Introduction
Rats are omnivore animals and forage in a great variety of habitats (Collier, Hirsch & Kanarek, 1977). Galef and Whiskin (1997) mentioned that animals do not have language and depend on conspecific observation to obtain information from them. One behavior vital for rats is to forage in adaptive ways. They must choose safe foods and avoid poisonous foods.
Sources of information that rats use are olfactory cues, olfactory stimuli (Arriaga et al, 2006; Rodas, 1974). Rats use olfaction to identify sources of food present in leaders breath returning from a foraging trip. Galef and Wigmore (1983) and Posadas-Andrews and Roper (1983) found in the same year and published in the same journal number, a finding that showed that rats, specifically Long Evans rats, were guided in adaptive foraging by olfactory cues present in demonstrator rats. A great number of studies found that this phenomenon is reliable (Galef, 1996; Galef & Whiskin, 2003) and that the essential ingredient of this social transmission of information was carbon disulfide, an element present in rats breath and the flavor of food eaten by demonstrators (Galef, Mason, Preti, & Bean, 1988). Galef and Durlach (1993) found absence of blocking (Kamin, 1969) in another study that evaluated the conditioned stimulus properties of the flavor of food that demonstrators had eaten. This absence of blocking demonstrated that rats are able to process at least two social stimuli in sequence, without the first interfering with the second.
Juárez-Maldonado et al. (2006) and Melchor-Hipólito et al. (2010) found serial position effects with a list of three demonstrators, with a technique that used this social learning model developed by Galef and Wigmore (1983) and Posadas-Andrews and Roper (1983).
Eichenbaum (2002) studying the involvement of the hippocampus in associative learning used this social transmission of food preference paradigm as an example of social olfactory learning and memory (Bunsey & Eichenbaum, 1995). He mentioned that this aspect of the social transmission of food preference task, the expression of memory in a situation different from the learning trial, is strongly consistent with the declarative property of representational flexibility.
The cholinergic system is essential in learning and memory. A muscarinic cholinergic antagonist like scopolamine may modify learning and memory in different tasks (Bammer, 1982; Blozovski & Hennocq, 1982; Brito, Davis, Stopp, & Stanton, 1983; Caine, Weingartner, Ludlow, Cudahy, & Wehry, 1981; Carnicella, Pain & Oberling, 2005; Decker, Tran, & McGaugh, 1990; Ennaceur & Meliani, 1992; Izquierdo, 1989; Mendez, Gilbert, Bizon, & Setlow, 2012; Petersen, 1977; Savage, Faust, Lambert, & Moerschbaecher, 1996). Several studies showed that scopolamine impairs learning (Caine et al., 1981; Petersen, 1977; Sitaram, Weingartner, & Gillin, 1978). However, Cruz-Morales, Durán-Arevalo, Diaz del Guante, Quirate, and Prado-Alcalá (1992) found that scopolamine impairs consolidation of passive avoidance. In another study Quirarte, Cruz-Morales, Cepeda, García-Montañez, Roldán-Roldán, and Prado-Alcalá (1994) found that a dose of 8mg/kg of scopolamine administered 30 min before training affected consolidation in a passive avoidance task. Agrawal, Tyagi, Saxena, and Nath (2009) also noted that scopolamine impairs consolidation rather than recall, in a passive avoidance task.
Izquierdo (1989) evaluating the effects of scopolamine in different studies wrote that the effects of scopolamine might vary depending of the task under study, and that verbal recall tasks may be different from passive avoidance tasks. Eichenbaum classified the task studied in this work as an example of declarative memory (e.g. 2002), so it is possible that the effects of a muscarinic cholinergic antagonist as scopolamine will have different effects to those seen with other tasks like passive avoidance that is an example of procedural memory.
Based on the studies mentioned it is possible that scopolamine will interfere with learning and memory of a trial in which social transmission of food preference usually develops. The purpose of this study was to assess the effect of a dose of scopolamine on learning and consolidation of this transmission of information.
Method
Subjects
Eighteen male Long Evans rats participated in this experiment. The age of the subjects varied between 2.3 and 3 months. Three rats served as demonstrators and 15 as observers. Demonstrators always ate the same flavor of food in each experiment. The rats obtained from the university vivarium were 60 days old at the beginning of the experiment. Their free-feeding body weights were in a range from 214 to 297 g. The rats were food deprived except when having free access to chow (Harlan, Mexico City) for 20 min each day. Rats lived in a room kept on a 12:12-hr cycle of light: darkness and an ambient temperature between 21° and 26°C.
Apparatus and Materials
The subjects were housed individually in steel boxes (Armexa, Mexico City) that were 18 cm high, 18 cm wide, and 24 cm long. The demonstrators received food in their home cages, powdered chow with flavor added. We mixed Food with one of three flavors: 1%cinnamon (McCormick, Mexico City), 2% cocoa (Hershey’s, Mexico City), or 0.5% vanilla (Flor de Maria, Mexico City), all by weight.
Demonstrations took place in a Plexiglas box that was 26 cm wide, 15 cm high and 36 cm long, the Demonstration Box. For the reference test, we placed flavored food in metal containers that were 2.4 cm high, 5 cm wide and 7.5 cm long. The containers were wired to the rear wall of a Plexiglas box that was 46 cm wide, 20 cm high, and 53 cm long, and were separated by Plexiglas lids, 1 cm wide, 1 cm high, and 15 cm long, called the Test Box.
The chamber and containers were cleaned after each observer had completed its 20-min test. On Ohaus balance, Model 310, with a .01-g resolution the experimenters weighed the amount of remaining food to calculate the amount consumed by subjects.
Procedure
We compared three groups of five subjects each one. In each group, we presented demonstrators on different days, in a fixed sequence: Cocoa, Vanilla, Cocoa, Cinnamon, and Cocoa. All subjects in the three groups passed through this sequence, once in each experiment. The difference between groups was the treatment administered. The observers in the Control group received no treatment. The observers in the Saline Group received an ip injection of a 2 ml/kg of saline, 15 min before demonstration. The observers in the Scopolamine Group received an ip injection with a dose of 8 mg/kg of scopolamine in 2 ml/kg volume of saline 15 min before demonstration. In all groups observers interacted with demonstrators for 15 min. After demonstration, observers received a preference test in the Test Box. Observers found three containers with 25 g of each of the flavored foods. The position of the flavored foods in the containers was counterbalanced. The preference test was 20 min long. After the preference test, we weighed the remaining food to determine preference and recall.
Results
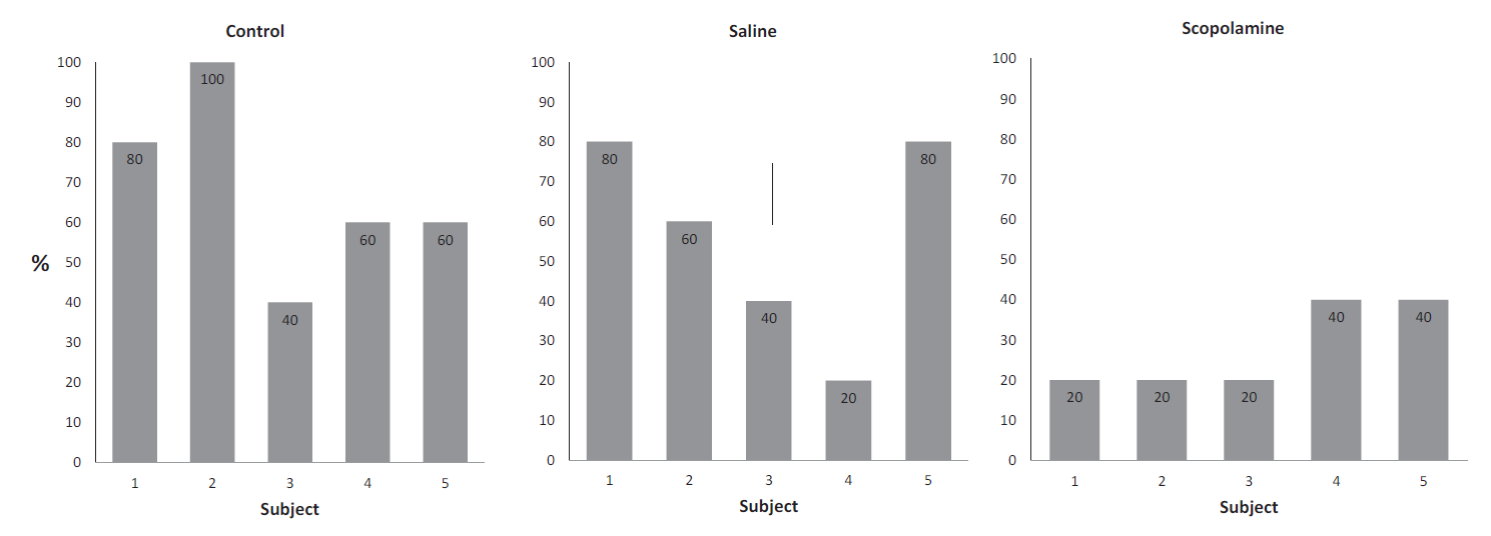
Figure 1 presents the mean of the proportion (± SE) of occasions in which social learning occurred in each subject in each group of the experiment.
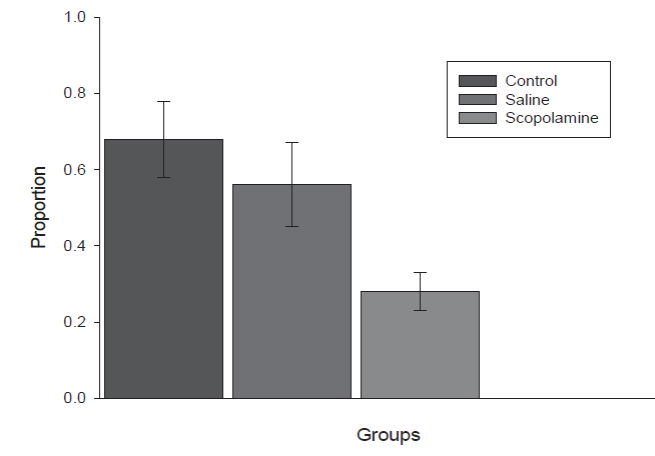
Figure 1 Mean proportion of occasions of transmission of preference (± SE) in each one of the groups in the Experiment.
The Control group and the Saline group had similar means. A chi squared made to the occasions that social transmission occurred in each group showed a reliable difference between the three groups, x 2 (2) = 9.21, the obtained value 9.75 shows a significant difference, p = .01. An analysis showed no difference between the control and saline groups, x 2 .90 (1) = 3.84, the value obtained was 3.30 for a p = .10. Figure 2 presents individual percentages.
Discussion
The purpose of this experiment was to assess if scopolamine would interfere with consolidation of memory of a trial of social transmission of food preference. Results showed that indeed scopolamine interferes with consolidation. The difference in number of occasions in which social learning occurred showed that scopolamine precluded the formation of the memory of the demonstrated event. This result is similar to the ones found by different researchers (Bammer, 1982; Blozovski & Hennocq, 1982; Brito et al., 1983; Caine et al., 1981; Carnicella et al., 2005; Decker et al., 1990; Izquierdo, 1989; Mendez et al., 2012; Petersen, 1977; Savage et al., 1996).
In some studies scopolamine interfered with learning (Caine et al., 1981; Petersen, 1977; Sitaram et al., 1978). In other studies, Agrawal et al. (2009), Cruz-Morales et al. (1992) and Quirarte et al (1994) , found that scopolamine impairs consolidation of memory of a passive avoidance trial. Izquierdo (1989) mentioned that the effects of scopolamine depend on the task under study.
Agrawal et al., Cruz-Morales et al. and Quirarte et al., found interference on consolidation in a passive avoidance task, the first group of authors with mice and the second and third with rats. The results from this study are similar to those obtained by Quirate et al. The task studied in present research is an example of declarative memory (Eichenbaum, 2002), and we found that scopolamine impaired consolidation of memory of trials in the social transmission of food preference paradigm.
The results from this study showed that the social transmission of food preference is a useful task to study learning and memory as Eichenbaum (2002) and Galef and Whiskin (2003) mentioned. Juárez-Maldonado et al. (2006) and Melchor-Hipólito et al. (2010) found serial position effects with a list of three demonstrators, a procedure also based on the food preference paradigm, a model that is an example of declarative memory. These results also showed that the cholinergic system is involved in memory formation in social transmission of food preferences. Because scopolamine affects central mechanisms of memory formation, in this work the reference is made to consolidation rather than learning.
Future studies may look for the effects of different intervals of drug administration that may deal with the different stages of memory acquisition and learning.











 nueva página del texto (beta)
nueva página del texto (beta)