Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Revista mexicana de ciencias agrícolas
versão impressa ISSN 2007-0934
Rev. Mex. Cienc. Agríc vol.13 no.spe28 Texcoco Set./Out. 2022 Epub 13-Jan-2023
https://doi.org/10.29312/remexca.v13i28.3279
Articles
Phosphorus and Bacillus subtilis in absorption and removal of micronutrients in Phaseolus vulgaris L.
1Facultad de Agricultura del Valle del Fuerte-Universidad Autónoma de Sinaloa. Calle 16 y Av. Japaraqui, Juan José Ríos, Ahome, Sinaloa, México. CP. 81110. Tel. 687 1387525. (chuyitaruelas@favf.mx).
2Hidrociencias-Colegio de Postgraduados. Carretera México-Texcoco km 36.5, Montecillo, Texcoco, México. CP. 56230. Tel. 55 13528662. (mendoza.candido@colpos.mx).
3Instituto de Ciencias Agrícolas-Universidad Autónoma de Baja California. Carretera a Delta s/n, ejido Nuevo León, Mexicali, Baja California, México. CP. 21705. Tel. 653 1160600. (fidel.nunez@uabc.edu.mx).
4Tecnológico Nacional de México-Campus Instituto Tecnológico de Torreón. Antigua carretera Torreón-San Pedro km 7.5, Torreón, Coahuila, México. (janethrocha17@gmail.com).
Common bean (Phaseolus vulgaris L.) is a very important legume that is part of the diet in Latin America and other countries. In northern Sinaloa, Mexico, the yield of this crop is mainly affected by management practices (irrigation and fertilization dose) and variability in climate. An experiment was established in the Fuerte Valley, north of Sinaloa, Mexico, with the purpose of researching the response of bean culture to different doses of phosphorus [(P) (0, 25, 50, 100 kg ha-1 P2O5)] and the influence of the Bacillus subtilis Q11 (Bs) strain on the absorption and removal of micronutrients. The experiment consisted of plots divided into randomized complete blocks with three repetitions. According to the results obtained, the doses of P significantly influenced the absorption of micronutrients in the following preferential order Fe> Mn> Zn> B> Cu. While inoculation with Bs increased the absorption process with respect to non-inoculated plants.
Keywords: extraction; fertilization; grow; inoculant
El frijol común (Phaseolus vulgaris L.) es una leguminosa muy importante que constituye parte de la alimentación en Latinoamérica y otros países. En el norte de Sinaloa, México, el rendimiento de este cultivo es afectado principalmente por prácticas de manejo (riego y dosis fertilización) y variabilidad en el clima. Se estableció un experimento en el valle del Fuerte, al norte de Sinaloa, México, con el propósito de investigar la respuesta del cultivo de frijol a diferentes dosis de fósforo [(P) (0, 25, 50, 100 kg ha-1 P2O5)] y la influencia de la cepa Bacillus subtilis Q11 (Bs) en la absorción y remoción de micronutrientes. El experimento consistió en parcelas divididas en bloques completos al azar con tres repeticiones. De acuerdo con los resultados obtenidos, las dosis de P influenciaron significativamente la absorción de micronutrientes en el siguiente orden preferencial Fe> Mn> Zn> B> Cu. Mientras que la inoculación con Bs incrementó el proceso de absorción con respecto a las plantas no inoculadas.
Palabras clave: cultivar; extracción; fertilización; inoculante.
Introduction
Common bean (Phaseolus vulgaris L.) is a very important legume cultivated worldwide and represents a food source rich in iron (Fe) and zinc (Zn) (Lima et al., 2016). The main restriction of yield potential is affected by biotic (soil pathogens) and abiotic factors such as low fertility (Amanullah et al., 2012; Iqbal et al., 2015), water deficit, temperature variability, inappropriate fertilization (Mweetwa et al., 2016) and interaction with chemical fertilizers (Hidayatullah et al., 2013).
Phosphorus (P) is the most limiting mineral despite the large amounts present in most soils (Khan et al., 2009). Its great ability to fix with the mineral fraction of the soil makes it not available for plant absorption. In recent years, the application of new technologies such as the use of inoculants based on plant growth-promoting rhizobacteria (PGPR), Pseudomonas, Azospirillum, Azotobacter, Klebsiella, Enterobacter, Alcaligenes, Arthrobacter, Burkholderia, Bacillus and Serratia is considered a vital component to increase nutrient availability (Ahemad et al., 2009), water absorption and natural fertility (Stajkovic et al., 2011).
According to several studies, rhizobacteria promote plant growth through different mechanisms that include the synthesis of substances (biological nitrogen fixation and phytohormone production), influence the availability of nutrients (production of low molecular weight organic acids and siderophores), induce resistance to stress events (drought, salinity, excess water, extreme temperatures) as well as the suppression of pathogens (García-Fraile et al., 2015).
Some works have shown that certain strains of Rhizobium increase the availability of Fe, Zn and P due to the secretion of low molecular weight iron chelators (Sridevi et al., 2007). Likewise, Rengel et al. (1999) found that inoculation with Rhizobium in legumes had a positive effect on plant growth and micronutrient concentration in parts of the plant. Reports with different rhizobacteria argue that their establishment and behavior are affected by environmental conditions (Ahemad and Kibret et al., 2014).
Some other reports specifically with B. subtilis mention that changes in temperature and nutrient scarcity limit their survival in soils (Qiao et al., 2017), which represent a key factor for PGPR-based inoculants to be effective (Compant et al., 2010). Finally, Fageria (2002) reported that all nutrient interactions in plants can be positive, negative or neutral, so they must be measured by plant growth and nutrient concentration in tissues.
Currently, bean fertilization in Sinaloa, Mexico, is mainly based on nitrogen applications, while phosphorus is applied without a specific dose to meet the demand of the crop. The objectives of this study were to evaluate different doses of phosphorous fertilizer and the behavior of Bacillus subtilis Q11 in the absorption of micronutrients and their concentration in seed.
Materials and methods
Description of the experiment
The work was carried out during the 2018 autumn-winter agricultural cycle in northern Sinaloa, Mexico (25° 45’ 49” north latitude, 108° 51’ 41” west longitude). The soil of the region has a clayey-loamy texture (50% clay, 30% silt and 20% sand), low organic matter content (<1%), bulk density of 1.15 g cm-3 and a volumetric moisture content of 0.155 cm3 cm-3. The sowing and tillage techniques used were those recommended by the technical guide of INIFAP (SIAP, 2013). Before fertilization, a soil sampling was carried out at a depth of 30 cm to determine physical and chemical characteristics. The sowing was carried out in wet soil and pest management was successfully controlled throughout the cycle.
Treatments
The treatments consisted of four doses of phosphorus (P2O5) (T1: 0 kg ha-1, T2: 25 kg ha-1, T3: 50 kg ha-1, T4: 100 kg ha-1) with and without Bacillus subtilis. The inoculation was performed by impregnating 5 ml (2x108 CFU ml-1) of the Bacillus subtilis Q11® strain per each kilogram of Azufrado Higuera seed. The dose of nitrogen (150 kg ha-1) and phosphorus was applied at pre-sowing using the Blaukorn® Classic source (12-8-16). For irrigation programming, the Irrimodel platform (Sifuentes, 2012) was used, which estimates the levels of soil moisture abatement in the root zone. Additionally, an irrigation criterion (IC) of 50% of the usable moisture was established and the moisture content was verified using TDR 300 (Time Domain Reflectometry).
Experimental design
The experiment consisted of plots divided into randomized complete blocks with three repetitions. The main plot consisted of the doses of phosphorus and its dimension was 64 m2, the sub-plots consisted of the inoculation of Bacillus subtilis Q11 and the control whose dimension was 32 m2 (four furrows arranged linearly within the main plot).
Micronutrient absorption
To estimate the nutrient absorption of the crop, vegetative analyses (destructive samplings) were carried out in different phenological stages (third trifoliate leaf, flowering, pod filling and physiological maturity). The sampling method consisted of the removal of the aerial biomass in a location (1 m) in each plot. Subsequently, the samples were taken to the laboratory where all the structures were separated (leaves, stems, pods) and subjected to drying (70 °C) in a forced air furnace for 72 h until reaching constant weight.
The concentration of micronutrients was determined with the filtered material that was obtained from wet digestion for phosphorus and then aliquots were placed for reading in a spectrophotometer following the protocol described in the Official Mexican Standard (NOM-021-RECNAT-2000). The absorption of micronutrients was estimated by multiplying the dry weight by its concentration (kg ha-1) (equation 1).
1).
DM (kg ha-1) represents dry matter in different phenological stages. The micronutrients in the seed were estimated with the dry weight of the grain multiplied by its concentration as indicated in equation 2.
2).
Data analysis
Data on nutritional absorption and seed concentration were subject to an appropriate analysis of variance (Anova) (Minitab, 2017). The mean difference in each treatment was performed using the Fisher’s LSD test (p≤ 0.05).
Results and discussion
Micronutrient absorption
No interaction effect was found between the evaluated factors, therefore all results are presented separately. The absorption pattern showed that all micronutrients gradually increased from flowering, absorption was maximized in pod filling and decreased at the end of the cycle.
Westermann et al. (2011) mentioned that the highest absorption occurred in a production system without input restrictions, which coincides with the results of this work, where plants under the doses of 50 and 100 kg of P exhibited the highest absorption range. The preference in total absorption was in the following order: Fe> Mn> Zn> B> Cu with 16, 4.3, 2.8, 1.2 and 0.54 kg ha-1. These data are consistent with what was reported by Chaudhary et al. (2008) and Bender et al. (2015), who mention the effect of P and the absorption of micronutrients in soybean (Glycine max L.). Iron absorption increased in response to P fertilization throughout the crop cycle (Figure 1A).
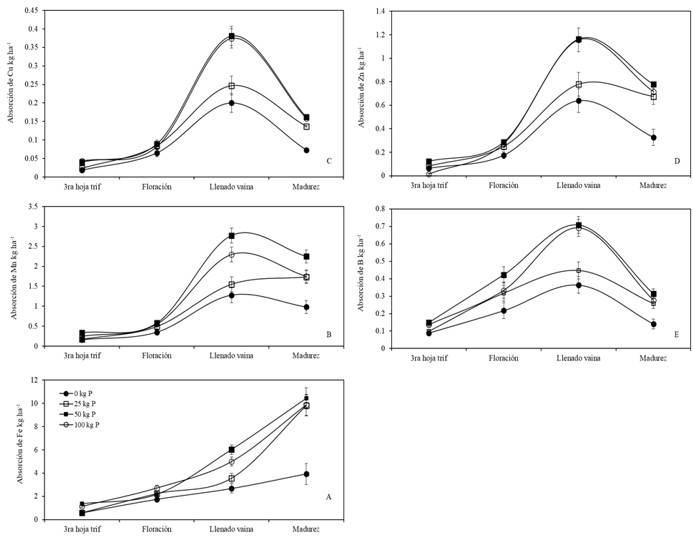
Figure 1 Absorption of micronutrients in phenological stages in bean because of the dose of phosphorus.
This absorption process was higher in the treatments under the doses of 50 kg and 100 kg of P (10.5 and 9.8 kg ha-1) respectively. Works by Tofiño-Rivera et al. (2016) mention that fertilization with P could increase the concentration of Fe, but not that of Zn in plant tissues or grains. It is worth mentioning that in many cases immobilization processes rather than scarcity in the soil affect the supply of Fe for plants. Inoculated plants increased 12.5% in the absorption of Fe over non-inoculated plants (Figure 2A).
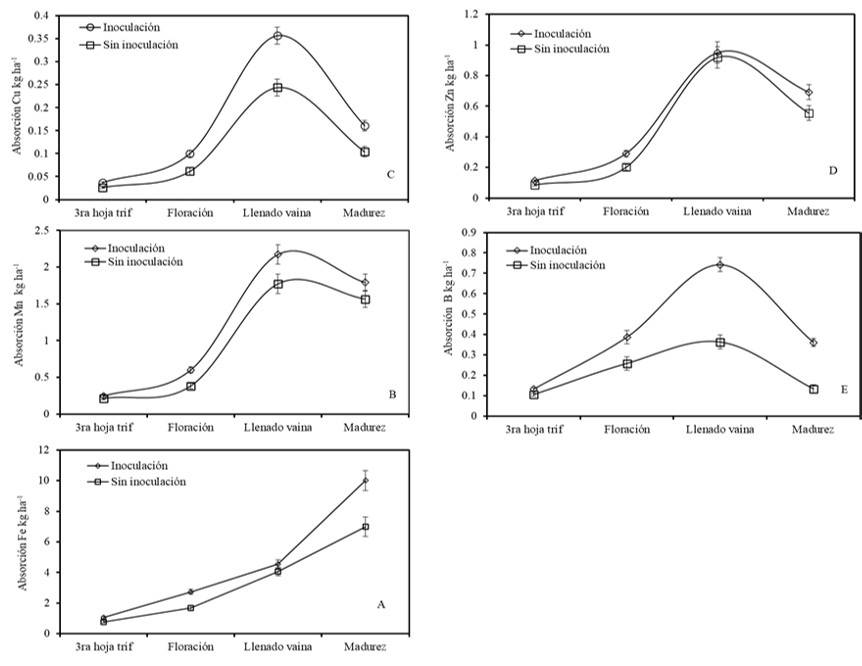
Figure 2 Absorption of micronutrients in phenological stages in bean due to the effect of Bacillus subtilis.
In that sense, Dehner et al. (2010) showed that inoculation with rhizobacteria increased the formation of siderophores (low molecular weight chelators) thus increasing the absorption of Fe in different cultures, since these act as solubilizers in organic and mineral compounds under limiting conditions of Fe (Indiragandhi et al., 2008).
It is also mentioned that siderophores not only complex Fe, but also form stable complexes with other metals such as Al, Cd, Cu, Zn and Pb (Neubauer et al., 2000). The absorption of manganese (Mn) was also increased by the supply of P during the crop cycle.
Plants under doses of 50 kg of P had higher absorption (2.3 kg ha-1), while plants under doses of 100 kg of P decreased this absorption at maturity (1.7 kg ha-1) (Figure 1B). As seen in the absorption pattern, Cu extraction was strongly influenced by P doses and increased consistently throughout the cycle. Likewise, the plants treated with the doses of 50 and 100 kg of P had the highest absorption range (0.38 kg ha-1), which declined in the stage of maturity (0.16 kg ha-1) (Figure 1C). The inoculation with Bs increased Cu absorption by 20% (Figure 2C).
The total absorption of Zn was lower than that of Mn (0.7 kg ha-1) (Figure 1D). According to Domínguez-Vivancos (1997), this process occurs due to high levels of P, which lead to physiological inhibition from the roots to the aerial parts. Findings reported by Astudillo and Blair (2008); Joshi et al. (2010); Velu et al. (2014) mention that when the content of P in the plant increases, that of Zn decreases. Inoculated treatments increased 5% in the absorption of Zn compared to non-inoculated treatments (Figure 2D). The total absorption of boron (B) was 0.3 kg ha-1, which was slightly lower in plants under doses of 100 kg of P.
It is mentioned that the boron absorption process is reduced in soils with good drainage and low moisture, as well as dry climate conditions (Havlin et al., 2005). In addition, the highest absorption (0.7 kg ha-1) occurred in plants with high P content in the pod filling stage (Figure 1E). On the other hand, the treatments inoculated with Bs substantially increased the absorption process compared to non-inoculated plants (Figure 2). Inoculated plants increased the absorption of Mn, Cu and B by 12, 25 and 35% compared to non-inoculated plants (Figure 2B, C and E).
Research conducted by Armada et al. (2014) showed that the absorption of micronutrients such as Zn 2+ , Mn2+ and Cu2+ increased by applying Bacillus megaterium, Bacillus thuringiensis and Bacillus spp., in lavender plants (Lavandula angustifolia L.) under water stress and in sage plants (Salvia divinorum L.). The results suggest that the stimulating effect of plant growth-promoting rhizobacteria on the absorption process can be attributed to their activity on nutrient solubilization (Ndakidemi et al., 2011). The effect of Bs could be related to the production of organic acids that bind metals such as Fe, Zn, Cu and Mn, which at the same time increase the availability for plant absorption (Havlin et al., 2005). Some authors have reported that individual application of inoculants to common bean has promoted greater nutritional absorption and accumulation in plant tissues.
Nutrient concentration in grain
Nutrient removal is a very important parameter in terms of quality for human nutrition. This is determined by the protein and mineral content present (Mune et al., 2013). According to the mineral content in bean, it has fluctuated depending on the genotype, crop management and storage conditions. When considering the concentration of micronutrients in bean grains, this concentration was higher for all treatments (application of P and treatments inoculated with Bs) compared to what was reported by Delfini et al. (2020). They found a maximum concentration of 115, 23, 17 and 47 mg kg-1 of Fe, Mn, Cu and Zn in 1512 accessions of common bean.
In this study, the data showed that there was no interaction between the factors evaluated (P × Bs) (Table 1). Except for Fe (p< 0.005), the rest of the micronutrients were not affected by the doses of P (p≥ 0.42). The maximum concentration of Fe was found in the treatments under the doses of 50 kg ha-1 P, and the lowest concentration was observed in the treatments that were not applied P. The contents of Mn, Cu, Zn and B in the grain were in the order of 139, 93, 315 and 114 mg kg-1. Astudillo and Blair (2008) showed that doses of P significantly increased concentrations of Fe and Zn in the grain.
One possible reason that may explain a high absorption of micronutrients is related to the production of nodules promoted by phosphorous fertilization, as mentioned by Amare et al. (2014); Shanka et al. (2018). Inoculation with PGPR has been associated with increasing micronutrient bio-fortification in common bean grains (Talaat et al., 2015; Jalal et al., 2021). Except for Zn, inoculation with Bs increased the concentration of micronutrients (Table 1).
Table 1 Nutrient content in bean seed (Phaseolus vulgaris L.).
| P2O5 (kg ha-1) | Fe | Mn | Cu | Zn | B |
| (mg kg-1) | |||||
| 0 | 300 ±43.4 b | 133 ±46.3 a | 87 ±21.6 a | 300 ±33.7 a | 108 ±24.1 a |
| 25 | 408 ±77.6 ab | 136 ±46.8 a | 97 ±19.6 a | 330 ±30.3 a | 108 ±31 a |
| 50 | 420 ±100.6 a | 150 ±59.7 a | 97 ±18.6 a | 333 ±20.6 | 128 ±33.7 a |
| 100 | 360 ±142 ab | 136 ±46.3 a | 92 ±23 a | 301 ±71.4 | 113 ±25.8 a |
| Probability | 0.005 | 0.81 | 0.71 | 0.42 | 0.43 |
| Without inoculation | 305 ±53.4 b | 112 ±36.7 b | 80 ±16.4 b | 311 ±29.7 a | 98 ±21.8 b |
| With inoculation | 440 ±34.7 a | 166 ±39.3 a | 105 ±17.9 a | 325 ±32.6 a | 131 ±24.6 a |
| Probability | 0.001 | 0.002 | 0.004 | 0.45 | 0.005 |
| P × Bs probability | 0.051 | 0.3 | 0.84 | 0.24 | 0.904 |
Values with different letters are significantly different. Least significant difference, Fisher’s test (p≤ 0.05), ± standard deviation.
The increase in micronutrients in inoculated plants compared to non-inoculated plants was in the order of 140, 54, 25, 33 mg kg-1, respectively for Fe, Mn, Cu and B. Although no effects were observed due to the inoculation of Bs on the concentration of Zn in grain, the values were even higher than those presented by Delfini et al. (2020). In some other crops such as wheat, the use of Bs has significantly increased the concentration of Zn in grain (68%) (Mumtaz et al., 2018).
Conclusions
The results of this study reveal that the application of phosphorous fertilizer in combination with Bacillus subtilis significantly increased the absorption and removal of micronutrients. The growth region between pod filling before the end of physiological maturity represented the maximum period for nutrient acquisition. The higher concentration of Zn and Fe in grain could represent a specific trait in future bio-fortification processes in common bean varieties.
Literatura citada
Ahemad, M.; Khan. M. S.; Zaidi, A. and Wani, P. A. 2009. Remediation of herbicides contaminated soil-using microbes. Microbes in Sustainable Agriculture. 261(5):1-84. [ Links ]
Ahemad, M. and Kibret, M. 2014. Mechanisms and applications of plant growth promoting rhizobacteria: Current perspective. J. King Saud University Science. 26(1):1-20. [ Links ]
Amare, G.; Assaye, D. and Tuma, A. 2014. The response of haricot bean varieties to different rates of phosphorus at Arba-Minch, Southern Ethiopia. ARPN J. Agric. Biol. Sci. 9(10):344-350. [ Links ]
Amanullah, M. A.; Almas, L. K.; Amanullah, J. Z. S.; Hidayatur, R. and Shad, K. K. 2012. Agronomic efficiency and profitability of fertilizers applied at different planting densities of maize in Northwest Pakistan. J. Plant Nutr. 35(3):331-341. [ Links ]
Armada, E.; Roldan, A. and Azcon, R. 2014. Differential activity of autochthonous bacteria in controlling drought stress in native lavandula and salvia plants species under drought conditions in natural arid soil. Microbial Ecol. 67(2):410-420. https://doi: 10.1007/s00248-013-0326-9. [ Links ]
Astudillo, C. y Blair, M. 2008. Contenido de hierro y cinc en la semilla y su respuesta al nivel de fertilización con fósforo en 40 variedades de frijol colombianas. Agron. Colomb. 26(3):471-476. [ Links ]
Bender, R. R.; Haegele, J. W. and Below, F. E. 2015. Nutrient uptake, partitioning and remobilization in modern soybean varieties. Agron. J. 107(2):563-573. https://doi.org/10.2134/agronj14.0435. [ Links ]
Compant, S.; Clément, C. and Sessitsch, A. 2010. Plant growth-promoting bacteria in the rhizo-and endosphere of plants: their role, colonization, mechanisms involved and prospects for utilization. Soil Biol. Biochem. 42(5):669-678. https://doi.org/10.1016/j.soilbio.2009.11.024. [ Links ]
Chaudhary, M. I.; Adu-Gyamfi, J. J.; Saneoka, H.; Nguyen, N. T.; Suwa, R.; Kanai, S.; El-Shemy, H. A.; Lightfoot, D. A. and Fujita, K. 2008. The effect of phosphorus deficiency on nutrient uptake, nitrogen fixation and photosynthetic rate in mashbean, mungbean and soybean. Acta Physiologiae Plantarum. 30(4):537-544. https://doi.org/10.1007/s11738-008-0152-8. [ Links ]
Dehner, C. A.; Awaya, J. D.; Maurice, P. A. and DuBois, J. L. 2010. Roles of siderophores, oxalate, and ascorbate in mobilization of iron from hematite by the aerobic bacterium Pseudomonas mendocina. Apply Environ. Microbiol. 76(7):2041-2048. https://doi.org/10.1128/aem.02349-09. [ Links ]
Delfini, J.; Moda-Cirino, V.; dos Santos, N. J.; Buratto, J. S.; Ruas, P. M. and Azeredo, G. L. S. 2020. Diversity of nutritional content in seeds of Brazilian common bean germplasm. PLoS ONE. 15(9):1-13. https://doi.org/10.1371/journal.pone.0239263. [ Links ]
Domínguez, V. A. 1997. Tratado de fertilización. 3ra . Ed. Ediciones Mundi-Prensa. 143 p. [ Links ]
Fageria, N. K.; Baligar, V. C. and Clark, R. B. 2002. Micronutrients in crop production. Advances in Agronomy. 77(1):189-272. https://doi.org/10.1016/S0065-2113(02)77015-6. [ Links ]
García, F. P.; Menéndez, E. and Rivas, R. 2015. Role of bacterial biofertilizers in agriculture and forestry. Bioengineering. 2(3):183-205. doi: 10.3934/bioeng.2015.3.183. [ Links ]
Havlin, J. L.; Beaton, J. D.; Tisdale, S. L. and Nelson, W. L. 2005. Soil Fertility and Fertilizers. An Introduction to Nutrient Management, 7th Ed. Pearson Prentice Hall, Upper Saddle River, NJ. 244-289 pp. [ Links ]
Hidayatullah, A.; Amanullah, A. and Shah, Z. 2013. Residual effect of organic nitrogen sources applied to rice on the subsequent wheat crop. Inter. J. Agron. Plant Produc. 4(4):620-631. [ Links ]
Indiragandhi, P.; Anandham, R.; Madhaiyan, M. and Sa, T. M. 2008. Characterization of plant growth-promoting traits of bacteria isolated from larval guts of diamondback moth Plutella xylostella (Lepidoptera: Plutellidae). Current Microbiology. 56(1):327-333. https://doi.org/ 10.1007/s00284-007-9086-4. [ Links ]
Iqbal, A.; Amanullah, A. and Iqbal, M. 2015. Impact of potassium rates and their application time on dry matter partitioning, biomass and harvest index of maize (Zea mays) with and without cattle dung application. Emirate’s J. Food Agric. 27(5):447-453. https://doi.org/10.9755/ ejfa.2015.04.042. [ Links ]
Jalal, A.; Galindo, F. S.; Boleta, E. H. M.; Oliveira, C. E.; Reis, A. R. D.; Nogueira, T. A. R.; Moretti, N. M. J.; Mortinho, E. S.; Fernandes, G. C. and Teixeira, F. M. C. M. 2021. Common Bean Yield and Zinc Use Efficiency in Association with Diazotrophic Bacteria Co-Inoculations. Agronomy. 11(5):959-979. https://doi.org/10.3390/agronomy11050959. [ Links ]
Joshi, A. K.; Crossa, J.; Arun, B.; Chand, R.; Trethowan, R.; Vargas, M. and Ortiz, M. I. 2010. Genotype × Environment interaction for zinc and iron concentration of wheat grain in eastern Gangetic plains of India. Field Crops Research. 116(3):268-277. https://doi.org/10.1016/j.fcr.2010.01.004. [ Links ]
Khan, M. S.; Zaidi, A.; Wani, P. A. and Oves, O. 2009. Role of plant growth promoting rhizobacteria in the remediation of metal contaminated soils. Environ. Chem. Letters. 7(1):1-19. https://doi.org/10.1007/s10311-008-0155-0. [ Links ]
Lima, R. T.; Nascimento, V. and Andreani, J. R. 2016. Densidade de plantas e fontes de nitrogênio no cultivo de feijoeiro. Brazilian J. Biosys. Eng. 10(3):327-338. https://doi.org/10.18011/bioeng2016v10n3p327-338 . [ Links ]
Minitab. 2017. Statistical software. PA: Minitab, Inc. [ Links ]
Mune, M. A.; Minkaa, M. R. and Mbombeb, I. L. 2013. Chemical composition and nutritional evaluation of a cowpea protein concentrate. Global Adv. Res. J. Food Sci. Technol. 2(3):35-43. [ Links ]
Mweetwa, A. M.; Chilombo, G. and Gondwe, B. M. 2016. Nodulation, nutrient uptake and yield of common bean inoculated with Rhizobia and Trichoderma in an acid soil. J. Agric. Sci. 8(12):61-70. https://doi.org/10.5539/jas.v8n12p61. [ Links ]
Mumtaz, M. Z.; Ahmad, M.; Jamil, M.; Asad, S. A. and Hafeez, F. 2018. Bacillus strains as potential alternate for zinc biofortification of maize grains. Inter. J. Agric. Biol. 20(8):1779-1786. https://doi.org/10.17957/ijab/15.0690. [ Links ]
Neubauer, U.; Furrer, G.; Kayser, A. and Schulin, R. 2000. Siderophores, NTA, and citrate: potential soil amendments to enhance heavy metal mobility in phytoremediation. Inter. J. Phytoremediation. 2(4):353-368. https://doi.org/10.1080/15226510008500044. [ Links ]
NOM-021-RECNAT. 2000. Norma Oficial Mexicana. Aprobada por el comité consultivo nacional de normalización para la conservación, protección, restauración y aprovechamiento de los recursos forestales de suelos y costas. 227 p. [ Links ]
Ndakidemi, P. A.; Bambara, S. and Makoi, J. 2011. Micronutrient uptake in common bean (Phaseolus vulgaris L.) as affected by Rhizobium inoculation, and the supply of molybdenum and lime. Plant Omics J. 4(1):40-52. https://doi.org/abs/10.3316. [ Links ]
Qiao, J.; Xiang, Y.; Xuejie, L.; Yongfeng, L.; Borriss, B. and Liu, L. 2017. Addition of plant-growth-promoting Bacillus subtilis PTS-394 on tomato rhizosphere has no durable impact on composition of root microbiome. BMC Microbiology. 17(1):1-12. https://doi.org/10.1186/s12866-017-1039-x. [ Links ]
Rengel, Z.; Batten, G. D. and Crowley, D. E. 1999. Agronomic approaches for improving the micronutrient density in edible portion of field crops. Field Crops Res. 60(1-2):27-40. https://doi.org/10.1016/S0378-4290(98)00131-2. [ Links ]
SIAP. 2013. Servicio de Información Agroalimentaria y Pesquera. www. siap.gob.mx. [ Links ]
Shanka, D.; Dechassa, N.; Gebeyehu, S. and Elias, E. 2018. Dry matter yield and nodulation of common bean as influenced by phosphorus, lime and compost application at southern Ethiopia. Open Agriculture. 3(1):500-509. https://doi.org/10.1515/opag-2018-0055. [ Links ]
Sifuentes, I. E.; Macías, C. J.; Quintana, J. and González, C. V. 2012. IrriModel 1.0: Programación integral y gestión del riego a través de internet. Folleto técnico. INIFAP-CIRNO-CEVAF. 52 p. [ Links ]
Sridevi, M.; Mallaiah, K. V. and Yadav, N. C. S. 2007. Phosphate solubilization by Rhizobium isolates from Crotalaria species. Journal of Plant Science. 2(6):635-639. [ Links ]
Stajkovic, O.; Delic, D.; Josic, D.; Kuzmanovic, D.; Rasulic, N. and Vukcevic, J. K. 2011. Improvement of common bean growth by co-inoculation with Rhizobium and plant growth-promoting bacteria. Romanian Biotechnological Letters. 16(1):5919-5926. [ Links ]
Talaat, N. B.; Ghoniem, A. E.; Abdelhamid, M. T. and Bahaa, T. S. 2015. Effective microorganisms improve growth performance, alter nutrients acquisition and induce compatible solutes accumulation in common bean (Phaseolus vulgaris L.) plants subjected to salinity stress. Plant Growth Regul. 75(1):281-295. https://doi.org/10.1007/s10725-014-9952-6 [ Links ]
Tofiño, R. A. P.; Pastrana, V. I. J.; Melo, R. A. E.; Beebe, S. y Tofiño, R. R. 2016. Rendimiento, estabilidad fenotípica y contenido de micronutrientes de genotipos de fríjol biofortificado en el Caribe seco colombiano. Corpoica. Ciencia Tecnología Agropecuaria. 17(3):309-329. https://doi.org/10.21930/rcta.vol17-num3-art:511. [ Links ]
Velu, G.; Ortiz, M. I.; Cakmak, I.; Hao, Y. and Singh, R. P. 2014. Bio-fortification strategies to increase grain zinc and iron concentrations in wheat. J. Cereal Sci. 59(3):365-372. doi.org/10.1016/j.jcs.2013.09.001. [ Links ]
Westermann, D. T.; Teran, H. X.; Muñoz, P. C. G. and Singh, S. P. 2011. Plant and seed nutrient uptake in common bean in seven organic and conventional production systems. Canadian J. Plant Sci. 91(6):1089-1099. https://doi.org/10.4141/cjps10114. [ Links ]
Received: June 01, 2022; Accepted: August 01, 2022











 texto em
texto em 


