Introduction
Cosmetidae C.L. Koch, 1839 is the second most diverse family in the suborder Laniatores, with about 126 genera and 719 species (Kury, 2013). However, its genera were traditionally defined by trivial features such as armature of dorsal scutum, and number of tarsomeres in legs I and II (García & Kury, 2017; Kury & Barros, 2014). The genus MeterginusPickard-Cambridge, 1905 is currently composed of 17 species, distributed from Mexico to Brazil (Kury, 2003). Its type species, Meterginus basalisPickard-Cambridge, 1905 (Fig. 1) was described from Tamahu, Guatemala with a drawing of the habitus, in dorsal view, and a ventral view of leg IV (Pickard-Cambridge, 1905; Table 53). After that, Goodnight and Goodnight (1942) recorded the species for Mexico (Veracruz), and Kury and Cokendolpher (2000:154) maintained the species as endemic to Veracruz. In the present work we present a redescription of M. basalis that includes some photographs and drawings of the species and the first SEM images of both male and female genitalia.
Materials and methods
Geographic coordinates were interpolated between square brackets in decimal degrees to indicate that they are estimates, using Google Maps and Geonames. The distribution map was made using SimpleMappr (Shorthouse, 2010). Color descriptions use the standard names of the 267 Color Centroids of the NBS/IBCC Color System (http://people.csail.mit.edu/jaffer/Color/Dictionaries#nbs-iscc) (Jaffer, 2001) as explained in Kury (2012). Scanning electron microscopy (SEM) was carried out with a JEOL JSM-6390LV at the SEM Lab of Marine Diversity of the Museu Nacional/UFRJ, and with a Zeiss DSM940 at Instituto de Biociências da Universidade de São Paulo/USP.
Individuals were photographed in different focal planes to create a multifocal stacked image using the Leica Application Suite with a Leica DFC450 camera attached to a Leica M205C stereomicroscope and a Sony Cybershot DSC-V1 camera. The resultant images were subsequently edited in Photoshop CC 2014 software. Drawings of the species were made using Inkscape 0.91 software. The holotype was imaged at the Sackler Biodiversity Imaging Laboratory of the Natural History Museum of London (BMNH) using a Canon EOS 700D mounted to a Leica MZ12.5. stereomicroscope. Montage images were created using Helicon Focus 5.3.
The morphological terminology follows Kury and Medrano (2016) for dorsal scutum terms, and Kury and Villarreal (2015) and Medrano and Kury (2017) for macrosetae of male genitalia. Morphometric abbreviations are: AL = maximum abdominal scutum length, AW = maximum abdominal scutum width, CL = carapace length, CW = maximum carapace width, DS = dorsal scutum, Fe = femur, IOD = interocular distance, MS = macrosetae of penis, Pa = patella, Ti = tibia, VP = ventral plate. All measurements are in mm unless otherwise noted.
The examined material is deposited in MZUSP (Museu de Zoologia-Universidade de São Paulo, São Paulo, Brazil. Curator: Ricardo Pinto-da-Rocha) and NHMUK (Natural History Museum United Kingdom (formerly BMNH, British Museum of Natural History), London, England. Curator: Janet Beccaloni)). Remarks. The type exemplars of M. basalis are labeled with the acronym BMNH. The acronym GDSLV (Collection Godman & Salvin) refers to a section of the NHMUK collection.
Results
MeterginusPickard-Cambridge, 1905
Cosmetus [part]: Gervais, 1842: 4, pl. 5; 1844: 116.
Erginus [part]: Simon, 1879: 207.
Meterginus: Kury, 2003:72; Mello-Leitão, 1923: 115; 1926: 335; 1932: 86; 1933: 107; 1935: 113; Pickard-Cambridge, 1905: 568; Roewer, 1912a:103; 1923: 379; 1927: 551; 1954: 69; 1963: 57 (type species Meterginus basalisPickard-Cambridge, 1905, by original designation).
Pararhaucus [part]: Roewer, 1912b: 142.
Rhaucus [part]: Sørensen, 1932: 347.
Meterginus basalisPickard-Cambridge, 1905
Meterginus basalis: Goodnight and Goodnight, 1942: 11; 1953: 56; Kury, 2003: 72; Kury and Cokendolpher, 2000: 154; Mello-Leitão, 1932: 86; Pickard-Cambridge, 1905: 568, pl. 53, fig. 12; Roewer, 1912a: 105, fig. 10; 1923: 380, figs. 460-461 (GDLSV [BMNH 3711-2]) male holotype and female paratype, pinned).
Taxonomic summary
Type locality: Guatemala. Tamahu [15.306542, -90.234716]
Record: Mexico. Veracruz. La Buena Ventura (Goodnight & Goodnight, 1942) (dubious record, see distribution section).
Examined material. Male holotype (BMNH 3711-2). Guatemala. Tamahu. Measurements: AL: 3.90; AW: 5.65; CL: 2.80, CW: 4.33; IOD: 1.56; legs: I: 20.0; II: 36.0; III: 27.0; IV: 35.0; FeIV: 11.0; tarsal count: 7(3)/15/9/10.
New records: 4 males 3 females (MZUSP 72206): Honduras. Cortés, San Pedro Sula. Parque Nacional Cusuco, (15.535000, -88.216667), 6.viii.2015. B. N. Damron leg. Measurements (n = 3): male: AL: 3.4; AW: 4.6; CL: 1.6; CW: 3.5; IOD: 1.5; FeIV: 10.1; tarsal count: 7(3)/14(3)/9/10. Female: AL: 3.2; AW: 4.0; CL: 2.1; CW: 3.2; IOD: 0.9; legs: I: 16.0; II: 30.1; III: 21.0; IV: 28.1; FeIV: 8.9; tarsal count: 7(3)/14(3)/9/10. Female: AL: 2.2; AW: 3.1; CL: 2.5; IOD: 1.0; legs: I: 14.5; II: 31; III: 21.5; IV: 30; FeIV: 9.5; tarsal count: 7(3)/14(3)/8/11.
Redescription (based on male holotype BMNH 3711-2 and one male of MZUSP 72206):
Dorsum (Figs. 1, 2A-C, 4). Dorsal scutum beta type, smooth, with yellow blots in a reticulated pattern always delimiting the posterior border of the cephalothorax and scutal area III (Figs. 1A, 2A), sometimes the mid-line of DS exhibits some perpendicular lines (Fig. 4). Carapace smooth (Fig. 2A). Ocularium low, without median depression and with some granules (Fig. 2A-C). Lateral margins of DS without granules (Fig. 2A, B). Mesotergum delimited, divided into 4 areas: area I with a pair of low mamilliform paramedian tubercles; area II and IV smooth; area III with a pair of paramedian, high spines (Fig. 2A-D). Posterior border of scutum substraight and smooth. Free tergites I-III with some granules (Fig. 2D).

Figure 2 Male holotype of Meterginus basalis (BMNH 3711-2). A, Dorsal view; B, lateral view; C, frontal view; D, posterior view; E, ventral view. Scale bars: A = 3 mm; B, E = 4 mm; C, D = 2.5 mm.

Figure 3 Female allotype of Meterginus basalis (BMNH 3711-2). A, Dorsal view; B, lateral view; C, frontal view; D, posterior view; E, ventral view. Scale bars: 3 mm.
Venter (Fig. 2E). Stigmatic area with a few granules. Stigmata large, oval, and transverse. Coxa I with 2 parallel rows of tubercles increasing in size distally and with an anterior smooth space for the pedipalp to rest; coxa II longer than coxa I, with a row of granules; coxa III longer than I and II, granulate, with posterior margin sigmoid; coxa IV directed backward, dorsally with a granular posterior process and ventrally with a row of tubercles in the distal margin. Genital operculum slightly granulate.
Chelicera (Fig. 5A-E). Chelicera swollen. Basichelicerite quadrate, with a lot of dispersed tubercles (Fig. 5A), lower on mesal face (Fig. 5B) than those on ectal face (Fig. 5C); one group of 4 tubercles of different sizes on the ectal face sharing a common base (Fig. 5C); one group of high tubercles on the ventral face (Fig. 5D). Hand with one tubercle near the joint of the movable finger and a group of sensiliae chaetica of different sizes. Fixed finger with the inner surface finely grooved, showing an elevation in the subdistal region. Movable finger with one trapezoidal sub-basal tooth, with the inner surface at distal portion finely grooved (Fig. 5E).
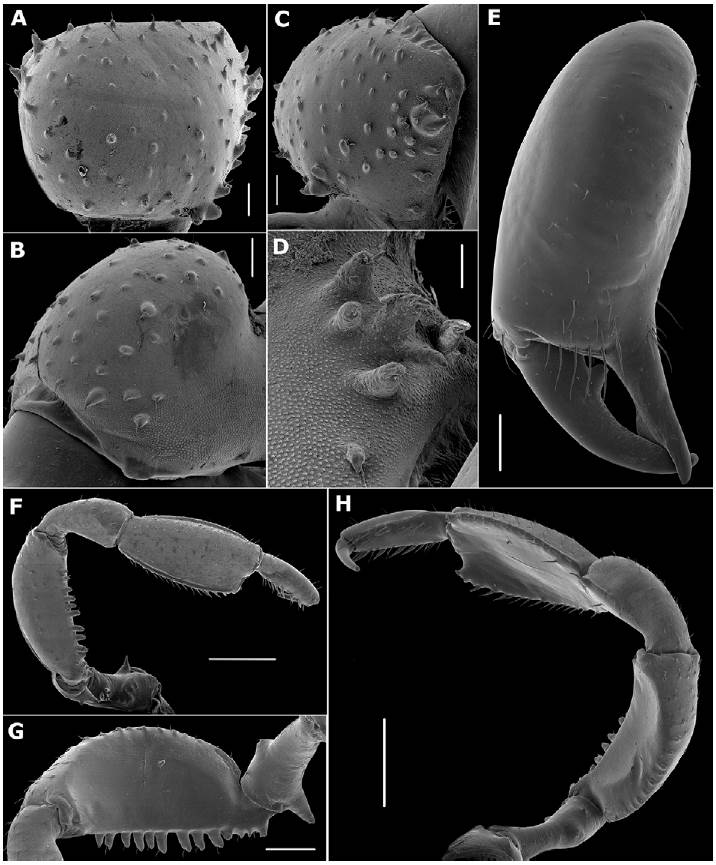
Figure 5 SEM images of Meterginus basalis male appendages (MZUSP 72206). Chelicera (A-E): A, basichelicerite, dorsal view; B, same, ectal view; C, same, mesal view; D, same, detail of ventral tubercles; E, hand, frontal view. Pedipalpus (F-H): F, ectal view; G, mesal view of trochanter and femur; H, oblique view. Scale bars: A-C = 200 μm; D = 100 μm; E, G = 500 μm; F, H = 1 mm.
Pedipalp (Fig. 5F-H). Coxa with one meso-distal granule dorsally and ventrally. Trochanter with one short dorsal tubercle and one acuminate ventral tubercle surrounded by 2 low tubercles (Fig. 5F-G). Femur compressed, with a row of dorsal tubercles and ventrally with a row of tubercles increasing in size distally, the 4 basal most lower than the others, the remaining 9 setiferous tubercles higher and conical (Fig. 5G). Patella distally depressed, with some granules; tibia depressed, oar-shaped, dorsally granulate, with lateral borders tuberculate and some spiniferous tubercles at distal portion (Fig. 5F, H); inner surface slightly striated (Fig. 5H). Tarsus long, conical, with some dorsal granules and ventrally with 2 rows of thickened setae. Claw long and sharp (Fig. 5H).
Legs (Figs. 1, 2A-D, 6). Coxae I-II each with an irregular dorsal anterior and dorsal posterior process; coxa III with one sub-basal prolateral granule (Fig. 2A); coxa IV with an irregular low shaped apophysis in the posterior margin and without clavi inguines (Fig. 2A, B). Trochanters I-III dorsally smooth and ventrally with a pair of tubercles in the distal margin; trochanter IV with some tubercles in the distal margin (Fig. 2E). Femora I-IV straight, I-III smooth; Fe IV dorsally with a row of median tubercles reaching the middle of the femur (Fig. 6A), ventrally with one row of high adjoining tubercles beginning at the base of the femur and posteriorly curving towards the retrolateral face, where they diminish in size and reach the distal part of the femur (Fig. 6A-C). Fe IV with a straight row of low tubercles arising from the middle of the structure and reaching the distal part of it and a row of small prolateral tubercles on the distal third of the femur (Figs. 1, 6B). Patellae I-IV dorsally granulate, curved; Tibiae I-IV straight and slightly granulate. Metatarsi I-IV with one ventrodistal seta. Tarsi I-II with one smooth claw; tarsi III-IV with 2 subparallel smooth claws and tarsal process (Fig. 6D).
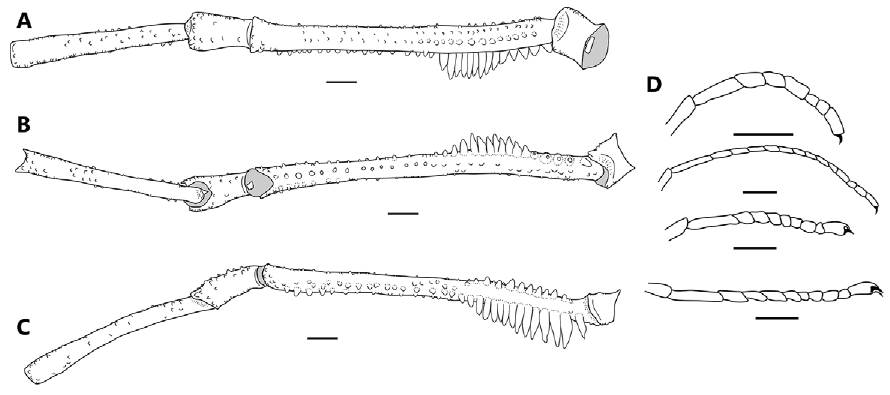
Figure 6 Leg IV and tarsomeres I-IV of Meterginus basalis male (MZUSP 72206). Leg IV, trochanter to tibia (A-C): A, dorsal view; B, ventral view; C, retrolateral view; D, tarsomeres I-IV. Scale bars = 1mm.
Penis (MZUSP 72206, Fig. 7A-C). Distal margin of ventral plate (VP) curved. Ventral surface of VP with 2 lateral, elongated, dense patches of type 4 microsetae. Lateral margins of VP widest at base closest to truncus, narrowing gradually until subapical region, then widening quickly (Fig. 7B). Two pairs of apical helicoidal macrosetae are present (type MS C), one long pair of setae just below these (type MS D1), a very reduced pair of type D located about halfway on the VP (MS D2), and 2 pairs of baso- lateral macrosetae (MS A). MS A1 (the more apical pair) is twice the length of MS A2. MS E are reduced and located ventro-laterally to MS D2 and MS D1 (Fig. 7C). MS B is polymorphic in this individual; one side has single reduced seta, but the left side has 2 (Fig. 7B). The glans and stylus are longer than the VP, glans with a thumb- like dorsal process. Stylus flattened dorso-ventrally with smooth apical edge. Stylus margins with lanceolate wattle barbels directed towards the basal portion of the structure (Fig. 7A).

Figure 7 SEM images of the genitalia of Meterginus basalis (MZUSP 72206). Male penis (A-C): A, dorso-apical view; B, ventro- apical view; C, latero-apical view. Female ovipositor (D-I): D, lateral view; E, apical view; F, general view of bifid seta; G, tip of seta; H, general view of bifid seta; I, tip of seta. Scale bars: A = 40 μm; B, C, E = 100 μm; D = 200 μm; F, H = 20 μm; G, I = 2 μm.
Female (Fig. 3). Similar to male, but differs by having anterior part of carapace narrower and coda divergent, scutal areas I and II lower, leg IV not armed, chelicerae not swollen.
Ovipositor (Fig. 7D-I). Apical portion of ovipositor is smooth, with 2 complete lobes, each with a small sulcus not completely dividing the lobe. There is a small protuberance, along the midline of the lobes, halfway between the end of the sulci and the lateral margin of the ovipositor (Fig. 7D). Ten apical macrosetae, 4 ventral (vs) and 6 dorsal (ds). The 3 pairs of ds are equidistant apart in the dorsal third of the lobe, with the most dorsal pair being next to the sulci dividing the lobes. The 2 pairs of vs show a similar pattern where they are equidistant from each other the most ventral pair being near to and in the sulcus. The macrosetae are all of similar length, but some are broken (Fig. 7E). The macrosetae are all striated in a ‘corkscrew’ pattern, with the distal tip twisting on itself (Fig. 7F, H). The macrosetae have forked tips but not all in the same pattern. Some appear to be an almost equal bifid pattern (Fig. 7G), whereas others have a prominent tip with a lateral spinule, or very reduced partner (Fig. 7I).
Distribution (Fig. 9). Guatemala and Honduras. Remarks: Several species of Meterginus have been recorded from Mexico (Kury, 2003). Besides that, the referees noted some important details: a) there are records from other species of the genus in Mexico; b) we did not study the material recorded by Goodnight and Goodnight from Mexico in order to confirm the presence of M. basalis in the country, and c) there are inaccuracies in several of Goodnight’s works about Mexican harvestmen (e.g., Goodnight & Goodnight [1942, 1953]), as commented in Kury and Cokendolpher (2000: 139). For these reasons, we decided to consider the records of Goodnight and Goodnight (1942) as dubious.
Discussion
About the penis. Male genitalia in invertebrates have been viewed as an important source of diagnostic information (Tanabe et al., 2001), and have been theorized to be under strong sexual selection by females of species (Eberhard, 1985). In harvestmen it has been drawn and described in a lot of publications, but only lately used as a source of information for identification keys (Townsend et al., 2010) and phylogenetic studies (Kury & Villarreal, 2015). For Cosmetidae, the penis morphology has been explored more widely in the last decade (e.g., García and Kury [2017] for RhaucusSimon, 1879; Kury and Barros [2014] for Taito; Medrano and Kury [2017] for EulibitiaRoewer, 1912). Here, we provide SEM images of the Meterginus basalis penis for the first time (Fig. 7A-C). In addition to this species, we imaged the male genitalia of Meterginus inermipes Roewer, 1947 (Fig. 8A, B) and Meterginus serratusRoewer, 1912 (Fig. 8C, D) for comparison. These species were selected based on the availability of specimens to the authors, superficial appearance of similarity to M. basalis, and their future use in a phylogenetic analysis of the family Cosmetidae (PhD thesis of second author).
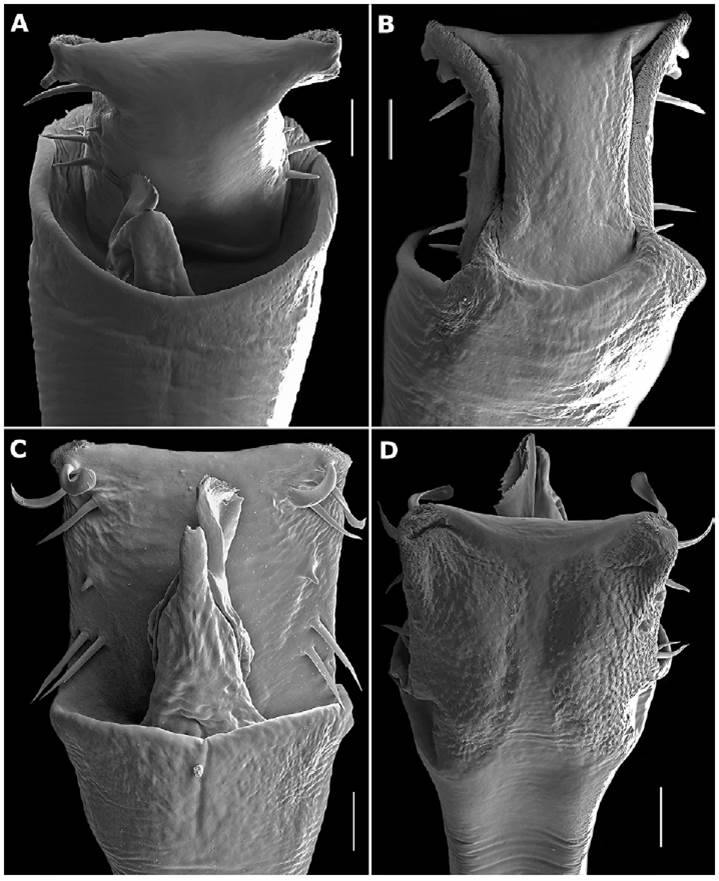
Figure 8 SEM images of the genitalia of other Meterginus species. Meterginus inermipes. Penis, apical portion: A, dorsal view; B, ventral view. Meterginus serratus. Penis, apical portion: C, dorsal view; D, ventral view. Scale bars: A-D = 40 μm.
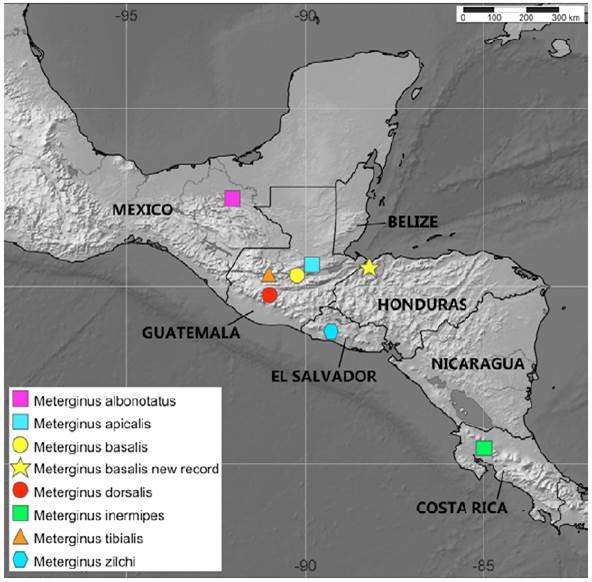
Figure 9 Map showing the distribution of Meterginus species from Central America according to type localities from Kury (2003). The records of Goodnight and Goodnight (1942) for Meterginus in Veracruz, Mexico were considered as dubious and are not indicated in the map.
Meterginus basalis and M. inermipes (type locality Costa Rica) have various similarities and differences in the penial structures. Both species have a curved apical margin, 2 MSC, a long proximate MSD, a reduced MSD2 halfway along the lateral margin, and 2 pairs of MSA, MSA1 longer than MSA2. Meterginus basalis and M. inermipes also have narrowing in the lateral margins and possess mats of microsetae (ms) type 4 along the lateral margins of the ventral surface of the VP (Fig. 8B). The significant differences between these 2 appear to be in the glans and stylus. Meterginus basalis has a glans and stylus longer than the VP length with a cteniform structure on the apical end of the stylus. Meterginus inermipes has an incredibly short glans and stylus, with the stylus presenting armature as a true caruncle, as in Ferreira and Kury (2010) (Fig. 8A).
The other species, M. serratus, iscommonlyencountered in Ecuador and Colombia. Its penis, however, differs significantly from the penis of M. basalis. Meterginus serratus and M. basalis have the same number of macrosetae on the lateral margin of the VP, but the 2 pairs of MSA at the base of the VP are similar in length in M. serratus, not of different lengths as in M. basalis and M. inermipes. The mats of type 4 ms on the ventral surface of the VP in M. serratus are more widely distributed than the ones observed in M. basalis, and almost meet at the center of the VP (Fig. 8D). The glans and stylus are similar in length to that observed in M. basalis but have a true caruncle on the apex of the stylus like M. inermipes (Fig. 8C).
The penial microsetae (ms) distribution in Gonyleptoidea was recently explored by Kury (2016), where some cosmetids were shown (Fig. 3). If we compare the arrangement of type 4 ms of some South American cosmetids -such as Rhaucus vulneratusSimon, 1879 (Fig. 3B) and Taito juruensis (Mello-Leitão, 1923) (Fig. 3D)- with type 4 ms of M. serratus presented by us, it matches entirely. In addition to that, the general square/rectangular shape of VP of M. serratus contrasts with the trapezoidal shape of the VP seen in M. basalis and M. inermipes, especially by not narrowing in the lateral margins. Finally, the body size, the DS shape, and the leg IV length of M. serratus differ widely from those of M. basalis and M. inermipes. These characteristics suggest that M. serratus does not belong to Meterginus; however, a phylogenetic analysis considering the other South American and Central American species of the genus is needed to confirm our suspicions.
About the ovipositor. Ovipositors have recently been providing characters to further help in defining new species (Villarreal & García, 2016; Walker & Townsend, 2014). Although it appears that there is a wealth of information to be found in female genitalia of Opiliones, few Cosmetidae species have been imaged and only one of those is in the genus Meterginus (excluding this work). In (Walker and Townsend 2014: Figs. 2C, 4D-F) M. inermipes was imaged and compared to other Cosmetidae species. When compared to M. basalis there are notable differences, as well as similarities. Meterginus inermipes and M. basalis both have smooth apical regions with no evidence of microsetae, the same number of peripheral macrosetae with various forms and degrees of striation, and an asymmetrical distribution (6 on anterior margin, 4 on posterior). Meterginus basalis’s ovipositor is different from that of M. inermipes in having 2 complete lobes that possess a small sulcus in each not completely dividing it, whereas M. inermipes possesses 4 complete lobes. The striation on the peripheral setae of M. basalis is also stronger than that of M. inermipes, creating strong lines and twisting of the distal tip. Meterginus basalis has forked tips on some of its peripheral setae and lateral spinules on others, whereas M. inermipes did not show any forked tips on its peripheral setae, but various forms of lateral spinules, from a singular spinule, to multiple, some sharing the same base.
At this time it is hard to say if the characters that the ovipositors of these 2 species share are indicative of relatedness. According to Walker and Townsend (2014), the number of peripheral setae can show some intraspecific variation, and so may not be a reliable character for comparative purposes. It should be noted however, that 5 M. inermipes ovipositors were examined in that study, all of which had 10 peripheral setae.
About the genus. Cosmetid genera are not well defined and/or delimited because of the problematic characters proposed by different authors (Goodnight & Goodnight, Mello-Leitão & Roewer), leading to systematic difficulties in the taxonomy and internal relationships of the family (García & Kury, 2017; Medrano & Kury, 2017). García and Kury (2017), in the revision of the Andean genus RhaucusSimon, 1879 stated that: (...) “To remedy this adverse situation and secure a grasp on the diversity in this family, and to achieve a natural composition of their genera, smaller building blocks are needed” (...). The present work intends to follow that precept to better understand Cosmetidae taxonomy, obtaining through modern techniques more informative morphological characters for Meterginus basalis, the type species of a genus that groups 17 species distributed from Mexico to Ecuador (Kury, 2003).
With the information here presented and some parallel projects (such as the preliminary results of the PhD dissertation of the second author, which shows that the species presented in this work are not closely related to one another), we can postulate that Meterginus is not a monophyletic genus. It appears that only the species restricted to mainland Central America are the “real” Meterginus species, and that those described from Colombia and Ecuador seem to be related to other genera (described or not) and may be exclusive of South America.











 nueva página del texto (beta)
nueva página del texto (beta)