Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Revista mexicana de ciencias agrícolas
versão impressa ISSN 2007-0934
Rev. Mex. Cienc. Agríc vol.6 spe 12 Texcoco Nov./Dez. 2015
Articles
Biological fertilization of maize
1Departamento de Riego y Drenaje, Universidad Autónoma Agraria Antonio Narro. Calzada Antonio Narro 1923, Saltillo, Coahuila, México. (hrr_homero@hotmail.com; abenmen@uaaan.mx; martincadena@uaaan.mx; camposmsg@hotmail.com).
2Palau Bioquim, S. A. de C. V. Saltillo, Coahuila, México. (cardenasjomar@gmail.com).
3Departamento de Horticultura, Universidad Autónoma Agraria Antonio Narro. Calzada Antonio Narro 1923, Saltillo, Coahuila, México. (hrr_homero@hotmail.com; abenmen@uaaan.mx; martincadena@uaaan.mx; camposmsg@hotmail.com).
4Departamento de Maquinaria Agrícola, Universidad Autónoma Agraria Antonio Narro. Calzada Antonio Narro 1923, Saltillo, Coahuila, México. (hrr_homero@hotmail.com; abenmen@uaaan.mx; martincadena@uaaan.mx; camposmsg@hotmail.com).
By the growing demand for agricultural products and increased costs of chemical fertilizers, associated to pollution that this may cause to the environment, it is necessary to develop other sustainable alternatives for agricultural production. Biofertilización could mitigate these problems, and in turn increase crop yields. The aim of this study was to evaluate the effect of the application of biological fertilizers derived from seaweed applied to the soil and foliage on forage maize (Zea mays L.). The response variables were: relative chlorophyll content, electron transport rate, plant height, stem diameter and dry weight of the plant. The research was conducted during the summer-fall cycle 2011 in Torreon, Coahuila, Mexico. Two treatments were evaluated. With and without biological fertilizers; the fertilizers derived from seaweed extracts were: 1 L ha-1 compound AlgaenzimsMR and 0.5 L ha-1 AlZincBMR. The experimental design was completely random. Each treatment was replicated 10 times. Plants receiving biological fertilization showed higher values (p≤ 0.05) for relative chlorophyll content (32.4%) and electron transport rate (17.8%), which resulted in an increase (p≤ 0.05) in plant height ( 17.3%), stem diameter (10.5%) and plant dry weight (14.8%) compared to plants without application of biological fertilizers.
Keywords: Zea mays L.; chlorophyll content; electron transport rate; seaweed
Por la creciente demanda de productos agrícolas y el incremento en los costos de los fertilizantes químicos, asociado a la contaminación que éstos pueden ocasionar al medio ambiente, es necesario desarrollar otras alternativas sustentables para la producción agrícola. Con la bofertilización, se podrían mitigar estos problemas, y a su vez incrementar el rendimiento de los cultivos. El objetivo de este estudio fue evaluar el efecto de la aplicación de fertilizantes biológicos derivados de algas marinas aplicados al suelo y al follaje en un maíz forrajero (Zea mays L.). Las variables de respuesta fueron: contenido relativo de clorofila, tasa de transporte de electrones, altura de planta, diámetro del tallo y peso seco de la planta. La investigación se realizó durante el ciclo verano-otoño 2011 en Torreón, Coahuila, México. Se evaluaron dos tratamientos. Con y Sin fertilizantes biológicos, Los fertilizantes derivados de extractos de algas marinas aplicados fueron: 1 L ha-1 del compuesto AlgaenzimsMR y 0.5 L ha-1 de AlZincBMR. El diseño fue completamente al azar. Cada tratamiento se repitió 10 veces. Las plantas que recibieron fertilización biológica mostraron valores mayores (p≤ 0.05) de contenido relativo de clorofila (32.4%) y tasa de transporte de electrones (17.8%), que resultó en un incremento (p≤ 0.05) en: altura de planta (17.3%), diámetro de tallo (10.5%) y peso seco de planta (14.8%) respecto a las plantas sin aplicación de fertilizantes biológicos.
Palabras clave: Zea mays L.; algas marinas; contenido relativo de clorofila; tasa de transporte de electrones
Introduction
Maize (Zea mays L.) is cultivated worldwide in more than 168 billion hectares with an annual production of more than 876 billion tons, so now this is the second most important crop in the world after wheat (Cotrisa, 2012). Its economic importance worldwide is due to the above fact as it is a fundamental grain for animal feed and in many countries, human (Zarkadas et al., 1995). Recently this grain has been used to produce ethanol, competing with traditional functions (Bothast and Schlierher, 2005; Vermis et al., 2007), so it is necessary to increase productivity. The application of biological fertilizers have been an alternative that allows to improve crop productivity by acreage and soil and water pollution is reduced (Carvajal-Muñoz and Mera-Benavides, 2010; Chisti et al., 2013).
The practice of biological fertilization based on seaweed species with agricultural value has shown increases in yield and good quality of crops from direct application or its derivatives (Painter, 1995; Canales-Lopez, 2001). Plants response to the application of seaweed are increased yields, better nutrients absorption, improve seed germination, increases chlorophyll content and leaves size (Metting et al., 1990; Crouch and van Staden, 1992).
Chlorophyll content in the leaf is closely and directly related to the nutritional status of the crop (Moran et al., 2000; Chang and Robinson, 2003; Berg and Perkins, 2004). Previous studies show that seaweed extracts contain cytokinins, auxins and betaines which significantly increase chlorophyll content in leaves (Whapham et al., 1993; Blunden et al., 1996; Schwab and Raab, 2004; Thirumaran et al., 2009).
Chlorophyll fluorescence is an important factor to determine the level of stress on photosynthetic processes in plants (Krause and Weis, 1991; Schreiber and Bilger, 1993) and to compare photosynthetic yield between plant species (Brodribb and Feild, 2000; Jones et al., 2010). Based on the measurement of chlorophyll fluorescence, the electron transport rate (ETR) in light saturation, which is also called photosynthetic capacity, is obtained and is used to express photosynthesis rate and physiological traits of leaves (Genty et al., 1989). ETR is the flow of electrons through the photosynthetic transport chain, so low levels of ETR may indicate that the plant is under stress which limits the photosynthesis rate. Comparisons of relative values of ETR between plants should be conducted under the same light conditions. The characteristics of light absorption by the leaves may vary depending on the species and the degree of water stress (Eichelman et al., 2004).
The aim of this study was to evaluate the effect of the application of biological fertilizer derived from seaweed applied to soil and foliage in forage maize (Zea mays L.), the relative chlorophyll content, electron transport rate and its relation with plant height, stem diameter and plant dry weight.
Materials and methods
Location and characteristics of the study area
The research was conducted during the summer-fall 2011 cycle in the locality "Ampuero" located in Torreon, Coahuila, Mexico (25° 27' north latitude and 103° 22' west longitude), at an altitude of 1 137 m. The physical and chemical soil analysis made in the substrate 0 to 0.3 m deep, clay-sandy texture moderately alkaline (pH= 8.5), no salt problems (EC of saturation extract 1.48 dS m-1), percentage of organic matter 1.59% and bulk density of 1.31 g cm-3.
Agronomical management
The yellow hybrid high-yield (HT-7887), which corresponds to a corn whose adaptation area is the northern and central Mexico, plant height is 2.5 to 2.7 m, intermediate cycle, with good yield and foliage quality. The trial was established on June 30, 2011. The seeds were placed in separate grooves 0.75 and 0.13 m between seeds for planting density of 102 564 plants ha-1. Two melgas of 10 m wide by 250 m long (2 500 m2) were established. Irrigation was applied pre planting on May 15. Subsequently three irrigations of an average sheet of 17 cm with an interval between irrigations of approximately 30 days using siphons to derive water from the channel into the plots; fall armyworm (Spodoptera frugiperda) was controlled with two applications of Palgus (Spinoteram) (125 mL ha-1). For pest control, Latigo (Chlorpyrifos-cypermethrin) (1.25 L ha-1) was applied. Weed control was performed by applying Primagram (Atrazine-Smetolachlor) (1.25 L ha-1) at 15 and 28 days after planting.
Treatments evaluated and experimental design
In one of the melgas described before the AlgaenzimsMR product was applied, which is a biological product based on macro and micro seaweed with alkaline pH (8.2). The application was made in the pre sowing irrigation at a dose of 1 L ha-1. Subsequently, to plants of the same melga was applied (foliar) 0.5 L ha-1 of AlZincBMR containing growth regulators such as auxins, gibberellins and cytokinins as an inherent part of seaweed extracts (Sargasuum spp.) plus trace elements like zinc (10%, w/w) and boron (0.5%, w/w). Applications were made 41 days after planting. The other melga was kept as control (without application of biological fertilization). Both melgas received mineral fertilization that the producer regularly applies, which consisted of 25 kg of ammonium sulfate and 100 kg of monoammonium phosphate, applied to the soil one day before planting.
A completely randomized design with two treatments (with and without application of biological fertilizers) and 10 replications was used; one plant represented the experimental unit. 10 plants in each melga were randomly selected in the central part of the plot. Means comparison was performed using Duncan test (p≤ 0.05).
Relative chlorophyll content
The effect of biofertilizer in relative chlorophyll content of leaves was performed spectrophotometrically using SPAD 502 (Minolta Inc.) sensor, which measures the relative chlorophyll concentration by light transmitted through the leaf at a wavelength of 650 to 940 nm (Sainz-Rozas and Echeverria, 1998). This sensor, estimates instantaneously relative chlorophyll content (ranging from 0 to 199) in leaves nondestructively with the following relationship:
Where: SPAD is the value of chlorophyll level; K is the extinction coefficient of the medium (leaf chlorophyll) to applied radiation (650 nm); IRt is the intensity of radiation that leaf transmits at 940 nm; IRo is the intensity of light emitted by the sensor at 940 nm; Rr is the intensity of radiation that leaf transmits at 650 nm; Ro is the intensity of light emitted by the sensor at 650 nm.
Measurements were made in 10 plants from the center of each melga. Eight measurements distributed along the third leaf of each plant (in four places on each side) were made. The data was obtained from the middle between the base and the apex. The records were made at 36 and 71 days after planting.
Electron transport rate (ETR)
The electron transport rate was determined through eight measurements per leaf on each of the 10 plants of each melga (with and without application of biofertilizers), 71 days after sowing (das). For this a pulse-modulated fluorometer FMS2 model (Hansatech Instruments) was used, which obtains ETR with the following equation:
Where: ETR is the electron transport rate (µmol m-2 s-1), ΦPSII is the quantum efficiency of photosystem II, α is the ratio of light absorption by the photosynthetic tissue (0.84), f is the proportion of light transferred to each of the photosystems (PSII and PSI), which corresponds to 0.54, and PAR is incident photosynthetically active radiation (µmol m-2 s-1).
The quantum efficiency of photosystem II is obtained by the following equation:
Where: FM 'is the real or effective maximum fluorescence and F is the initial fluorescence.
Plant growth components evaluated
Plant height was measured from ground surface to the last knot from the stem. The first evaluation was performed at 71 and the second at 99 das. 10 plants were selected randomly from each treatment. Stem diameter was measured at 5 cm from ground surface with digital vernier surtek (Electronic digital caliper 122200). Measurements were made in 10 plants per treatment, randomly selected at 99 das. Plants from each melga were harvested at ground level at 100 das these were placed in a drying oven at 70 oC for 24 h to obtain plant dry weight.
Results and discussion
Relative chlorophyll content
Application of biological fertilizers seaweed based had a significant effect (p≤ 0.05) in the relative chlorophyll content of leaves (Figure 1). In the first assessment (36 das), chlorophyll content was 8.1% higher in plants treated with biological fertilizers compared to treatment without biological fertilizer. In the second assessment (71 das) the difference between treatments increased to 32.4% (Figure 1).
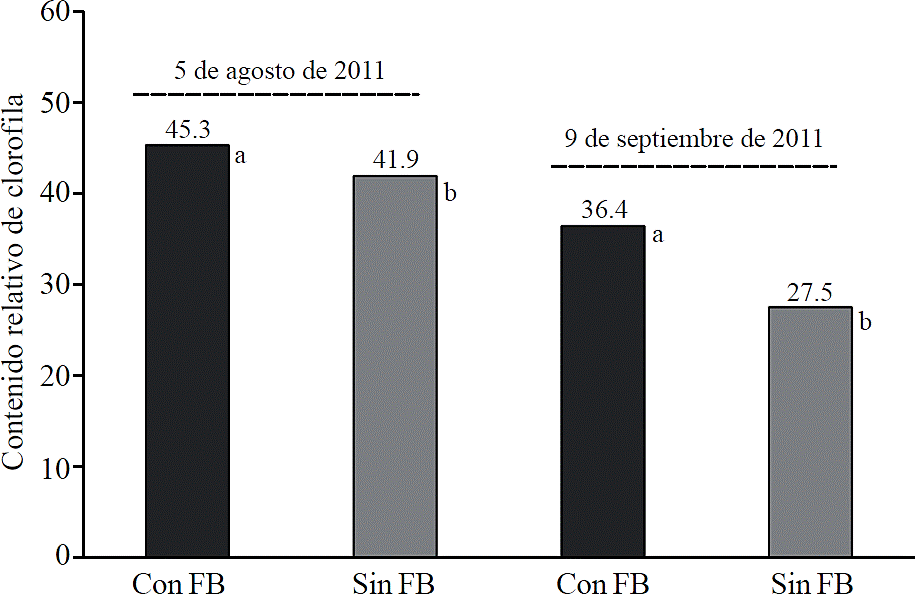
Figure 1 The relative content of chlorophyll in leaves of corn plants with biological fertilization (FB) (AlgaenzimsMR and ALZincBMR) and biological plants without fertilization (no FB), observed on August 05 (36 days after planting) and 9 September (71 days after sowing), 2011, Torreon, Coahuila, Mexico.
Previous studies have shown that seaweed extracts contain significant amounts of cytokinins, auxins and betaines, when applied to soil or crop increases chlorophyll concentration in the leaves (Whapham et al., 1993; Blunden et al., 1996; Schwab and Raab, 2004).
Electron transport rate (ETR)
The application of biofertilizers had a significant effect (p≤ 0.05) in the electron transfer rate (ETR) in leaves (Figure 2). At 71 days after sowing, ETR on leaves from treated plants was 17.8% higher than plants without application of biofertilizer (Figure 2). This result also indicated that the degree of stress in treated plants was lower, since the electron transport rate is an indicator of plant stress (Krause and Weis, 1991; Wajahatullah et al., 2009).

Figure 2 Electron transport rate (ETR) in leaves of corn plants with biological fertilization (FB) (AlgaenzimsMR and ALZincBMR) and biological plants without fertilization (no FB) observed on 9 September (71 days after seeding) 2011, Torreon, Coahuila, Mexico.
In this regard, Nabati et al. (1994) reported an increased tolerance to salinity in Kentucky blue grass (Poa pratensis L.) in response to the application of seaweed extract. Similarly, Nahed et al. (2011) reported that the application of seaweed extract (Ascophyllum nodosum) in amaranth (Amaranthus tricolor L.) reduces salinity stress and increases length, stem diameter, root length and leaf number. While Spann and Little (2011) found that the application of seaweed extract (Ascophyllum nodosum) to the soil or foliar to orange (Citrus sinensis L.) improves tolerance to water stress.
Plant height
Figure 3 shows that the application of biological fertilizer increased plant height (p≤ 0.05). In the first assessment (71 das), plants with biological fertilizer were 17.3% higher than plants without applications. In the second evaluation (99 das) height difference was 7.7% in plants with biological fertilization. These results can be attributed to marine algae are a natural source of auxins and cytokinins, which are a group of bio-regulators (Crouch and van Staden, 1993). Similar results were reported by Zodape et al. (2011) finding that foliar application seaweed sap (Kappaphycus alvarezii) (5%), increased 34% plant height and 45% root length in tomato plants. Similarly Sivasankari et al. (2006) reported that the application of seaweed (Sargassum wightii and Caulerpa chemnitzia) increased plant growth on beans (Phaseolus vulgaris L.).
Stem diameter
Stem diameter also showed increases (p≤ 0.05) by the application of biological fertilizers, since at 99 das, stem diameter was 10.5% higher than that observed in plants without biological fertilization (Figure 4). This can be attributed to seaweed extracts provide to plants major and minor elements content, plus enzymes and growth regulators (Canales-López, 1997). In this regard, some studies report that seaweed extracts are a source of macro and micro nutrients, amino acids, vitamins, cytokinins, auxins and abscisic acid (ABA), which affect cell metabolism in plants leading crops to higher growth and yields (Durand et al., 2003; Stirk et al., 2003; Ordog et al., 2004).
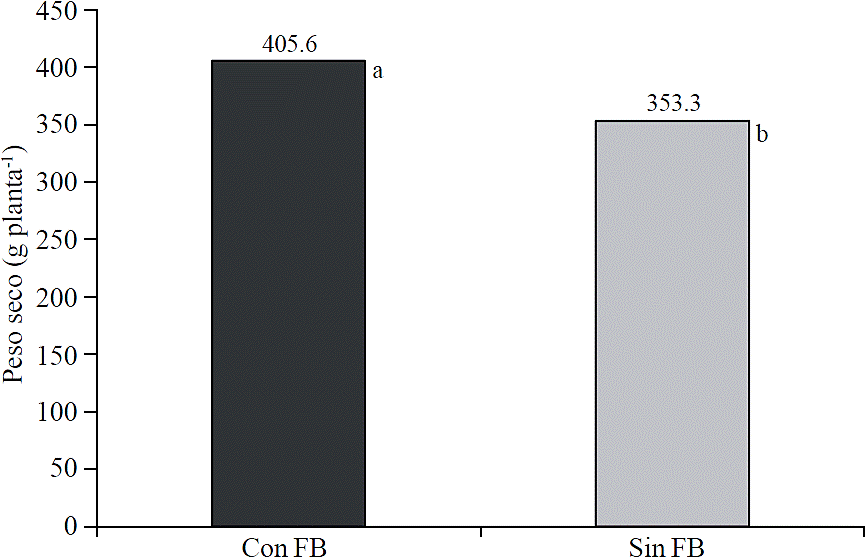
Plant dry weight
Application of biofertilizers derived from seaweed increased plant dry weight (p≤ 0.05) (Figure 5). At 99 days after transplantation, the average plant dry weight was 405.6 g plant-1, and without applications to plants was 353.2 g plant-1, this represented an increase of 14.8%. Rathore et al. (2009) reported higher growth and grain yield on soybean (Glycine max L.) with foliar application of seaweed extracts. Similarly, Jeannin et al. (1991) found an increased fresh weight on corn seedlings treated with seaweed extracts.
Literatura citada
Berg, A. K. and Perkins, T. D 2004. Evaluation of a portable chlorophyll meter to estimate chlorophyll and nitrogen contents in sugar maple (Acer saccharum Marsh.) leaves. Forest Ecol. Manag. 200:113-117. [ Links ]
Blunden, G.; Jenkins, T. and Liu, Y. 1996. Enhanced leaf chlorophyll levels in plants treated with seaweed extract. J. Appl. Phycol. 8:535-543. [ Links ]
Bothast, R. J. and Schlierher, M. A. 2005. Biotechnology processes for conversion of corn into ethanol. Appl. Microbiol. Biotechnol. 67:19-25. [ Links ]
Brodribb, T. J. and Field, T. S. 2000. Stem hydraulic supply is linked to leaf photosynthetic capacity: evidence from new caledonian and tasmanian rainforests. Plant Cell Environ. 23:1381-1388. [ Links ]
Canales-López, B. 1997. Las algas en la agricultura orgánica. Consejo Editorial Trillas S. A. de C. V. México, D. F. 28 p. [ Links ]
Canales-López, B. 2001. Uso de los derivados de algas marinas en la producción de papa, tomate, chile y tomatillo: Resultados de investigación. Coahuila: Palau Bioquím S. A. 24 p. [ Links ]
Carvajal-Muñoz, J. S. y Mera-Benavides, A. C. 2010. Fertilización biológica: técnicas de vanguardia para el desarrollo agrícola sostenible. Producción + limpia. 5(2):77-96. [ Links ]
Chang, S. X and Robinson, D. J. 2003. Nondestructive and rapid estimation of hardwood foliar nitrogen status using the SPAD502 chlorophyll meter. Forest Ecol. Management. 181:331-338. [ Links ]
Chesti, M. U. H.; Qadri, T. N.; Hamid, A.; Qadri, J.; Azooz, M. M. and Ahmad, P. 2013. Role of Bio-fertilizers in crop improvement: In: crop improvement new approaches and modern techniques. Rehman, K. H.; Paravaiz, A. and Munir, O. (Eds.). Springer science + business media, LLC, New York, NY. 189-208 pp. [ Links ]
Chirinos, J.; Leal, A. and Montilla, J. 2006. Uso de insumos biológicos como alternativa para la agricultura sostenible en la zona sur del estado Anzoátegui. Revista Digital Ceniap. 11:1-7. [ Links ]
Cotrisa (2012) Cotrisa (2012) http://www.cotrisa.cl/mercado/maiz/internacional/detalle.php (Consultado octubre, 2012). [ Links ]
Crouch, I. J. and Staden, J. van. 1992. Effect of seaweed concentrate on the establishment and yield of greenhouse tomato plants. J. Appl. Phycol. 4:291-296. [ Links ]
Crouch, I. J and Staden, J. van. 1993. Evidence for the presence of plant growth regulators in commercial seaweed products. Plant Growth Regulation. 13:21-29. [ Links ]
Durand, N; Briand, X. and Meyer, C. 2003. The effect of marine bioactive substances (NPRO) and exogenous cytokinins on nitrate reductase activity in Arabidopsis thaliana. Physiologia Plantarum 119:489-493. [ Links ]
Eichelman, H; Oja, V.; Rasulov, B.; Padu, E.; Bichele, I.; Pettai, H.; Niinemets, O. and Laisk, A. 2004. Development of leaf photosynthetic parameters in betual pendula roth leaves: correlation with photosystem I density. Plant Biol. 6:307-318. [ Links ]
Genty, B; Briantais, J. M. and Baker, N. R. 1989. The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochimica et Biophysica Acta. 990:87-92. [ Links ]
Jeannin, I.; Lescure J. C. and Morot-Gaudry, J. F. 1991. The effects of aqueous seaweed sprays on the growth of maize. Botánica Marina 34:469-473. [ Links ]
Jones, T. J.; Luton, C. D.; Santiago, L. S. and Goldstein, G. 2010. Hydraulic constraints on photosynthesis in subtropical evergreen broad leaf forest and pine woodland trees of the Florida Everglades. Trees: Structure and Function 24:471-478. [ Links ]
Krause, G. H. and Weis, E. 1991. Chlorophyll fluorescence and photosynthesis: the basics. Ann. Rev. Plant Physiol. Plant Mol. Biol. 42:313-349. [ Links ]
Metting, B; Zimmerman, W. J.; Crouch, I. J. and Staden, J. van 1990. Agronomic uses of seaweed and microalgae. In: introduction to applied phycology. Akatsuka, I. (Ed.). SPB Academic Publishing. The Hague, Netherlands. 269-627 pp. [ Links ]
Moran, J. A.; Mitchell, A. K.; Goodmanson, G. and Stockburger, K. A. 2000. Differentiation among effects of nitrogen fertilization treatments on conifer seedlings by foliar reflectance: a comparison of methods. Tree Physiology. 20:1113-1120. [ Links ]
Nabati, D. A; Schmidt, R. E. and Parrish, D. J. 1994. Alleviation of salinity stress in Kentucky bluegrass by plant growth regulators and Iron. Crop Sci. 34:198-202. [ Links ]
Nahed, G. A.; Mona, H. M. and Hanan, S. S. 2011. Growth, flowering and chemical constituents performance of amaranthus tricolor plants as influenced by seaweed (Ascophyllum nodosum) extract application under salt stress conditions. J. Appl. Sci. Res. 7(11):1472-1484. [ Links ]
Ordog, V; Stirk, W. A.; Staden, J. V.; Novak, O. and Strnad, M. 2004. Endogenous cytokinins in the three genera of microalgae from the Chlorophyta. J. Phycol. 40:88-95. [ Links ]
Painter, T. J. 1995. Biofertilizers: exceptional calcium binding affinity of a sheath proteoglycan from the blue-green soil alga Nostoc calcicola. Carbohydrate Polymers 26(3):231-233. [ Links ]
Rathore, S. S; Chaudhary, D. R.; Boricha, G. N.; Ghosh, A.; Bhatt, B. P.; Zodape, S. T. and Patolia, J. S. 2009. Effect of seaweed extract on the growth, yield and nutrient uptake of soybean (Glycine max) under rainfed conditions. South Afr. J. Bot. 75:351-355. [ Links ]
Sainz-Rozas, H. and Echeverría, H. E. 1998. Relación entre las lecturas del medidor de clorofila (Minolta SPAD 502) en distintos estadios del ciclo del cultivo de maíz y el rendimiento en grano. Rev. Fac. Agron. La Plata. 103(1):37-44. [ Links ]
Schreiber, U. and Bilger, W. 1993. Progress in chlorophyll fluorescence research: major developments during the past years in retrospect. Progress Bot. 54:151-173. [ Links ]
Schwab, W. and Raab, T. 2004. Developmental changes during strawberry fruit ripening and physicochemical changes during postharvest storage. In: production practices and quality assessment of food crops, ‘qualityhandling and evaluation’. Dris, R. and Jain, S. M. (Eds.). Kluwer Academic Publishers, Netherlands. 341-369 pp. [ Links ]
Sivasankari, S.; Venkatesalu, V.; Anantharaj, M. and Chandrasekaran, M. 2006. Effect of seaweed extracts on the growth and biochemical constituents of Vigna sinensis. Bio. Technol. 97(14):1745-1751. [ Links ]
Spann, T. M. and Little, H. A. 2011. Applications of a commercial extract of the brown seaweed Ascophyllum nodosum increases drought tolerance in container-grown ‘Hamlin’ sweet orange nursery trees. Am. Soc. Hortic. Sci. 46(4):577-582. [ Links ]
Stirk, W. A.; Novak, M. S. and Staden, J. van 2003. Cytokinins in macroalgae. J. Plant Growth Reg. 41:13-24. [ Links ]
Thirumaran, G.; Arumugam M.; Arumugam, R. and Anantharaman, P. 2009. Effect of seaweed liquid fertilizer on growth and pigment concentration of Cyamopsis tetragonoloba (L) Taub. Am. Eur. J. Agron. 2(2):50-56. [ Links ]
Vemis, W.; Saballos, A.; Ejeta, G.; Mosier, N. S.; Landisch, M. R. and Carpita, N. C. 2007. Molecular breeding to enhance etanol production form corn and sorghum stover. Crop Sci. 47:142-153. [ Links ]
Wajahatullah, K; Rayirath, U. P.; Subranian, S.; Jithesh, M. N.; Rayorath, P.; Hodges, D. M.; Critchley, A. T.; Craigie, J. S.; Norrie J. and Prithiviraj, B. 2009. Seaweed extracts as biostimulants of plant growth and development. J. Plant Growth Reg. 28:386-399. [ Links ]
Whapham, C. A.; Bluden, G.; Jenkins, T. and Hankins, S. D. 1993. Significance of beteines in the increased chlorophyll content of plants treated with seaweed extract. J. Appl. Phycol. 5:231-234. [ Links ]
Zarkadas, C. G.; Yu, Z.; Hamilton, R. I.; Patison, P. L. and Rose, N. G. 1995. Comparison between protein quality of northern adapted cultivars of common maize and quality protein maize. J. Agric. Food Chem. 43:84-93. [ Links ]
Zodape, S. T; Gupta, A.; Bhandari, S. C.; Rawat, U. S.; Chaudhary, D. R.; Eswaran, K. and Chikara, J. 2011. Foliar application of seaweed sap as biostimulant for enhancement of yield and quality of tomato (Lycopersicon esculentum Mill). J. Sci. Ind. Res. 70:215-219. [ Links ]
Received: March 2015; Accepted: June 2015











 texto em
texto em