Serviços Personalizados
Journal
Artigo
Indicadores
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Revista mexicana de biodiversidad
versão On-line ISSN 2007-8706versão impressa ISSN 1870-3453
Rev. Mex. Biodiv. vol.79 no.2 México Dez. 2008
Taxonomía y Sistemática
Morphological and genetic comparative analyses of populations of Zoogoneticus quitzeoensis (Cyprinodontiformes: Goodeidae) from Central Mexico, with description of a new species
Análisis comparativo morfológico y genético de diferentes poblaciones de Zoogoneticus quitzeoensis (Cyprinodontiformes:Goodeidae) del Centro de México, con la descripción de una especie nueva
Domínguez–Domínguez Omar1*, Pérez–Rodríguez Rodolfo1 and Doadrio Ignacio2
1 Laboratorio de Biología Acuática, Facultad de Biología, Universidad Michoacana de San Nicolás de Hidalgo, Fuente de las Rosas 65, Fraccionamiento Fuentes de Morelia, 58088 Morelia, Michoacán, México
2 Departamento de Biodiversidad y Biología Evolutiva, Museo Nacional de Ciencias Naturales, José Gutiérrez Abascal 2, 28006 Madrid, España.
*Correspondent:
odoming@jupiter.umich.mx
Recibido: 17 septiembre 2007
Aceptado: 24 enero 2008
Abstract
A genetic and morphometric study of populations of Zoogoneticus quitzeoensis (Bean, 1898) from the Lerma and Ameca basins and Cuitzeo, Zacapu and Chapala Lakes in Central Mexico was conducted. For the genetic analysis, 7 populations were sampled and 2 monophyletic groups were identified with a genetic difference of DHKY= 3.4% (3–3.8%), one being the populations from the lower Lerma basin, Ameca and Chapala Lake, and the other populations from Zacapu and Cuitzeo Lakes. For the morphometric analysis, 4 populations were sampled and 2 morphotypes identified, 1 from La Luz Spring in the lower Lerma basin and the other from Zacapu and Cuitzeo Lakes drainages. Using these 2 sources of evidence, the population from La Luz is regarded as a new species Zoogoneticus purhepechus sp. nov. The new species differs from its sister species Zoogoneticus quitzeoensis in having a shorter preorbital distance (Prol/SL  = 0.056, SD = 0.01), longer dorsal fin base length (DFL/SL
= 0.056, SD = 0.01), longer dorsal fin base length (DFL/SL  = 0.18, SD = 0.03) and 13–14 rays in the dorsal fin. The new species differs from both members of its sister taxon (Zoogoneticus tequila and Z. quitzeoensis) at 10 fixed nucleotide positions in the cytochrome b gene. We have determined that Zoogoneticus purhepechus is distributed in the lower Lerma, upper Ameca, Armeria and Santiago river basins, and Chapala Lake. This new species should be considered endangered of extinction according to the criteria of the MER (Aii,Bi,Ci,Di) and for the IUCN (A–1,b,c,e).
= 0.18, SD = 0.03) and 13–14 rays in the dorsal fin. The new species differs from both members of its sister taxon (Zoogoneticus tequila and Z. quitzeoensis) at 10 fixed nucleotide positions in the cytochrome b gene. We have determined that Zoogoneticus purhepechus is distributed in the lower Lerma, upper Ameca, Armeria and Santiago river basins, and Chapala Lake. This new species should be considered endangered of extinction according to the criteria of the MER (Aii,Bi,Ci,Di) and for the IUCN (A–1,b,c,e).
Key words: Zoogoneticus, cytochrome b, new species, Mesa Central, Mexico, morphometry.
Resumen
Se realizó un estudio genético y morfométrico en poblaciones de Zoogoneticus quitzeoensis (Bean, 1898) pertenecientes a las cuencas de los ríos Lerma y Ameca y los lagos de Cuitzeo, Zacapu y Chapala en el centro de México. Para el análisis genético se analizaron 7 poblaciones, identificándose 2 grupos monofiléticos bien diferenciados, con distancias genéticas entre ellos de DHKY = 3.4% (3–3.8%), uno de los grupos se distribuye por las cuencas de los ríos Ameca y bajo Lerma y en el lago de Chapala, mientras que el otro incluye las poblaciones de los lagos de Zacapu y Cuitzeo. Se emplearon 4 poblaciones para los análisis morfométricos identificándose 2 morfotipos, 1 de la localidad del manantial La Luz en la cuenca del bajo Lerma y el otro a los lagos de Zacapu y Cuitzeo. Con estas 2 fuentes de evidencia, la población de La Luz es considerada como una nueva especie Zoogoneticus purhepechus n. sp. La especie nueva difiere de su especie hermana, Z. quitzeoensis por tener una distancia preorbital más corta (PrOL/SL  = 0.05 –0.06), la base de la aleta dorsal más larga (DFL/SL
= 0.05 –0.06), la base de la aleta dorsal más larga (DFL/SL  = 0.17–0.20) y presentar entre 13 y 14 radios en la aleta dorsal. La especie nueva difiere de las 2 especies descritas en el género (Zoogoneticus tequila y Z. quitzeoensis) en 10 posiciones nucleotídicas fijadas para el gen citocromo b. Zoogoneticus purhepechus se distribuye por las cuencas de los ríos Ameca, Armería, Santiago y bajo Lerma, así como en el lago de Chapala. Z. purhepechus debe ser considerada en peligro de extinción de acuerdo a los criterios del MER (Aii,Bi,Ci,Di) y de la UICN (A–1,b,c,e).
= 0.17–0.20) y presentar entre 13 y 14 radios en la aleta dorsal. La especie nueva difiere de las 2 especies descritas en el género (Zoogoneticus tequila y Z. quitzeoensis) en 10 posiciones nucleotídicas fijadas para el gen citocromo b. Zoogoneticus purhepechus se distribuye por las cuencas de los ríos Ameca, Armería, Santiago y bajo Lerma, así como en el lago de Chapala. Z. purhepechus debe ser considerada en peligro de extinción de acuerdo a los criterios del MER (Aii,Bi,Ci,Di) y de la UICN (A–1,b,c,e).
Palabras clave: Zoogoneticus, cytochrome b, Mesa Central, México, morfometría.
Introduction
The Mesa Central of Mexico is characterized by its high diversity of freshwater fishes (Barbour, 1973; Echelle and Echelle, 1984; Domínguez–Domínguez et al., 2005). A total of 100 native species have been reported, of which 70% are endemic (Guzmán–Arroyo, 1994). This important biological diversity has been attributed to the complex geological and zoogeographic history of central Mexico (Miller and Smith, 1986; Domínguez–Domínguez et al., 2006a). Of the endemic fish fauna of the Mesa Central, the cyprinodontiform fish subfamily Goodeinae (family Goodeidae) is one of the most diverse and interesting. The subfamily exhibits internal fertilization, matrotrophy and viviparity (Parenti, 1981; Grudzien et al., 1992). When the genus Zoogoneticus Meek, 1902, was described, the 14 species of goodeines were included in the Poeciliidae, which also comprised the presently recognized families Profundulidae, Fundulidae, Rivulidae, Cyprinodontidae, and Anablepidae (sensu Parenti, 1981). Meek (1904) placed Fundulus robustus Bean 1892, Platypoecilus quitzeoensis Bean, 1898 and Fundulus dugesii Bean, 1887 in Zoogoneticus, and simultaneously described 2 new species (Z. diazi Bean, 1887 and Z. miniatus Bean, 1887).
Regan (1908) proposed the synonymy of Z. miniatus with Z. diazi and Z. maculatus Regan 1904 with Z. robustus (Bean, 1892). The revision by Hubbs andTurner (1939), based on the anatomy of the ovary and the trophotaeniae, restricted the genus to include only Z. quitzeoensis (Bean, 1898), removing other taxa to what are presently 3 different genera of goodeines (Allotoca Hubbs and Turner, 1937, Alloophorus Hubbs and Turner, 1937 and Allodontichthys Hubbs and Turner, 1937). Based on molecular characters the genus Zoogoneticus is currently placed in the tribe Chapalichthyini (sensu Doadrio and Domínguez, 2004). The genus is currently comprised of the species Z. quitzeoenesis and Z. tequila Webb and Miller, 1988. Zoogoneticus quitzeoensis is widely distributed in Central Mexico whereas Z. tequila has a restricted distribution. The former is considered endangered and the second has been considered extinct in the wild (Espinosa–Pérez et al., 1993; Webb and Miller, 1998; SEMARNAT 2002), although a small and restricted population was recently reported (De la Vega–Salazar et al., 2003). Genetic studies have shown that populations of Z. quitzeoensis have a geographical structure with a consistently high degree of genetic divergence among populations (Doadrio and Domínguez, 2004; Domínguez–Domínguez et al., 2007).
The causes of the ancient population structure may be explained by several volcanic and tectonic events during the Plio–Pleistocene; the population has been subject to different events of dispersion and vicariance that differ in spatial and temporal scale (Domínguez–Domínguez et al., 2006a). Thus, genetic and morphological differences within and between populations have been observed in other non–goodeid fishes from Central Mexico including Poeciliopsis infans (Woolman, 1894) (Mateos et al., 2002) and the genus Notropis Rafinesque 1818 (Schönhuth and Doadrio, 2003).
According to a phylogenetic hypothesis proposed by Doadrio and Domínguez (2004), the westernmost populations of Z. quitzeoensis are genetically different from populations from the Lake Zacapu and La Mintzita, the latter in the Lake Cuitzeo systems. These results suggest that a morphological and more extensive molecular revision of Zoogoneticus needs to be conducted to better establish the taxonomic identity of the different populations of the genus that still exist in nature. These patterns of variation between population in the lower and middle Lerma drainages, with evidence of separated closely related clades, are paralleled by the sister species Skiffia lermae Meek 1902, and S. multipunctata (Pellegrin, 1901). It is hypothesized that both groups of species owe their origins to the same vicariant event between 1 and 3.5 Mya (Domínguez–Domínguez et al., 2006a).
The purpose of this study, therefore, is to analyze the morphologic and genetic differences among populations of the genus Zoogoneticus and to provide the description of a newly recognized species.
Materials and methods
The study was based on specimens collected using hand and seine nets and electrofishing. All of the sampled specimens were preserved in 70% ethanol. Voucher specimens are housed at the Universidad Michoacana de San Nicolás de Hidalgo (CPUM), the Museo Nacional de Ciencias Naturales de Madrid (MNCN) and the Instituto de Biología, Universidad Nacional Autónoma de México (CNP). The tissues used in genetic analysis are housed at the Museo Nacional de Ciencias Naturales de Madrid (voucher numbers MEX 38, 4271, 4272, 506 and 508).
Morphological analysis. Specimens from the type locality (San Cristobal, Cuitzeo Lake, Michoacan) and 3 other populations of Z. quitzeoensis, (La Luz–Spring, Zamora, Michoacan; La Mintzita Spring, Morelia, Michoacan and Zacapu Lake, Zacapu, Michoacan) were analyzed (Table 1 and Fig. 1). Twenty morphometric characters were measured with digital calipers (0.01 mm) and 4 meristic variables were recorded using a stereoscopic microscope. The abbreviations used for morphometric variables are: SL, standard length; HL, head length; PrOL, preorbital length; ED, eye diameter; InOW, interorbital width; BD, body depth; BLD, body least depth; PAD, pelvic–anal fin distance; PDD, pelvic–dorsal fin distance; PODE, pelvic fin origin to dorsal fin posterior extent distance; DAD, dorsal–anal fin distance; DOAE, dorsal fin origin to anal fin posterior extent distance; DFL, dorsal fin length; DEAO, dorsal fin posterior extent to anal fin origin distance; AFL, anal fin length; AEDE, anal fin posterior extent to dorsal fin posterior extent distance; EDUP, end of dorsal fin–upper extreme of caudal peduncle distance; EDLP, end of dorsal fin–lower extreme of caudal peduncle distance; EAUP, end of the anal fin–upper extreme of caudal peduncle distance; EALP, end of the anal fin– lower extreme of caudal peduncle distance. The abbreviations for meristic characters are: D, dorsal fin rays; A, anal fin rays; P, pectoral fin rays; GR, gill rakers. All the measurements are in millimetres.
A two–way analysis of variance (ANOVA), comparing both morphometric and meristic characters, was conducted to test sexual dimorphism and variation between populations. Burnaby's method was used to correct size effect (Burnaby, 1966; Rohlf and Bookstein, 1987; Doadrio et al., 2002). All analyses were conducted with the corrected matrix. To identify the variables that contributed most to the variability between populations, a principal components analysis (PCA) was conducted using the covariance matrix for morphometric characters and the correlation matrix for meristic characters. The classificatory hypothesis obtained by PCA was tested by a discriminate function analysis (DFA). All analyses were conducted with the statistics packages NTSYS v.2.1 (Rohlf, 2000) and SPSS v.13.0. Both sets of characters, morphometric and meristic, were analyzed independently. Because of sexual dimorphism involved in the morphometric measurements, these were analysed separately for males and females.
Genetic analysis. Six sequences of the gene Cytochrome b of Zoogoneticus spp. from different localities (Table 1 and Fig. 1) were obtained from GenBank (AF510751–AF510755 and AF510757) and 1 was obtained for the outgroup (Xenoophorus captivus (Hubbs, 1924), AF510758). The other sequences (including specimens from the type locality of the new species and specimens from the type locality for Z. quitzeoensis) were obtained using the following protocol. Total cellular DNA was isolated from tissues by a standard proteinase K and phenol/chloroform extraction method (Sambrook et al., 1989). Two overlapping fragments of the cytochrome b gene (total of 1140 bp) were ampliï¬ed via polymerase chain reaction (PCR) for each individual DNA sample.
The primers used for cytochrome b in all species were those discussed in Machordom and Doadrio (2001). The amplification process was conducted as follows: 94 °C (2 min), 35 cycles at 94 °C (45 s), 48 °C (1 min), 72 °C (90 s), and 72 °C (5 min). PCR mixtures were prepared in 25 µl reactions with a final concentration of 0.4 µM of each primer, 0.2mM of each dNTP, 1.5mM MgCl2, and 1U of Taq DNA polymerase (Biotools). PCR products were checked on 1.5% agarose gels, and cloned using the pGEM–T vector (Promega) into Escherichia coli JM109. Positive clones were sequenced using the Big Dye Deoxy Terminator cycle–sequencing kit (Applied Biosystems). DNA sequences of both strands were obtained using M13 universal (forward and reverse) sequencing primers. All samples were sequenced on an Applied Biosystems 3700 DNA sequencer following manufacturer's instructions. Chromatograms and alignments were visually checked.
The model of DNA substitution that best fitted the data set was selected using Modeltest 3.7 (Posada and Crandall 1998) using the Bayesian information criterion (BIC). The aligned data were analysed with the Bayesian inference method with the program Mr. Bayes 3.1.1 (Hueselsenbeck and Ronquist, 2001) by simulating a Markov chain for 1,000,000 cycles. Based on the HKY+G model obtained by Modeltest, a genetic distance between the 2 groups was obtained using the program Sequencer 6.1.0 (written by B. Kessing and available at http://nmg.si.edu/).
Results
Morphometrics. Analysis of variance showed significant differences (< 0.05) between species for most of the morphometric variables mainly due to differences in standard length, except for the dorsal–fin origin to end of the anal fin. We inferred from this result that the longer dorsal–fin base in Z. purhepechus n. sp. influences the DOAE measurement 0.32–0.25 ( = 0.29) (Table 2). ANOVA for sexual dimorphism showed significant differences in all variables except in the end of the anal–fin to end of the dorsal–fin distance, thus showing that females have a narrower caudal peduncle than males.
= 0.29) (Table 2). ANOVA for sexual dimorphism showed significant differences in all variables except in the end of the anal–fin to end of the dorsal–fin distance, thus showing that females have a narrower caudal peduncle than males.

The interaction between populations and sexes in most of the morphometric characters do not show significant differences, except for preorbital length. On the contrary, in the meristic characters, the significant differences are only between populations (Table 2). These results justify the separation of morphometric but not meristic analyses by sex.
In an exploratory PCA with the morphometric measurements, PCI explains 90.29% of the variation in males and 90.03% in females; for both sexes, the eigenvectors show closed values with the same symbol, suggesting an influence of the standard length in the results (Doadrio et al., 2002). A second PCA with a Burnaby corrected matrix, accumulates 45.53% of the variance in the PCII in males and 52.42% in females. For males, the high values in eigenvectors were preorbital length, dorsal fin length and anal fin length in the PCI, and interorbital width and FAD–ESPC in the PCII. For females, the high values in eigenvectors were preorbital length, dorsal fin length and FAD–ESPC in the PCI, and anal fin length in the PCII (Table 3). In both sexes, the variation patterns were determined by the preorbital length and dorsal fin length in the PCI. This analysis shows a clear tendency to form 2 groups (ellipses in Fig. 2 A and B), from the lower Lerma basin (La Luz Spring) and middle Lerma (San Cristobal and La Mintzita Springs in the Cuitzeo drainage and Zacapu Lake). With respect to the PCA with meristic characters, PCII accumulates 76.05% of the explained variance, and the high values in the eigenvectors were dorsal fin rays and gill rakers in the PCI, and pectoral fin rays in the PCII (Table 4). Similar to morphometric characters, the meristic characters show a variation pattern with the formation of 2 groups (ellipses in Fig. 3).

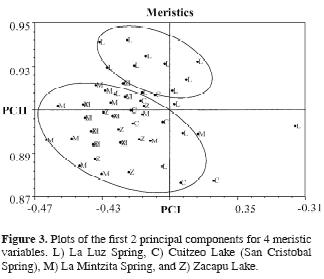
Starting with the classificatory hypothesis (formation of 2 groups), the DFA shows a significant difference (α < 0.05) with the intermediate distances of morphometric characters in males (P = 0.003) and females (P = 0.010), and with the meristic characters (P = 0.000). These results agree with the proposed classificatory hypothesis and corroborate the variation pattern found in the PCA.
Genetics. In the data set, 106 characters were variable, and 35 were parsimony informative. Third codon positions were the most informative characters (24 informative characters), followed by the first codon position (10 characters). Saturation of transition and transversion changes was checked by plotting the absolute number of changes of each codon position against patristic distances. There was no ingroup evidence of saturation at any of the 3 positions (not shown). The HKY–G model was selected as the best fit to the data set. Rate matrix parameters were: –lnL = 2205.0105; K = 5; BIC = 4456.7324. The base frequencies were: freqA = 0.2522; freqC = 0.2685; freqG = 0.1390; freqT = 0.3402. Among–site rate variation was approximated with gamma distribution shape parameter α = 0.1976. The phylogenetic tree obtained by the Bayesian analysis after discarding the initial 500 burn–in chains (Fig. 4) showed the formation of 2 well differentiated groups, with a posterior probability of the branches of 100 for the Z. quitzeoensis clade (including the type locality) and 96 for the clade which contains the new species Z. purhepechus sp nov. The genetic distance obtained by the model HKY–G within groups was 3.4% (3–3.8%).
Description
Zoogoneticus purhepechus n. sp. (Figures 5 A–B, Table 5)

D = (13) 14; A = 13–14; P = (11–12) 13–15; GR = (7) 9–12. Morphometric measurements are shown in Table 5. Body relatively deep, laterally compressed and elongated, maximum height  = 3.1 (range = 2.8–3.6) times the standard length in males and
= 3.1 (range = 2.8–3.6) times the standard length in males and  = 3.3 (range = 2.9–3.7) times in females. Minimum body height
= 3.3 (range = 2.9–3.7) times in females. Minimum body height  = 6.5 (range = 6.2–7) times the standard length for males and
= 6.5 (range = 6.2–7) times the standard length for males and  = 7 (range = 6.2–7.7) in females. Head short, cephalic length
= 7 (range = 6.2–7.7) in females. Head short, cephalic length  = 3.5 (range = 3.3–3.7) times standard length in males and
= 3.5 (range = 3.3–3.7) times standard length in males and  = 3.7 (range = 3.3–4.1) in females. Preorbital distance short, preorbital distance
= 3.7 (range = 3.3–4.1) in females. Preorbital distance short, preorbital distance  = 16.6 (range = 14.6–21.8) times standard length in males and
= 16.6 (range = 14.6–21.8) times standard length in males and  = 20 (range = 12.8–27.4) in females. Anal fin inserted before origin dorsal fin at same axis. Dorsal fin length long
= 20 (range = 12.8–27.4) in females. Anal fin inserted before origin dorsal fin at same axis. Dorsal fin length long  = 5.2 (range = 3.4–7) standard length in males and
= 5.2 (range = 3.4–7) standard length in males and  = 5.8 (range = 5.3–6.6) in females.
= 5.8 (range = 5.3–6.6) in females.
Pigmentation pattern. When alive, both sexes exhibit a light brown coloration, with dark brown and moderately large spots on the posterior part of the body, starting at the base of the caudal fin. In the anterior part of the body, a mottling pattern of small spots can be distinguished at the top of the ventral region. They show a pair of dark brown spots laterally aligned at the base of the caudal peduncle, in the region of the hypural plate. In males, during the breeding season, these spots could not be distinguished. The ventral region lacks spots. Adult males are slightly darker than females and may show a slightly bluish or greenish hue on the lateral side of the body and some scales can produce iridescence. The males from the type locality show an intense red band at the end of the pelvic and dorsal fins. In specimens from other localities, this band may be an intense orange. Alcohol preserved specimens are light brown on the body with the abdominal region yellowish, and from the caudal peduncle approximately to the insertion of the anal fin showing moderately large, dark brown spots. From the anal fin forward, spots are smaller and irregularly shaped. The red band in the dorsal and anal fins and the slightly bluish or greenish hue are faint or are less intense.
Sexual dimorphism and reproduction. As in all members of the subfamily Goodeinae, males have the first 2 to 7 anal rays shorter. These rays form a short lobe that is inferred to function in sperm transfer (Parenti, 1981) (Fig. 5A). Differences in size were found between sexes, with females being larger than males. As in Z. quitzeoensis, the caudal peduncle is narrower (SL/BLD  = 7.0 , range= 6.2–7.7) and longer (SL/EDUP
= 7.0 , range= 6.2–7.7) and longer (SL/EDUP  = 4.1, range= 3.4–4.7) in females than in males (SL/BLD x = 6.5 , range= 6.1–7.0 and SL/EDUP
= 4.1, range= 3.4–4.7) in females than in males (SL/BLD x = 6.5 , range= 6.1–7.0 and SL/EDUP  = 3.8 , range= 3.5–4.4). The males have a stripe with intense red coloration at the end of the dorsal and anal fin. The sex of males can be distinguished at a few weeks after birth. In captive conditions (temperature 21°C ± 2°C), the males and females can become sexually mature between 8 and 12 weeks. Gestation requires 7 to 9 weeks. Reproduction occurs throughout the year, but peaks when the temperature is between 20 and 21°C. The number of offspring per reproductive event in captivity oscillates between 15 and 45. The fry are usually 9 to 12 mm in standard length at birth and feed on the second day after birth. First parturition females usually have a low number of offspring.
= 3.8 , range= 3.5–4.4). The males have a stripe with intense red coloration at the end of the dorsal and anal fin. The sex of males can be distinguished at a few weeks after birth. In captive conditions (temperature 21°C ± 2°C), the males and females can become sexually mature between 8 and 12 weeks. Gestation requires 7 to 9 weeks. Reproduction occurs throughout the year, but peaks when the temperature is between 20 and 21°C. The number of offspring per reproductive event in captivity oscillates between 15 and 45. The fry are usually 9 to 12 mm in standard length at birth and feed on the second day after birth. First parturition females usually have a low number of offspring.
Taxonomic summary
Material examined. Holotype: (Table 5, Fig. 5A) CPUM 1509, adult male 34.12 mm SL, La Luz Spring (lower Lerma), Zamora, Michoacán, México, Ludo Couvreur, Jan de Moree, Kees de Jong, Juan C. Merino and Luis Escalera–Vázquez, November 2002.
Paratypes: CPUM 1055, (10 individuals); MNCN 246184 (15 individuals); CNP–IBUNAM 14425–14427 (3 individuals). Collected with the holotype.
Distribution: in accordance with the molecular and morphometric diagnostic characters, we hypothesized that the populations from Ameca, Santiago and Armería basins and Chapala Lake, identified as Z. quitzeoensis, should henceforth be considered as Z. purhepechus. Thus, the distribution of Z. purhepechus occupies the lower Lerma basin, the upper part of the Armeria, Santiago and Ameca basins and Chapala Lake (Fig. 1).
Habitat: the type locality of Z. purhepechus is La Luz Spring. La Luz is a permanent spring of lentic and clear waters and forms a small pond of approximately 1500 m2. Once, the water of this spring flowed to the Duero River, which forms part of the lower Lerma River basin. Currently, the water of this spring is used for irrigation and as a water supply to the population. It has an average depth of 1.5 m with a maximum depth of 3.5 m. The bottom is rocky in its periphery and muddy in most of the pond. Aquatic vegetation is Iris sp. and Ceratophyllum sp, emergent vegetation Typha sp. and Scirpus sp., and the introduced species Eichhornia sp, and terrestrial vegetation is of the subtropical forest type.
The associated fish fauna are the native species, Alloophorus robustus (Bean, 1892); Chapalichthys encaustus (Jordan and Snyder, 1899); Goodea atripinnis Jordan 1880; Skiffia multipunctata (Goodeidae); Poeciliopsis infans (Poeciliidae); and Lampetra geminis (Alvarez, 1964) (Petromyzontidae); and the introduced species Poecilia mexicana Steindachner 1863; Xiphophorus hellerii Heckel 1848, (Poeciliidae); Oreochromis spp (Cichlidae); and Cyprinus carpio Linnaeus 1758, Ctenopharingodon idella (Valenciennes 1844) (Cyprinidae).
Conservation: although this species has been taken from a number of localities, and is widely distributed in different drainages along the occidental part of Central Mexico, in the last 5 years, a reduction in its distribution of almost 75% of the historical occurrence points has been observed (Fig. 1). The most common alterations reported in the localities where the species has disappeared are the introduction of exotic species, water pollution and desiccation (De la Vega–Salazar et al., 2003; Domínguez–Domínguez et al., 2005; Domínguez–Domínguez et al., 2006b). Genetic erosion related with human perturbations of the population of this new species was demonstrated recently (Domínguez–Domínguez et al., 2007). This species should be considered as in danger of extinction, following the criteria and categories of the MER–Aii,Bi,Ci,Di (SEMARNAT 2002) and the International Union for the Conservation of Nature and Natural Resources IUCN–A,1a,c,e. (IUCN, 2001–http://app.iucn.org/webfiles/doc/SSC/RedList/RedListGuidelines.pdf).
Etymology: the name "purhepechus" comes from purhepecha, the name of the indigenous ethnic group which inhabited part of the distribution range of this species, including the type locality.
Remarks
Zoogoneticus purhepechus n. sp. differs from its sister species, Z. quitzeoensis by the following combination of characters: 13–14 branched rays in the dorsal fin (vs. 12, rarely 11 or 13 branched dorsal rays in Z. quitzeoensis), long dorsal fin base length (DFL/SL = 0.18, SD = 0.03 vs.  = 0.16, SD = 0.01 in Z. quitzeoensis) and short pre–orbital length (Prol/SL
= 0.16, SD = 0.01 in Z. quitzeoensis) and short pre–orbital length (Prol/SL  = 0.056, SD = 0.01 vs
= 0.056, SD = 0.01 vs  = 0.066, SD = 0.008 in Z. quitzeoensis); 2 conspicuous dark brown spots in the hypural plate region, except in males within the reproductive season; 10 molecular autapomorphies in the cytochrome b gene also differentiate Zoogoneticus purhepechus sp. nov. from Z. tequila and Z. quitzeoensis (Table 6). Divergences in the cytochrome b gene is DHKY = 3.4% (3–3.8%) compared to Z. quitzeoensis and DHKY = 11% (9–13%) compared to Z. tequila.
= 0.066, SD = 0.008 in Z. quitzeoensis); 2 conspicuous dark brown spots in the hypural plate region, except in males within the reproductive season; 10 molecular autapomorphies in the cytochrome b gene also differentiate Zoogoneticus purhepechus sp. nov. from Z. tequila and Z. quitzeoensis (Table 6). Divergences in the cytochrome b gene is DHKY = 3.4% (3–3.8%) compared to Z. quitzeoensis and DHKY = 11% (9–13%) compared to Z. tequila.
The genus Zoogoneticus is characterized by the presence of some distinctive characteristics, exhibiting 2 to 6 melanic patches in the post–ventral region of the body (Webb and Miller, 1998). Two dark brown spots in the hypural plate region are characteristic, more evident in Z. quitzeoensis and Z. purhepechus than in Z. tequila, in which the spots are less evident or are fused. The trophotaenia is a ribbon–type with 9–14 termini. The genetic and morphometric variation pattern within the populations of the 2 species analysed here show the separation of 2 well–defined groups, supporting previous findings made with molecular characters (Doadrio and Domínguez, 2004). The ANOVA analysis between populations shows that DOAE distance differs between them. We inferred that this is a result of the larger DFL distance in Z. purhepechus and this inference is supported by the PCA. In the same manner, differences in PrOL were obtained and supported by the minus preorbital length obtained as a diagnostic character in PCA. The morphometric diagnostic characters are preorbital length and dorsal fin length in the PCI. Accordingly, with this classification, which was corroborated by the DFA, the Z. quitzeoensis populations (San Cristobal Spring and La Mintzita Spring in the Cuitzeo drainage and Zacapu Lake) have a larger superior mandible and a shorter base of the dorsal fin. On the other hand Z. purhepechus (La Luz Spring) exhibits a shorter upper mandible, a larger dorsal fin base and a higher number of dorsal rays.
This grouping model is congruent with the results of the Bayesian analysis, and the high genetic divergences between Z. quitzeoensis and Z. purhepechus (DHKY= 3.4%, range= 3–3.8%). These values are higher than those described for the family Goodeidae, in which an intraspecific pairwise uncorrected "p" distance of 0.001 to 1.7% was found, and are similar to those found in interspecific distances of 1.7 to 11% (Doadrio and Domínguez, 2004). In the same manner, the middle Lerma and lower Lerma basins are considered to have undergone a pattern of isolation and union, which correlates with vicariant events inferred to promote cladogenetic processes in at least 2 pairs of sister species within the Goodeinae (e.g. Skiffia lermae–Skiffia multipunctata and Zoogonetius quitzeoensis–Z. purhepechus) (Domínguez–Domínguez et al., 2006a).
Acknowledgments
The authors are grateful to Luis Escalera, Asdrubal Molina, Hugo Mejia, Rogelio Rosas, Jen Nightingale, Jean de Moree, Ludo Couvreur, Kees de Jong, Juan Carlos Merino and Adrian Pompa for their help during field trips. Partial funds to conduct this research were provided by the COECyT Michoacán and Chester Zoo Garden, England to OD and by the project CGL2006–12325/BOS. We thank Ivan Dibble for his support to the Hobbyist Aqualab Conservation Project (HALCP) that receives funds from European and North American aquarist associations. OD and RP thank the Consejo Nacional de Ciencia y Tecnología for the scholarship.
Literature cited
Barbour, C. D. 1973. A biogeographical history of Chirostoma (Pisces: Atherinidae): A species flock from the Mexican Plateau. Copeia 1973:533–556. [ Links ]
Burnaby, T. P. 1966. Growth–invariant discriminant functions and generalized distances. Biometrics 22: 96–110. [ Links ]
De la Vega–Salazar, M., E. Avila–Luna and C. Macías–García. 2003. Ecological evaluation of local extinction: the case of 2 genera of endemic Mexican fish, Zoogoneticus and Skiffia. Biodiversity and Conservation 12:2043–2056. [ Links ]
Doadrio, I., J. A. Carmona and C. Fernández–Delgado. 2002. Morphometric study of the Iberian Aphianus (Actinopterygii, Cyprinodontiformes), with description of a new species. Folia Zoologica 51:67–79. [ Links ]
Doadrio, I. and O. Domínguez. 2004. Phylogenetic relationships within the fish family Goodeidae based on cytochrome b sequence data. Molecular Phylogenetics and Evolution 31:416–430. [ Links ]
SEMARNAT (Secretaría de Medio Ambiente y Recursos Naturales). 2002. Norma Oficial Mexicana NOM–059–ECOL–2001, Protección ambiental–Especies nativas de México de flora y fauna silvestres–Categorías de riesgo y especificaciones para su inclusión, exclusión o cambio–Lista de especies en riesgo. Diario Oficial de la Federación. 6 de marzo de 2002, Primera sección. México, DF. [ Links ]
Domínguez–Domínguez, O., N. Mercado–Silva, J. Lyons and H. J. Grier. 2005. The viviparous Goodeid fish. In Proceedings of the second international symposium of viviparous fishes, M. C. Uribe and H. J. Grier (eds.). New Life Publications, Homestead, Florida. p. 505–549. [ Links ]
Domínguez–Domínguez, O., I. Doadrio and G. Pérez–Ponce de León. 2006a. Historical biogeography of some river basins in Central Mexico evidenced by their goodeiine freshwater fishes: A preliminary hypothesis using secondary Brooks Parsimony Analysis (BPA). Journal of Biogeography 30:1437–1447. [ Links ]
Domínguez–Domínguez O., E. Martínez–Meyer, L. Zambrano and G. Pérez–Ponce de León. 2006b. Using ecological–niche modeling as a conservation tool for freshwater species: the live–bearing fishes (Goodeidae) in Central Mexico. Conservation Biology. 20: 1730–1739. [ Links ]
Domínguez–Domínguez O, L. Boto, F. Alda, G. Pérez–Ponce de León and I. Doadrio. 2007. Human impacts on Basins of the Mesa Central of Mexico, and its genetic effects on an endangered fish, Zoogoneticus quitzeoensis. Conservation Biology 21:168–180. [ Links ]
Echelle, A. A. and A. F. Echelle. 1984. Evolutionary genetics of a "species flock:" Atherinid fishes on the Mesa Central of Mexico. In Evolution of fish species flocks, A. A. Echelle and I. Kornfield (eds.). University of Maine Press, Orono. p. 93–110. [ Links ]
Espinoza–Pérez, H., M. T. Gaspar–Dillanes and P. Fuentes–Mata. 1993. Listados faunísticos de México. III. Los peces dulceacuícolas mexicanos. Instituto de Biología, Universidad Nacional Autónoma de México, México, D.F. 98 pp. [ Links ]
Grudzien, T. A., M. M. White and B. J. Turner. 1992. Biochemical systematics of the viviparous fish family Goodeidae. Journal of Fish Biology 40:810–814. [ Links ]
Guzmán–Arroyo, F. 1994. Osteología y variación no geográfica de la suspensión de la aleta anal de Goodea luitpoldi, (Osteichthyes: Goodeidae). Universidad, Ciencia y Tecnología 3:33–41. [ Links ]
Hubbs, C. L. and C. L. Turner. 1939. Studies of the fishes of the Order Cyprinodontes. XVI. A revision of the Goodeidae. Miscellaneous Publications of the Museum of Zoology. University of Michigan 42:1–80. [ Links ]
Hueselsenbeck, J. P. and F. R. Ronquist. 2001. MrBayes: Bayesian inference of phylogeny. Bioinformatics 17:754–755. [ Links ]
IUCN (International Union for the Conservation of Nature and Natural Resources). 1994. IUCN Red List Categories. Prepared by the IUCN Species Survival Commission. Gland: IUCN, Switzerland. [ Links ]
Machordom, A. and I. Doadrio. 2001. Evidence of a Cenozoic Betic–Kabilian connection based on freshwater sh phylogeography (Luciobarbus, Cyprinidae). Molecular Phylogenetics and Evolution 18:252–263. [ Links ]
Mateos, M., O. I. Sanjur and R. C. Vrijenhoek. 2002. Historical biogeography of the livebearing fish genus Poeciliopsis (Poeciliidae: Cyprinodontiformes). Evolution 56:972–984. [ Links ]
Meek, S. E. 1904. The fresh–water fishes of Mexico north of the Isthmus of Tehuantepec. Field Columbian Museum Publications 5:1–252. [ Links ]
Miller, R. R. and M. L. Smith. 1986. Origin and geography of the fishes of central Mexico. In The zoogeography of the North American Freshwater Fishes, C. H. Hocutt and E. O. Wiley (eds.). John Wiley and Sons, New York. p. 487–517. [ Links ]
Parenti, L. 1981. A phylogenetic and biogeographic analysis of cyprinidontiform fishes (Teleostei, Atherinomorpha). Bulletin of the American Museum of Natural History 168:335–557. [ Links ]
Posada, D. and K. A. Crandall. 1998. Modeltest: testing the model of DNA substitution. Bioinformatics 14: 817–818. [ Links ]
Regan, C. T. 1908. Pisces. In Biologia Centrali–Americana, F. D. Godman and O. Salvin (eds.). London, United Kingdom. p. 1–203. [ Links ]
Rohlf, F. J. and F. L. Bookstein. 1987. A comment on shearing as a method for size correction. Systematic Zoology 36:356–367. [ Links ]
Rohlf, F. J. 2000. NTSYS–pc: Numerical taxonomy and multivariate analysis system, version 2.1. Setauket, New York. [ Links ]
Sambrook, J., E. F. Fritsch and T. Maniatis. 1989. Molecular Cloning: A Laboratory Manual. New York: Cold Spring Harbor Laboratory. [ Links ]
Schönhuth, M. S. and I. Doadrio. 2003. Phylogenetic relationships of Mexican minnows of the genus Notropis (Actinopterygii, Cyprinidae). Biological Journal of the Linnean Society 80:323–337. [ Links ]
Webb, S. A. and R. R. Miller. 1998. Zoogoneticus tequila a new goodeid fish (Cyprinodontiformes) from Ameca drainage of Mexico, and a rediagnosis of the genus. Occasional Papers of the Museum of Zoology, University of Michigan 725:1–23. [ Links ]














