Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista mexicana de ciencias agrícolas
versión impresa ISSN 2007-0934
Rev. Mex. Cienc. Agríc vol.9 spe 20 Texcoco abr./may. 2018
https://doi.org/10.29312/remexca.v0i20.991
Articles
Effect of elicitors of natural origin on tomato plants subjected to biotic stress
1Antonio Narro Autonomous Agrarian University-Department of Horticulture
2Food Department. Calzada Antonio Narro # 1923, Saltillo, Coahuila, Mexico. CP. 25315. Tel. 01 (844) 4110200, ext. 2303. (emlaugaren@gmail.com; abenmen@gmail.com; armando.robledo@outlook).
3Arysta LifeScience of Mexico. Boulevard Jesus Valdés Sánchez # 2369, Fracc. Europe, Saltillo, Coahuila, Mexico. CP. 25290. Tel. 01 (844) 4380500, ext. 6703 (susana.solis@arysta.com).
4CONACYT-Antonio Narro Autonomous Agrarian University. Calzada Antonio Narro # 1923, Saltillo, Coahuila, Mexico. CP. 25315. Tel. 01(844) 4110200, ext. 2303.
The elicitors are substances from various inorganic and organic sources that can induce physiological effects and changes such as the activation of defensive responses and the accumulation of phytoalexins in the organism to which they are applied, therefore, the objective of this investigation was to know the effect of the application of two elicitors of natural origin on the vigor of plants and the quality of tomato fruits in conditions of biotic stress caused by Fusarium oxysporum f. sp. lycopersici (FOL). Elicitors of natural origin (E1 and E2) were applied at 7, 15 and 56 ddt (days after transplantation); FOL inoculation was performed at 19 ddt, using a concentration of 1 × 106 spores mL-1. The vigor in the plants consisted of measuring: the incidence and severity of the disease, SPAD units, stomatal conductance during the development of the plants, height, total dry weight and yield; as well as the pH, soluble solids, firmness and vitamin C content in the fruits. The application of the elicitors decreased the severity of the symptoms of the disease. The elicitor E2 increased the height of the treated plants, the accumulation of biomass and avoided the reduction in yield, also decreased the content of soluble solids in the fruits. It is concluded that the application of elicitors of natural origin from plant extracts had a positive effect on the vigor of the tomato plants inoculated with FOL by increasing the height and accumulation of total biomass, in addition to that some quality variables were modified of the fruit.
Keywords: Fusarium oxysporum f. sp. lycopersici; fruit quality; systemic resistance in plants
Los elicitores son sustancias de diversas fuentes tanto inorgánicos como orgánicos que pueden inducir efectos fisiológicos y cambios como la activación de respuestas defensivas y la acumulación de fitoalexinas en el organismo al cual son aplicados, por lo cual, el objetivo de esta investigación fue conocer el efecto de la aplicación de dos elicitores de origen natural sobre el vigor de plantas y la calidad de frutos de tomate en condiciones de estrés biótico causado por Fusarium oxysporum f. sp. lycopersici (FOL). Los elicitores de origen natural (E1 y E2) se aplicaron a los 7, 15 y 56 ddt (días después del trasplante); la inoculación del FOL se realizó a los 19 ddt, usando una concentración de 1×106 esporas mL-1. El vigor en las plantas consistió en medir: la incidencia y severidad de la enfermedad, unidades SPAD, conductancia estomática durante el desarrollo de las plantas, altura, peso seco total y rendimiento; así como el pH, sólidos solubles, firmeza y el contenido de vitamina C en los frutos. La aplicación de los elicitores disminuyó la severidad de los síntomas de la enfermedad. El elicitor E2 incrementó la altura de las plantas tratadas, la acumulación de biomasa y evitó la reducción en el rendimiento, también disminuyo el contenido de sólidos solubles en los frutos. Se concluye que la aplicación de elicitores de origen natural provenientes de extractos de plantas tuvo un efecto positivo sobre el vigor de las plantas de tomate inoculadas con FOL al incrementar la altura y la acumulación de biomasa total, además de que se modificaron algunas variables de calidad del fruto.
Palabras clave: Fusarium oxysporum f. sp. lycopersici; calidad de fruto; resistencia sistémica en plantas
Introduction
Plants are organisms that have evolved to grow and develop in environments where there are different stress factors, this due to the development of specific mechanisms that allow them to detect changes in the environment and respond to them, minimizing possible damage and in turn conserving resources that allow him to continue with his development (Atkinson and Urwin, 2012). Biotic stress is caused by the attack of a wide range of pests and pathogens that include fungi, bacteria, viruses, nematodes and herbivorous insects (Hammond-Kosack and Jones, 2015). One of the phytopathogenic fungi that stand out for its distribution and its devastating effects is Fusarium oxysporum f. sp. lycopersici, cause of vascular wilt, recognized as the main disease that causes problems in the tomato crop and that is responsible for the decrease of almost 60% in yield, besides affecting the quality of the product (Ascencio et al., 2008).
The strategies for the control of this disease in crops consist of the use of fungicide products and the development of resistant varieties; however, they have been ineffective due to the appearance of new breeds of the pathogen (Mandal et al., 2009). Under these circumstances, the demand for alternative products as biological products for the control of pests and diseases or the use of inducing molecules increases called elicitors, which are capable of triggering defense responses in plants (Nasir et al., 2014). This option is emerging as a promising alternative due to the wide variety of compounds in the plants that are generated as part of their development and that could act as elicitors (da Cruz et al., 2013); however, it must be considered that both the magnitude and extent of the induced response will depend on the type of molecule, the signal and its mobility, or ability to induce secondary signaling within the tissue (Eder and Cosio, 1994).
For this reason, it was proposed to study the effect of the application of two elicitors of natural origin on the vigor of the plant and the quality of the tomato fruits produced in conditions of biotic stress.
Materials and methods
Vegetal material
This research was carried out in the facilities of the Department of Horticulture of the Antonio Narro Autonomous Agrarian University (UAAAN), in the city of Saltillo, Coahuila, Mexico. Tomato seedlings of the Rio Grande variety of determined growth were used, which were planted in polystyrene trays with a mixture of mineral perlite of Hortiperl granulometry type, of the brand Termolita and peat moss (Klasman ST1) in a 1:1 ratio. (v/v), for germination were placed in a bioclimatic room at a temperature of 23° and a photoperiod of 14 h light and 10 h of darkness, 25 days later were transferred to the greenhouse for conditioning and subsequent transplantation to black bags of 10 L polyethylene with peat moss-perlite mixture (1:1 v/v), nutrition was provided through a Steiner solution (Steiner, 1961) according to the phenological stage.
Description of treatments
In this study four treatments were evaluated, designated as follows: a control treatment (TA) with plants without inoculation; two treatments that consisted of the inoculation with Fusarium oxysporum f. sp. lycopersici (FOL) and the application of elicitors (E1) formulated from algae extracts and (E2) from organic compounds derived from plants; a control treatment of the incidence of (FOL). The application of the elicitors was made at 7, 14 and 56 ddt, for which said elicitors were prepared in aqueous solution and sprayed on the leaves of the plants, with a dose of 3.75 mL L-1 for E1 and 1.5 mL L-1 for E2, it should be noted that the E1 and E2 elicitors are prototype formulations of the company Arysta LifeScience and the doses applied are those recommended by the company.
Inoculation of FOL
The strain of F. oxysporum f. sp. lycopersici, was isolated from roots of tomato plants with symptoms of vascular wilt. To obtain the inoculum, the reproduction of the pathogen in submerged culture was carried out, the concentration was standardized in 1 × 106 spores mL-1, the inoculation was performed at 19 ddt, making wounds in the roots of the plants and adding 50 mL of inoculum per plant.
Evaluation of variables
Agronomic variables. The evaluation of the variables of interest was carried out at different dates of the development of the plants (Figure 1). The incidence of the disease was evaluated considering the presence of healthy and diseased plants; the severity of the symptoms of the disease caused by FOL was evaluated according to the scale proposed by Diener and Ausubel (2005), which is described as follows, 0= dead plant (100%); 1= dead old leaves and young leaves with arrested growth (80%); 2= old chlorotic leaves and young leaves with arrested growth (60%); 3= old leaves with vascular chlorosis and young leaves with arrested growth (40%); 4= leaf petioles with arrested growth (20%); 5= no visible symptoms (0%).

Figure 1 Schedule of applications of the elicitors and evaluation of variables (the points indicate the number of evaluations performed per variable analyzed).
The evaluation of the SPAD units was made with a SPAD brand Minolta model 502DL and the stomatal conductance with a Decagon Devis model SC-1 porometer, the data was obtained from the measurement of the third physiologically mature leaf, making two readings per sheet per each plant evaluated, the record of the readings was made during the first hours of the morning.
The height of the plants was measured with a tape measure, considering from the base of the stem to the apex of the same. The total dry weight of the plants was carried out by destructive sampling of the plants, then they were placed in paper bags and kept in a Lindberg Blue drying oven model GO1350C-1, for 72 h at 80 °C, and subsequently, the weight of the samples was determined on a scale of Oahus model OHA PIONEER PB1, for obtaining yield per plant they were harvested and weighed the fruits produced to the second cluster.
Fruit quality variables. The evaluation of fruit quality variables was carried out on the fruits of the first bunch, considering one fruit per plant, when they presented more than 90% of the red surface, called stage six (USDA, 1997) and consisted of obtaining of the firmness of the fruits by means of a penetrometer brand Wagner Instruments model FDK, using the 8 mm strut, while the values of pH and total soluble solids were determined in the fruit mash with the help of a potentiometer mark Daigger model 550 and a digital refractometer brand Atago model Pal1, the quantification of vitamin C content was carried out according to the technique of AOC (1990), for which 10 g of fresh sample was weighed, crushed in a mortar adding 10 mL of 2% hydrochloric acid, the mixture was filtered and adjusted to 100 mL with distilled water, then a 10 mL aliquot was taken and placed in a flask, the sample was titrated with 2, 6 dichlorophenolindophenol solution, until the first pink tone that persisted for 30 seconds appeared, with the data obtained from the titrations of the samples and the blank, the vitamin C content was calculated, substituting the values in the following formula:
Where: Vm= volume spent on the sample; Vb= volume spent on the target; M= molarity of 2,6-dichlorophenolindophenol (0.001 N); FC= conversion factor of 1 mL of 2,6-dichlorophenolindophenol to 0.088 mg of vitamin C; W= sample weight in mg; V= total volume; a= aliquot.
Statistical analysis
The analysis of the data was carried out by means of analysis of variance and tests of comparison of means by means of Tukey (p≤ 0.05), a completely randomized experimental design was used, considering 4 treatments with 5 repetitions and a plant as experimental unit, for the severity variable the values obtained in percentages were adjusted according to the angular transformation of Bliss (Steel and Torrie, 1985).
Results and discussion
Agronomic variables. The incidence (Table 1) and the severity of the disease caused by FOL (Figure 2) were reduced by the application of elicitors of natural origin compared to the control treatment, in the evaluation performed at 57 ddt, a decrease was presented of 5% for the incidence of the disease and 66% for the severity of the symptoms shown for the treatment of E1, while for E2, a 3% decrease in the incidence and 44% of the severity of the disease was observed.
Table 1 Percentage of the incidence of diseased plants inoculated with FOL and treated with elicitors of natural origin.
| ddt | TA | E1 | E2 | FOL |
| 57 | 0% | 90% | 95% | 95% |
| 80 | 0% | 100% | 100% | 100% |
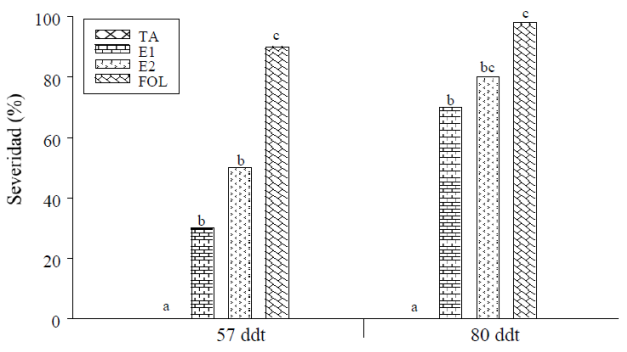
Figure 2 Severity of the disease caused by FOL in tomato plants treated with elicitors of natural origin (according to the scale used the plants that do not present symptoms have a value of zero). Means with the same letter do not differ statistically (Tukey, p≤ 0.05).
In the sampling carried out at 80 ddt, the incidence among the treatments inoculated with the pathogen showed the same values for the disease, although the severity of the symptoms increased over time, during this sampling the severity presented by the treatments with the application of the elicitors was lower 28% for E1 and 18% for E2.
The application of plant extracts from various sources promotes the induction of resistance in treated plants, they also have an effect on the growth and development of the pathogen (Arzoo et al., 2012) as observed in this study, where the incidence of disease caused by FOL and the severity of its symptoms during the first evaluation were reduced by the application of the elicitors of natural origin, this could be due to the fact that the applied compounds activated some mechanism related to the defense of the plant, to cope with conditions of biotic stress.
This response can be from the induction of some physical barriers or biochemical reactions that alert the uninfected cells to danger (Glazebrook, 2005). Although the severity continued to be lower in the treatments where the elicitors were applied, the incidence at the end of the experiment was the same for all the treatments inoculated with FOL. This may be conditioned to the number of applications made, as well as to the regularity of the same, since it is reported that the weekly use of the extract of C. paradisi, allowed a better control of the disease caused by Fusarium, which when applied in only one occasion (Rodríguez and Montilla, 2002).
The evaluation of the SPAD units (Figure 3) showed an increase during the development of the tomato plants, the readings obtained at 15 ddt, which corresponds to the second application of the elicitors, did not show significant differences between the treatments evaluated, finding values for this variable between 43 and 45 SPAD units. At 23 ddt, values between 49 and 52 SPAD units were observed, although no significant difference was found between the treatments, otherwise for the readings recorded at 57 ddt, where highly significant differences were found between the treatments, observing that the treatments were inoculated with the pathogen showed a decrease for this variable. The SPAD units for this sampling were similar between the plants of the control treatment and the E1 treatment, while the treatments E2 and FOL showed a decrease of 31% and 65%, respectively in comparison with the control.

Figure 3 Effect of elicitors of natural origin on the SPAD units in tomato plants subjected to biotic stress. Means with the same letter do not differ statistically (Tukey, p≤ 0.05).
On the other hand, stomatal conductance (Figure 4) showed a decreasing tendency through the development of plants. For the measurement made at 15 ddt no statistical differences were observed between the treatments, while the data obtained at 23 ddt, showed an increase in this variable for the treatments that were inoculated by the pathogen, being the plants corresponding to the treatment with the application of E2 showed the highest values, with an increase of 22%, while the treatment E1 and FOL showed an increase of 11% with respect to the control. Conductance readings at 57 ddt, showed similar values between the absolute control and the treatment with the application of E1, treatments E2 and FOL showed a decrease of 28% and 65% respectively in comparison with the control.
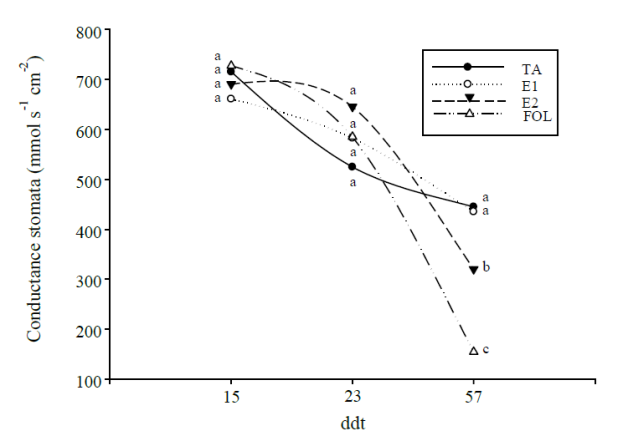
Figure 4 Effect of elicitors of natural origin on stomatal conductance in tomato plants subjected to biotic stress. Means with the same letter do not differ statistically (Tukey, p≤ 0.05).
The decrease of the SPAD units and the stomatal conductance in the plants inoculated with the pathogen, is directly related to the development of the disease, since it has been described that one of the symptoms caused by this disease is yellowing of the leaves followed by its detachment from the plant (González et al., 2012). Foliage of the foliage also occurs as a result of the invasion of the root by the pathogen with the subsequent colonization of the xylem vessels severely compromising the transport of water that ends in complete wilting of the individual (Michielse and Rep, 2009), which coincides with the results obtained by Dong et al. (2015) who indicate that in banana plants inoculated with F. oxysporum a significant decrease was found for this pair of variables in the last dates evaluated by the advance of the disease.
The plants inoculated with the pathogen and treated with the elicitor E2 showed similar values in the total dry weight accumulation with approximately 166 g (Figure 5), while the plants inoculated and treated with E1 showed 3% less than the control plants and the treatment subjected to biotic stress showed a decrease of this variable 39% with respect to the absolute control.
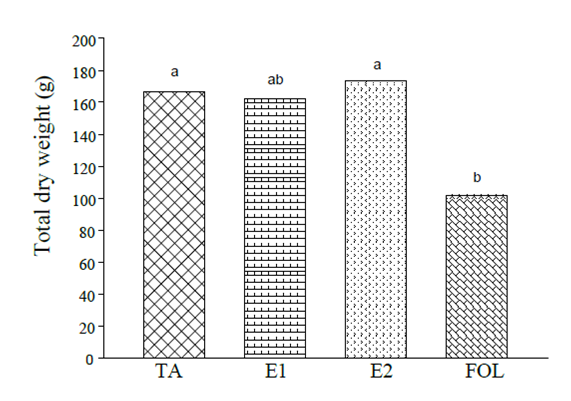
Figure 5 Effect of elicitors of natural origin on total dry weight in tomato plants subjected to biotic stress. Means with the same letter do not differ statistically (Tukey, p≤ 0.05).
The application of the elicitors of natural origin in the plants inoculated by FOL promoted the increase in the height of the plant (Figure 6), showing an increase of 6% for the plants treated with E1 and 13% for the plants treated with E2, in comparison with the height of the plants used as a control, whose height was 89.4 cm, otherwise it happened for the FOL treatment, where there was a 6% decrease in the height of the plants.
The positive effect of the application of the elicitors of natural origin was not only observed in the reduction of the symptoms of the disease; it was also found that its application promoted the accumulation of total dry biomass, the increase in height and a lower yield loss per plant compared to the inoculated plants that were not treated. This is because some plants have the capacity to
synthesize and accumulate secondary metabolites in their organs, which intervene in complex interactions between living organisms and which also have stimulating effects on the growth of other plants (Rodríguez and Hechevarría 2004).

Figure 6 Effect of elicitors of natural origin on height in tomato plants subjected to biotic stress. Means with the same letter do not differ statistically (Tukey, p≤ 0.05).
The stimulating effect of growth induced by plant extracts is reported in several species, such as the increase in biomass accumulation of maize plants under salinity conditions with the use of a biostimulant derived from alfalfa plants (Ertani et al., 2013). On the other hand, the application of extracts of H. longipes in tomato seedlings increased the height and the biomass accumulation and when subjected to conditions of biotic stress, the decrease of these variables was lower than in the untreated plants (González et al., 2015).
The disease caused by F. oxysporum is recognized as one of the causes of crop losses (Agrios, 2005), this could be verified with a 27% reduction in the yield of the plants inoculated with the pathogen and that did not receive control. The results of the effect caused by the elicitors on the yield with the application of elicitors, coincide with those obtained by Rodríguez and Montilla (2002) who indicate that there is a direct relationship between the decrease of wilting in plants and their production, since as the severity of the symptoms of the disease decreased, the decrease in performance was lower.
Quality variables the fruit. The presence of the pathogen and the application of the elicitors induced changes in the fruit quality variables (Table 2). The pH values obtained showed no changes for any of the treatments, while the content of total soluble solids was modified in the fruits of plants treated with the pathogen and the application of the elicitors, in the same way, the firmness was affected by the presence of the disease, although there was a loss of firmness less of the fruits from the plants to which the elicitors were applied, no significant differences were found in the vitamin C content for the fruits of the different treatments.
Table 2 Effect of the application of elicitors of natural origin on quality parameters in tomato fruits subjected to biotic stress.
| Treatment | pH | Total soluble solids (°Brix) |
Firmness (kg cm-2) |
Vitamin C (mg 100 g-1) |
| TA | 4.3 a* | 6.3 a | 1.8 a | 30.26 a |
| E1 | 4.4 a | 5.4 b | 1.36 bc | 29.14 a |
| E2 | 4.4 a | 5 b | 1.62 ab | 28.98 a |
| FOL | 4.4 a | 5.7 ab | 1.34 c | 29.3 a |
The results obtained in this study for pH (4.3-4.4) and for total soluble solids (5-6.7) coincide with those reported by García et al. (2014) for tomato fruits produced in greenhouses. The attributes that confer quality to the fruit such as color, total soluble solids, acidity index, pH and firmness, are affected by genotypic variations, growth conditions, irradiance, growing season, nutrition, irrigation, temperature, diseases and post-harvest conditions (Turhan and Seniz, 2009; Fischer et al., 2016).
The loss of firmness in the harvested fruits of the plants inoculated with the pathogen may be influenced by the lack of generalized water in the plant as a consequence of the infection of the pathogen; likewise, it would affect the transport of photoassimilates in the fruit, observing the decrease in total soluble solids.
Literatura citada
Agrios, G. N. 2005. Control of plant disease. In: plant pathology. Fifth (Ed.). Elsevier Academic Press. New York. 293-353 pp. [ Links ]
AOAC. 1990. Oficial Methods of Analysis of AOAC (Association of Oficial Analytical Chemist). 1 15th edition. Vol. II. Association of Oficial Analytical Chemist. Washington, DC. USA. 829-830 pp. [ Links ]
Arzoo, K.; Biswas, S. K. and Rajik, M. 2012. Biochemical evidences of defence response in tomato against Fusarium wilt induced by plant extracts. Plant Pathol. J. 11(2):42-50. [ Links ]
Ascencio, Á. A.; López, B. A.; Borrego, E. F.; Rodríguez, H. S. A.; Flores, O. A.; Jiménez, D. F. y Gámez, V. A. J. 2008. Marchitez vascular del tomate: I. Presencia de razas de Fusarium oxysporum f. sp. lycopersici (Sacc.) Snyder y Hansen en Culiacán, Sinaloa, México. Rev. Mex. Fitopatol. 26(2):114-120. [ Links ]
Atkinson, N. J. and Urwin, P. E. 2012. The interaction of plant biotic and abiotic stresses: from genes to the field. J. Exp. Bot. 63(10):3523-3543. [ Links ]
da Cruz, C. L.; Pinto, V. F. and Patriarca, A. 2013. Application of plant derived compounds to control fungal spoilage and mycotoxin production in foods. International J. Food Microbiol. 166(1):1-14. [ Links ]
Diener, A. C. and Ausubel, F. M. 2005. Resistance to Fusarium oxysporum 1, a dominant arabidopsis disease-resistance gene, is not race specific. Genetics. 171(1):305-321. [ Links ]
Dong, X.; Wang, M.; Ling, N.; Shen, Q. and Guo, S. 2016. Potential role of photosynthesis-related factors in banana metabolism and defense against Fusarium oxysporum f. sp. cubense. Environmental and Experimental Botany. 129: 4-12. [ Links ]
Eder, J. and Cosio, E. G. 1994. Elicitors of plant defense responses. International review of cytology. 148:1-36. [ Links ]
Ertani, A.; Schiavon, M.; Muscolo, A. and Nardi, S. 2013. Alfalfa plant-derived biostimulant stimulate short-term growth of salt stressed Zea mays L. plants. Plant and Soil.364(1-2):145-158. [ Links ]
Fischer, G.; Parra-Coronado, A. y Miranda, D. 2016. La calidad poscosecha de los frutos en respuesta a los factores climáticos en el cultivo. Agron. Colomb. 1:S1415-S1418. [ Links ]
García, E. E. L.; La Rosa, I. D.; Mendoza, V. R.; Quezada, M. M. R. y Arellano, G. M. 2014. Efecto de una película plástica modificada en algunos aspectos bioquímicos de un cultivo de tomate (Solanum lycopersicum L.). Ecos. Rec. Agrop. 1(2):151-162. [ Links ]
Glazebrook, J. 2005. Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu. Rev. Phytopathol. 43:205-227. [ Links ]
González, I.; Yailén, A. y Peteira, B. 2012. Aspectos generales de la interacción Fusarium oxysporum f. sp. lycopersici-tomate. Rev. de Protección Vegetal. 27(1):1-7. [ Links ]
González, M. S.; Benavides, M. A.; García, E. E. L.; Rodríguez, C. E. M. y Flores, O. A. 2015. Efecto de las alcamidas como inductores de tolerancia al estrés biótico en tomate. Rev. Mex. Cienc. Agríc. Vol. Esp. 12: 2371-2382. [ Links ]
Hammond, K. K.E.; Jones, J. D.G.; Buchannan, B.; Gruissem, W. and Jones R. 2015. Response to plant pathogens. In: Biochemistry and molecular biology of plants. Buchanan, B. B.; Gruisem, W. and Russell, L. J. (Eds). Second (Ed.) Jhon Wiley & Sons. Oxford, UK. 984-1050 pp. [ Links ]
Mandal, S.; Mallick, N. and Mitra, A. 2009. Salicylic acid-induced resistance to Fusarium oxysporum f. sp. lycopersici in tomato. Plant Physiol. Biochem. 47(7):642-649. [ Links ]
Michielse, C. B. and Rep, M. 2009. Pathogen profile update: Fusarium oxysporum. Mol. Plant Pathol. 10(3):311-324. [ Links ]
Nasir, M. N.; Polo, L. D.; Luzuriaga, L. W.; Deleu, M.; Lins, L.; Ongena, M. and Fauconnier, M. L. 2014. New alternatives to chemical pesticides: deciphering the action mechanisms of lipid based plant elicitors via complementary biophysical and biological approaches. In: 19th National Symposium on Applied Biological Sciences. Gembloux, Bélgica. (https://orbi.uliege.be/handle/2268/165417). [ Links ]
Rodríguez, D. A. y Montilla, J. O. 2002. Disminución de la marchitez causada por Fusarium en tomate con extracto de Citrus paradisi. Manejo integrado de plagas. 63:46-50. [ Links ]
Rodríguez, G. H. y Hechevarría S. I. 2004. Efectos estimulantes del crecimiento de extractos acuosos de plantas medicinales y gel de Aloe vera (L.) NL Burm. Rev. Cubana de Plantas Medicinales, 9(2):0-0. [ Links ]
Steel, R. y J. Torrie. 1985. Análisis de la varianza II: clasificaciones múltiples. En Bioestadística: principios y procedimientos. McGraw-Hill Latinoamericana, Bogotá, Colombia.188-230 pp. [ Links ]
Steiner, A. A. 1961. A universal method for preparing nutrient solutions of a certain desired composition. Plant Soil. 15(2):134-154. [ Links ]
Turhan, A. and V. Seniz. 2009. Estimation of certain chemical constituents of fruits of selected tomato genotypes grown in Turkey. Afr. J. Agric. Res. 4(10):1086-1092. [ Links ]
USDA (United States Department of Agriculture). 1997. United States standards for grades of fresh tomatoes. United States Departament of Agriculture, Agricultural Marketing Service, Fruits and Vegetable Division, Fresh Products Branch. Washington, D.C. USA. 13 p. [ Links ]
Received: January 2018; Accepted: March 2018











 texto en
texto en 


