 | 9.84 | 0.38 | 2.00 | 1.07 | 1.59 | 0.53 |
| Quercus texana Buckley | 0.33 | 0.44 | 13.19 | 0.51 | 1.00 | 0.53 | 1.48 | 0.49 |
| Yucca filifera Chabaud | 0.15 | 0.19 | 4.41 | 0.17 | 2.00 | 1.07 | 1.43 | 0.48 |
| Celtis laevigata Willd. | 0.11 | 0.15 | 3.09 | 0.12 | 2.00 | 1.07 | 1.33 | 0.44 |
| Delonix regia (Bojer) Raf. | 0.11 | 0.15 | 2.13 | 0.08 | 2.00 | 1.07 | 1.30 | 0.43 |
| Schinus molle L. | 0.29 | 0.39 | 8.92 | 0.34 | 1.00 | 0.53 | 1.26 | 0.42 |
| Diospyros texana Scheele | 0.22 | 0.29 | 2.62 | 0.10 | 1.00 | 0.53 | 0.93 | 0.31 |
| Platanus rzedowskii Nixon & J.M.Poole | 0.11 | 0.15 | 3.06 | 0.12 | 1.00 | 0.53 | 0.80 | 0.27 |
| Eucalyptus globulus Labill. | 0.04 | 0.05 | 5.00 | 0.19 | 1.00 | 0.53 | 0.77 | 0.26 |
| Helietta parvifolia (A. Gray ex Hemsl.) Benth. | 0.11 | 0.15 | 0.58 | 0.02 | 1.00 | 0.53 | 0.70 | 0.23 |
| Pinus halepensis Mill. | 0.04 | 0.05 | 2.35 | 0.09 | 1.00 | 0.53 | 0.67 | 0.22 |
| Vitex agnus-castus L. | 0.04 | 0.05 | 1.40 | 0.05 | 1.00 | 0.53 | 0.64 | 0.21 |
| Sapindus saponaria L. | 0.04 | 0.05 | 1.27 | 0.05 | 1.00 | 0.53 | 0.63 | 0.21 |
| Ficus microcarpa L.f. | 0.04 | 0.05 | 0.93 | 0.04 | 1.00 | 0.53 | 0.62 | 0.21 |
| Caesalpinia mexicana A.Gray | 0.04 | 0.05 | 0.86 | 0.03 | 1.00 | 0.53 | 0.62 | 0.21 |
| Tabebuia rosea (Bertol.) DC. | 0.04 | 0.05 | 0.66 | 0.03 | 1.00 | 0.53 | 0.61 | 0.20 |
| Prunus persica (L.) Batsch | 0.04 | 0.05 | 0.59 | 0.02 | 1.00 | 0.53 | 0.61 | 0.20 |
| Punica granatum L. | 0.04 | 0.05 | 0.29 | 0.01 | 1.00 | 0.53 | 0.59 | 0.20 |
| Citrus sinensis (L.) Osbeck | 0.04 | 0.05 | 0.26 | 0.01 | 1.00 | 0.53 | 0.59 | 0.20 |
| 75.43 | 100.00 | 2 611.31 | 100.00 | 187.00 | 100.0 | 300.0 | 100.00 | |
*The species follow a decreasing order according to their importance value.
The density of individuals per hectare according to the diameter classes registered in the study showed that most of the individuals evaluated are concentrated in category II (14.5 - 29 cm) with a total of 1 011 N ha-1 (Figure 2).

Densidad = Density; Clases diaméticas = Diametric classes.
Figure 2 Density of individuals according to diametric classes in the study areas.
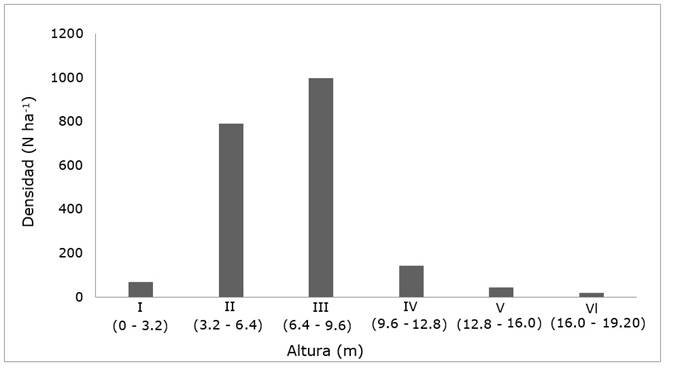
The mensuration variable of total height was calculated based on the density of individuals by height category. Category III (6.4 - 9.6 m) was the one that predominated with a total of 998 individuals per hectare, which corresponds to 48.3 % of all registered individuals, while category VI (16 - 19.2 m) was the lowest number of individuals with only 19 (0.91 %) (Figure 3).
The evaluated area showed values of Margalef's Index of DMg = 5.24 and of Shannon-Wiener of H´ = 1.99, and the val H'max = 3.17.
Discussion
In the inventory of urban trees 2 066 specimens were registered, belonging to 24 families and 41 species. This number of species is close to the data of Zamudio (2001) in the center of Linares, who identified the existence of 39 species, with 19 families in 1995 and 49 species with 27 families in 1999. It is low compared to the study conducted by Alanís (2005), who in an investigation in the metropolitan area of Monterrey, detected 115 species, grouped in 37 families, which surely responds to the difference in the sampled surface.
On the other hand, Alanís et al. (2014) observed a similar number of species (39) to the one registered in this study, by restricting the area to the university campus of the Faculty of Forestry Sciences of the UANL.
The Fabaceae family was the best represented with six species (three native and three exotic), followed by Fagaceae with four species (three exotic), Moraceae with three species (two exotic), and Arecaceae with three species (all exotic). These data coincide with those of Alanís et al. (2014), who highlighted Fabaceae with 10 species (nine native). It also coincides with the information of Alanís (2005), in which Fagaceae and Moraceae are the most important in the Metropolitan Area of Monterrey.
The species with the greatest ecological weight for this study was Fraxinus americana, which concentrates 30.91 % of the Importance Value Index, followed by Quercus virginiana, Carya illinoinensis, Whasingtonia robusta and Ligustrum japonicum Thunb. This percentage is higher than that calculated by Alanís et al. (2014) who granted Fraxinus americana 18.21 % of IVI. Zamudio (2001) defined the latter within the four species with the greatest ecological weight (13.40 % of IVI).
In the study described here, dominant were Fraxinus americana (46.93 %), Quercus virginiana (21.14 %), Carya illinoinensis (9.79 %) and Washingtonia robusta (6.17 %). For Carabias and Herrera (1986), these percentages are not satisfactory, since they establish that no species should exceed 5 % of the total population in the public trees of a city.
Only 13 species are native (36.6 %) and 27 are exotic (63.4 %) (Table 2). The opposite is revealed by Alanís et al. (2014), since of its total number (39), 12 are exotic and 27 are native; this corresponds to a higher percentage of species of the second origin in the urban trees of the campus of the forest school. For the Metropolitan Area of Monterrey, Alanís (2005) accounted for 61 introduced species (53 %) and 54 native species (47 %).
The evaluated plant community has a species richness of DMg = 5.24 and a diversity of H ́ = 1.99. These values are lower than those of Alanís et al. (2014), DMg = 7.62 and H ́ = 3.05. When comparing the results observed in this research with those of Zamudio (2001) for diversity, for the year 1995 the value obtained was H ́ = 2.54 and for the year 1999 it was H ́ = 2.27.
If the results of diversity and richness determined in the present investigation are contrasted only with the native species consigned by Alanís et al. (2014), it can be observed that these authors estimated high values of DMg = 5.80 and H ́ = 2.82, with respect to those here calculated of DMg = 2.12 and H ́ = 0.8127, which are lower.
Conclusions
The urban trees of the of Linares include a high number of exotic species; Fraxinus americana, Quercus virginiana, Carya illinoinensis and Washingtonia robusta var. Gracilis are dominant, the first one of which is the most representative with a total of 703 individuals, which are equivalent to more than 45 % of its green areas.
The actual research study revealed data about the number and surface areas of the squares, ridges and parks in the city of interest, as well as of the species and families of all the trees of the urban forest. This information is useful in the detection of priority areas for reforestation, as this is a reliable inventory that contributes to the management of public trees.
Acknowledgements
To the Facultad de Ciencias Forestales (School of Forest Sciences) of the Universidad Autónoma de Nuevo León (Autonomous University of Nuevo Leon), as well as to the Linares municipality, Nuevo León, for all the facilities granted for the establishment and development of the research, the authors also thank all the people who actively participated in field work.
REFERENCES
Acharya, A. K. 2011. Urbanization and spatial changes in demographic characteristics in Monterrey Metropolitan Region. Caminhos de Geografia 12(39): 271-282. [ Links ]
Alanís F., G. J. 2005. El arbolado urbano en el área metropolitana de Monterrey. Ciencias UANL 8(1):20-32. [ Links ]
Alanís, E., J. Jiménez, A. Mora-Olivo, P. Canizalez y L. Rocha. 2014. Estructura y composición del arbolado urbano de un campus universitario del noreste de México. Revista Iberoamericana de Ciencias 1 (7): 93-101. [ Links ]
Benavides M., H. M. 1992. Current situation of the urban forest in Mexico City. Journal of Arboriculture 18(1): 33-36. [ Links ]
Benavides M., H. M. y D. Young F. G. 2012. Estructura del arbolado y caracterización dasométrica de la segunda sección del Bosque de Chapultepec. Madera y Bosques 18(2):51-71. [ Links ]
Carabias, J. y A. Herrera. 1986. La ciudad y su ambiente. Cuadernos Políticos 45:56-60. [ Links ]
Carbó-Ramírez, P. and I. Zuria. 2011. The value of small urban greenspaces for birds in a Mexican city. Landscape and Urban Planning 100(3): 213-222. [ Links ]
Chace, J. F. and J. J. Walsh. 2006. Urban effects on native avifauna: a review. Landscape and Urban Planning 74(1): 46-69. [ Links ]
Checa-Artasu, M. M. 2016. Las áreas verdes en la Ciudad de México. Las diversas escalas de una geografía urbana. Biblio 3W 21 (1.159):1-21. [ Links ]
Dobbs, C., F. Escobedo J. and W. Zipperer C. 2011. A framework for developing urban forest ecosystem services and goods indicators. Landscape and Urban Planning 99(3): 196-206. [ Links ]
Escobedo, F. J., T. Kroeger and J. Wagner E. 2011. Urban forests and pollution mitigation: analyzing ecosystem services and disservices. Environmental Pollution 159(8): 2078-2087. [ Links ]
Jim, C. Y. and H. Zhang. 2013. Species diversity and spatial differentiation of old-valuable trees in urban Hong Kong. Urban Forestry & Urban Greening 12(2): 171-182. [ Links ]
Jiménez J., P., G. Cuéllar R. y E. Treviño G. 2013. Áreas Verdes del Municipio de Monterrey. In: Gobierno Municipal de Monterrey (ed.). Estudio de Áreas Verdes del Municipio de Monterrey. Facultad de Ciencias Forestales de la UANL. Gobierno Municipal de Monterrey 2012-2015. Monterrey, N.L., México. 21 p. [ Links ]
Lososová, Z., M. Horsák, M. Chytry, T. Čejka, J. Danihelka, K. Fajmon and Z. Otypková. 2011. Diversity of Central European urban biota: effects of human‐made habitat types on plants and land snails. Journal of Biogeography 38(6): 1152-1163. [ Links ]
McDonnell, M. J. and A. K. Hahs 2009. Comparative ecology of cities and towns: past, present and future. In: McDonnell, M. J., A. K. Hahs and J. H. Breuste (eds.). Ecology of cities and towns: a comparative approach. Cambridge University Press. Cambridge, UK. 736 p. [ Links ]
Mora O., A. y J. G. Martínez-Á. 2012. Plantas silvestres del bosque urbano, Cd. Victoria, Tamaulipas, México. Universidad Autónoma de Tamaulipas. Coordinación Editorial Dolores Quintanilla. Saltillo, Coah., México. 139 p. [ Links ]
Mostacedo, B. y T. Fredericksen S. 2000. Manual de métodos básicos de muestro y análisis en ecología vegetal. Proyecto de Manejo Forestal Sostenible BOLFOR. Santa Cruz, Bolivia. 87 p. [ Links ]
Municipios Mx. 2018. Nuevo León. Información sobre Linares. http://www.municipios.mx/nuevo-leon/linares/ (1 de junio 2018). [ Links ]
Nagendra, H. and D. Gopal D. 2011. Tree diversity, distribution, history and change in urban parks: studies in Bangalore, India. Urban Ecosystems 14(2): 211-223. [ Links ]
Niemelä, J., J. H. Breuste, T. Elmqvist, G. Guntenspergen, P. James and N. E. McIntyre. 2011. Urban ecology: patterns, processes and applications. Oxford University Press. Oxford, UK. 376 p. [ Links ]
Oleyar, M. D., A. I. Greve, J. C. Withey and A. M. Bjorn. 2008. An integrated approach to evaluating urban forest functionality. Urban Ecosystems 11(3): 289-308. [ Links ]
Pesola, L., X. Cheng, G. Sanesi, G. Colangelo, M. Elia and R. Lafortezza. 2017. Linking above-ground biomass and biodiversity to stand development in urban forest areas: a case study in Northern Italy. Landscape and Urban Planning 157: 90-97. [ Links ]
Ramalho, C. E. and R. J. Hobbs. 2012. Time for a change: dynamic urban ecology. Trends in Ecology & Evolution 27(3): 179-188. [ Links ]
Riley, C. B., D. Herms A. and M. Gardiner M. 2017. Exotic trees contribute to urban forest diversity and ecosystem services in inner-city Cleveland, OH. Urban Forestry & Urban Greening 29:367-376. http//dx.doi.org/10.1016/j.ufug.2017.01.004 (15 de noviembre de 2017). [ Links ]
Zamudio C., E. 2001. Análisis del comportamiento del arbolado urbano público durante el período de 1995 a 1999 en la ciudad de Linares, NL. Tesis de Maestría. Facultad de Ciencias Forestales. Universidad Autónoma de Nuevo León. Linares, N L, México. 117 p. [ Links ]
Zhang, H. and C. Jim Y. 2014. Contributions of landscape trees in public housing estates to urban biodiversity in Hong Kong. Urban Forestry & Urban Greening 13(2): 272-284. [ Links ]
Received: March 21, 2018; Accepted: June 18, 2018











 texto em
texto em