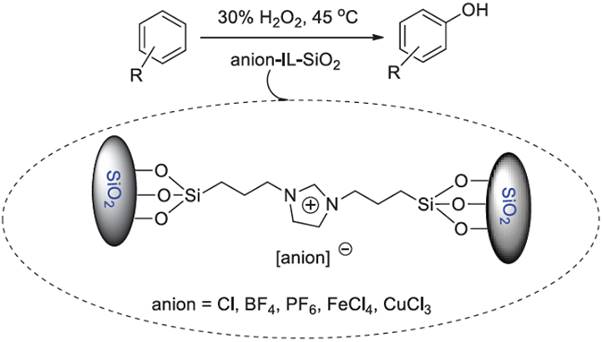
Introduction
Phenols are important industrial intermediates and have been widely used for the production of a variety of chemicals [1, 2]. Generally, phenols are mainly produced by the three-step cumene method process [3]. However, this process suffers from some problems such as complex operations, poor selectivity, high process costs, and environmental hazards. Therefore, the direct hydroxylation of aromatics to phenols in a single-stage reaction has attracted much attention in chemical industry in recent years, both from economic and environmental standpoint [4]. Nowadays, the green processes for the hydroxylation of aromatics are mostly focused on using clean oxidants like H2O2, N2O, O2, and H2-O2 system [4-8]. Among these oxidants, H2O2 is a good choice since it is an environmentally benign oxidant, quite inexpensive, ready-made, easily handled, and efficient with nontoxic water as the sole by-product. A large number of catalysts, including NiII(tepa) [9], mesostructured V/mp-C3N4[10], VPO@GO [11], CuCr2O4 spinel nanoparticles [12], AlPO4/ZnAlPO4[13], nanostructured CuFe [14], Vanadium-containing mesoporous carbon [15], V-MimSaIm-PMoV [16], nanostructured V2O5/ SnO2[17], PMO/VO(acac)2[18], and other complexes [19-24], have been developed for this catalytic conversion. Some of these procedures, however, are often affected by a low conversion or poor selectivity. Therefore, explore novel catalytic system with improved conversion rates for the direct hydroxylation of aromatics to phenols remains an interesting research topic.
Ionic liquids (ILs) have been acclaimed as green reaction media and catalysts as they are non-volatile, highly solvating, excellent ionic conductivity, good electrochemical properties, etc [25-30],29). Examples of the applications of ILs in the hydroxylation of aromatics to phenols were seldom studied [31, 32]. Although good catalytic activities of these ILs presented, they have some disadvantages such as partially ILs wasted and difficulties in the separation and recycle. A useful method to surmount these shortages is the development of supported ILs catalysts [33-39], which could transfer the catalytic reactions from homogeneous catalysis to a type of heterogeneous catalysis. In this case, attractive features such as gaining high catalyst efficiency and stability, facilitating the catalyst recovery and excellent reusability have been achieved [37-39]. In this work, we designed and synthesized a type of supported ILs anion-IL-SiO2, which exhibited good thermal and chemical stability, and were utilized as a new heterogeneous solid catalyst in the hydroxylation of aromatics to phenols with hydrogen peroxide as oxidant (Scheme 1). As well as this, the reusability of the silica supported functionalized ILs was also studied.
Results and discussion
Characterization of the supported ILs was studied using FT-IR, SEM, EDX, XRD and TG analysis. The FT-IR spectra of the supported catalysts are presented in Fig. 1. All ionic liquid materials exhibit absorptions at about 1072 and 965 cm-1, which correspond to the stretching vibration of Si−O−Si. Characteristic peaks at the positions of about 1465, 1530, and 1650 cm-1, correspond to C−H bending vibrations, C=C and C=N stretching vibrations of the imidazolium ring, indicating that imidazole group was successfully attached to the surface of silica gel [40]. For b and c, absorptions in the B-F (1045 cm-1) and P-F (865 cm-1) bands are observed, indicating the interaction between heteropoly acid anions and the SiO2−IL support. The characteristic peak at 798 cm-1 was attributed to the C-Cl (Fig. 1a). Also, the peaks observed at 725 cm-1 (Fig. 1d) and 570 cm-1 (Fig. 1e) are related to vibrational modes of FeCl4 and CuCl3, respectively.
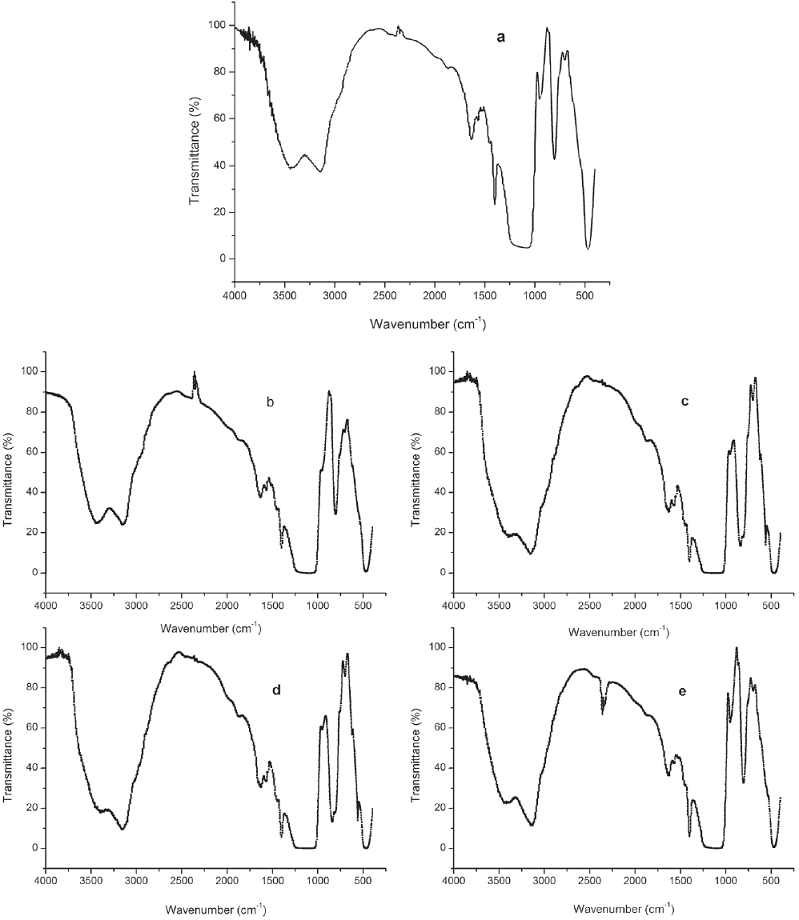
Fig. 1 FT-IR spectra of Cl-IL-SiO2 (a), BF4-IL-SiO2 (b), PF6-IL-SiO2 (c), FeCl4-IL-SiO2 (d), and CuCl3-IL-SiO2 (e).
The surface morphology and chemical composition of the supported ionic liquid catalysts were studied by SEM (Fig. 2) and EDX (Fig. 3). Fig. 2g is the SEM image of silica gel, Fig. 2a-e is the SEM images of the silica gel supported ILs, Cl-IL-SiO2 (Fig. 2a), BF4-IL-SiO2 (Fig. 2b), PF6-IL-SiO2 (Fig. 2c), FeCl4-IL-SiO2 (Fig. 2d), CuCl3-IL-SiO2 (Fig. 2e), it clearly observed that all the IL particles were well-defined, and the particle size of the IL is smaller to that of silica gel. It can be seen that the supported ILs were made up of uniform nanometre-sized particles. The chemical composition of the supported ILs was investigated by EDX analysis (Fig. 3), which showed the presence of the expected elements in its structure. XRD pattern of the supported IL catalysts were studied in a domain of 5-90° (Fig. 4). As shown in Fig. 4, all the catalysts had characteristic peak at 2θ ≈ 22.8°, which was the presence of the amorphous nature of SiO2 support [41]. The absence of obvious characteristic peaks of anions of ILs in the XRD patterns suggests that these anions units are well-dispersed on the surface and in the channels of the SiO2-IL supports.

Fig. 2 SEM images of Cl-IL-SiO2 (a), BF4-IL-SiO2 (b), PF6-IL-SiO2 (c), FeCl4-IL-SiO2 (d), CuCl3-IL-SiO2 (e), four-times recycled CuCl3-ILSiO2 (f), and silica gel (g).
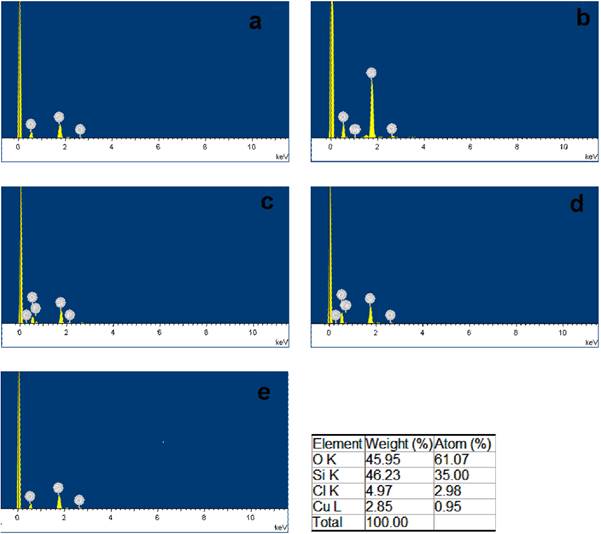
Fig. 3 EDX images of Cl-IL-SiO2 (a), BF4-IL-SiO2 (b), PF6-IL-SiO2 (c), FeCl4-IL-SiO2 (d), and CuCl3-IL-SiO2 (e).
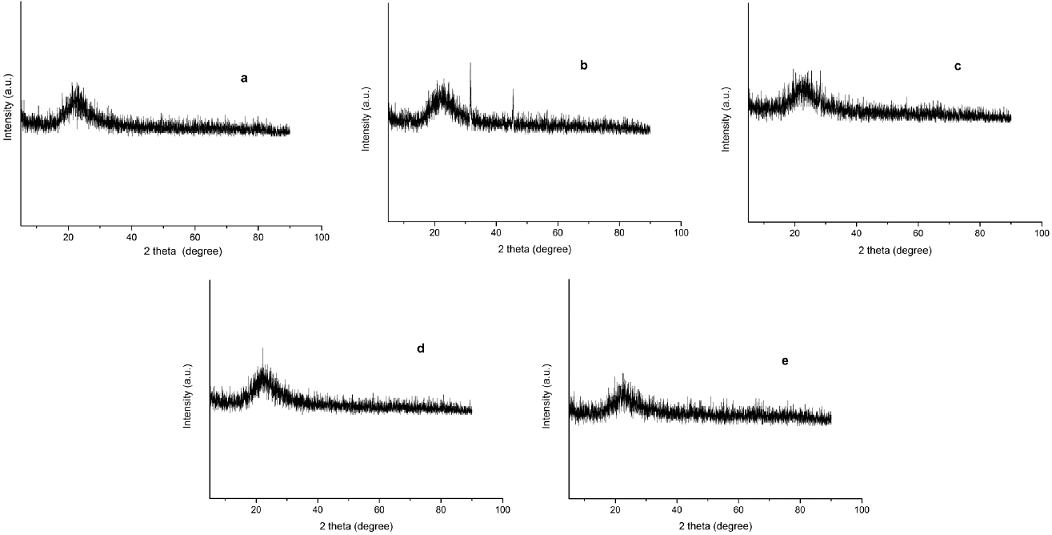
Fig. 4 Wide-angle XRD patterns of Cl-IL-SiO2 (a), BF4-IL-SiO2 (b), PF6-IL-SiO2 (c), FeCl4-IL-SiO2 (d), and CuCl3-IL-SiO2 (e).
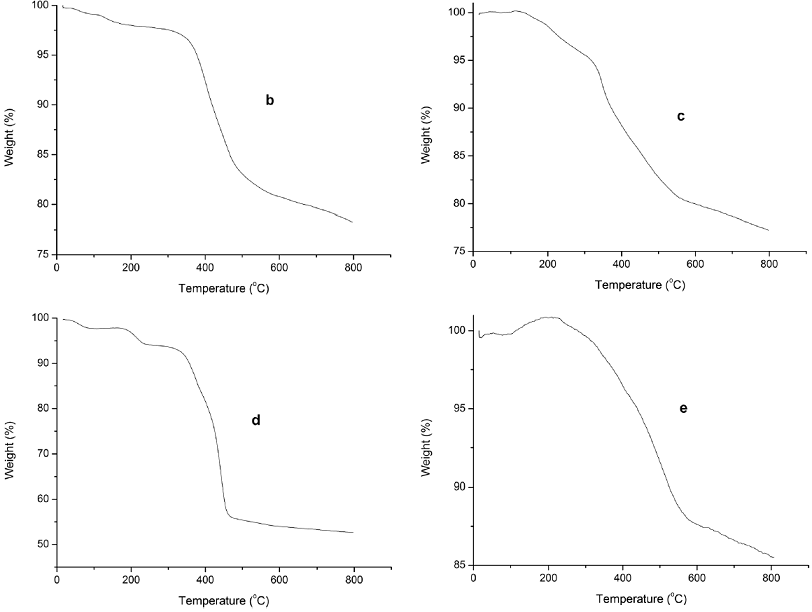
The stability of the supported IL catalysts, BF4-IL-SiO2, PF6-IL-SiO2, FeCl4-IL-SiO2, and CuCl3-IL-SiO2, were determined by thermogravimetric analysis, and the TG analysis results are shown in Fig. 5. In the TG curve, the observed organic materials weight loss of the sample showed a mass weight loss of ~21.8 (Fig. 5b), 22.7 (Fig. 5c), ~47.4% (Fig. 5d), ~14.5% (Fig. 5e) on heating to 800 °C, respectively. The small weight loss before 260 °C is related to the desorption of adsorbed water, indicating a considerably thermal stability up to 260 °C. The drastic weight loss between 260 and 580 °C is attributed to the breakdown of the organic moieties. From the curve depicted, it can be seen that the supported ionic liquid catalysts are thermally stable below 260 °C.
In order to optimize the reaction conditions and test the catalytic activity of supported ILs, we selected the hydroxylation of benzene with H2O2 as a reaction model and the effect of IL catalysts were examined for this reaction (Table 1). Similar to a non-catalyst system, the supported IL precursor Cl-IL-SiO2 did not give any products (Table 1, entries 1 and 2). The other supporting ILs, such as BF4-IL-SiO2, PF6-IL-SiO2, Fe-Cl4-IL-SiO2, and CuCl3-IL-SiO2, were tested as catalysts in the hydroxylation (Table 1, entries 3-6), and CuCl3-IL-SiO2 demonstrated the highest efficiency in terms of selectivity and yield (Table 1, entry 6). Over CuCl3-IL-SiO2, a yield of 59.4 % was achieved (Table 1, entry 6), although FeCl4-IL-SiO2 showed to be more active in the hydroxylation of benzene, nevertheless a much lower selectivity of 84.7 % was obtained (Table 1, entry 5). The corresponding controlled experiments were conducted with bare CuCl2 and FeCl3 (Table 1, entries 7 and 8). The CuCl2 exhibits 26.5% yield and 93.2% of selectivity, and 45.6% yield and 72.8% of selectivity are obtained over FeCl3. Thus, based on the results obtained, the CuCl3-IL-SiO2 catalyst was chosen to undergo further investigation for the different parameters in this reaction.
Table 1 Hydroxylation of benzene to phenol with H2O2 over various catalysts.
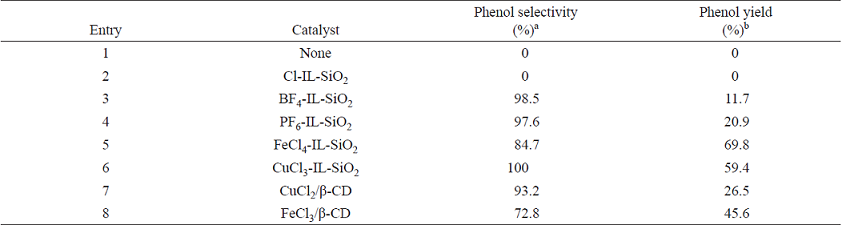
The reactions were carried out with benzene (0.1 mol), 30% H2O2 (0.09 mol), catalyst (1.5 g) at 45 °C for 140 min.
a Isolated selectivity.
b Isolated yield.
The effects of other parameters such as the amount of the catalyst, mole ratio, reaction temperature, and reaction time were also investigated, and the results are presented in Figs. 6-9. The effect of the catalyst amount on the hydroxylation is shown in Fig. 6. The phenol yield and selectivity raised up as the amount of catalyst increased to 1.5 g. However, further increasing in the amount of catalyst had a reverse effect on the hydroxylation. Therefore a catalyst loading of 1.5 g was considered to be suitable for the reaction. The effect of mole ratio on benzene conversion was studied over CuCl3-IL-SiO2 catalyst varying amounts of H2O2 to a fixed amount of benzene from 0.5:1 to 1.5:1 ratios (H2O2: benzene) and the results are shown in Fig. 7. The yield and selectivity increased as the mole ratio increased from 0.5 to 0.9 and thereafter leveled off. The decreasing in yield when the mole ratio increased from 0.9 to 1.5 was due to the over oxidation of phenol, which was detected by GC analysis in all the cases. Thus, the mole ratio 0.9: 1 (H2O2: benzene) would be the optimum one. The activities of CuCl3-IL-SiO2 at various temperatures on the hydroxylation of benzene are plotted in Fig. 8. Results indicate that the phenol yield initially increased with increasing reaction temperature. The highest reaction activity was obtained at a temperature of 45 °C, the phenol yield and selectivity are 59.4 % and 100%, respectively. As the temperature continued to increase, there was a notable decreasing in the phenol yield, mostly because of the spontaneous decomposition of H2O2 at high temperatures and then a trend to descend with the increase of reaction temperature. These data indicated that a temperature of 45 °C was the most favorable. The effects of reaction time are investigated and shown in Fig. 9. The phenol yield increased with the increase of the reaction time, and reached a maximum at 140 min. Thus, 140 min may be considered as a moderate time.
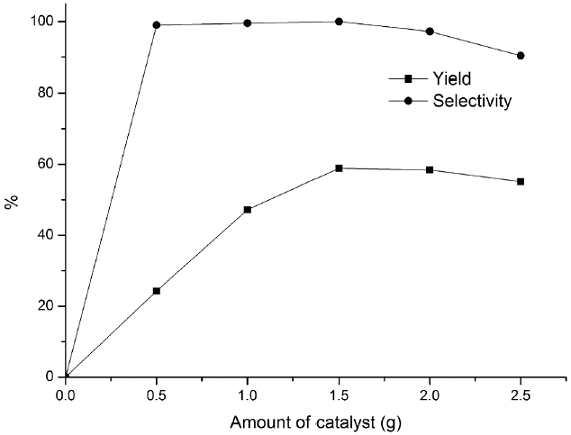
Fig. 6 Influence of catalyst amount on hydroxylation of benzene with H2O2. Reaction conditions: benzene (0.1 mol), 30% H2O2 (0.09 mol), catalyst CuCl3-IL-SiO2, 45 °C, 140 min.
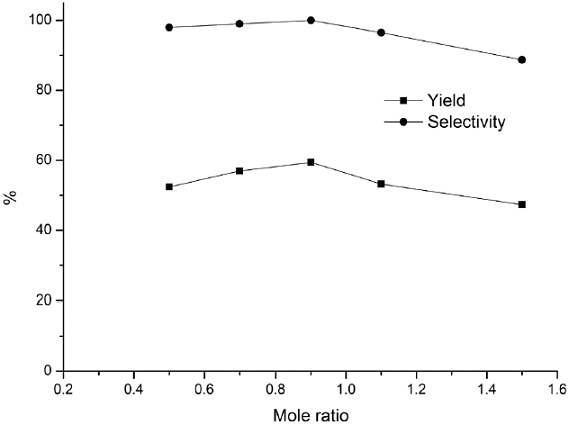
Fig. 7 Effect of mole ratio (H2O2: benzene) on hydroxylation of benzene. Reaction conditions: benzene, 30% H2O2, CuCl3-IL-SiO2 (1.5 g), 45 °C, 140 min.
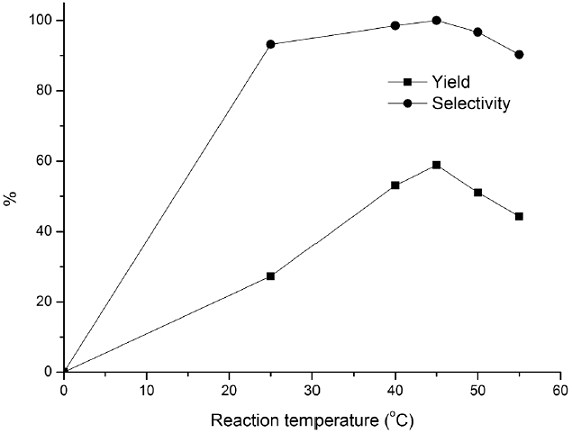
Fig. 8 Influence of reaction temperature on hydroxylation of benzene. Reaction conditions: benzene (0.1 mol), 30% H2O2 (0.09 mol), CuCl3-IL-SiO2 (1.5 g) 140 min.
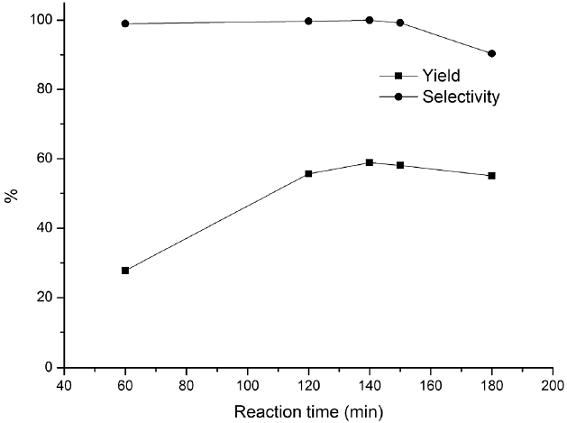
Fig. 9 Influence of reaction time on hydroxylation of benzene. Reaction conditions: benzene (0.1 mol), 30% H2O2 (0.09 mol), CuCl3-IL-SiO2 (1.5 g), 45 °C.
The heterogeneous CuCl3-IL-SiO2 catalyst can be easily separated and recovered by filtration after the reaction. The hydroxylation results by recovered CuCl3-IL-SiO2 after four cycles are shown in Fig. 10. Results indicate that the phenol selectivity in each run is above 95% with no notable decline in catalytic efficiency during the consecutive four runs, suggesting that the catalyst could be recovered and reused up to 4 runs with no significant loss of activity. The recovered catalyst after four runs had no obvious change in the morphology and size, referring to the SEM image in comparison with fresh catalyst (Fig. 2). To show the merit of CuCl3-IL-SiO2 in comparison with other reported catalysts, we compared the results of the hydroxylation reactions of benzene with other reported in the literature. As shown in Table 2, our catalyst showed milder reaction conditions and good yield and selectivity compared to the other catalysts.
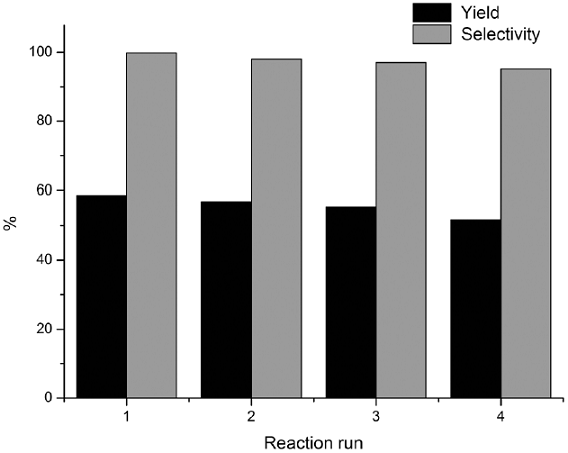
Fig. 10 The repeating hydroxylation reactions of benzene (0.1 mol), 30% H2O2 (0.09 mol) in the presence of the recycled CuCl3-IL-SiO2.
Table 2 Comparison of different catalysts for the hydroxylation of benzene to phenol with H2O2.
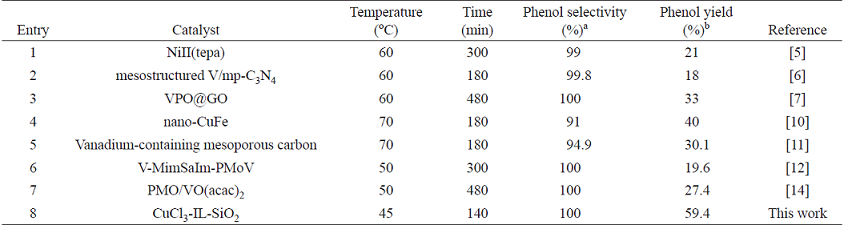
a Isolated selectivity.
b Isolated yield.
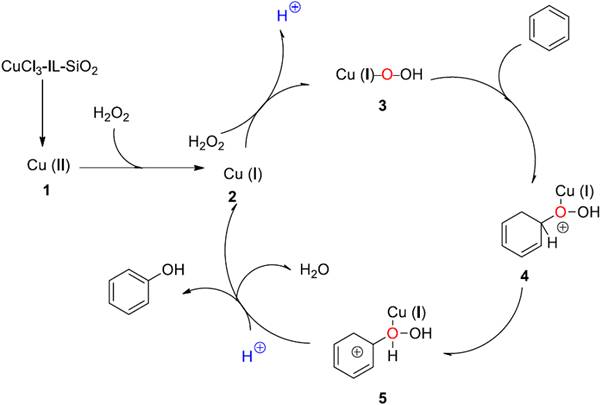
According to the literatures [17, 18, 42] and the observations in our reactions, we propose a possible mechanism as shown in Scheme 2. In the reaction, the catalyst CuCl3-IL-SiO2 provides a source of Cu (II), which reacts with H2O2 to form Cu (I) and Cu (I)-O-OH (3). Then, 3 reacts with benzene to form transition state 4 and 5. Then, 5 very rapidly affords Cu (I) to yield phenol. The monovalent copper ion is then re-oxidized to Cu (I)-O-OH by H2O2 in ionic liquid to complete the catalytic cycle.
To demonstrate generality and efficiency of CuCl3-IL-SiO2, hydroxylation of other aromatics were tested (Table 3). Various aromatics were hydroxylated to the corresponding phenols in good to excellent yields, whereas benzenes with electron-withdrawing substituents were less reactive and more drastic reaction conditions were needed (Table 3, entries 6−8). To our delight, the hydroxylation of aromatics with electron-donating substituents gave phenols in excellent yields with the distribution of isomers of ortho-(o) and para-(p), and no meta-(m) products (Table 3, entries 2-5).
Conclusion
In conclusion, a series of silica supported functionalized ionic liquid catalysts were prepared and tested in the direct hydroxylation of aromatics to phenols with H2O2 as the oxidant. Among these catalysts, CuCl3-IL-SiO2 is the most active catalyst in the titled reaction. The catalyst leads to a liquid-solid heterogeneous catalysis system, showing high activity, easy recoverability and steady reuse. Compared to the synthetic methods reported in previous literatures, not only the yield and selectivity of the product were enhanced or greatly improved, but also the operating units were easy workup.
Experimental
General
The reagents and solvents used in this work were of analytical grade. FT-IR spectra were conducted on a Nicolet Nexus 470 Fourier transform infrared spectrometer. Powder X-ray diffraction (XRD) analysis was conducted on a Rigaku Ultima IV diffractometer with high-intensity Cu Ka radiation. Energy dispersive X-ray analysis (EDX) was used to conduct the elemental analysis of the supported ILs. Scanning electron microscopy (SEM) studies were investigated by a JSM-7500F Scanning Electron Microscope. Thermogravimetric analysis (TGA) were determined by a Netzsch Thermoanalyzer STA 449. GC analyses were performed on a Shimadzu-14B gas chromatography.
Preparation of Supported ILs
The preparation processes were according to known process [37-39] and shown in Scheme 3. (i) Preparation intermediate 1: Imidazole (13.6 g, 0.2 mol), (3-chloropropyl) triethoxysilane (48.2 g, 0.2 mol) and toluene (350 mL) were added into a 1 L round flask and the mixture was refluxed for 22 h. Then, triethylamine (20.2 g, 0.2 mol) was added dropwise and the resulting mixture was refluxed for another 2 h. Then, the slurry was filtrated and the filter cake was washed with toluene (3x20 mL). The solvent was removed to afford 1 as a viscous liquid. (ii) Preparation intermediate 2: 1 (27.2 g, 0.1 mol), (3-chloropropyl) triethoxysilane (24.1 g, 0.1 mol) and toluene (80 mL) were stirred under reflux conditions for 12 h. The solvent was evaporated to give 2 as a viscous white liquid. (iii) Preparation IL 3: 2 (25.8 g, 0.05 mol), activated silica (40 g) and toluene (250 mL) were stirred under reflux conditions for 24 h. Then, the mixture was filtrated and washed with dichloromethane (3x20 mL) and dried under vacuum at 60 °C to afford Cl-IL-SiO2 3 as a white powder. (iv) Preparation ILs 4 and 5: 9.0 g Cl-IL-SiO2 was dispersed throughout 100 mL dichloromethane and treated with 0.66 g NaBF4 or 1.1 g KPF6, then the mixture was stirred under reflux conditions for 24 h. After cooled down to room temperature, the solid was isolated by filtration and washed with dichloromethane/distilled water (3x20 mL). The resulting material was dried under vacuum at 80 °C to give the supporting ionic liquid BF4-IL-SiO2 (4, gray powder) or PF6-IL-SiO2 (5, yellow powder). (vi) Preparation ILs 6 and 7: 10.0 g Cl-IL-SiO2 was dispersed in 100 mL acetonitrile and stirred vigorously with 3.0 g FeCl3 or 2.0 g CuCl2 for 24 h under reflux conditions. Then the sample was filtrated and washed with dichloromethane (3x20 mL). The resulting material was dried under vacuum at 80 °C to afford the supporting ionic liquid FeCl4-IL-SiO2 (6, yellow powder) or CuCl3-IL-SiO2 (7, yellow-green powder).
Typical Procedure for the Catalytic Hydroxylation of Aromatics to Phenols
In a typical experiment, benzene (0.1 mol) and catalyst CuCl3-IL-SiO2 (1.5 g) were added to a 100 mL flask reactor. Then, aqueous 30% H2O2 (0.09 mol) was added dropwise in 30 min. Then, the mixture was stirred at 45 °C for 140 min. The reaction progress was monitored by GC. In this catalytic system, only phenol product was detected, and no other GC signals were found, i.e. the phenol selectivity is 100%. The catalyst was separated by filtration, washing with benzene (2x5 mL) and dried under vacuum. Fresh benzene and H2O2 were then recharged to the recovered catalyst and recycled. All products were known and identified by comparing their GC data with those of commercial materials.











 nueva página del texto (beta)
nueva página del texto (beta)