Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Tropical and subtropical agroecosystems
On-line version ISSN 1870-0462
Trop. subtrop. agroecosyt vol.13 n.1 Mérida Jan. 2011
Artículos de investigación
Evaluation of selected soil fertility management interventions for suppression of Fusarium spp. in a maize and beans intercrop
Evaluación de algunas acciones selectas de manejo de la fertilidad del suelo para la supresión de Fusarium spp. en un cultivo intercalado de maíz y frijol
Sheila A. Okoth1* and Elizabeth Siameto2
1 School of Biological Sciences, University of Nairobi P.O Box 30197, Nairobi. * Corresponding author E-mail dorisokoth@yahoo.com
2 Moi University, School of Sciences, Narok University College P. O Box 861, Narok.
Submitted February 15, 2010
Accepted May 25, 2010
Revised received June 10, 2010
Abstract
Fusarium root rot of maize and beans is a common problem in Taita District, Kenya causing economic losses to the small scale farmers. The pathogen attacks maize and beans at all growth stages causing rot at the seedling stage, yellowing of the leaves, stunted growth, and death if severe. Potentially effective crop rotations to maintain the pathogen at low levels are not currently practical due to the small size of farms while fungicides are out of reach to the small scale farmer due to high prices. This study aimed at assessing alternatives to fungicides in controlling root infection by Fusarium sp. in maize and beans cropping systems.
Field trials were done in Taita District where agriculture contributes to 95% of household income with limited use of any soil fertility amelioration by farmers. The following were tested in the trials; three types of inorganic fertilizers, cow manure, and Trichoderma inoculan! Planting was done during the long and short rains. Soil and roots were collected from the rhizosphere during harvesting and assessed for inoculum density while the roots were evaluated for incidence of infection by Fusarium spp. The most common species in both soil and roots were F. oxysporum (Schlecht) Snyd.et Hans, and F. sporotrichoides Sherb. Addition of soil amendments had a positive effect of reduced root infection and in some cases lowering inoculum density in the soil. Of the four integrated soil fertility interventions, Mavuno fertilizer had the highest yield and was the most effective in suppressing root colonisation by Fusarium spp.
Keywords: Fusarium spp.; root infection; fertilizers; Trichoderma; soil amendments.
INTRODUCTION
Maize (Zea mays L.) is a staple food of the majority of inhabitants of sub Saharan Africa. In Kenya, maize is grown as an intercrop with common bean (Phaseolus vulgaris L.), an important source of protein. Apart from providing families with cheapest source of starch and protein, maize and bean harvest was source to generate incomes. Production of these crops is constraint by pests and diseases. Most farmers are small scale and cannot afford expensive inputs for crop protection. Moreover these pesticides are not environmentally friendly. Fungal infection of maize and beans not only results in reduced yields through rotting, but may also lead to mycotoxin production. Currently maize ear rot ranks highly as a maize production constraint in Kenya and is caused by a variety of fungi that belong to several genera which include Fusarium spp, Stenocarpella spp, Penicillium spp and Aspergillus spp (Kedera, et al, 1992, 1998; MacDonald and Chapman, 1997). Several phytopathogenic species of Fusarium are found to be associated with maize including F. verticillioides (Sacc.) Nirenberg, F. proliferatum (Matsushina) Nirenberg, F. graminearum Schwabe and F. anthophilum (A. Braun) Wollenweber (Scott, 1993; Munkvold and Desjardins, 1997). Root rot severely constrains bean production in Kenya especially where soil fertility is low and bean production is intensive (Otsyula et al, 1998; CIAT, 1992). Root rot is primarily caused by Fusarium solani fsp. phaseoli, Rhizoctonia solani, and Pythium species. (Nderitu et al, 1997). Root rot pathogens attack beans at all growth stages and cause damping-off at the seedling stage, yellowing of the leaves, stunted growth, and death if severe. Fusarium species are ubiquitous in soils and are considered as field fungi invading more than 50% of maize grains before harvest (Robledo-Robledo, 1991). Crop rotations which contribute to minimization of Fusarium inoculums in soils is not feasible due to scarcity of land and cultural values (Hall and Phillips, 1992).
The purpose of this study was therefore to evaluate the efficacy of soil amendments in management of Fusarium sp. in maize and beans cropping systems.
MATERIALS AND METHODS
Description of the study site
The study was carried out in Taita Taveta District within the global UNEP-GEF funded project: CSM-BGBD (Conservation and Sustainable Management of belowground biodiversity) project. The District is located within the Taita Hills (lat 3°25'; long 38°20') situated in South-Eastern Kenya, Coast Province. The altitude ranges between 1200 to 2000m with mean annual rainfall of 800 - 2000mm. The district covers an area of 16,965 Km2 and is divided into five divisions, Wundanyi, Tausa, Voi, Taveta and Mwatate. The study site was in Werugha and Wumingu locations of Wundanyi Division where majority of the farmers are small-scale.
Establishing of the field trials
Field trials were established at the Agricultural Training Centre (ATC) and on selected farms. The experiment at the ATC was laid out in a Randomized Complete Block Design (RCBD) with treatments replicated five times. These treatments were established in selected twelve farms that were 500m apart and considered as replicates. One block constituted by the treatments described earlier were installed in each farm and measured 5 x 10m. Maize variety (H516) was sown at a spacing of 90 x 30cm with two seeds per hole. The bean variety was Mwezi moja planted at a spacing of 75 x 25 cm and two seeds per hole. The treatments were Triple superphosphate combined with Calcium ammonium nitrate (TSP + CAN), Mavuno fertilizer (a blend of fertilizer containing 11 nutrients) and Mijingu Rock Phosphate (MRP) fertilizers, Cow manure, and Trichoderma seed coating (Table 1). The fertilizers were added by broadcasting during planting and top dressing of CAN and Mavuno done after first round of weeding. Planting was done during the long rains which occur between March and May and short rains between October and December. Soil and root samples were collected during harvesting from each treatment. Samples were bulked from five points in maize root and bean root rhizosphere respectively. The soils were transported in a cool box to the laboratory.

Assessment of Fusarium Density in Soil
One gram of the sampled soil was added to nine ml of 0.05% water agar (10 -1) and shaken. One milliliter of the first dilution was pippeted and added to nine ml of 0.05% water agar (10-2) and shaken. From this last soil dilution 1.0 ml was taken and pippeted to each of two Malachite Green Agar (MGA) plates and incubated at room temperature for six days. The colonies formed were counted (Leslie and Summerell, 2006).
A small piece of growth at the edge of the colonies identified as Fusarium was transferred to Potato Dextrose Agar (PDA) plates incubated at room temperature for five days and then transferred to Spezieller Nahrstoffarmer Agar (SNA) and Carnation Leaf Agar (CLA) for identification. Isolates of Fusarium spp obtained from SNA and CLA media were identified using the text references and taxonomic keys of Burgess et al., (1994) and Booth (1971).
Assessment of Fusarium Infection Incidence in Plant Roots
From each soil sample, 20 small pieces of the thinnest roots were cut approximately one centimeter long, washed in 1% sodium hypochlorite for 30 seconds and sterilized distilled water before drying in sterilized paper towels. Five root pieces were transferred to two MGA plates each and incubated at room temperature for five days. Total infection incidence were calculated by considering the total number of root pieces as the 100% and the number of roots infected as the percentage of incidence (Singleton et al., 1992). A small piece of agar at the edge of the colonies identified as Fusarium was transferred to a PDA plate incubated at room temperature for five days and transferred to SNA and CLA for identification.
Statistical Analysis
Analysis of variance tests were done to establish the effect of soil amendments on the occurrence of the fungus, soil fungal density and root infection. Fisher's Least Significance Difference (LSD) was used to compare treatment group means. Shannon's diversity indices were applied to compare fungal species diversity. Species accumulation curves were generated by plotting the total number of species recorded per sample from bean and maize rhizosphere soils and roots from all the treatments.
RESULTS
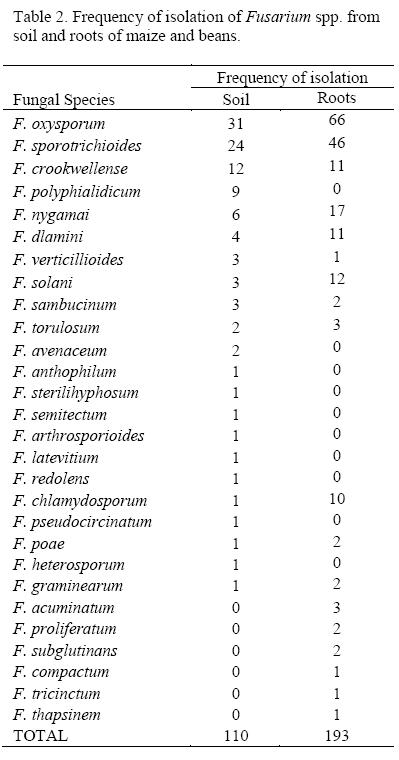
A total of 303 isolates oí Fusarium sp. were recovered, of which 164 were from the roots representing 18 species while 110 were from the soil and represented 22 species (Table 2). The most frequently isolated species were F. oxysporum and F. sporotrichioides in both soil and roots. The frequency of isolation and diversity oí Fusarium varied with treatment (Table 3). The fungus was more abundant and diverse in plots treated with Trichoderma and Manure. Plots treated with Mijingu + CAN recorded the least frequency of isolation and diversity. Plots treated with Trichoderma had the highest frequency of isolation and diversity of the fungus in the roots too. Mavuno and Mavuno + Trichoderma also recorded values higher than the control. Fusarium was most rare and least diverse from roots in Mijingu + CAN treated plots.
Fusarium inoculum density in soil varied significantly across treatments (p<0.001, Table 4). Plots treated with Mijingu + CAN fertilizer had the least amount of inoculum followed by those treated with Trichoderma seed coat and Manure. The highest inoculum density was recorded in plots treated with TSP + CAN. Mavuno,and Mavuno + Trichoderma recorded the lowest inoculum levels.. Root infection varied significantly with treatment (p=0.052). Mijingu + CAN had the highest infection incidence while TSP + CAN the least. The mean values (Table 5) showed variation with Trichoderma treatment recording the highest root infection for beans while Mijingu + CAN treatment recorded the highest root infection for maize. There was a significant interaction between crop type and treatment on Fusarium root infection . Soil inoculum density was highest from the TSP + CAN for both bean and maize rhizosphere and least in Mijingu + CAN. This difference was significant at pO.001. Crop type alone did not significantly affect the soil Fusarium abundance.
Soil amendments significantly influenced Fusarium richness in the roots , but not in the soil rhizosphere (Table 6). Trichoderma treated plots had the highest number of species in the roots followed by Mavuno + Trichoderma. Plots treated with TSP + CAN and Manure recorded levels lower than Control while Manure + Trichoderma treatment presented the lowest root infection. Mavuno and Mijingu + CAN plots had levels of root infection similar to Control plots according to Fisher's LSD grouping. Species diversity was influenced by crop type with bean rhizosphere being more diverse with Fusarium compared to maize rhizosphere as shown by Shannon indices (Table 7) and species accumulation curve (Fig 1a). However, the fungus was more diverse (Table 7) and rich (Fig 1b) in maize than bean roots. Fertilizer treatment influenced species accumulation in soils and roots with Trichoderma and manure treated plots having the highest number of species both in the soil and roots (Fig 2a, b).
DISCUSSION
Fusarium is of widespread distribution in soils and root tissues. Soil fertility management influenced the occurrence, diversity and abundance of this fungus. Plots treated with Mavuno fertiliser recorded the least root infection followed by TSP + CAN, Mavuno + Trichoderma and Manure + Trichoderma. These fertilizers may have controlled root infection by improving plant growth due to nutrient availability (Kapkiyai et al., 1996). Low soil fertility has been reported to cause poor bean production in many parts of Kenya because of root rot by Fusarium solani sp. phaseoli, Rhizoctonia solani and Pythium species (CIAT, 1992, Gitu,1992). Application of cultural methods such as crop rotations to maintain the pathogen at low levels have not been successful due to the small acerages of smallholder farms leaving the use of farm amendments as the only affordable option (Hall and Phillips, 1992; Mutitu et al., 1985 and 1989; Otsyula and Ajanga,1994)
Addition of fertilizer or organic manures may also affect the pathogens themselves. The population of soil Fusarium was markedly controlled in plots treated with Mijingu + Can fertilizer followed by Manure and Trichoderma However these treatments did not reduce root infection. TSP + CAN and manure promoted occurrence and diversity of Fusarium sp. in soil while controlling root infection. The effect of the fertilizer on the fungus could be encouraging competitive fungi to grow in the soil thereby reducing population of Fusarium. Alternatively the fertilizer may have made the environment of the soil non- conducive for Fusarium proliferation. Kimani et al., (2001) reported that manures or other organic inputs applied to the soil control the rate, pattern and extent of growth and activity of soil organisms and provide the source of carbon, energy and nutrients for the synthesis of soil organic matter. Manure can increase the humus content of soils by 15-50%, depending on soil type, in addition to increasing soil aggregate stability and root permeability (Kapkiyai et al., 1996). Kapkiyai et al. (1996) reported that manuring restocks the particulate organic matter fraction better than fresh crop residues. Manure also acts as a buffer, thus improving nutrient uptake for crops grown in acid soils. Using manures alleviates aluminium toxicity and improves the availability of nutrients such as P, particularly in soils with a high phosohorus (P) fixation, and sulphur (S). Manure also supplies essential elements such as Mg, and trace elements which may not be available in commonly used inorganic fertilizers. This indicates that manure still remains an important fertilizer and source of nutrients both to the plant and soil organisms.
Trichoderma as a bioherbicide also had promising effects. The bio-inoculant, Trichoderma, and the organic amendment, manure were second to Mijingo + CAN in controlling the density of soil Fusarium. However, roots from these treatments recorded high infection incidences. Addition of Trichoderma with other fertilizers reduced root infection more than the bio-inoculant or the fertilizer applied alone. Trichoderma as seed coat alone is not sufficient as a control of Fusarium. The fungus should be mixed with other fertilizers as manure and mavuno to promote its growth and effectiveness.
The interaction between crop rhizosphere and soil management influenced the diversity of Fusarium. Crop type alone did not have an effect. Fusarium species diversity was higher in bean than maize rhizosphere. However maize roots showed higher diversity of Fusarium root infestation than beans suggesting that the type of crop species is important when choosing a disease management strategy.
CONCLUSION
Integrated soil fertility management interventions could be used to control Fusarium root infections. The type of intervention chosen could be based on crop type and the fungus to be controlled. Overall, Mavuno fertilizer rated highest in this case followed by TSP + CAN which implied that chemical fertilizer use and low fertility could be the major cause for high Fusarium incidence in soil and use of the fertilizers would reduce the abundance.
REFERENCES
Booth, C, 1971. The Genus Fusarium. Commonwealth Mycological Institute. Kew Surrey, England. [ Links ]
Burgess, L.W., Summerell, B. A., Bullock, S., Gott, K. P., and Backhouse, D., 1994 'Laboratory manual for Fusarium Research.' (Fusarium Research Laboratory, University of Sydney and Royal Botanic Gardens: Sydney). [ Links ]
CIAT, 1992. Pathology in Africa. CIAT Annual Report, 1992. Bean Programme, Cali, Colombia. 385pp. [ Links ]
Gitu, K.W., 1992. Agriculture Data Compendium. Technical Paper 92-10. Long Range Planning Division, Ministry of Planning and National Development, Government of Kenya. [ Links ]
Hall, R. and Phillips, L.G., 1992. Effects of crop sequence and rainfall on population dynamics of Fusarium solani f. sp. phaseoli in soil. Canadian Journal of Botany 70:2005-2008. [ Links ]
Kapkiyai J., Woomer, P. Oireshi I, Smithson, P. and Karanja N., 1996. Effects of Fertiliser and Organic Inputs on Soil Organic Matter and Nutrient Dynamics in Kenyan Nitisol. Paper presented to the International Symposium "Carbon and Nutrient Dynamics in Natural and Agricultural Tropical Ecosystems", Harare, Zimbabwe, 29 April-4 May 1996. [ Links ]
Kedera, C.J., R.D. Platter and A.E. Desjadins, 1998. Incidence of Fusarium spp and levels of fumonisins in maize in Western Kenya. Applied and Environmental Microbiology. 65: 41-44. [ Links ]
Kedera, C.J., J.F. Leslie and L. E. Claflin, 1992. Systematic Infection of corn by Furasium moniliforme (abstr.) Phytopathology, 82: 1138. [ Links ]
Kimani SK, Mángale, N. Gichuru, M., Palm, C and Wamuongo, J., 2001. lntergrated Use and Effects of Manures with modest Application of Inorganic Fertilisers on soil properties and maize production in the central Kenya Highlands Final Technical Report to the Rockfeller Foundation May 2001. [ Links ]
Leslie J and Summerell B., 2006. The Fusarium Laboratory Manual. Blackwell Publishing 388 pages. [ Links ]
MacDonald, M.V. and R. Chapman, 1997. The incidence of Fusarium moniliforme on maize from Central America, Africa and Asia during 1992-1995. Plant Pathology: 46:112-126. [ Links ]
Munkvold GP, Desjardins AE., 1997. Fumonisins in maize. Can we reduce their occurrence? Plant Dis. 81 556-584. [ Links ]
Mutitu, E.W., Mukunya, D.M. and Keya, S.O., 1989. Effect of organic amendments of Fusarium yellow disease on the bean host. Discovery Innovation 1:67-70. [ Links ]
Mutitu, E.W., Mukunya, D.M and Keya, S.O., 1985. Biological control of Fusarium yellows on beans caused by Fusarium oxosporium Schl. F. sp. phaseoli Kendrick and Synder using organic amendments locally available in Kenya. Acta Horticulture 153:267-274. [ Links ]
Nderitu J.H, Buruchara R.A, Ampofo K.O., 1997. lntergrated pest management of beans. African Highland Initiative. [ Links ]
Otsyula, R.M. and Ajanga, S.I., 1994. Control strategy for bean roor rot in Western Kenya Proceedings of the fourth KARI Scientific Conference held in Nairobi, Kenya. [ Links ]
Otsyula, R.M., Ajanga R.A. Buruchara and C.S. Wortmann, 1998. Development of an intergrated bean root rot control strategy for Western Kenya. African Crop Science Journal 6: 61-67. [ Links ]
Robledo-Robledo E., 1991. Strategies for the prevention and control of fungi and mycotoxins in Central and South America. In: Champ BR, Highely E. Hocking AD, Pitt JI. Eds. Fungi and mycotoxins in stored products. Proceedings of an international conference, Bangkok, Thailand, 23-26 April 1991. p 39-46. [ Links ]
Scott PM., 1993. Fumonisins, Int. J. Food Microbiol. 18: 257-270. [ Links ]
Singleton, L. L., Mihail J. D. and Rush C. M., 1992. Methods for Research on soilborne phytopathogenic fungi. St Paul, MN, USA, American Phytopathological Society Press. 264 pages. [ Links ]














