Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Revista mexicana de ciencias pecuarias
versão On-line ISSN 2448-6698versão impressa ISSN 2007-1124
Rev. mex. de cienc. pecuarias vol.10 no.2 Mérida Abr./Jun. 2019
https://doi.org/10.22319/rmcp.v10i2.4759
Technical notes
Genome association with Cooperia punctata resistance in crossbreed cattle in the sub-humid tropics of Mexico
a Instituto Nacional de Investigaciones Forestales Agrícolas y Pecuarias (INIFAP). Centro Nacional de Investigación Disciplinaria en Fisiología y Mejoramiento Animal. Ajuchitlán, Querétaro.
b Universidad Nacional Autónoma de México (UNAM). Centro de Enseñanza, Investigación y Extensión en Ganadería Tropical. Martínez de la Torre, Veracruz. México.
c Instituto Nacional de Investigaciones Forestales Agrícolas y Pecuarias (INIFAP). Sitio Experimental las Margaritas, Hueytamalco, Puebla. México.
d Instituto Nacional de Investigaciones Forestales Agrícolas y Pecuarias (INIFAP). Campo Experimental La Posta, Paso del Toro, Veracruz. México.
e Instituto Nacional de Investigaciones Forestales Agrícolas y Pecuarias (INIFAP). Centro Nacional de Investigación Disciplinaria en Salud Animal e Inocuidad. Jiutepec, Morelos. México.
This study is an evaluation of resistance to natural infection by Cooperia spp. in Zebu x Holstein crossbreed calves in the tropics. Fourteen four-month-old calves were dewormed and moved to pastures naturally infested with gastrointestinal nematodes under sub-humid tropical conditions. Fecal samples were collected from each animal every seven days for three months to quantify the number of eggs per gram of feces (EPG), and nematode species identified with end-point PCR. Hair samples were collected for genotyping using the GeneSeek Genomic Profiler HD-V3 panel, which contains 139,376 SNP markers. Variation in EPG per individual ranged from a minimum of 7+7 to a maximum of 4,657+1,886 EPG. The PCR identified breed differences between the Zebu x Holstein crossbreeds. Genome-wide association studies detected five statistically significant haplotypes (P<0.001). The haplotype in chromosome 2 includes four markers, that in chromosome 10 includes three, that in chromosome 15 includes two, that in chromosome 23 includes four and chromosome X includes three. Of these regions only chromosome 23 was found to be associated with parasite resistance, measured as EPG phenotypes. The remaining chromosomes exhibited no association in the studied animals. These regions could be sequenced and tested for gene expression against Cooperia and other gastrointestinal nematodes.
Key words: SNP markers; Genomic association studies; Nematode resistance; Crossbreed calves
El objetivo del presente estudio fue evaluar la resistencia a la infección natural por Cooperia spp. en bovinos jóvenes cruzados Cebú x Holstein, en trópico. Catorce (14) becerros de cuatro meses de edad fueron tratados con desparasitante y trasladados a potreros naturalmente contaminados con nematodos gastrointestinales bajo condiciones de trópico sub-húmedo. Muestras de heces se colectaron cada siete días durante tres meses por individuo, para cuantificar el número de huevos por gramo de heces (HPG) e identificar el género por PCR de punto final. Se colectaron muestras de pelo para llevar a cabo estudios de asociación genómica utilizando la plataforma GeneSeek Genomic Profiler HD-V3 que contiene139,376 marcadores SNP. Los resultados obtenidos indican variación en el número de HPG por individuo (mínimo 7+7 y máximo 4657+1886 HPG). En los análisis de componentes principales se identificaron diferencias raciales entre los animales con genes de las razas Cebú y Holstein. Los estudios de asociación del genoma completo detectaron 5 haplotipos estadísticamente significativos (P<0.001). El haplotipo del cromosoma 2 incluye cuatro marcadores, el 10 incluye a tres, el 15 incluye a dos, el 23 cuatro y el del cromosoma X incluye tres. De estas regiones sólo el cromosoma 23 se encontró asociado a resistencia por parásitos, medidos como fenotipo de HPG; los cromosomas restantes identificados no mostraron asociación en ninguno de los individuos en estudio. Se considera que estas regiones podrían ser secuenciadas y probar la expresión de genes para la resistencia contra este nematodo y de otros nematodos del complejo gastrointestinal.
Palabras clave: Marcadores SNP; Asociación genómica; Cooperia spp.; Bovinos; Cruzamientos
Gastrointestinal (GI) nematode infection in young cattle is one of the main health problems affecting livestock under grazing conditions in tropical climates1. The Cooperia species are GI parasites which negatively affect growth and productivity in cattle by compromising small intestine function2. Several mechanisms have been studied to combat Cooperia infection, including pharmaceuticals and genetic resistance3. Availability of high density DNA marker panels for single nucleotide polymorphisms (SNP) has made it possible to map the loci of traits of commercial interest and thus increase the presence of desirable genes in a population. Haplotypes are regions of the genome (comprising between 100 and 150 base pairs) that are jointly inherited in a specific population and may have significant effects on genes located within a region of the chromosome4. Genome-wide association studies (GWAS) allow identification of these regions in genetic material. Recently, genome regions have been identified that indicate resistance to disease and parasites5.
The present study objective was to identify regions of the genome associated with parasite load (number of eggs per gram of feces) of Cooperia species in crossbreed cattle (Zebu x Holstein and vice versa) in a grazing system in the tropics of Mexico.
The study was carried out in the Center for Education, Research and Extension in Tropical Livestock (Centro de Enseñanza, Investigación y Extensión en Ganadería Tropical - CEIEGT), of the Faculty of Veterinary Medicine and Zootechnic, National Autonomous University of Mexico, located in the municipality of Tlapacoyan, in the state of Veracruz, Mexico. Study area altitude is 112 m asl. Its climate is warm humid, with rains year round and no defined dry season (Af(m) w”(e)6, an average temperature of 23.5 ± 0.5 °C, and average annual rainfall of 1,991 mm ± 392 mm. Gastrointestinal (GI) nematode prevalence in the area exceeds 75% year round7. The experimental animals were fourteen crossbreed calves (– Zebu x ¼ Holstein [n = 6] and ¼ Zebu x – Holstein [n = 8]), four months of age in a grazing system. Prior to weaning, both calves and mothers were treated with 7.5 mg Levamisole by intramuscular injection to eliminate any possible GI nematode contamination.
All animals were naturally infected with Cooperia punctata (L3) infectious larvae (confirmed by end-point PCR as a dominant genus) by ingestion. The first record of eggs per gram of feces (EPG) was taken 21 days after infection by grazing was initiated, and then continued every seven days for three months. Eggs counts were done following the McMaster technique. Samples were collected directly from the rectum to prevent contamination and processed immediately. Average EPG was calculated during sampling, and the results included in a cases and controls study, considering cases to be animals exhibiting a >200 EPG8,9. Infection level was classified by EPG counts for parasitic nematodes (Table 1). Blood samples were taken to measure the percentage of the package cell volume (PCV), using 24% as the level for a non-anemic individual9. Levels for PCV were measured using a 22 + 1.6% minimum and a 28 + 3.3% maximum. It is important to mention that the Cooperia genus exhibits tissue-feeding habits and thus does not directly affect PCV percentage. This genus was prevalent throughout the three-months study period, meaning that PCV low percentage was a response to animal nutritional status; this was how six individuals were identified as resilient.
Table 1: Selection of resistant, susceptible and resilient calves vis-a-vis Cooperia punctata based on eggs per gram of feces (EPG) and package cell volume (PCV) percentage
| ID | EPG Mean ± SE |
PCV (%) Mean ± SE |
C |
|---|---|---|---|
| 62 | 7 ± 7 | 24 ± 2.2 | RR |
| 58 | 164 ± 89 | 24 ± 1.1 | RR |
| 61 | 200 ± 107 | 26 ± 2.2 | RR |
| 57 | 178 ± 154 | 23 ± 2.8 | RR |
| 55 | 86 ± 291 | 22 ± 0.7 | Rs |
| 56 | 650 ± 291 | 28 ± 1.7 | Rs |
| 51 | 557 ± 291 | 24 ± 3.3 | Rs |
| 67 | 607 ± 165 | 22 ± 1.5 | Rs |
| 65 | 764 ± 283 | 23 ± 1.5 | Rs |
| 63 | 1193 ± 636 | 25 ± 3.3 | Rs |
| 59 | 1221 ± 558 | 25 ± 0.8 | SS |
| 53 | 1378 ± 291 | 26 ± 2.7 | SS |
| 54 | 3100 ± 291 | 22 ± 2.3 | SS |
| 64 | 4657 ± 1886 | 23 ± 2.1 | SS |
| X ± SE | 1055 ± 1155 | 24 ± 2.0 |
ID= individual identification; SE= standard error; C= individual phenotype classification; RR= resistant; Rs= resilient; SS= susceptible.
Hair samples including the follicle were taken from all animals for DNA isolation. These were then genotyped using the GeneSeek Genomic Profiler HD-V3 platform, which contains 139,376 SNP. A genotype quality control analysis was run, eliminating markers with a call rate <0.90, an allele frequency <0.05 and a Hardy-Weinberg equilibrium value of P<0.001. After quality control, 120,405 SNP were included in identification of 3,054 haplotypes. As a correction for the effect of source breed, principal component analyses (PCA) were done and included as an adjustment factor in the association analyses. The GWAS were implemented with a logistic regression model, corrected using the Bonferroni test and the false discovery rate. All genomic analyses were run with the SVS-Golden Helix ver. 8.6 software10.
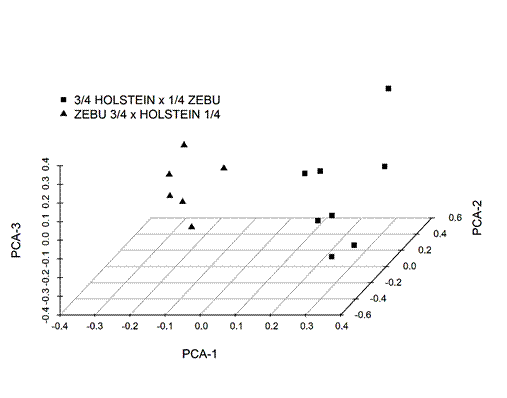
Molecular identification of the GI nematode genera showed Cooperia to be the dominant genus in the region, since no other genera were identified during the three-months follow-up. Individual infection level ranged from a minimum of 7 ± 7 to a maximum of 4,657 ± 1,886 (Table 1). The PCA showed that differences in animal breed composition could be identified (Figure 1), with those having more Zebu genes to the left and those with more Holstein genes to the right.
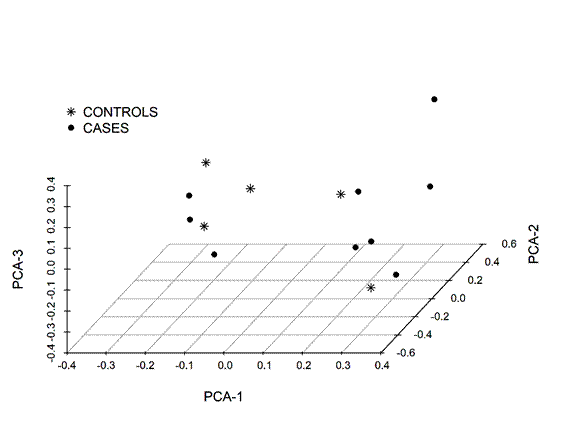
Some breeds, crosses or specific populations exhibit genetic predispositions for some traits and/or diseases11. However, no such tendencies were found to be associated with the presence of Cooperia cases and controls by crossing (Figure 2).
The GWAS identified five significant haplotypes (P≤0.001; Figure 3). Chromosome 2 includes four markers (BTA-55603-no-rs, BovineHD4100001149, BovineHD0200010866 and BovineHD0200010867); chromosome 10 includes three (BovineHD1000009272, ARS-BFGL-NGS-44351 and BovineHD10000 30584); chromosome 15 includes two (ARS-BFGL-NGS-18481 and ARS-BFGL-NGS-103542); chromosome 23 includes four (BovineHD2300011368, BovineHD2300011369, BovineHD2300011370 and BovineHD2300011371); and chromosome X includes three (BovineHD3000031659, BovineHD3000031667 and BovineHD3000031676).
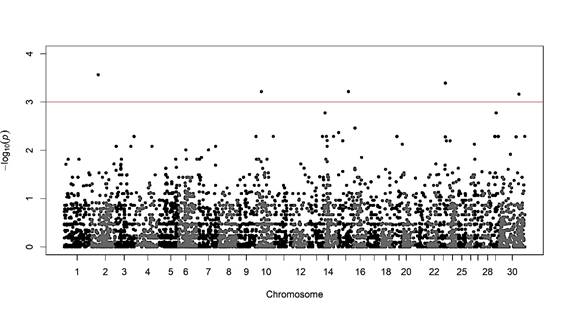
Figure 3: Manhattan graph of association analysis of haplotypes identified in Cooperia cases and controls study. Chromosome X is indicated as chromosome 30
Due to the limited number of genomic association studies to disease resistance, there are few annotations in the genome for references. Of the significant regions found in the present study for chromosomes 2, 10, 15 and X, no genes were identified that could be related to the found regions (results verified in NCBI database)12. Only the significant region of chromosome 23 has a previous report associated with the infection rate for trypanosome in N'Dama and Boran cattle in West a protozoan parasite in Africa13. Since most of the identified regions are not associated with EPG traits, nor specifically with Cooperia, they might be candidates for sequencing and testing the existence of genes for resistance to this nematode and other GI parasites.
In conclusion, despite of the low number of calves included in the present study, the results suggest existence of SNP associated with nematode resistance, mainly on chromosome 23. However, more data is required to define this relationship. Study of the host/parasite interaction implies greater knowledge of the diversity of immune evasion mechanisms, and anthelmintic detoxification in nematodes. The present contribution to understanding the factors associated with the host genome and Cooperia punctata infection processes suggests that in the future, mechanisms may be found that make feasible the use of markers associated with nematodes in cattle under grazing conditions in the tropics.
Acknowledgements
The research reported here was financed by Proyecto Fiscal INIFAP No. 16573433030. Thanks are due the CEIEGT, FMVZ-UNAM for their support.
REFERENCES
1. Borges FA, Almeida GD, Heckler RP, Lemes RT, Onizuka MKV, Borges, DGL. Anthelmintic resistance impact on tropical beef cattle productivity: effect on weight gain of weaned calves. Trop Anim Health Prod 2013;45:723‐727. [ Links ]
2. Stromberg BE, Gasbarre LC, Waite A, Bechtol DT, Brown MS, Robinson NA, Newcomb H. Cooperia punctata: Effect on cattle productivity? Vet Parasitol 2013;183(3-4):284-291. [ Links ]
3. Geary TG, Hosking BC, Skuce PJ, von Samson-Himmelstijerna G, Maeder S, Holdsworth P, Pomroy W, Vercruysse J. World Association for the Advancement of Veterinary Parasitology (W.A.A.V.P.) Guideline: Anthelmintic combination products targeting nematode infections of ruminants and horses. Vet Parasitol 2012;190:306-316. [ Links ]
4. Fritz S, Capitan A, Djari A, Rodriguez SC, Barbat A. Detection of haplotypes associated with prenatal death in dairy cattle and identification of deleterious mutations in GART, SHBG and SLC37A2. PLoS ONE 2013;8(6):e65550. [ Links ]
5. Coppieters W, Mes TH, Druet T, Farnir F, Tamma N, Schrooten C, Ploeger HW. Mapping QTL influencing gastrointestinal nematode burden in Dutch Holstein-Friesian dairy cattle. BMC Genomics 2009;10(1):96. [ Links ]
6. García E. Modificaciones al Sistema de Clasificación Climática de Köppen. 3a. ed. Instituto de Geografía. UNAM. México. D.F. 1981. [ Links ]
7. Alonso-Díaz MA, Arnaud-Ochoa RA, Becerra-Nava R, Torres-Acosta JFJ, Rodriguez-Vivas RI, Quiroz-Romero RH. Frequency of cattle farms with ivermectin resistant gastrointestinal nematodes in Veracruz, Mexico. Vet Parasitol 2015;212:439-443. [ Links ]
8. Coles GC, Bauer C, Borgsteede FHM, Geerts S, Klei TR, Taylor MA, Waller P, World Association for the Advancement of Veterinary Parasitology (W.A.A.V.P.) methods for the detection of anthelmintic resistance in nematodes of veterinary importance. Vet Parasitol 1992;44:35-44. [ Links ]
9. Encalada-Mena L, Tuyub-Solis H, Liébano-Hernández E, Ramirez-Vargas G, Mendoza-de-Gives P, Aguilar-Marcelino L, López-Arellano ME. Phenotype and genotype traits of gastrointestinal nematodes resistant to benzimidazole in infected calves from tropical regions of Campeche State, Mexico. Vet Parasitol 2014;205:246-254. [ Links ]
10. Golden Helix Inc. SNP & Variation Suite Manual Version 8.6.0 Copyright © 2000-2016 Golden Helix, Inc. 2016. [ Links ]
11. Zhang H, Wang Z, Wang S, Li H. Progress of genome wide association study in domestic animals. J Anim Sci Biotech 2012;3:26. [ Links ]
12. National Center for Biotechnology Information. The National Center for Biotechnology Information The National Center for Biotechnology Information https://www.ncbi.nml.nih.gov Accessed Jul 15, 2017. [ Links ]
13. Gelhaus A, Horstmann R, Teale A. Mapping of quantitative trait loci controlling trypanotolerance in a cross of tolerant West African N’Dama and susceptible East African Boran cattle. Proc Nat Acad Sci USA 2003;100(13):74. [ Links ]
Received: January 30, 2018; Accepted: May 08, 2018











 texto em
texto em