Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista mexicana de ciencias pecuarias
versión On-line ISSN 2448-6698versión impresa ISSN 2007-1124
Rev. mex. de cienc. pecuarias vol.7 no.4 Mérida oct./dic. 2016
Articles
Cooling at the summer onset to mitigate the heat stress in Holstein cows from the northwest Mexico
a Departamento de Ciencias Agronómicas y Veterinarias del Instituto Tecnológico de Sonora. Calle 5 de Febrero 818 Sur, Colonia Centro, Cd. Obregón, Sonora, México. C.P. 85134.
b Department of Animal Science, Colorado State University, CO. 80523. USA.
c Department of Animal Science, University of California, Davis CA. 95616. USA.
d Instituto de Ciencias Agrícolas, Universidad Autónoma de Baja California. México.
e Department of Animal Science, New Mexico State University, USA.
The objective of this study was to determine the physiological, endocrine and productive response in lactating dairy cows under several series of artificial cooling at the beginning of the summer (June to July, 2011). Fourteen multiparous Holstein cows were assigned in one of two treatments: Cooling (C) before milking (0700 and 1700 h) and additional cooling (AC) each 2 h (from 0700 to 1700h) in the holding pen of the milking parlor. Milk yield (MY), and serum levels of prolactin (PRL) and IGF1 were measured. Rectal temperature and respiratory rate were registered and included as physiological markers of heat stress. Ambient temperature and relative humidity were also collected and used to calculate temperature-humidity index (THI). Both markers of heat stress showed a significant interaction (P<0.001) between treatment and time/week, which were lower in AC treatment (-3.8 breaths per minute and -0.2 °C) during wk 6 and 7. Serum levels of PRL were higher under additional cooling (AC: 35.1 vs C: 29.4 ng/ml; P=0.004), while IGF1 serum levels showed variation only through the sampling weeks (P=0.014). The MY increased (P<0.05) in AC only during wk 6 (+3.1 kg/d) and 7 (+3.1 kg/d). It was concluded that additional cooling at the beginning of the summer reduced heat stress in dairy cows allowing a better productive performance at the end of the study because such effect relied on the variable time/week.
Key words: Cooling; IGF1; Performance; Prolactin
El objetivo de este estudio fue determinar la respuesta fisiológica, endócrina y productiva en vacas lactantes sometidas a varias series de enfriamiento artificial al inicio de verano (junio a julio de 2011). Se asignaron 14 vacas Holstein multíparas a uno de dos tratamientos: enfriamiento (E) antes de la ordeña (0700 y 1700 h) y enfriamiento adicional (EA) cada 2 h de 0700 a 1700 h en el corral de espera a la ordeña. Se midió la producción de leche (PL), niveles de prolactina (PRL) e IGF1, mientras que la temperatura rectal y frecuencia respiratoria fueron registradas y consideradas como indicadores fisiológicos de estrés por calor. La temperatura ambiental y humedad relativa también fueron registradas y utilizadas para calcular el índice de temperatura-humedad (ITH). Los indicadores de estrés calórico mostraron una interacción significativa (P<0.001) entre tratamiento y tiempo/semana, los cuales fueron menores con EA (-3.8 respiraciones por minuto y -0.2 °C) sólo durante las semanas 6 y 7. Los niveles séricos de PRL fueron mayores bajo enfriamiento adicional (EA: 35.1 vs E: 29.4 ng/ml; P=0.004), mientras que los de IGF1 solo variaron através de las semanas de muestreo (P=0.014). La PL se incrementó (P<0.05) en EA durante las semanas 6 (+3.1 kg/día) y 7 (+3.1 kg/día). Se concluye que la mayor frecuencia de baños mitigó la condición de estrés calórico al inicio de verano, permitiéndole a la vaca expresar un mejor desempeño productivo al final del estudio, ya que dicho efecto dependió de la variable tiempo/semana.
Palabras clave: Enfriamiento; IGF1; Producción; Prolactina
Introduction
In warm regions as northwest Mexico, the combination of humidity and temperature during summer exceeds thermal requirements of Holstein cattle1,2. Once the cows leave the thermoneutral zone, they activate a complex system of endocrine and metabolic adjustments, that is regulated by genes, to improve cellular functions affected by heat stress3, that finally compromises dairy cattle production due to such adjustments imply an energy consumption associated to the increase of respiration rate4.
Body temperature in cattle oscillates from 37.5 to 38.9 ºC in healthy animals under thermoneutral conditions, but in warm climates its increase is a result of the self-generated and environmental heat load5. Such hyperthermia has been negatively correlated to growth, lactation and fertility in Holstein cattle 6,7. The respiratory system is an evaporative pathway to eliminate body heat because the animal increases the respiratory rate as a regulatory mechanism of body temperature under warm climates8,9.
Prolactin and IGF1 are involved in energy metabolism, growth, fertility, lacto genesis and mammary development3,10,11. Moreover, such hormones are also related to the thermoregulatory process in ruminants, and its increases in blood levels is a sensitive response to a warm environment12,13,14.
Artificial cooling is an alternative to reduce negative effects from high temperatures on physiology and productivity of dairy cattle2,15; however, under moderate to high humidity conditions the efficiency of a high pressure cooling system (i.e., humidifiers) is limited8. Therefore, hair and skin soaking combined with forced ventilation help to reduce body heat through conductive and convective ways16,17,18.
Once that environmental humidity and temperature increase at the beginning of the summer in the Yaqui Valley, Sonora, the productive efficiency in lactating cattle decreases because the weather become more adverse19. An increase of bath frequency during the day can help to reduce some physiological indicators of heat stress to avoid a sudden reduction in Holstein cattle milk production as the weather conditions become worst during summer. Therefore, the objective of this study was to evaluate physiological, endocrine and reproductive responses in Holstein dairy cattle subjected to direct cooling at the beginning of the summer.
Material and methods
Experimental location and animals
The study was conducted at the Unidad academica de investigación en producción lechera of the Instituto Tecnologico de Sonora (ITSON), located in block 910 (N: 27°21’ O: 109°54’) of the Yaqui Valley, Sonora, Mexico, during June and July, 2011. Twenty-eight (28) multiparous Holstein cows with 150 ± 25.9 d in milk and 3.5 ± 0.35 of body condition score (BCS) were selected for this study. Cows were maintained in shaded holding pens with free access to water, and they were fed twice a day (i.e., 0800 to 0900 h and 1700 to 1800 h) with a ration made up with 48.8 % corn silage, 16.2 % alfalfa hay, and 35 % commercial energy supplement added with vitamins and minerals premix. All cows received bovine somatotropin (BST; 500 mg S.C.) each 14 d during lactation, from wk 9 postpartum until 15 d before to start the dry period.
Treatments
Cows were randomly assigned in two treatments: cooling (C) before milking (two times per day, 0700 and 1700 h) and additional cooling (AC) each 2 h (six times per day, from 0700 to 1700 h). Therefore, cows from treatments C and AC entered daily into the holding pen of the milking parlor, and they received 2 and 6 cooling periods that were equivalent to 36 and 108 min/d of cumulative cooling per treatment, respectively. Each cooling period included a sequence of three soaking cycles alternated with three ventilation cycles.
Cooling system
Each cooling cycle included 3 min of water aspersion (~ 15 L per cow/cycle) with a system made up by 16 sprayers (located at 2.73 m from the bottom) which allowed soaking the back and side of each cow, alternated with 3 min of forced ventilation with three ½ HP electric fans located at 2.73 m from the bottom. All cows received 8.5 m2 of shadow inside the holding pens.
Climatic data
Ambient temperature (AT) and relative humidity (RH) data were collected from a nearby (~500 m) and automatic climatic station. Such information was used to calculate the temperature-humidity index (THI), that estimates the level of heat stress at which the cows were exposed, using the formula7: THI= (0.8*AT)+[(RH/ 100)*(AT”14.4)]+46.4. In order to obtain accuracy climatic values during the sampling period, climatic data were registered each hour from 0900 through 1700 h, which were averaged and combined to information from the last 2 d before each sampling date4.
Physiological markers of heat stress
Respiratory rate (RR; breaths/min) and rectal temperature (RT; °C) were collected in both groups at 1200 h twice per week during the study. The RR was measured by visual counting of intercostal movements1, while RT was collected using a digital thermometer (TES-1310®) with a contact sounding line (type K; 9-cm large), that was introduced into the animal to touch the rectal mucosa20.
Dynamic of blood hormone levels
Blood samples (i.e., 8 ml) were collected weekly from the coccygeal vein using Vacutainer tubes without anticoagulant. Once the samples were collected (i.e., from 1000 to 1100), each tube was labeled and transported (5 °C) to the Reproductive Biotechnology Lab from the Department of Veterinary and Agronomic Sciences at ITSON (5 °C) and centrifuged at 2,500 rpm/30 min to obtain the blood serum. Then, serum samples were transported to the Endocrinology Lab of New Mexico State University, USA, in order to measure concentrations (ng/ml) of prolactin (PRL) and insulin-like growth factor 1 (IGF1), using radioimmunoassay (Packard Instrument Company Inc., Meriden, CT, USA) of double antibody11,12 under a coefficient of variation of 9.4 and 13.4 % for PRL and IGF1, respectively.
Milk yield
Cows were milked twice per day (0700 and 1700) and milk production (MY; kg) was recorded daily using an electronic system (Metraton 21™, GEA. Westfalia-Surge Farm Technologies, Siemensstraße, Bönen Germany).
Statistical analyses
The procedures PROC UNIVARIATE and PROC GLM (Levene’s test) were used to test normal distribution and equality of variances, respectively. Repeated and random statements from the MIXED procedure were processed using the SAS software V9.421, in order to analyze repeated measures per week of RR, RT, PRL, IGF1 and MY. The mixed model included treatment, time, treatment*time interaction, and lactation number as fixed effects, and the cow as random effect. Days in milk was included in the model as covariant, which resulted as significant source of variation (P<0.05). According to Bayesian and Akaike information criterion, the auto-regressive covariance resulted as the best fitted structure for repeated measures in the mixed model. If the term time resulted as significant source of variation (P<0.05), then a pairwise comparison of the minimum square means was executed using the PDIFF option in SAS.
Results and discussion
Climatic data
Ambient temperature showed a variation from 30.4 to 36.6 °C (Table 1). The maximum thermic threshold for Holstein cattle is 25 °C7, and thermic fluctuations from 21 to 32 °C lead to a reduction in milk yield22. The RH level fluctuated during the day from 18.1 y 32.8 % through the experiment. Effect of humidity is non-significant at 24 °C; however, an important negative impact appears when daily temperature increases above 34 °C23, and evaporative functions (i.e., panting and sweating) of thermic regulation will be limited when RH is higher than 45 %. Although in this study HR was lower, it tended to intensify early in the summer which represents the beginning of regular conditions of heat stress in this region. The average THI during the day was 79.1 units that is considered as moderate heat stress15, and it became more adverse after wk 4. Collier et al25 showed that milk yield in high producer cows starts to decline when the THI reaches 68 units.
Heat stress markers
Treatment by time interaction was significant (P<0.001) for RR and RT (Table 2), which are considered as indicators of heat stress. It was expected that an increase in the number of showers improves RR and RT; however, the effect of the treatment on these variables was only detected at the end of the study. It coincided with the beginning of the summer (wk 6 and 7); therefore, the favorable effect after to increase the number of showers depended of the variable time/week which involves the climatic variables. Berman20 tested different cooling levels at 38 °C and 47 % of humidity including until seven cycles of shower and ventilation to reduce rectal temperature in Holstein cows, but it did not improve after increase cycles from eight to ten. Spiers et al9 reported a group of cows which under thermo neutral conditions (AT of 19 °C and RH ~55 %) showed 59.6 breaths/min of RR and 39 °C of RT; however, once the cows were heat-stressed (AT of 29 °C and RH ~50 %), their RR and RT increased 32.8 and 2.5 %, respectively. In this study, RR was similar between treatments (P>0.05) during the first 5 wk (Figure 1), but it was lower in AC group in comparison to C group (P<0.05) in wk 6 (78.5 vs 82.9 breaths/ min) and 7 (69.5 vs 69.9 breaths/min), which coincided with the time when climatic conditions became more severe. Likewise, the RT in AC cows was lower (P<0.05) than C cows only in wk 6 (38.5 vs 39.0 °C) and 7 (38.6 vs 39.1 °C).
Table 2 Least squares means and probability for treatment, time and its interaction effects in Holstein cows subjected to different frequencies of cooling

RR= Respiratory rate (breaths per minute); RT= Rectal temperature (°C); PRL= Prolactin levels (ng/ml); IGF1= Insulin like growth factor-1(ng/ml); MY= Milk yield (kilograms per day).
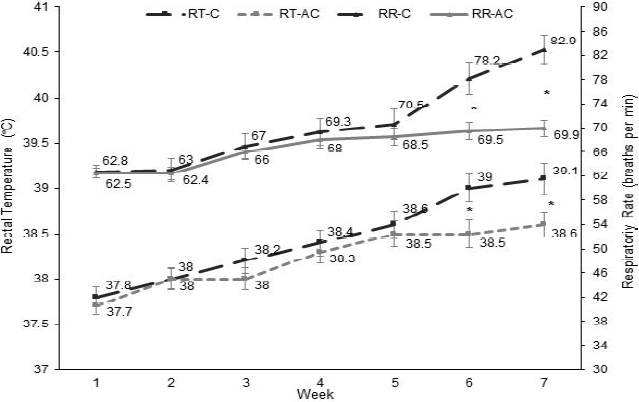
* Difference (P<0.05) of the treatment*time fixed effect. RT= Rectal temperature; RR= Respiratory rate; C= Cooling; AC= Additional cooling.
Figure 1 Respiratory rate (RR) and rectal temperature (RT) in Holstein cows subjected to different frequencies of cooling at the summer onset
During the study, cows from C group increased (P<0.001) 32.0 and 3.4 % their RR and RT, respectively, while cows from AC group only increased 11.8 and 2.3 %, respectively. Differences between treatments are lower because the two groups received cooling. However, heat stress conditions start once the cow exceeds a RT of 39.2 °C and RR of 70 breaths/min. Under these conditions, the objective of the artificial cooling is to maintain RT and RR closer to a physiological threshold. At the end of this study, both groups maintained a RT <39.2 °C, even though RR in C cows was higher than 70 breaths/min.
Dynamic of blood hormone levels
Serum levels of PRL differed by treatment (P=0.004) and sampling time (P<0.001; Table 2). During the experiment, PRL average values were higher for AC treatment (35.1 ± 1.3) in comparison with C treatment (29.4 ± 1.1 ng/ ml). In contrast, Igono et al22 reported a reduction in PRL levels from cows that received cooling and shadow, in comparison to those cows that only received shadow, and they concluded that serum PRL declined in response to heat stress reduction and the improvement of the environmental comfort of the cows. However, the higher values of PRL found in this study in cows that received additional cooling could be associated to lactation process, because the blood samples were collected in the morning after milking. Therefore, we assume that this early sampling masked the effect of heat stress on PRL levels in cows without additional cooling.
Regarding to the time of sampling, the Figure 2 depicts a gradual reduction of serum PRL (P<0.05) on wk 4 and 5, which become steady on wk 6 and 7. In contrast to such results, Scharf et al14 have reported that PRL concentrations increase in warm climates, because it is an homoeothermic hormone associated to heat stress events26,27. Likewise, PRL stimulates the expression of heat shock proteins (HSP)28, which avoid protein denaturalization in bovine body cells threatened by environmental heat stress29. The unexpected PRL response found in middle July was probably due to the short experimental time, because the negative effect of the weather in this area is more severe after July19 due to the accumulation of heat load in cows; then, activation of mechanisms involving PRL would be more evident at the end of the summer. Moreover, the covariant of days in milk could influence PRL levels at the end of the study, because this variable resulted as significant source of variation (P<0.05).
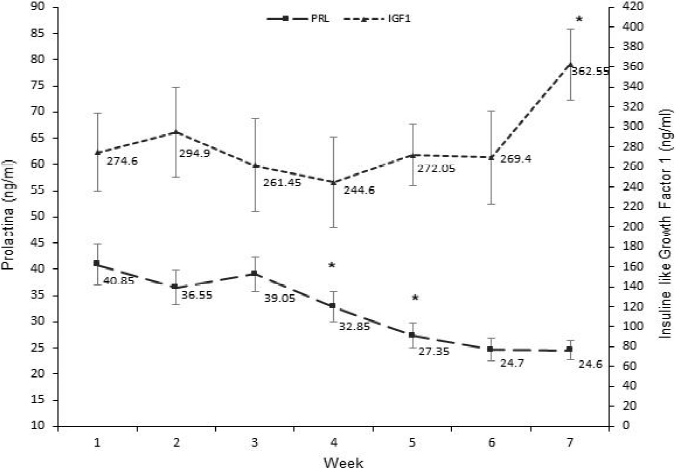
As effect of time [*] indicate statistical difference (P<0.05) in relation to the previous week on average level per week (i.e., including both treatments; ng/ml) of PRL and IGF1 during the study.
Figure 2 Dynamic of the prolactin (PRL) and insulin like growth factor-1 (IGF1) levels (ng/ml) during the study
The serum concentrations of IGF1 were only effected by time (P=0.0143; Table 2), and tended to increase at the end of the study (P<0.05; Figure 2). Rhoads et al10 have reported a small IGF1 reduction in cows exposed to warm climate conditions. Growth hormone (GH) and IGF1 have a close relationship on metabolic processes associated to milk yield11, then GH measurements would help to explain the environmental effect on IGF1 levels and its relationships with milk production. It is important to mention that cows from this study received a subcutaneous application of bovine somatotropin (STB) as part of the herd management. The STB is a synthetic hormone with similar function to GH in bovine cattle, and its exogenous application is a potential enhancer of heat load in lactating cows under heat stress13,30,31, as well as blood concentrations of IGF132-35. Therefore, administration of STB in this study could induce a direct effect on IGF1 levels, and consequently on PRL levels.
Although levels of IGF1 were constant during the study, it showed a significant increasing (P<0.05) until to reach its higher concentration on wk 7, which coincided with the hottest period of the experiment.
Growth hormone has alternative pathways to mobilize energy from other tissues in bovine cattle exposed to a high energetic demand needed for maintenance (i.e., thermoregulation) and lacto genesis in milking cows10. Metabolic regulation of GH and IGF1 allows to prioritize some emerging functions such as the adaptive response to a warm climate in transition for lactation maintenance of milking cattle. The PRL activates milk synthesis in ruminants, however, it is not essential for lactation maintenance36, while the IGF1 pathway stimulates galactophoyesis and interacts by separate with PRL and STB37. It explains the reduction in PRL levels and the increasing in IGF1 levels during the warmest period of this study.
Milk yield
The treatment by time interaction was significant (P=0.002) for milk yield (Table 2). A higher production was observed in cows from AC group in comparison with cows from C group on wk 6 (17.9 vs 21.0 kg) and 7 (18.0 vs 21.1 kg), when the heat stress represented by the ITH was higher (Figure 3). Direct cooling (shower) and forced ventilation under warm and moist climates is able to reduce negative effects from a summer heat season on cows physiology16,18, which contributes to hormonal status12 and help to correct energy metabolism13 in order to improve their productive performance1. A 9.3 % improvement in milk production has been reported after to increase cooling series from 5 to 8 in the waiting parlor area17. Low pressure cooling in this area before milking can avoid a milk reduction of 0.76 kg/d per cow, when ambient temperature fluctuates from 27 y 46 °C15.

MY= Milk yield. [*] In means of columns indicate the effect of treatment by week (P<0.05).THI= Temperature-Humidity Index.The treatments C= Cooling and AC= Additional cooling.
Figure 3 Effect of additional cooling on milk yield (MY) performance of Holstein cows
In this experiment, the reduction in MY started at the third week when the cows were exposed to an THI level of 76.6 units (Figure 3). Collier et al25 demonstrated that cows producing >35 kg/d need artificial cooling once the THI reaches 68 units, because at this threshold dairy cows reduce milk production 2.2 kg/d. Leyva et al19 found in Sonora a milk yield reduction of 0.65 kg/d per cow, once the threshold for THI reached 71.2 units.
While the negative effect of summer was approaching, higher conditions of heat stress where mitigated with the rise of cooling sessions in AC cows during the day.
This represents an advantage if we consider that all physiological and metabolic changes that the cows experienced trying to regulate body temperature by themselves could be diminished using additional cooling. Serum PRL levels and milk yield had the same trend in both treatments (Figures 2 and 3), then we assume that PRL was associated to milking instead of thermoregulation. However, both the increase in IGF1 levels and the additional cooling probably allowed the cow to mobilize energy to maintain lactation as the cows experienced a higher energy requirement to support thermoregulation the last 2 wk of this study.
Conclusions and implications
The increase of shower frequency in Holstein cows mitigated the impact of the physiological indicators of heat stress at the beginning of the summer, which was reflected in the maintenance of milk production in a summer season characterized by a gradual increase of THI levels. Serum levels of PRL and IGF1 were unexpected, which could be due to the short experimental period and the early-morning sampling time in case of PRL, and due to the application of commercial STB in case of PRL and IGF1. Milk yield increased 3.1 L per cow at wk 6 and 7, which represents an additional income of $23.25 Mexican pesos per cow. However, such profit maybe is not enough to compensate expenses due to the water supply needed for additional cooling, which would not be a problem if a water recycling system is available. Therefore, an initial study of economic implications is suggested before to start an additional cooling system. Moreover, the identification of cattle with a superior threshold for heat stress tolerance is proposed as an economic and environmental strategy to increase milk production during summer without a rise in the water supply required for additional cooling.
Literatura citada
1. Avendaño RL, Álvarez FD, Correa CA, Saucedo QJS, Robinson PH, Fadel JG. Effect of cooling Holstein cows during the dry period on postpartum performance under heat stress conditions. Liv Sci 2006;105:198-206. [ Links ]
2. Correa CA, Leyva C, Avendaño L, Rivera F, Díaz R, Álvarez FD, Ardon F, Rodríguez F. Effect of artificial cooling and its combination with timed artificial insemination on fertility of Holstein heifers during summer. J Appl Anim Res 2009;35:59-62. [ Links ]
3. Collier RJ, Collier JL, Rhoads RP and Baumgard LH. Invited review: Genes involved in the bovine heat stress response. J Dairy Sci 2008;91:445-454. [ Links ]
4. West JW, Mullinix BG, Bernard JK. Effects of hot, humid weather on milk temperature, dry matter intake, and milk yield of lactating dairy cows. J Dairy Sci 2003;86:232-242. [ Links ]
5. Finch VA. Body temperature in beef cattle: its control and relevance to production in the tropics. J Anim Sci 1986;62(2):531-542. [ Links ]
6. Dikmen SE, Cole JB, Null DJ, PJ Hansen. Heritability of rectal temperature and genetic correlations with production and reproduction traits in dairy cattle. J Dairy Sci 2011;95:3401-3405. [ Links ]
7. Hahn GL. Dynamic responses of cattle to thermal heat loads. J Anim Sci 1999;77:10-20. [ Links ]
8. Berman A. Predicted limits for evaporative cooling in heat stress relief of cattle in warm conditions. J Anim Sci 2009;87(10):3413-3417. [ Links ]
9. Spiers DE, Spain JN, Sampson JD, Rhoads RP. Use of physiological parameters to predict milk yield and feed intake in heat stressed dairy cows. J Thermal Biol 2004;29:759-764. [ Links ]
10. Rhoads ML, Rhoads RP, VanBaale MJ, Collier RJ, Sanders SR, Weber WJ, et al. Effects of heat stress and plane nutrition on lactating Holstein cows: I. Production, metabolism, and aspects of circulating somatotropin. J Dairy Sci 2009;92:1986-1997. [ Links ]
11. Rhoads ML, Kim JW, Collier RJ, Crooker B, Boisclair YR, Baumgard LH, Rhoads RP. Effects of heat stress and nutrition on lactating Holstein cows: II. Aspects of hepatic growth hormone responsiveness. J Dairy Sci 2010;93:170-179. [ Links ]
12. DoAmaral BC, Connor EE, Tao S, Hayen JM, Bubolz JW, Dahl GE. Heat stress abatement during the dry period influences prolactin signaling in lymphocytes. Dom Anim Endo 2010;38:38-45. [ Links ]
13. Wheelock JB, Rhoads RP, VanBale MJ, Sanders SR , Baumgard LH. Effect of heat stress on energetic metabolism in lactating Holstein cows. J Dairy Sci 2010;93:644-655. [ Links ]
14. Scharf B, Carroll JA, Riley DG, Chase CC, Coleman SW, Keisler DH, et al. Evaluation of physiological and blood serum differences in heat-tolerant (Romosinuano) and heat-susceptible (Angus) Bos Taurus cattle during controlled heat challenge. J Anim Sci 2010;88(7):2321-2336. [ Links ]
15. Armstrong DV. Heat stress interaction with shade and cooling. J Dairy Sci 1994;77:2044-2050. [ Links ]
16. Flamenbaum I, Wolfenson D, Mamen M, Berman A. Cooling dairy cattle by a combination of sprinkling and forced ventilation and its implementation in the shelter system. J Dairy Sci 1986;69:3140-3147. [ Links ]
17. Honig H, Miron J, Lehrer H, Jackoby S, Zachut M, Zinou A, Portnick Y, Moallem U. Performance and welfare of high-yielding dairy cows subjected to 5 or 8 cooling sessions daily under hot and humid climate. J Dairy Sci 2012;95:3736-3742. [ Links ]
18. Legrand A, Schütz KE, Tucker CB. Using water to cool cattle: Behavioral and physiological changes associated with voluntary use of cows showers. J Dairy Sci 2011;94:3376-3386. [ Links ]
19. Leyva JCC, Félix PV, Luna NP, Morales MIP, Grageda JG. Impacto de las condiciones climáticas de verano en el valle del Yaqui sobre el desempeño productivo del ganado lechero. Reunión internacional sobre producción de carne y leche en climas cálidos. Mexicali BC, México. 2008:233-237. [ Links ]
20. Berman A. Forced heat loss from body surface reduces heat flow to body surface. J Dairy Sci 2010;93:242-248. [ Links ]
21. SAS Institute Inc. 2014. SAS/STAT® 13.2 User‘s Guide. Cary, NC: SAS Institute Inc. [ Links ]
22. Igono MO, Bjotvedt G, Sanford-Crane HT. Environmental profile and critical temperature effects on milk production of Holstein cows in desert climate. Int J Biometeorol 1992;36:77-87. [ Links ]
23. Johnson HD, Vanjonack WJ. Effects of environmental and other stressors on blood hormone patterns in lactating animals. J Dairy Sci 1976;59:1603-1617. [ Links ]
24. Berman A. Extending the potential of evaporative cooling for heat-stress relief. J Dairy Sci 2006;89:3817-3825. [ Links ]
25. Collier RJ, Zimbelman RB, Rhoads RP, Rhoads ML, Baumgard LH. A Re-evaluation of the impact of temperature humidity index (THI) and black globe humidity index (BGHI) on milk production in high producing dairy cows. Western Dairy Management Conf. Reno, NV. USA. 2011:113-125. [ Links ]
26. Wettemann RP, Tucker HA, Beck TW, Meyerhoeffer DC. Influence of ambient temperature on prolactin concentrations in serum of Holstein and Brahman x Hereford heifers. J Anim Sci 1982;55:391-394. [ Links ]
27. Beede DK, Collier RJ. Potential nutritional strategies for intensively managed cattle during thermal stress. J Anim Sci 1986;62:543-554. [ Links ]
28. Peroti C, Liu R, Parusel CT, Böcher N, Schultz J, Bork P, et al. Heat shock protein-90-alpha, a prolactin-STAT5 target gene identified in breast cancer cells, is involved in apoptosis regulation. Breast Cancer Res 2008;10:6/R93. [ Links ]
29. Collier JL, Abdallah MB, Hernandez LL, Norgaard JV, Collier RJ. Prostaglandins A1 (PGA1) and E1 (PGE1) alter heat shock protein 70 (HSP-70) gene expression in bovine mammary epithelial cells (BMEC) [abstract]. J Dairy Sci 2007;90(Suppl 1):62. [ Links ]
30. Elvinger F, Natzke RP, Hansen PJ. Interaction of heat stress and bovine somatotropin affecting physiology and immunology of lactating cows. J Dairy Sci 1992;75:449-462. [ Links ]
31. Settivari RS, Spain JN, Ellersieck MR, Byatt JC, Collier RJ, Spiers DE. Relationship of thermal status to productivity in heat-stressed dairy cows given recombinant bovine somatotropine. J Dairy Sci 2007;90:1265-1280. [ Links ]
32. Gallo GF, Block E. Effects of recombinant bovine somatotropin on nutritional status and liver function of lactating dairy cows. J Dairy Sci 1990;73:3276-3286. [ Links ]
33. Bilby CR, Bader JF, Salfen BE, Youngquist RS, Murphy CN, Garverick HA, et al. Plasma GH, IGF-I and conception rate in cattle treated with low doses of recombinant bovine GH. Theriogenology 1999;51:1285-1296. [ Links ]
34. Rhoads RP, La Noce AJ, Wheelock JB, Baumgard LH. Short communication: Alterations in expression of gluconeogenic genes during heat stress and exogenous bovine somatotropin administration. J Dairy Sci 2011;94:1917-1921. [ Links ]
35. Davey HW, Xie T, McLachlan MJ, Wilkins RJ, Waxman DJ, Grattan DR. STAT5b is required for GH-induced liver IGF-1 gene expression. Endocrinology 2001;142:3836-3841. [ Links ]
36. Yang J, Zhao B, Baracos VE, Kennelly JJ. Effects of bovine somatotropin on â-casein mRNA levels in mammary tissue of lactating cows. J Dairy Sci 2005;88:2806-2812. [ Links ]
37. Miller AR, Stanisiewski EP, Erdman RA, Douglass LW, Dahl GE. Effects of long daily photoperiod and bovine somatotropin (Trobest) on milk yield in cows. J Dairy Sci 1999;82:1716-1722. [ Links ]
Received: July 14, 2015; Accepted: August 26, 2015











 texto en
texto en 



