Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista mexicana de biodiversidad
versión On-line ISSN 2007-8706versión impresa ISSN 1870-3453
Rev. Mex. Biodiv. vol.81 no.3 México dic. 2010
Notas científicas
A new record of Atractus boettgeri (Serpentes: Colubridae), with notes on taxonomy and natural history
Un registro nuevo de Atractus boettgeri (Serpentes: Colubridae), con comentarios sobre la taxonomía e historia natural de la especie
Jorge Salazar–Bravo1*, Julieta Vargas2, Agustín Jimenez–Ruiz3 and Jay M. Savage4
1 Department of Biological Sciences, Texas Tech University, Lubbock, Texas 79409, USA. *Correspondent: j.salazar–bravo@ttu.edu
2 Colección Boliviana de Fauna, casilla 8706, La Paz, Bolivia
3 Department of Zoology, Southern Illinois University, Carbondale, Illinois 62901–6501, USA.
4 Rana Dorada Enterprises, S.A., San Diego, CA 92116–2490, USA.
Recibido: 06 febrero 2009
Aceptado: 20 enero 2010
Abstract
We report a range extension of Atractus boettgeri, a rare snake endemic to Bolivia. This species differs from Atractus taeniatus by a higher segmental count (well outside the range for A. taeniatus) and by having only 6 maxillary teeth as opposed to 8–9 in A. taeniatus. In addition, A. boettgeri differs from A. emmeli in having 6–6 supralabials (versus 7–7) and fewer ventrals (175–177 versus 181–189 in females). All known records of A. boettgeri indicate an association between this species and the Cerrado vegetation of central Bolivia. This report is also unique in that the specimen reported herein was found in the stomach of a Common Long–Nosed Armadillo (Dasypus novemcinctus).
Key words: Atractus boettgeri, Atractus taeniatus, Atractus emmeli, Bolivia.
Resumen
Se reporta una extensión de en la distribución de la especie Atractus boettgeri, una serpiente endémica de Bolivia. Esta especie se diferencia de A. taeniatus por tener un número segmental muy por encima del intervalo descrito para A. taeniatus y por tener sólo 6 dientes maxilares, en vez de 8–9 en A. taeniatus. Además, A. boettgeri se diferencia de A. emmeli por tener escamas supralabiales en patrón 6–6 (en vez de 7–7) y tener un conteo de escamas ventrales menor en hembras (175–177 versus 181–189). Incluyendo éste, todos los registros conocidos para A. boettgeri parecen estar asociados con la vegetación del Cerrado en Bolivia. El ejemplar se encontró en el estómago del tatú común (Dasypus novemcinctus).
Palabras claves: Atractus boettgeri, Atractus taeniatus, Atractus emmeli, Bolivia.
The genus Atractus includes over 100 species of semi–fossorial colubrids distributed from eastern Panama (Myers, 2003) to southern Brazil and northern Argentina (Giraudo and Scrocchi, 2000; Schargel and Castoe, 2003). At present, the taxonomy of Atractus is poorly understood, in part due to the paucity of museum records, but also due to the lack of a modern revision on a continental scale (Passos et al., 2005). The available data suggest that most species in the genus have restricted distributions (Jorge da Silva Jr et al., 2005), although it has also been suggested that this may be a sampling artifact of their semi–fossorial habits (see Myers, 2003 for a discussion on Panamanian species). Regardless, little is known about the ecology or evolution of many of its species (see Zaher et al., 2005 for an exception). Atractus boettgeri is a Bolivian endemic (González and Reichle, 2003) currently known from only 2 localities in central Bolivia: the type locality in Cochabamba (Cunha and Nascemento, 1983) and a second locality in the Department of Santa Cruz (McCoy, 1971). Here we report a third locality and specimen of A. boettgeri from the Department of El Beni; this new record extends the known range of the species almost 100 km east of the type locality and more than 300 km north of the Santa Cruz de la Sierra record.
A specimen of Atractus boettgeri was found in the stomach of a Common Long–Nosed Armadillo (Dasypus novemcinctus) at the following specific locality: Bolivia; Department Beni: 1.4 km N of San Pablo; 15° 14' 32"S and 63° 52' 43" W (GPS datum WGS 84), 195 m elev. Both, the position of the snake in the digestive tract of the armadillo and the condition of the snake suggest that it had been ingested no more than a couple of hours before the armadillo was shot (at ca. 5 a.m.) by local farmers. The snake is currently cataloged in the Herpetology Division of the Texas Natural History Collections, University of Texas at Austin (TNHC 61951). The skull of the armadillo is cataloged in the Division of Mammals at the Museum of Southwestern Biology, University of New Mexico (MSB 99077).
The snake is a female with a standard length of 244 mm and a tail length of 19 mm (7.2% of total length). There had been some digestion of the anterior head region (Fig 1), but it is clearly a representative of the colubrid genus Atractus because it has only 1 pair of genials and 1+2 temporals. Other scutellation features are: dorsal scales smooth in 15–15–15 rows; rostral smaller than either prefrontal; internasals small, much less than half the size of either prefrontal; prefrontals long, at least one and a half times as long as broad; loreal elongate, bordering orbit; no preocular; postoculars 1–1; temporals 1+2/1+2; 6–6 supralabials, 3–4/3–4 bordering orbit; infralabials 7–7, 4 bordering genials; subcloacal plate entire; ventrals 175; subcaudals 25; ventrals plus subcaudals 200; 6 maxillary teeth.
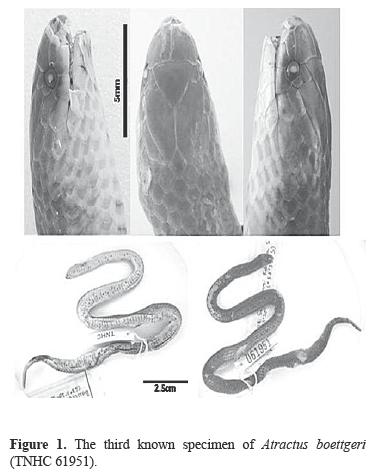
A brown head cap extends from the tip of the snout back to the level of the posterior margin of the orbit. The cap is followed by a lighter brown head band extending across the posterior part of the head onto the first dorsal scale row. This appears to be a light head band probably suffused with darker pigment ontogenetically. The dorsal and caudal surfaces are brown with considerable dark pigment concentrated at the tip of some scales to produce scattered dark spots. These spots tend to line up as a faint dark middorsal stripe on the posterior part of the body. The labials, chin, and throat are light with some brown spotting. The venter is yellowish and speckled with brown pigment which tends to be concentrated as irregular broken lines on the posterior margin of each ventral. The subcaudals are heavily marked with brown.
On the specific identity of TNHC 61951.
Three species described originally from Bolivia were considered as possibly conspecific with our specimen: Atractus boettgeri Boulenger, 1896 (Bolivia: Cochabamba: Sierra de las Yungas), Atractus emmeli Boettger, 1888 (Bolivia: La Paz: Río Mapiri, 490 m), and Atractus taeniatus Griffin, 1916 (from Bolivia: Santa Cruz: Santa Cruz de la Sierra, 480 m). The type specimens for these names agree with TNHC 61951in having 15–15–15 dorsal scale rows, and in the number and conformation of the head scales, except they have 2–2 postoculars instead of 1–1. Moreover, A. emmeli differs from the other 2 species and TNHC 61951 in having 7–7 supralabials. Segmental counts of the type specimens of the 3 candidate species are presented in Table 1. The types of both A. emmeli and A. boettgeri have essentially uniform dark upper surfaces, but that of A. taeniatus has a light head band bordered posteriorly by a dark band, a dark middorsal stripe, and a dark midventral stripe on a light ground color.
Boulenger (1896) listed additional examples of Atractus emmeli from Bolivia with the following segmental counts: 2 males with 158–167 ventrals, 27 subcaudals; 185–194 totals; and 3 females with 181–189 ventrals, 21–26 subcaudals; 202–215 totals. He also referred a female specimen from Belem do Para, Brazil to A. emmeli. The segmental counts (161 ventrals, 16 subcaudals, and 177 total) are well below those for females from Bolivia (181–189; 21–26; 202–215) and this snake doubtless represents a different species. Schmidt and Walker (1943) identified a specimen at the Universidad de Arequipa from an unknown locality in Peru as A. emmeli. The snake is a female with 188 ventrals, 22 subcaudals; 210 totals, and has dark spots in its dorsal pattern. We suspect that this is an undescribed Peruvian species and have not used its characteristics in comparisons.
McCoy (1971) reported on an additional specimen (CM 2822, a female) of a putative A. boettgeri from Bolivia: Santa Cruz: Santa Cruz de la Sierra (480 m), with 175 ventrals, 22 subcaudals (total 197). He also reported a male of A. taeniatus (CM 2932) with 151 ventrals, 23 subcaudals (total 174) from Bolivia: Santa Cruz: Buenavista, 400 m. The upper surfaces of the male were uniform brown, a light head band was present, and the ventrals were light with dark edges. The upper surfaces of the female were brown and the venter spotted with brown. McCoy (1971) concluded that the differences in scale counts between A. boettgeri and A. taeniatus were due to sexual dimorphism and that the presence of a light head band was a juvenile feature as the Buenavista specimen was 149 mm in total length. On this foundation, he consequently synonymized A. taeniatus with A. boettgeri.
Williams and Gudynas (1991) described samples from Argentina (Buenos Aires, Entre Ríos and Missiones Provinces), which conformed closely in coloration with the type of A. taeniatus. In their series of 2 males and 4 females the segmental counts were 146–150 ventrals, 24–33 subcaudals and 141–153 ventrals, 25–30 subcaudals, respectively. Maxillary teeth numbered 8–9 and all had a light head band. The dorsal pattern consisted of a regular to irregular mid–dorsal dark stripe or a series of elongate dark spots corresponding to an interrupted mid–dorsal stripe; the venter was yellow with only small brown spots not organized into a distinct midventral stripe. The type of A. boettgeri and McCoy's Santa Cruz de la Sierra specimen (both females) had 177 and 175 ventrals, 20 and 22 subcaudals, respectively, for a total of 197 in both examples. Williams and Gudynas (1991) concluded that A. boettgeri was a valid taxon as there was no overlap in segmental counts with the larger sample of A. taeniatus from Argentina. Both of these species co–occur at Santa Cruz de la Sierra, further confirming their validity.
Atractus emmeli (Boettger, 1888; Boulenger, 1896) differs from both A. boettgeri and A. taeniatus in having 158–170 ventrals in males and 181–189 in females compared with A. boettgeri with 175–177 ventrals in females, and A. taeniatus with 146–152 ventrals in males, 141–153 in females. It also has 7–7 supralabials versus 6–6 in the other 2 species.
The snake reported here (TNHC 61951) has segmental counts (175 ventrals, 25 subcaudals; 200 totals) well outside the range for A. taeniatus. It further differs from A. taeniatus in having 6 maxillary teeth in contrast to 8–9 in the latter. It differs from A. emmeli in having 6–6 supralabials (versus 7–7) and fewer ventrals (175–177 versus 181–189 in females). We thus conclude that it is the third known representative of A. boettgeri. Interestingly, the dorsal pattern in this snake is reminiscent of that in A. taeniatus, having a series of interrupted elongate spots in place of the mid–dorsal dark stripe, while the type of A. boettgeri and the specimen reported by McCoy (1971) have an essentially uniform pattern. As pointed out by Savage (1960), this is not an uncommon level of intraspecific variation in coloration for Atractus.
Detailed comparisons between the holotype of A. boettgeri and various specimens of A. albuquerquei were conducted by Zaher et al. (2005). Except for the fact that the snake reported here (TNHC 61951) shows the highest count of subcaudals of any of the 3 known specimens of A. boettgeri (25, instead of 20 in the holotype and 23 in the specimen reported by McCoy, 1971), and that it shows only 1 postocular scale on both sides (versus 2+2 in both the holotype and McCoy's specimen), all differences reported in Zaher et al. (2005) between boettgeri and albuquerquei are also evident for TNHC 61951. Moreover, our specimen resembles the holotype of A. boettgeri depicted in Zaher et al. (2005) in all qualitative characters.
The 3 known records of A. boettgeri are associated with the Cerrado savannas of central Bolivia (Fig 2). Interestingly, the Cerrado biome was recognized by Myers et al. (2000) as a center of high endemism for several groups of vertebrates, including reptiles (>20% of endemism). In fact, the Cerrado is included among the 25 biodiversity hotspots of the world because of its large number of endemic species and the high levels of human disturbance (Myers et al., 2000).
Snakes have been reported from the diet of the Common Long–Nosed Armadillo in Alabama and Florida (Breece and Dusi, 1985; Wirtz et al., 1985), but apparently not from South America (Redford, 1985; Anacleto, 2007). Several general accounts of armadillo ecology mention the fact that some species feed on small vertebrates. Our report and that of Bezerra et al. (2001) support these claims at least for colubrid snakes and mice, respectively. Large quantities of soil and a few insects (Formicidae, Scarabeidae) were also found in the stomach of the Long–Nosed Armadillo.
We thank the Colección Boliviana de Fauna for granting the permits to work in Bolivia. Special thanks to Jon Dunnum (Division of Mammals, Museum of Southwestern Biology, University of New Mexico) for confirming the identification of the armadillo. This research was supported in part by a grant from the National Science Foundation to M.L. Jameson and F.C. Ocampo (NSF–DBI 0500767).
Literature cited
Anacleto, T. 2007. Food habits of four armadillo species in the Cerrado area, Mato Grosso, Brazil. Zoological Studies 46:529–537. [ Links ]
Bezerra, A. M. R., F. H. G. Rodrigues and A. P. Carmignotto. 2001. Predation of rodents by the yellow armadillo (Euphractus sexcinctus) in Cerrado of the Central Brazil. Mammalia 65:86–88. [ Links ]
Boettger, O. 1888. Beitrag zur Reptilien des oberen Beni in Bolivia. Bericht der Senckenbergischen Naturforschenden Gesellschaft in Frankfurt am Main 1888:191–199. [ Links ]
Boulenger, G. A. 1896. Catalogue of the snakes in the British Museum (Natural History). Vol. 3. Taylor & Francis, xiv + 727 p. [ Links ]
Breece G. A. and J. L. Dusi. 1985. Food habits and home ranges of the common long–nosed armadillo Dasypus novemcinctus in Alabama. In Ecology of armadillos, sloths, and vermilinguas, G. G. Montgomery (ed.). Smithsonian Institution Press. Washington, D.C. p. 419–428. [ Links ]
Cunha, O. R. Da and F.P. Do Nascimento. 1983, Ofídios da Amazônia XX–as espécies de Atractus Wagler, 1828, na Amazônia oriental e Maranhão. (Ophidia, Colubridae). Boletim Museu Paraense E. Goeldi, nova serie Zoologia 123:1–38. [ Links ]
Giraudo, A. R. and G. J. Scrocchi. 2000. The genus Atractus (Serpentes: Colubridae) in north–eastern Argentina. Herpetological Journal 10:81–90. [ Links ]
González, L. and S. Reichle. 2003. Lista de reptiles presentes en Bolivia. Anexo 3. In Biodiversidad: La riqueza de Bolivia. Estado de conocimiento y conservación, P. Ibisch and G. Mérida (eds.). Ministerio de Desarrollo Sostenible. Editorial FAN, Santa Cruz de la Sierra, Bolivia. p. 587–589. [ Links ]
Griffin, L. E. 1916. A catalog of the Ophidia from South America at present (June, 1916) contained in the Carnegie Museum, with descriptions of some new species. Memoirs of the Carnegie Museum 7:163–228. [ Links ]
Jorge Da Silva Jr, N., H. L. R. Silva, R. S. Ribeiro, I. Souza and C. D. A. Souza. 2005. A new species in the genus Atractus Wagler, 1928 (Colubridae: Dipsadinae) in the Cerrado region of Central Brazil. Papeis Avulsos de Zoologia (Sao Paulo) 45:33–39. [ Links ]
Josse, C., G. Navarro, P. Comer, R. Evans, D. Faber–Langendoen, M. Fellows, G. Kittel, S. Menard, M. Pyne, M. Reid, K. Schulz, K. Snow and J. Teague. 2003. Ecological systems of Latin America and the Caribbean: A working classification of terrestrial systems. Natureserve, Arlington. 47 p. [ Links ]
McCoy, C. J. 1971. Comments on Bolivian Atractus (Serpentes, Colubridae). Herpetologica 27:314–316. [ Links ]
Myers, C. W. 2003. Rare snakes – Five new species from eastern Panama: Reviews of northern Atractus and southern Geophis (Colubridae: Dipsadinae). American Museum Novitates 391:1–47. [ Links ]
Myers, N., R. Mittermier, C. Mittermier, G. Fonseca and J. Kent 2000. Biodiversity hotspots for conservation priorities. Nature 403:853–858. [ Links ]
Passos, P., R. Fernandes and N. Zanella. 2005. A new species of Atractus (Serpentes: Colubridae) from southern Brazil. Herpetologica 61:209–218. [ Links ]
Redford, K. H. 1985. Foods habits of armadillos (Xenarthra: Dasypodidae). In Ecology of armadillos, sloths, and vermilinguas, G. G. Montgomery (ed.). Smithsonian Institution Press, Washington, D.C. p. 429–437. [ Links ]
Savage, J. M. 1960. A revision of the Ecuadorian snakes of the colubrid genus Atractus. Miscellaneous Publications Museum of Zoology, University of Michigan 112:1–86. [ Links ]
Schargel, W. E. and T. A. Castoe. 2003. The hemipenes of some snakes of the semifossorial genus Atractus, with comments on variation in the genus. Journal of Herpetology 37:718–721. [ Links ]
Schmidt, K. P. and W. F. Walker, Jr. 1943. Peruvian snakes from the University of Arequipa. Zoological Series of Field Museum of Natural History 24:279–296. [ Links ]
Williams, J. D. and E. Gudynas. 1991. Revalidation and redescription of Atractus taeniatus Griffin, 1916 (Serpentes: Colubridae). Contribuciones en Biologia Centro Investigacion y Promocion Franciscano y Ecologia Centro Educacion Don Orione 15:1–8. [ Links ]
Wirtz W. O., D. H. Austin and G. W. Dekle. 1985. Food habits of the common long–nosed armadillo Dasypus novemcinctus in Florida, 1960–1961. In Ecology of armadillos, sloths, and vermilinguas, G. G. Montgomery (ed.). Smithsonian Institution Press, Washington, D.C. p. 439–451. [ Links ]
Zaher, H., I. Souza, D. J. Gower, E. Hingst–Zaher and N. Jorge Da Silva, Jr. 2005. Redescription of Atractus albuquerquei (Serpentes: Colubridae: Dipsadinae), with comments on geographical distribution and intraspecific variation. Papeis Avulsos de Zoologia (São Paulo) 45:19–32. [ Links ]














