Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Tropical and subtropical agroecosystems
versión On-line ISSN 1870-0462
Trop. subtrop. agroecosyt vol.14 no.3 Mérida sep./dic. 2011
Artículos de investigación
Response of chile pepper (Capsicum annuum L.) to salt stress and organic and inorganic nitrogen sources: III. Ion uptake and translocation
Respuesta de plantas de chile (Capsicum annuum L.) al estrés salino y fuentes orgánicas e inorgánicas de nitrógeno: III absorción y translocación de iones
Marco A. Huez-López1*, April L. Ulery2, Zohrab Samani2, Geno Picchioni2, Robert P. Flynn2
1Departamento de Agricultura y Ganadería. Universidad de Sonora. Rosales y Luis Encinas. C.P. 83000. Hermosillo, Sonora. México. e-mail: mhuez@guayacan.uson.mx.
2New Mexico State University. Las Cruces, New Mexico. USA.
* Corresponding Author
Submitted April 10, 2010
Accepted September 22, 2010
Revised received March 27, 2011
Abstract
The combined effect of salinity and two N sources on content, uptake rate and translocation of nutrients by chile pepper plants (Capsicum annuum L.) cv. Sandia was investigated in a greenhouse experiment. Either an organic-N liquid fertilizer extracted from grass clippings or ammonium nitrate, an inorganic fertilizer, were combined with three different soil salinity treatments (1.5, 4.5, and 6.5 dS m-1). Fertilizer treatments were two rates of organic-N fertilizer (120 and 200 kg ha-1) and 120 kg ha-1 of inorganic fertilizer. The combination of each N rate and source with the three salinity levels were arranged in a randomized complete block design replicated four times. The use of the organic-N source produced greater cation contents (Ca2+, Mg2+, Na+ and K+) in roots, shoots, and enhanced the uptake rates and translocation of cations to shoots compared to plants fertilized with inorganic -N. The root and shoot concentration, uptake rates and root-to-shoot transport of Cl- increased at increasing salinity. Higher contents of Cl- and cations in chile pepper shoots in relation to roots were observed. It was also observed that high N rate from the organic source enhanced the cation contents in both roots and shoots. Salinity diminished N content, N uptake rate and root to shoot transport in both roots and shoots.
Key words: green pepper; salinity; organic fertilizer; ion uptake rate; root-to-shoot ion transport.
Resumen
El efecto combinado de salinidad y dos fuentes nutrimentales a base de N en el contenido, tasa de absorción y translocación de nutrientes en plantas de chile (Capsicum annuum L.) cv. Sandia fue investigado en un experimento bajo condiciones de invernadero. Tres diferentes salinidades del suelo (1.5, 4.5, and 6.5 dS m-1) fueron combinados con un fertilizante orgánico nitrogenado líquido extraído de residuos de zacates o con un fertilizante inorgánico de nitrato de amonio. Los tratamientos de fertilización fueron dos dosis de fertilizante de N orgánico (120 y 200 kg ha-1) y 120 kg ha-1 de fertilizante inorgánico. La combinación de cada dosis y fuente de N con los tres niveles de salinidad fueron arreglados en un diseño de bloques completos al azar repetidos cuatro veces. El uso de la fuente orgánica de N produjo los mayores contenidos de cationes (Ca2+, Mg2+, Na+ and K+) en la raíz y parte aérea y mejoró las tasas de absorción y translocación de cationes a la parte aérea comparado con las plantas fertilizadas con N inorgánico. La concentración, tasa de absorción y transporte raíz-parte aérea de Cl- aumentó al incrementar la salinidad. Un mayor contenido de Cl- y cationes en la parte aérea en relación a la raíz fue observado. También fue observado que altas dosis de N de la fuente orgánica incremento los contenidos de cationes tanto en raíces como en la parte aérea. La salinidad disminuyó el contenido, tasa de absorción y transporte raíz-parte aérea de N tanto en raíces como en la parte aérea.
Palabras clave: chile verde; salinidad; fertilizante orgánico; tasa de absorción de iones; transporte de iones raíz-parte aérea.
INTRODUCTION
The inhibitory effects of salinity on plant growth and yield are caused by both lowering of the water potential of the root environment (and hence restricted water and ion uptake by plants) and the accumulation of ions in plant tissues at concentrations that may be toxic or to give rise to nutritional imbalances (Greenway and Munns, 1980; Grattan and Grieve, 1994). These nutrient imbalances in salt-stressed plants may result from the effect of salinity on nutrient availability, competitive uptake, transport or partitioning within the plants (Grattan and Grieve, 1999). Competition and interaction between Cl- and NO3- , and Na+ and Ca2+ in the substrate as well as within the plant frequently lead to ion imbalances that may result in nutrient deficiencies (Grieve and Shannon, 1999). The salt-stressed plant's requirements for the major ions (K+, Na+ and Ca2+ are determined both by the rate of growth and by the uptake of these solutes in the leaves (Pitman, 1984). Changes in nutrient content (uptake) of roots and shoots is one way of measuring the uptake rate of nutrients by plants (Fageria et al, 1991), and this uptake rate expressed per unit of total root dry weight is an index of the nutrient uptake efficiency of roots (Hunt, 1990).
Pepper (Capsicum annuum L.) is classified as a moderately sensitive crop (Maas and Hoffman, 1977), and some adverse effects of salinity on macronutrient content in its different plant parts have been reported (Gómez et al, 1996; Günez et al, 1996; Cornillon and Palloix, 1997; Tadesse et al, 1999; Chartzoulakis and Klapaki, 2000; De Pascale et al, 2003; Navarro et al, 2003).
Nutrient uptake and transport may be affected by the simultaneous presence of salts and nutrient elements (Alam, 1994). Salt ions (i.e. Na+ or Cl-) in high concentration in the soil solution are taken up at high rates by plants, which may lead to excessive accumulation in the tissue (Cramer et al, 1991). Salt uptake competes with the uptake of other nutrient ions, such as K+, Ca2+, N, P resulting in nutritional disorders and eventually, reduced yield and quality (Grattan and Grieve, 1999). So, the primary purpose of this study was to determine the effects of application of two N sources on the uptake rate, transport and accumulation of Ca2+, Mg2+, Na+, K+, Cl-, and N on roots and shoots of chile pepper plants grown at three salinity levels.
MATERIALS AND METHODS
Seedlings (three weeks old) of chile pepper {Capsicum annuum L.) cv. "Sandia" were transplanted to plastic pots filled with 15 kg of a non saline (ECe = 0.9 dS m-1), air dried soil passed through a 2-mm sieve. This soil is a Brazito sandy loam (Mixed thermic Typic Torripsamment) (Bulloch and Neher, 1980). The soil saline treatments consisted of three targeted soil salinity levels (Low: 1.5 dS m-1, Moderate: 4.5 dS m-1, and High: 6.5 dS m-1). The three salinity levels were prepared by adding solutions of a mixture of Na and Ca chloride (1:1 equivalent weight ratio), which were added to the original soil to obtain the desired soil salinity levels (electrical conductivity, ECe, dS m-1). Each pot was salinized one-time with the addition of saline solutions a day before seedling transplant.
Fertilizer treatments consisted of two N sources (inorganic and organic plant-based fertilizer). The inorganic source was ammonium nitrate. The organic liquid fertilizer was obtained from grass clippings, whose nutrients were extracted with water through an anaerobic bioleaching process (Saha, 2002). A rate of 120 kg N ha-1 (low N rate) inorganic fertilizer and two rates (120 and 200 kg N ha-1) organic, plant-based fertilizer were applied.
The experiment was initiated on July 15, 2001. Before transplanting, four seedlings were selected at random for initial root, stem, and leaf dry weights and leaf area. At the end of the experiment (72 days after transplanting), plants were collected by cutting their base close to the ground and separated into roots, stems, leaves, and washed with deionized water and then dried in a forced-air oven at 70 °C for 72 h. Dried plant material was ground in a Wiley mill and passed through a 40-mesh sieve.
The elements were determined on 0.1 g tissue samples of each part plant combined with 5 mL deionized water, 5 mL HNO3 (70%) and 2 mL H2O2 (30%) and digested in a microwave accelerated reaction system (MARS5, CEM Corp., Matthews, NC). Digestion solutions were allowed to cool to room temperature and adjusted to a final volume of 50 mL with deionized water. Elemental concentrations of Ca2+, Mg2+, K+, and Na+ were determined by ICP-OES (Inductively Coupled Plasma-Optical Emission Spectrometer, Perkin Elmer Instruments Model Optima 4300DV).
Tissue Cl ion concentrations were determined in 1.0 g subsamples of dry plant material extracted in 25 mL of deionized water and analyzed using silver ion titration with a Buchler-Cotlove chloridometer (Digital chloridometer, Labconco Corporation, Kansas City). The total N concentration in plant parts was determined by the Kjeldahl method, which used wet digestion in concentrated sulfuric acid for 3.5 h at 400 °C (Gavlak et al. 1994). Analyses were made colorimetrically for NH4-N using an automated Technicon Autoanalyzer II (Technicon Instruments, Tarry town, NY).
Plant nutrient contents (mg plant-1) were calculated as the product of concentration (mg g-1) in the different parts and the plant material dry weights (g plant"). The specific uptake rates (SUR) of ions were calculated according to the relationship described by Hackett (1969):
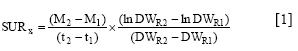
and the transport between root and shoot (SARRS) by
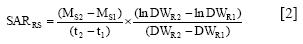
where M is the average amount of nutrient per plant calculated from nutrient concentration and plant dry weight (DW, g) at times (/, days) 1 (transplanting) and 2 (end of experiment); DWR is the mean root dry weight (g) at times 1 and 2, and Ms is the average amount of nutrient x in the shoot at times 1 and 2.
The experiment was arranged in a completely randomized block design, and the data were analyzed following the General Linear Model (GLM) procedure of the SAS (SAS Inst, Inc., Cary, NC, 2002). If differences were significant, means separation was done by Duncan's Multiple Range Test, and a probability level < 0.05 was considered significant. Orthogonal contrasts were applied to determine if a linear or quadratic trend for nutrient content, uptake rate and root-shoot transport existed among the soil salinity treatments for each N rate and source combinations.
RESULTS AND DISCUSSIONS
Root and shoot content of macro-elements Calcium
Significant decreases were observed in root Ca2+ content of pepper plants fertilized with the low inorganic-N rate, as well as in plants fertilized with the high organic-N rate (Table 1). The first case presented a significant quadratic trend, and the second, a linear trend. The only statistical differences in root Ca2+ content of pepper plants grown at moderate soil salinity were observed between the N rate and source combination treatments. On the other hand, increased soil salinity did not affect shoot Ca2+ content when plants were fertilized with any N rate and source combination (Table 2). However, plants grown at low and high soil salinity levels showed statistical differences between fertilizer treatments. Whether significant or not, plants fertilized with the organic-N source had higher Ca2+ content in roots and shoots than those fertilized with the inorganic-N source.
Cabañero et al. (2004) observed that Ca concentration in roots of pepper plants grown in nutrient solution did not show significant differences with plants treated with NaCl (50 mM) + CaCl2 (10 mM) but decreased in plants treated with 50 mM NaCl alone. Gomez et al. (1996) also observed that an interaction between salinity and N fertilization reduced Ca2+ uptake in leaves of sweet pepper plants. Contrarily, Aktas et al. (2005) showed that Ca2+ uptake in leaves of pepper plants was not different in salinity levels of 2.2 and 4.0 dS m-1, but increased significantly at salinities of 7.5 dS m-1. Flores et al. (2001) reported that Ca concentration in leaves of salt-stressed tomato plants was not affected by salinity but N fertilization had a significant effect.
Calcium is a divalent ion and as ions increase in valence, uptake decreases (Marschner, 1995). High concentrations of Na+ in soils can have an adverse effect on Ca2+ uptake so increasing salinity decreases Ca2+ uptake, and its transport to the shoot as a result of the high Na/Ca ratio in the soil solution. Another consequence of these Na:Ca interactions is the reduction of K+ content in salinized plants. Calcium deficiency did not occur in our experiments due to the application of saline solutions at equivalent ratio of Na:Ca (1:1). However, supplementation with the high N increased the plant Ca2+ content mainly in the shoots.
Magnesium
Root as well as shoot Mg2+ content followed a similar pattern to root and shoot Ca2+ content (Tables 1 and 2). However, Mg2+ contents were lower than Ca2+ contents in both roots and shoots. Calcium is strongly competitive with Mg2+ and binding sites on the root plasma membrane appear to have lower affinity for the highly hydrated Mg ion than for Ca2+ (Marschner, 1995; Garg, 1998). Also, the uptake of Mg2+ is seriously affected by an excess of K+ (Mengel and Kirkby, 1987). Flores et al. (2001) reported that leaf Mg2+ concentration in salt-stressed tomato plants was not affected by increasing salinity but increased NO3" :NH4+ ratio reduced leaf Mg2+ concentration. Also, Gómez et al. (1996) reported an interactive effect between salinity and N rate in leaf Mg2+ content of salt-stressed sweet pepper plants where first, salinity up to 25 meq L-1 NaCl increased the Mg2+ content and then decreased progressively at salinities of 50 and 100 meq L-1 NaCl. These results agree with those observed by Fernandez-Garcia et al. (2004) in two tomato cultivars where Mg concentration in leaves was significantly increased up to a salinity level of 5.3 dS m-1 and then decreased at a salinity level of 8.3 dS m-1.
Our data showed that not only the uptake but also the transport of Mg2+ from the roots to the shoot was restricted by K+ and Ca2+. However, the classical intervenial yellowing or chlorosis Mg2+ deficiency symptoms were not observed in our experiments.
Potassium
In this study, root K+ content did not differ between soil salinity treatments and it was also not affected by fertilizer treatments (Table 1). However, comparison across fertilizer treatments showed that only shoot K+ content of plants fertilized with the high organic-N rate exhibited statistical differences as soil salinity increased (Table 2). No significant effect existed in shoot K+ content between N rate and source combination treatments. Aktas et al. (2005) found no differences in leaf K+ content of pepper plants grown at salinities up to 7.5 dS m"1. Contrarily, Gómez et al. (1996) reported that leaves of salt-stressed sweet pepper plants fertilized with 2 meq L-1 N decreased the K+ content but that additional N applications (15 meq L-1 N) increased leaf K+. Also, Giines et al. (1996) found that shoot K+ content in pepper plants was significantly reduced at increasing salinities from 2.1 to 9.5 dS m-1.
In a saline environment, plants take up excessive amount of Na+ at the cost of K+. A reduction in K+ concentration could inhibit growth by reducing the capacity of osmotic adjustment and turgor maintenance (Rengel, 1992). Our data indicate that root and shoot K+ content were greater than Na+ content suggesting that K+ may act as the major monovalent cationic osmoregulator. The ability of plants to retain K+ and to maintain K+/Na+ selectivity has been considered a key feature of salt tolerance (Munns, 2002; Tester and Davenport, 2003).
Sodium
Means comparison between fertilizer treatments and between soil salinity levels showed no differences in root Na+ content (Table 1). A major characteristic of salt-stressed plants is the accumulation of Na+ and impairment of K+ nutrition (Cakmak, 2005). However, the addition of adequate Ca2+ influences the K+/Na+ selectivity by shifting the uptake ratio in favour of K+ at expense of Na+ (Grattan and Grieve, 1999). Increasing Ca2+ concentration restores selectivity to the K+ channels, thereby reducing Na+ toxicity and also K+ deficiency (Reid and Smith, 2000). So, similar to the results of Chartzoulakis and Klapaki (2000), the Na+ exclusion combined with the ability to maintain relatively a high K+ concentration in leaves provided pepper plants with a successful mechanism for tolerance to low and moderate salinity levels.
Regarding shoot Na+ content, no significant differences were observed between N rate and source combination treatments in any soil salinity level (Table 2). However, plants fertilized with the low organic-N rate exhibited a decrease in shoot Na+ content at moderate salinity returning to beginning levels at high salinity.
Increases of shoot Na+ content of pepper plants were reported by Günes et al. (1996) when salinity increased from 2.1 to 9.5 dS m-1. Similar results were reported by Gómez et al. (1996) in leaves of sweet pepper plants grown at salinities up to 100 meq L-1 NaCl and fertilized with 2 meq N L-1. However, the addition of N up to 15 meq L-1 decreased the leaf Na content in their study. Navarro et al. (2003) also found that the Na+ concentration in the leaf sap of pepper plants increased significantly with salinity from 2 to 8 dS m-1. Results of our experiment show that Na+ content in the shoots of pepper plants were higher than Na+ content in roots. This agrees with Blom-Zandstra et al. (1998) who found that the Na+ content in stems were greater than that in roots of pepper plants exposed to 15 mM NaC1.
In addition to individual nutrient concentrations, the ratios of various nutrients are also important in determining plant productivity. E.g., Ca deficiency is a critical nutritional imbalance problem which occurs as a result of either high soluble Na+/Ca2+ or Mg2+/Ca2+ ratios in the growth medium (Garg, 1998). In our study, the application of organic-N fertilizer enhanced the uptake of Ca2+ and Mg2+ in the roots and shoots of pepper plants compared to those fertilized with inorganic-N.
The presence of high concentrations of Na+ and Cl" in the soil solution may depress nutrient-ion activities and produce extreme ratios of Na+/Ca2+, Na+/K+, Ca2+/Mg2+, and C17NO3" (Grattan and Grieve, 1999) and could produce a high uptake of these ions and contribute to their increased flux into xylem (Navarro et al., 2003). Salt tolerance in plants is usually associated with the ability to restrict the uptake and/or transport of saline ions (Na+ and Cl") from roots to shoots.
Net nutrient uptake rate and root-shoot transport
Calcium
Ca2+ uptake rates (Table 3) and root-shoot transport (Table 4) in plants fertilized with the low inorganic-N rate increased in response to salinity. Linear trends were observed in both parameters with an additional quadratic component in Ca2+ root-shoot transport whereas no significant differences were observed in salt-stress plants fertilized with organic-N. However in plants grown at high soil salinity, both Ca2+ uptake rate and root-shoot translocation were lower in the inorganic-N treatment than at either rate of the organic-N fertilizer.
Magnesium
The rate of Mg2+ uptake per unit weight of root per day and its transport to the shoot both increased with salinity in plants fertilized with the low inorganic-N rate (Tables 3, and 4). At high application rates of the organic-N fertilizer, these parameters decreased at moderate salinity and increased again with high salinity stress. For plants grown in high soil salinity, the Mg2+ uptake rate and root-to-shoot transport were higher in the organic-N treatments, regardless of application rate.
According to Marschner (1995), the uptake rate of Mg2+ can be strongly reduced by other cations such as K+, NH4+ and Ca2+.
Potassium
With the exception of plants fertilized with the high organic-N rate, analysis of variance indicated no effect of soil salinity or fertilizer treatments on K+ uptake rate (Table 3). Contrary to these results, Taleisnik and Grunberg (1994) found that the net K uptake rate in whole plants decreased significantly in two salt-stressed tomato cultivars compared to non salt-stressed plants.
Potassium root-to-shoot transport was not affected by either N rate or source (Table 4). The high salt-stressed plants fertilized with organic-N had higher root-to-shoot transport of K+ than those treated with inorganic N (Table 17). Although root-to-shoot K+ transport in plants fertilized at the higher N rate of either source did not differ, plants fertilized with the organic-N source had greater root-shoot K+ transport.
Sodium
Plants fertilized with the organic-N source at both rates had increased Na+ uptake rates as salinity increased (Table 3). Both organic-N rates displayed linear trends, and those plants fertilized with the low organic-N rate also showed a quadratic trend. Moderately salt-stressed plants fertilized at the high rate with organic-N had higher Na+ uptake than plants grown at the lower N rates regardless of source. Contrasting results were obtained by Taleisnik and Grunberg (1994) in two tomato cultivars where increasing salinity decreased significantly both net Na uptake rate and translocation to shoots in whole plants. Rubio et al. (2003) found that adding K+ and Ca2+ to the nutrient solution, the net Na+ uptake was inhibited in pepper plants. They stated that a balanced mineral nutrition can contribute to increasing pepper salt tolerance.
Content, uptake rate, and translocation of chloride
As expected, an increased Cl- accumulation in response to salinity was observed in this study. In general, root and shoot Cl" contents increased as salinity. However, only plants fertilized with the high organic-N rate showed statistical differences in the amount of root Cl- with increasing salinity (Table 5). On the other hand, shoots accumulated higher amounts of Cl" than roots. Marked differences were observed in shoot Cl- content for any N rate and source combination treatment as a result of salt stress. The amount of shoot Cl" in moderate and high salt-stressed plants fertilized with the low inorganic-N rate was 5 to 7 times higher than the amount of Cl- in the shoots of low salt-stressed plants. In plants fertilized with the same organic-N rate, the increases were 4 to 5 times more in moderate and high salt-stressed plants compared to those of low salt-stressed plants. In plants fertilized with the high organic-N rate, shoot Cl- content increased 2 to 3 times in moderate and high salt-stressed plants compared to low salt-stressed plants. Significant linear trends were observed in those treatments that showed statistical differences. Only shoot Cl- content of the low salt-stressed plants showed statistical differences between N rate and source combinations. The highest Cl- contents were found in roots and shoots of plants fertilized with the inorganic-N source. The high concentration of Cl- within the plant could be directly or indirectly responsible for reduced plant growth, either depressing the uptake of other anions such as nitrate (Bernal et al., 1974; Grattan and Grieve, 1999) or taking up excessive amounts of Cl- which may cause leaf injury, thereby decreasing photosynthesis and productivity (Alam, 1994; Chartzoulakis and Klapaki, 2000). De Pascale et al. (2003) reported that Cl- accumulation in leaves and fruits of salt-stressed pepper plants significantly decreased total and marketable yield.
Chloride is taken up very rapidly by plants and its uptake rate depends primarily on the concentration in the soil solution (Mengel and Kirby, 1987). Similar to shoot Cl" content, the Cl- uptake rate and transport to the shoot showed significant differences for any N rate and source combination under soil salinity treatments (Table 5). Both Cl" uptake rate and Cl" transport to the shoot parameters increased as soil salinity increased. However, no significant differences were observed in these two parameters between fertilizer treatments in any salinity level.
Data analysis from this study shows that Cl- contents were higher in shoots than in roots. Chartzoulakis and Klapaki (2000) found that Cl- contents were higher in roots than leaves of two hybrids of pepper plants grown in salinities from 1.2 to 17.8 dS m-1. They suggest that pepper plants have the ability to limit uptake and/or transport of Cl- and Na ions from the root to aerial parts. Also, Navarro et al. (2003) reported a reduction in Cl- content in leaves of pepper plants grown in salinities from 2 to 8 dS m-1. Contradictory results were reported by Lessani and Marschner (1978) who noted a progressive absorption of Cl" with increasing salinity. The same was reported by Günes et al. (1996) in shoots of salt-stressed pepper plants, and by Papadopoulos and Rendig (1983) and Flores et al. (2001) in leaves of salt-stressed tomato plants.
According to White and Broadley (2001), the critical tissue Cl" concentration for toxicity is about 4-7 and 15-15 mg g-1d-1 for Cl-sensitive and Cl--tolerant plant species. In our study, the lower values of Cl- transport to the shoot observed show that pepper plants restricted Cl- transport to the shoot withstanding Cl- toxicity.
Content, uptake rate, and translocation of nitrogen
The N content in roots and shoots of chile pepper plants are summarized in Table 6. Salinity had a significant effect on root N content of plants fertilized with the low organic-N rate, reducing it linearly as much as 30% and 35% in moderate and high salt-stressed plants, respectively, while at the high organic -N rate, root N content decreased linearly about 30% in both moderate and high salt-stressed plants. Although not significant, the root N content of pepper plants receiving inorganic-N also decreased as soil salinity increased. Plants fertilized with the inorganic-N source had significantly greater root N content than those fertilized with the organic-N source at the low N rate.
Nitrogen content in shoots followed a similar pattern as that of the root N content but the trends were quadratic, that is, decreasing at moderate salinity and increasing at the high salinity level (Table 6). In comparison to the organic-N source, the application of the inorganic-N source significantly increased the shoot N content at all three salinity levels. Günes et al. (1996) also reported that shoot N content of salt-stressed plants grown between 6.1 and 9.6 dS m-1 decreased significantly compared to those grown at 2.1 dS m-1. However, De Paséale et al. (2003) observed that pepper plants grown at 4.4 and 8.5 dS m-1 had statistically similar N content in the leaves than those grown in nonsaline soils. Similar results were observed by Papadopoulos and Rendig (1983) in leaves of salt-stressed tomato plants at increasing N rates.
The uptake rate in whole pepper plants and root to shoot transport of N increased with increasing salinity in plants fertilized with the high organic-N rate (Table 6). In this fertilizer treatment, there were no differences between low and moderate salt-stressed plants in both root N uptake rate and transport to shoot. However, high salt-stressed plants had substantially higher N uptake and transport values. The inorganic-N applied to moderate or high salinity-stressed plants produced higher N uptake rates and transport than fertilization with the organic-N source. In general, comparisons within of each N source, showed that the high N applications gave higher N uptake rates and root to shoot transport than the low N applications. Similar results were observed by Flores et al. (2004) who reported that the N uptake rate in salt-stressed tomato plant fertilized with an inorganic -N source was higher with the application of 4.0 mmol NO3- L-1 than with the application of 2.0 or 0.5 mmol NO3- L-1.
CONCLUSIONS
The results of this experiment indicated that soil salinity and N-fertilizers had confounding effects on nutrient content, uptake rate and transport in chile pepper plants. These parameters were affected not only by salinity but also by the source and rate of N application.
The accumulation and translocation of Cl was enhanced under increased salinity. While in most cases there was no significance, the content of Ca, Mg, and Na in roots and shoots increased with increasing salinity, the content of K was not affected.
Salinity diminished N content, N uptake rate and root to shoot transport in both roots and shoots of chile pepper plants. Although these parameters were less decreased with the inorganic-N source application, the organic-N supply enhanced the effectiveness of nitrogen use which is vital to enhance the fruit yield in plants.
In general, it was observed that the application of organic-N fertilizer produced greater cation content in roots, shoots, cation uptake rates and root-to-shoot transport than the inorganic-N. Also, high N rates increased the values of these parameters. The increased content of these elements may have resulted from their addition in the extracted grass clippings. So, this experiment shows organic liquid fertilizer from grass clippings to be an effective source of nutrition for pepper and other vegetable crops.
REFERENCES
Alam, S.M. 1994. Nutrient uptake by plants under stress conditions. In: Handbook of plants and crop stress. Edited by M. Pessarakli. Mercel Dekker, Inc. New York. [ Links ]
Aktas, H., L. Kami, D. Chang, E. Turhan, A. Bar-Tal, and B. Aloni. 2005. The suppression of salinity-associated oxygen radicals production, in pepper {Capsicum annuum) fruit, by manganese, zinc and calcium in relation to its sensitivity to blossom-end rot. Physiologia. Plantarum. 123:67-74. [ Links ]
Bernal, C.T., .T. Bingham, and J. Oertli. 1974. Salt tolerance of Mexican wheat. II Relation to variable sodium chloride and length of growing season. Soil Science Society American Proceeding. 38:777-780. [ Links ]
Blom-Zandstra M, Vogelzang G, Nublat SA, Veen BW. 1998. Sodium fluxes in sweet pepper exposed to varying sodium concentrations. Journal of Experimental Botany. 49:1863-1868. [ Links ]
Broadley, M.R., H.C. Bowen, H.L. Cotterril, J.P. Hammond, M.C. Meacham, A.Mead, and P.J. White. 2003. Variation in the shoot calcium content of angiosperms. Journal of Experimental Botany. 54:1431-1446. [ Links ]
Bulloch, H.E, and R.E. Neher. 1980. Soil survey of Dona Ana County Area, New Mexico. United States. Department of Agriculture, Soil Conservation Service in cooperation with United States Department of the Interior, Bureau of Land Management. New Mexico Agricultural Experiment Station. [ Links ]
Cabañero, F.J., V. Martinez, and M. Carvajal. 2004. Does calcium determine water uptake under saline conditions in pepper plants, or is it water flux which determines calcium uptake. Plant Science. 166:443-450. [ Links ]
Cakmak, I. 2005. The role of potassium in alleviating detrimental effects of abiotics stresses in plants. Jornal of Plant Nutrition and Soil Science 168:521-530. [ Links ]
Chartzoulakis, K., and G. Klapaki. 2000. Response of two greenhouse pepper hybrids to NaCl salinity during different growth stages. Scientia Horticulturae. 86:247-260. [ Links ]
Cornillon, P., A. Palloix. 1997. Influence of sodium chloride on the growth and mineral nutrition of pepper cultivars. Journal of Plant Nutrition. 20:1085-1094. [ Links ]
Cramer, G.R., E. Epstein, and A. Lauchli. 1991. Effects of sodium, potassium and calcium on salt-stressed barley. II. Elemental analysis. Physiologia Plantarum. 81:197-202. [ Links ]
De Pascale, E., C. Ruggiero, G. Barbieri, and A. Maggio. 2003. Physiological responses of pepper to salinity and drought. Journal of the Amererican Society of Horticultural Science. 128:48-54. [ Links ]
Fageria, N.K., V.C. Baligar, and C. A. Jones. 1991. Growth ad mineral nutrition of field crops. Marcel and Dekker, Inc. [ Links ]
Fernández-Garcia, N., V. Martinez, and M. Carvajal. 2004. Effect of salinity on growth, mineral composition, and water relations of grafted tomato plants. Journal of Plant Nutrition and Soil Science. 167:616-622. [ Links ]
Flores, P., M. Carvajal, A. Cerda, and V. Martinez. 2001. Salinity and ammonium/nitrate interactions on tomato plant development, nutrition, and metabolites. Journal of Plant Nutrition. 24:1561-1573. [ Links ]
Garg, B.K. 1998. Role of calcium in plants under salt stress. Annals of Arid Zone. 37:107-118. [ Links ]
Gomez, I., J. Navarro-Pedrero, R. Moral, M.R. Iborra, G. Palacios, and J. Mataix. 1996. Salinity and nitrogen fertilization affecting the macronutrient content and yield of sweet pepper. Journal of Plant Nutrition. 19:353-359. [ Links ]
Grattan, S. R., and CM. Grieve. 1999. Salinity-mineral nutrient relations in horticultural crops. Scientia Horticulturae. 78:127-157. [ Links ]
Grattan, S. R., and CM. Grieve. 1994. Mineral nutrient acquisition and response by plants grown in saline environments. In: Handbook of plants and crop stress. Edited by M. Pessarakli. Mercel Dekker, Inc. New York. [ Links ]
Greenway, H, and R. Munns. 1980. Mechanisms of salt tolerance in non-halophytes. Annual Review of Plant Physiology and Plant Molecular Biology. 31:149-190. [ Links ]
Grieve, CM., and M.C. Shannon. 1999. Ion accumulation and distribution in shoot components of salt-stressed eucalyptus clones. Journal of the American Society of Horticultural Science. 124:559-563. [ Links ]
Günes, A., A. Inal, and M.Alpasian. 1996. Effect of salinity on stomatal resistance, proline, and mineral composition of pepper. Journal of Plant Nutrition. 19:389-396. [ Links ]
Hackett, C. 1969. A study of the root system of barley. II. Relationships between root dimensions and nutrient uptake. New Phytologist. 68:1023-1030. [ Links ]
Hunt, R. 1990. Basic growth analysis: plant growth analysis for beginners. Academic Press, London, UK. [ Links ]
Lessani H., and H. Marschner. 1978. Relation between salt tolerance and long-distance transport of sodium and chloride in various crop species. Austalian Journal of Plant Physiology. 5:27-37. [ Links ]
Maas, E.V., and G.J. Hoffman. 1977. Crop salt tolerance-Current assessment. Journal of Irrigation and Drainage Divsion. ASCE 105:115-134. [ Links ]
Marschner, H. 1995. Mineral nutrition of higher plants. Academic Press: London. [ Links ] Mengel, K, and E.A. Kirkby. 1987. Principles of plant nutrition. 4th Edition. International Potash Institute. Switzerland. [ Links ]
Munns, R. 2002. Comparative physiology of salt and water stress. Plant and Cell Environment. 25:239-250. [ Links ]
Navarro, J.M., C. Garrido, V. Martinez, and M. Carvajal. 2003. Water relations and xylem transport of nutrients in pepper plants grown under two different salt stress regimes. Plant Growth Regulation. 43:237-245. [ Links ]
Papadopoulos, I., and V.V. Rendig. 1983. Interactive effects of salinity and nitrogen on growth and yield of tomato plants. Plant and Soil. 73:47-57. [ Links ]
Pitman, M.G. 1984. Transport across the root and shoot/root interactions. In: Salinity tolerance in plant strategies for crop improvement. R. C. Staples and G. H. Toenniessen (Eds.) John Wiley & Sons, Inc. [ Links ]
Reid, R.J., and F.A. Smith. 2000. The limits of sodium/calcium interactions in plant growth. Australian Journal of Plant Physiology. 27:709-715. [ Links ]
Rengel, Z. 1992. The role of calcium intoxicity. Plant and Cell Environment. 15:625-632. [ Links ]
Rubio, F., P. Flores, J.M. Navarro, and V. Martinez. 2003. Effects of Ca2+, K+, and cGMP on Na+ uptake in pepper plants. Plant Science. 165:1043-1049. [ Links ]
Saha, R.K. 2002. Liquid fertilizer from organic waste using anaerobic process. [MSc thesis]. Las Cruces, NM. New Mexico State University. 96 p. Available from: Branson Library. NMSU. [ Links ]
Tadesse, T., M.A. Nichols, and K.J. Fisher. 1999. Nutrient conductivity effects on sweet pepper plants grown using a nutrient film technique. 2. Blossom-end rot and fruit mineral status. New Zealand Journal of Crop Horticultural Science. 27:239-247. [ Links ]
Taleisnik, E., and K. Grunberg. 1994. Ion balance in tomato cultivars differing in salt tolerance. I. Sodium and potassium accumulation and fluxes under moderate salinity. Physiologia Plantarum. 92:528-534. [ Links ]
Tester, M., and R. Davenport. 2003. Na+ tolerance and Na+ transport in higher plants. Annals of Botany. 91:503-527. [ Links ]
White, P.J., and M.R. Broadley. 2001. Chloride in soils and its uptake and movement within the plant: A review. Annals of Botany. 88:967-988. [ Links ]














