More than 260 species of arbuscular mycorrhizal fungi (AMF) have been described and almost 25% of them (ca. 70 species) correspond to the genus Glomus (Glomerales, Glomeraceae) which forms glomoid spores with a single wall with few layers (Schüßler & Walker, 2010); it is distributed from xeric to humid environments worldwide. During a survey of the diversity of AMF in the Bajo Mixe region, Oaxaca, at the Gulf of Mexico coastal plain (17°29’ N, 95°37’ W; 100-300 m asl), Mexico, rhizospheric soil samples were collected from natural savannas (fide, A. Gómez-Pompa). This savanna is recognized by the herbaceous stratum dominated by the grasses Paspalum spp., Panicum spp., Hyparrhenia spp. (Poaceae) and Cyperus spp. (Cyperaceae) (Reyes & Zamora, 1977), with isolated trees of Quercus oleoides Schltdl. & Cham., Byrsonima crassifolia (L.) Kunth, and Curatella americana L.
Spores were extracted from the soil by wet sieving followed by sucrose gradient (20-60% w/v) centrifugation. Morphological characteristics and subcellular structures of the specimens were described from spores mounted in polyvinyl alcohol-lactic acid-glycerol with and without Melzer´s reagent. Observations were made with a Nomarski interference contrast microscope (Nikon Optiphot-II), and photographs were taken with a Toupcam camera (UCMOS02000KPB; ToupTek ToupView software version x64.3.7.1460). The taxonomical identification was made by comparison and contrast of morphological characters of the spores and with glomoid species at the International Collection of Vesicular and Arbuscular Mycorrhizal Fungi (INVAM) (http://invam.wve.edu/), the web page of Prof. Janusz Blaszkowski (http://www.zor.zut.edu.pl/Glomeromycota/), and Glomus species newly described (Goto et al., 2012). Voucher specimens have been deposited at the TLXM Herbarium, Centro de Investigación en Ciencias Biológicas, Universidad Autónoma de Tlaxcala.
Glomus trufemii B.T. Goto, G.A. Silva & Oehl, 2012 Sporocarps formed as loose clusters, 364-572 µm in diameter; orange-red (2.5YR5/8: Munsell Soil Color Chart) (U.S. Department of Agriculture, 1975) by reflected light, peridium and amorphous material absent. Spores formed apically on short branches of straight hyphae, elliptic, 67-94 × 90-103 µm, to subglobose, 69-97 µm, orange to orange-red (2.5YR6/8) to dark reddish brown (10R4/4) by transmitted light. Spores abortive globose, 34-63 µm, hyaline to light yellow (≈ 2.5Y8/3) when examined with transmitted light, spore wall with 2 layers sw1 and sw2 flexible. Spore wall on mature spores consisting of 2 layers, the sw1 is hyaline, fragile, 1- < 2 µm wide; the sw2 is orange brown (2.5YR7/8) to lighter orange-brown (5YR7/8) at its inner surface, 6-15 µm, laminated. Both layers continued through hyphae at the base of the spore. Pore closure a bridging septum from the sw2. Melzer’s reaction absent. Supporting hyphae light yellow (≈ 2.5Y8/4), 5-12 µm wide at the base of the spore, hyaline and narrowing to 4-10 µm beyond spore, wall 2-4 µm wide (Figs. 1-5).
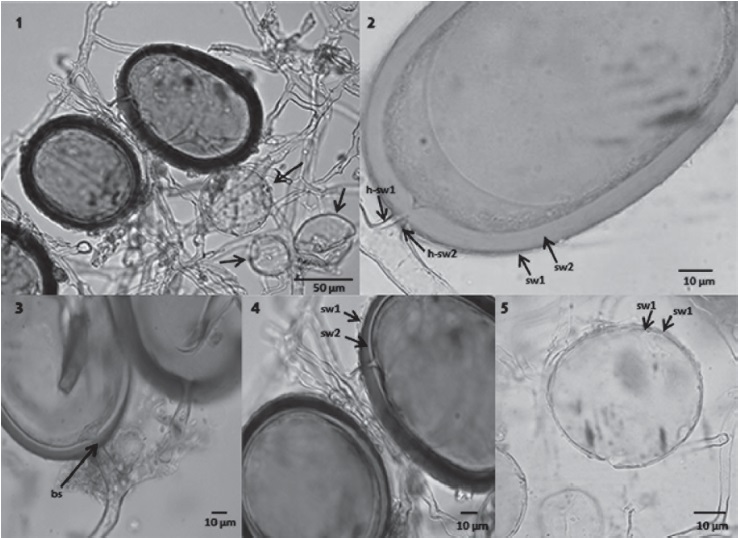
Figs. 1-5 Glomus trufemii. 1, Sporocarp with mature and abortive spores (arrows); 2, mature spore showing the 2 layers of the spore wall (sw1, sw2) and hyphal wall (h-sw1, h-sw2); 3, mature spores showing closure of the bridging septum (bs); 4, wall layer showing the external hyaline layer (sw1) and the innermost laminated layer (sw2); 5, yellowish abortive spore showing 2 flexible walls (sw1, sw2).
The orange-brown sporocarps without peridium or amorphous material, spores abortive and 2 layered spores without Melzer’s reaction are the outstanding features of Glomus trufemii. Similar glomoid sporocarpic species are G. aureum Oehl & Sieverd., which contains amorphous material, and the layer sw1 of the spore wall staining with Melzer’s reagent (Oehl et al., 2003); Funneliformis badium (Oehl, D. Redecker & Sieverd.) C. Walker & Schüßler which forms a central mass of hyphae with cystidia-like projections; G. brohultii Herrera-Peraza, Ferrer & Sieverd., which has hyphae recurved, semi-bulbose or bifurcated at the base of the spores (Herrera-Peraza et al., 2003).
Glomus trufemii was described from spores recovered from soils of sand dune shrub land in Brazil (Goto et al., 2012). Until now, this species had not been recorded from anywhere else; however, it now appears that it may have a broader tropical distribution in the Americas. The host species of G. trufemii are still unknown; however, they are likely grasses (Poaceae and Cyperaceae), due to their dominance in savanna ecosystems.
Thanks to the IPN for the logistical support (SIP 20161449), to J. Błaszkowski and B. Goto for the review of photographs of the specimens and comments, to anonymous reviewers of a previous version of the manuscript, to J. Newhall Williams for the revision of the language. Jonás Álvarez-Lopeztello acknowledges to Conacyt for his Doctorate Scholarship 266682.











 nova página do texto(beta)
nova página do texto(beta)


