Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Ingeniería mecánica, tecnología y desarrollo
versión impresa ISSN 1665-7381
Ingenier. mecáni. tecnolog. desarroll vol.4 no.6 México mar. 2014
Artículos
Mechanical Characterization of Femoral Cartilage Under Unicompartimental Osteoarthritis
Vidal-Lesso A.1, Ledesma-Orozco E.2, Daza-Benítez L.3, Lesso-Arroyo R.1
1 Departamento de Ingeniería Mecatrónica, Instituto Tecnológico de Celaya. Celaya, Guanajuato, México.
2 Departamento de Ingeniería Mecánica, Campus Irapuato-Salamanca, Universidad de Guanajuato. Salamanca, Guanajuato, México.
3 Unidad Médica de Alta Especialidad HGP No. 48, IMSS, León, Guanajuato, México.
Fecha de recepción: 12-12-2013
Fecha de aceptación: 25-02-2014
Abstract
The aim of this study was to determine the mechanical properties and thickness of articular cartilage in the unaffected femoral regions in cases of unicompartimental osteoarthritis on the knees. The specimens were tested using a 3mm plane-ended cylindrical indenter and a displacement of 0.5mm was applied at specific points in seven femoral knee cartilages with unicompartimental osteoarthritis. The thickness, stiffness, elastic modulus, shear modulus and bulk modulus were obtained. These properties and thickness were compared with those reported from previous studies.
Our findings showed that mechanical properties reported herein present a major magnitude (up to 53%) than those from the previous studies on healthy cartilage.
However, the thickness reported in previous studies is larger (up to 69%) than the thickness measured in cartilage specimens of this work. In addition, results showed that analyzed points related to the displacement line in the kinematic flexion axis of the compartments, have significantly (p<0.05) similar thickness and elastic, shear and bulk modulus.
Mechanical properties reported into this work for knees with unicompartimental osteoarthritis could be used to analyze cases of unicompartimental knee arthroplasty by means of numerical simulation and to predict the biomechanics behavior of the joint.
Keywords: osteoarthritis, cartilage, mechanical properties, knee joint, biomechanics.
Resumen
El objetivo de esta investigación fue determinar las propiedades mecánicas y el espesor de cartílago articular en regiones femorales no afectadas en casos de osteoartritis unicompartimental de rodilla. Se realizaron pruebas en los especímenes utilizando un indentador cilíndrico plano de 3mm de diámetro y se aplicó un desplazamiento de 0.5mm en puntos específicos de cartílago femoral de siete rodillas con osteoartritis unicompartimental. Se obtuvieron el espesor, la rigidez, el módulo elástico, modulo cortante y volumétrico. Estas propiedades y espesores fueron comparados con trabajos previos de otros autores para cartílago sano.
Los resultados mostraron que las propiedades mecánicas obtenidas en este trabajo para cartílago articular con osteoartritis unicompartimental presentan una mayor magnitud (hasta 53%) respecto a las obtenidas para cartílago sano en trabajos previos. Sin embargo, se encontró que el espesor de cartílago reportado para cartílago sano es mayor (hasta 69%) que el medido en los especímenes de este trabajo. Adicionalmente, los resultados mostraron que los puntos de análisis correspondientes a la línea de desplazamiento en el eje de flexión cinemática de los cóndilos presentan una significativa (p<0.05) similitud en espesor y módulos elástico, cortante y volumétrico.
Las propiedades mecánicas obtenidas en este trabajo para cartílago de rodilla en casos de osteoartritis unicompartimental pueden ser utilizadas para analizar la artroplastia unicompartimental de rodilla a través de simulación numérica y predecir el comportamiento biomecánico de la articulación.
Palabras clave: osteoartritis, cartílago, propiedades mecánicas, rodilla, biomecánica.
Introduction
The articular cartilage works as a mechanical damping surface in the synovial joints, experiencing finite deformation due to physiological loading [1, 2]. In unicompartimental osteoarthritis (UOA), a disease of the synovial joints, it is assumed that one compartment is damaged and the other one is normal, however, several studies [3, 4] have reported that the visual and histological appearance of the cartilage is not a good indicator of performance in a joint and it requires a biomechanical evaluation of the tissue.
Mechanical properties of each component in the knee, specifically the femur and tibia, have been as consider as orthotropic material by many research articles [5, 6]. In addition, elastic properties and models have been used to reproduce the mechanical behaviour of articular cartilage. Currently most research uses stress analysis on articular cartilage to predict a one phase, isotropic and homogeneous linearly elastic behaviour [5-8]. In these cases the mechanical properties considered were an elastic modulus (E) of 15MPa and a Poisson ratio (ν) of 0.475. Values of 5MPa for E and 0.46 for ν have been reported in finite element analysis for articular cartilage to determine the structural effect of menisci damage [9]. Also, an elastic modulus of 12MPa was presented in a tri-dimensional finite element analysis to study the structural response of cartilage and unicompartimental prosthesis in cases of unicompartimental knee arthroplasty [1]. The finite element analysis mentioned before were performed in order to study the biomechanics of the knee considering mechanical properties of healthy cartilage. In these studies, they considered the cartilage behaviour as an isotropic, linear material and homogeneous.
In addition, thickness and mechanical properties has been reported in other works. One of them reported thicknesses of 2.51±1.04mm for unaffected cartilage in cases with osteoarthritis and 3.23±0.63mm for control healthy compartments of the tibial plateau [3]. The mechanical properties and morphological characteristics of articular cartilage from tibial plateau of human knees including regions covered by the meniscus have been reported values from 2.13±0.74 MPa up to 5.13±1.91 MPa for elastic modulus and 4.87±3.75 N/mm up to 20.38±5.32 N/mm for cartilage stiffness have been reported [10]. Micro-indentation techniques were used to determine the permeability, aggregate and shear modulus of the porcine cartilage in the anterior, posterior, medial, lateral and central regions of each porcine compartment [11].
Currently, the dynamic response of femoral knee cartilage with no visual damage in cases under unicompartimental osteoarthritis has been reported. They found a maximum stiffness value of 83.34±38.32 N/mm for cycle 2 located in the medial point. They also reported a maximum value of 2.42±0.27 MPa for shear relaxation modulus and 23.32±2.66 MPa for bulk relaxation modulus [12].
The distribution of mechanical properties of femoral cartilage has not been reported in previous studies for femoral cartilage. Therefore, this study presents a study of the mechanical properties distribution and thickness of femoral cartilage in cases of unicompartimental osteoarthritis. This distribution can be used in finite element analysis to reproduce a more realistic behaviour of cartilage for the study of the biomechanics of the knee and unicompartimental prosthesis in cases of unicompartimental osteoarthritis.
Materials and Methods
Specimens
Seven femoral knee specimens were obtained from female patients diagnosed with unicompartimental osteoarthritis, which came from a non-cadaveric population with ages ranging from 59 to 73 years. The specimens were removed during surgery for total knee replacement, so it was possible to obtain the cartilage with subchondral bone of femoral compartment; they were immersed in a solution of formaldehyde in water and after two hours they were tested. The knee specimens were donated for scientific and medical research under administrative control of the Mexican Social Security Institute (IMSS) corresponding to the Institutional Review Board (IRB)/Ethics Committee.
Groups
Mechanical testing on cartilage of lateral femoral compartments was performed using an impermeable plane-ended cylindrical indenter. Five points were selected for cartilage indentation testing on the surface of each femoral compartment. Each point was identified and associated with a group (see Figure 1), as follows: AP, anterior point; CP, central point; LP, lateral point; MP, medial point and PP correspond to the posterior point.

Groups AP, CP, PP and MP, correspond to points whose load and wear in tibio-femoral contact was predominant. Group LP corresponds to points where the least load and wear was found due to the nature of loading on the knee.
The Schematics points were located in the specimens of lateral compartment removed from the femur for mechanical testing. The medial and lateral compartments were removed during surgery.
Indentation test
Elastic properties of different materials can be obtained through the indentation technique, which has been used for measuring the compressive properties of cartilage [13, 14].
In this study, instantaneous modules were determined rather than those in steady state because it behaves approximately as an incompressible elastic material of a single phase immediately after application of load, since the fluid inside of the articular cartilage is unable to flow due to the short time of load application [14-16]. It is important to mention that cartilage indentation in this study was performed following previous similar testing protocols of several studies [14, 15, 17]. Complete cartilage specimens with subchondral bone without any additional cutting were tested; this avoids any induced mechanical effects on the test site. A displacement of 0.5mm, corresponding to a real physiological displacement of 25-30% [18], was applied to the cartilage in axial direction with a 3mm diameter indenter attached to a 500N load cell of a materials testing machine.
We applied cycles of displacement, the applied strain rate of 0.21mm/s used in this work would correspond to a loading frequency of 0.1Hz. This value was used in other studies to analyze the mechanical response of bovine articular cartilage at physiological stress levels [19] and to study the enzymatic degradation under dynamic unconfined compression loading [20]. Also, this value is between the frequencies ranging from 0.01 to 2Hz, which keep the majority of interstitial fluid inside of the cartilage; these values reproduce the phenomenon created by the real physiological frequencies in the lower limbs of human body [19, 21-24]. Thus, the strain rate used in the present work to achieve the maximum displacement, reproduce the effect of physiological loading in the human knee joint.
Previous analyses [3, 14] have suggested that the indenter diameter should be small enough compared with the specimen diameter size, when plugs of cartilage with subchondral bone are taken; this is to avoid the stiffness effect of the subchondral bone on cartilage measurements. In this experiment, the method of testing the complete femoral compartment was similar to those tests performed in previous studies on tibia [10]. Plugs were not taken out of cartilage to avoid any induced mechanical effects on the test site and the stiffness effect of the subchondral bone on cartilage measurements.
During indentation testing, the specimen was fixed in a platform where it could be carefully adjusted to any position so that the cartilage surface was just in contact and perpendicular to the longitudinal axis of the indenter (Figure 2). Also, it is important to mention that before and after testing all plugs were immersed in a solution of formaldehyde in water, however during testing at room temperature of 23oC, the plugs were taken out and cartilage was hydrated applying a drop of distilled water every 30 seconds to prevent dehydration of cells and indentation surface, this reproduce the physiological normal state of hydration.

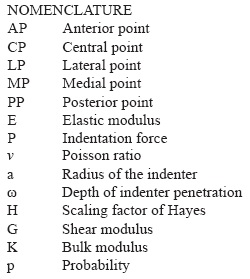
Finally, test runs were performed and force, time and displacement parameters were recorded during each test run (see Figure 3).
Cartilage thickness measurement
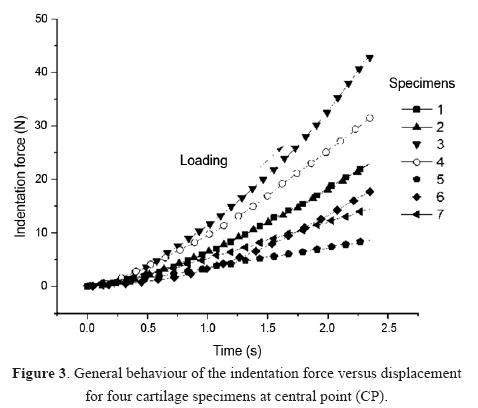
The cartilage thickness was determined using the needle method reported in previous works [10], which penetrates the cartilage with a thin needle at low constant speed of 0.21mm/s. The thickness was calculated as the distance between the detection of force on the surface and the rapid increase of it when the needle reaches the transition zone of calcified cartilage and subchondral bone (see Figure 4).
Mechanical properties
The parameters recorded in mechanical testing were used to obtain four mechanical properties: stiffness (N/mm), elastic modulus (MPa), shear modulus (MPa) and bulk modulus (MPa). Force values obtained in the indentation test were normalized to the corresponding depth of indenter penetration to obtain stiffness values. This procedure to calculate the stiffness was also reported in previous works presented by Thambyah A. et al. [10] and Lyyra T. et al. [15].
The elastic modulus was obtained using experimental data from indentation test, the geometrical characteristic of the indentation region in the specimen and the mathematical model proposed by Hayes et al. (Eq. 1) for indentation analysis of articular cartilage on a rigid body, which has been used in previous studies [10, 25, 26].

where E is the instantaneous elastic modulus; P, the applied load; , the Poisson ratio; a, the radius of the indenter; , the depth of indenter penetration and H is the scaling factor of Hayes et al. [25].
The scale factor values were reported by Hayes et al. [24] as a function of the ratio "a / h" and " " where "a" is the radius of the indenter, "h", the cartilage thickness and " " the Poisson ratio. In this model the Poisson ratio was taken as 0.45, considering the cartilage as a nearly incompressible material [1, 7, 14-16] and considering that the load application is in a short period of time [27]. The scale factors were obtained for all test points of each plug, so for this study these values were from 1.89 to 5.52.
The model reported by Hayes et al. [25] was related with the shear modulus considering the isotropic relation between them. Thus, the shear modulus (G) can be determined by:

In addition, the bulk modulus was obtained using the following relationship with the shear modulus:

where K represents the bulk modulus.
Results
Table 1 summarizes mean values and standard deviations (SD) of stiffness, elastic modulus, shear and bulk modulus and thickness obtained in different test groups for the lateral femoral compartment. Results showed that the maximum stiffness value was obtained at MP point, with a maximum variation of 42.71% respect to the lowest value located in LP point. The minimum variation values were presented in AP, CP and PP points; the CP value was stiffer compared to AP and PP, with a difference of 14.47% and 17.57% respectively. LP point has less stiffness (25.71%) and MP point is 29.67% stiffer than the central point, CP.

Similarly, the maximum elastic instantaneous modulus was located in MP point being 47.49% larger than the minimum value presented at the LP point. AP, CP and PP points have similar (p<0.05) elastic modulus with a maximum difference of 5.29% at AP and 4.37% at PP respect to CP point. It has an elastic modulus 45.35% larger than LP point and decrease 23.67% compared to the MP point.
Cartilage thickness measurements showed that points AP and PP were the thickest, while point LP was the thinnest. The maximum difference of cartilage thickness was 23% presented between AP and PP points respect to LP. Points AP, CP and PP have a very small difference between their thicknesses, being that CP is 6.5% less thick than AP and a similar difference was present between CP and PP. Cartilage thickness at points LP and MP are 17.64% and 4.81% thinner than cartilage of CP point, respectively.
The maximum shear modulus was located in the MP point (see Table 1), while the lowest value was located in LP point. PA, PC and PP presented similar values of shear modulus, with a maximum difference of 5.29% between them.
Bulk modulus presented in Table 1, showed that MP presented the largest bulk modulus and LP the smallest modulus, this was a similar behavior as the one presented for the shear modulus. In addition, AP, CP and PP showed values with a maximum difference between them of 5.24%.
Discussion
The Stiffness values showed in Table 1 for lateral compartment of cartilage in knees with unicompartimental osteoarthritis can be compared in magnitude with previous results of healthy cartilage [10, 15]. In these previous studies, authors measured the displacement obtained with the application of a constant force (0.5N) in healthy cartilage of a population with ages ranging from 62-80 years [10]. They reported stiffness values of 4.87 N/mm to 20.38 N/mm in different areas of cartilage. Moreover, force values have been reported in healthy cartilage of 2.4N and 3.1N. These values when normalized to displacement represent stiffness values of 8N/mm and 10N/mm for the medial and lateral compartment, respectively, in patients with mean age of 26 years [15]. In that study, an indentation of 300 microns was performed and the force response was measured. The present work reported values from 43.16N/mm to 75.34N/mm with a maximum displacement of 0.5mm. The difference in magnitudes that is presented in comparison with other authors is attributed to different reasons. The most obvious is that cartilage that was analyzed in other studies is healthy cartilage [10, 15] while in this study is on cartilage of knees with unicompartimental osteoarthritis. Furthermore, another reason to explain this difference is due to the ages of patients. One of the previous studies [15] referred its analysis to patients with mean ages of 26 years while in the present study the ages were between 59 to 73 years.
Displacement values applied to the specimens are another cause of the differences presented, due to the cartilage stiffness change according with them [28]. Analyzing loading curves obtained from the specimens in this work, it is possible to obtain stiffness values at different displacements between 0 and 0.5mm.
A maximum stiffness mean value of 42.64±10.58N/mm was obtained in the MP test point and a minimum mean value in the LP point of 22.75±8.45N/mm. These values correspond to a displacement between 0 to 0.15mm. Also, these values were compared with those reported for healthy cartilage [10] of 4.87N/mm and 20.38N/mm obtained with a displacement of 0.10mm and 0.02mm. It was observed that values obtained in the present work are larger than those obtained for healthy cartilage and these values tend to increase when the depth of penetration increases.
Moreover, a statistical analysis was performed using the Kruskal-Wallis test with the null hypothesis that the medians were similar between points (AP, CP and PP) that are in the displacement line of the kinematic axis of flexion. The degree of freedom of the Kruskal-Wallis test (N=7) was 2 and the critical chi-square value was 5.991 (α=0.05). Results of this test showed that AP, CP and PP have significantly similar (p<0.05) thickness values, elastic modulus, shear and bulk modulus. These points correspond to the displacement line of the kinematic axis of flexion and areas of cartilage-cartilage contact and load in the knee [29-31].
In addition, cartilage thickness measured in this study was compared with those obtained in previous studies [30, 32, 33]. The distribution of thicknesses reported by Li G. et al. [30] on femur were obtained using magnetic resonance techniques (MRI) on healthy knees of people with average ages of 27 years, these values are thicker than those obtained in the present study (see Table 2) for an older population with ages ranging from 59-73 years.
Moreover, using the same technique (MRI), an average thickness value of 2.2mm in healthy knees was reported [32]. Currently, several techniques have been used for image processing from MRI [33] for thickness measurement where the distribution of healthy cartilage thickness has been reported on femur. In all cases, the thickness values found in the present study for femoral cartilage are minor in a range of 6 to 40%, to those found in healthy knees in other studies (see Table 2). It is important to note that points AP, CP and PP in the present study are those with the thickest cartilage and corresponds to areas of cartilage-cartilage contact [30]; also these points correspond to loading [31] and displacement areas of the knee around the kinematic axis of flexion [29].
The instantaneous elastic modulus determined in this work considers the resistive force to indentation, the displacement of the indenter and the cartilage thickness at the test point according to the Hayes model et al. [25]. Previous studies have reported elastic modulus values in the range from 2.13 ± 0.74 to 5.13 ± 1.91MPa for healthy cartilage [10]. In a similar way, other cases have reported average values of 5MPa [7, 9, 34]. The distribution of instantaneous elastic modulus obtained in this study (see Table 3) shows that the values at points AP, CP, PP and MP are larger in magnitude (up to 40%) than those obtained in previous studies for healthy cartilage [7, 9, 10, 34]. Also, it can be observed that the instantaneous elastic modulus values in the displacement line of flexion AP, CP and PP showed a minimum variation in magnitude, while the points LP and MP differ significantly (p<0.05).
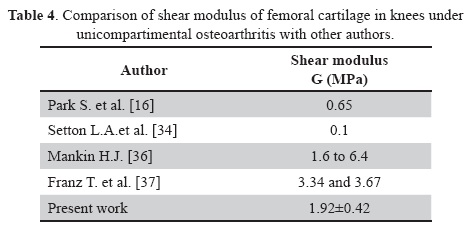
Previous studies showed that the human femoral compartments [36] shear modulus values were the range of 1.6 to 6.4MPa which changes based on damage Mankin score reported for these cases from 1 to 7 [37, 38]. Average shear modulus values of 3.34 and 3.67MPa in the medial and lateral compartment have been reported for Mankin score of 4 and 4.5, respectively. Maximum values obtained in the present study can be compared with these values. Table 1 show a maximum shear modulus of 2.48±0.90MPa and a minimum of 1.30±0.47MPa. It is noted that values in this work were near and between the range from 1.6 to 6.4MPa reported in human femoral compartment [36], since the cartilage analyzed in this study were also from human femoral specimens but in this case the specimens corresponds to knees under unicompartimental osteoarthritis (see Table 4).
Conclusions
This study has shown the distribution of mechanical properties and thickness of femoral cartilage in knees under unicompartimental osteoarthritis. These values were compared with results of previous studies finding that the mechanical properties reported herein were larger (up to 53%) than those of previous works on healthy cartilage. However, the thickness reported in previous work was larger (up to 69%) than the thickness measured in cartilage specimens in this work. The decrease in thickness in the different analyzed specimens provides evidence of wear in cartilage of knees under unicompartimental osteoarthritis. This may destroy the thin layer of proteins, collagen fibbers and cells highly packed and oriented that cover between 10 to 20% of the thickness from the surface, this diminish the cartilage recovery leading to osteoarthritis [40-41].
In addition, results have shown that thickness values and elastic, shear and bulk modulus were significantly similar (p<0.05) in points located in the line of cartilage-cartilage contact, loading and displacement in the kinematic axis of flexion.
Finally, experimental values presented in this work could be used to include the distribution of mechanical properties in femoral cartilage simulation and for the validation of a numerical model of cartilage behaviour for the study of biomechanics of the knee and prosthesis in cases of unicompartimental osteoarthritis and arthroplasty.
Acknowledgements
This work was carried out with support from the Mexican Social Security Institute (IMSS), the Biomechanics Laboratory of the Technological Institute of Celaya (ITC) and the Department of Mechanical Engineering of the University of Guanajuato (UG).
References
[1] Hopkins A.R., New Andrew M., Rodriguez-y-Baena F., Taylor M., Finite element analysis of unicompartimental knee arthroplasty, Med Eng Phys 32(2010) 14–21. [ Links ]
[2] Huang C., Soltz M.A., Kopacz M., Mow V.C., Ateshian G.A., Experimental verification of the roles of intrinsic matrix viscoelasticity and tension-compression nonlinearity in the biphasic response of cartilage, J Biomech Eng 125(2003) 84–93. [ Links ]
[3] Obeid E.M.H., Adams M.A., Newman J.H., Mechanical properties of articular cartilage in knees with unicompartimental osteoarthritis, J Bone Joint Surg 76-B(1994) 315-319. [ Links ]
[4] Armstrong C.G., Mow V.C., Variations in the intrinsic mechanical properties of human articular cartilage with age, degeneration, and water content, J Bone Joint 64-A(1982) 88-94. [ Links ]
[5] Vidal Lesso A., Lesso Arroyo R., Rodriguez Castro R., Garcia Miranda S., Daza Benitez L., Analysis, simulation and prediction of contact stresses in articular cartilage of the knee joint, Modelling in Medicine and Biology VII, Southampton, UK, 2007, pp. 55-64. [ Links ]
[6] Haut D.T.L., Hull M.L., Rashid M.M., Jacobs C.R., A finite element model of the human knee joint for the study of tibio-femoral contact, J Biomech Eng 124(2002) 273-280. [ Links ]
[7] Li G., Gil J., Kanamori A., Woo S.L., A validated three-dimensional computational model of a human knee joint, J Biomech Eng 121(1999) 657-662. [ Links ]
[8] Wei H.W., Sun S.S., Jao S.H.E., Yeh C.R., Cheng C.K., The influence of mechanical properties of subchondral plate, femur and neck on dynamic stress distribution of the articular cartilage, Med Eng Phys 27(2005) 295-304. [ Links ]
[9] Peña E., Calvo B., Martínez M.A., Doblaré M., A three-dimensional finite element analysis of the combined behaviour of ligaments and menisci in the healthy human knee joint, J Biomech 39(2006) 1686-1701. [ Links ]
[10] Thambyah A., Nather A., Goh J., Mechanical properties of articular cartilage covered by the meniscus, Osteoarthritis Cartilage 14(2006) 580-588. [ Links ]
[11] Lu X.L., Mow V.C. and Guo X.E., Proteoglycans and mechanical behavior of condylar cartilage, J Dent Res 88(2009) 244-248. [ Links ]
[12] Vidal-Lesso A., Ledesma-Orozco E., Lesso-Arroyo R., Rodriguez-Castro R., Dynamic response of femoral cartilage in knees with unicompartimental osteoarthritis, J Appl Res Tech 9(2011) 173-187. [ Links ]
[13] Mow V.C., Gibbs M.C., Lai W.M., Zhu W.B., Athanasiou K.A., Biphasic indentation of articular cartilage–II. A numerical algorithm and an experimental study, J Biomech 22(1989) 853-861. [ Links ]
[14] Mak A.F., Lai W.M., Mow V.C., Biphasic indentation of articular cartilage-I. Theoretical analysis, J Biomech 20(1987) 703-714. [ Links ]
[15] Lyyra T., Kiviranta I., Vaatainen U., Helminen H.J., Jurvelin J.S., In vivo characterization of indentation stiffness of articular cartilage in the normal human knee, J Biomed Mater Res 48(1999) 482-487. [ Links ]
[16] Garcia J.J., Altiero N.J., Haut R.C., An approach for the stress analysis of transversely isotropic biphasic cartilage under impact load, J Biomech Eng 120(1998) 608-613. [ Links ]
[17] Jun-Kyo S., Zhengfang L., Savio L-Y. W., Dynamic behavior of a biphasic cartilage model under cyclic compressive loading, J Biomech 28(1995) 357-364. [ Links ]
[18] Herberhold C., Faber S., Stammberger T., Steinlechner M., Putz R., Englmeier K. H., Reiser M., Eckstein F., In situ measurement of articular cartilage deformation in intact femoropatellar joints under static loading, J Biomech 32(1999) 1287-1295. [ Links ]
[19] Park S., Hung C. T., Ateshian G. A., Mechanical response of bovine articular cartilage under dynamic unconfined compression loading at physiological stress levels, Osteoarthritis Cartilage 12(2004) 65-73. [ Links ]
[20] Park S., Nicoll S. B., Mauck R. L., Ateshian G. A., Cartilage mechanical response under dynamic compression at physiological stress levels following collagenase digestion, Ann Biomed Eng 36(2008) 425-434. [ Links ]
[21] Huang C.Y., Mow V.C., Ateshian G.A., The role of flow independent viscoelasticity in the biphasic tensile and compressive responses of articular cartilage, J Biomech Eng 123(2001) 410-417. [ Links ]
[22] Lee R.C., Frank E.H., Grodzinsky A.J., Roylance D.K., Oscillatory compressional behavior of articular cartilage and its associated electromechanical properties, J Biomech Eng 103(1981) 280-92. [ Links ]
[23] Soltz M.A., Ateshian G.A., Interstitial fluid pressurization during confined compression cyclical loading of articular cartilage, Ann Biomed Eng 28(2000) 150–159. [ Links ]
[24] Barker M. K., Seedhom B. B., Articular cartilage deformation under physiological cyclic loading—Apparatus and measurement technique, J Biomech 30(1997) 377-381. [ Links ]
[25] Hayes W.C., Keer L.M., Herrmann G., Mockros L.F., A mathematical analysis for indentation tests of articular cartilage, J Biomech 5(1972) 541-551. [ Links ]
[26] Zhang M., Zheng Y.P., Mak A.F., Estimating the effective Young's modulus of soft tissues from indentation tests - nonlinear finite element analysis of effects of friction and large deformation, Med Eng Phys 19(1997) 512-517. [ Links ]
[27] Donzelli P.S., Spilker R.L., Ateshian G.A., Mow V.C., Contact analysis of biphasic transversely isotropic cartilage layers and correlations with tissue failure, J Biomech 32(1999) 1037-1047. [ Links ]
[28] Barker M.K., Seedhom B.B., The relationship of the compressive modulus of articular cartilage with its deformation response to cyclic loading: does cartilage optimize its modulus so as to minimize the strains arising in it due to the prevalent loading regime?, Rheumatology 40(2001) 274-84. [ Links ]
[29] Smith P.N., Refshauge K.M., Scarvell J.M., Development of the concepts of knee kinematics, Arch Phys Med Rehabil 84(2003) 1895-902. [ Links ]
[30] Li G., Park S.E., DeFrate L.E., Schutzer M.E., Ji L., Gill T.J., Rubash H.E., The cartilage thickness distribution in the tibiofemoral joint and its correlation with cartilage-to-cartilage contact, Clin Biomech 20(2005) 736–744. [ Links ]
[31] Adam C., Eckstein F., Milz S., Putz R., The distribution of cartilage thickness within the joints of the lower limb of elderly individuals, J Anat 193 (1998) 203-214. [ Links ]
[32] Bingham J. T., Papannagari R., Van de Velde S.K., Gross C., Gill T.J., Felson D.T., Rubash H.E., Li G., In vivo cartilage contact deformation in the healthy human tibiofemoral joint, Rheumatology 47(2008) 1622-1627. [ Links ]
[33] Carballido-Gamio J., Bauer J.S., Stahl R., Lee K.Y., Krause S., Link T.M., Majumdar S., Inter-subject comparison of MRI knee cartilage thickness, Med Image Anal, 12(2008) 120-135. [ Links ]
[34] Guoa Y., Zhanga X., Chena W., Three-dimensional finite element simulation of total knee joint in gait cycle, Acta Mech. Sol. Sin. 22(2009) 347-351. [ Links ]
[35] Setton L.A., Mow V.C., Howell D.S., Mechanical behavior of articular cartilage in shear is altered by transection of the anterior cruciate ligament, J Orthop Res 13(1995) 473-82. [ Links ]
[36] Franz T., Hasler E.M., Hagg R., Weiler C., Jakob R.P., Mainil-Varlet P., In situ compressive stiffness, biochemical composition, and structural integrity of articular cartilage of the human knee joint, Osteoarthritis Cartilage 9(2001) 582-592. [ Links ]
[37] Yuehuei H. A., Kylie L.M., Handbook of histology methods for bone and cartilage, Totowa, New Jersey; Humana Press 2003 411-421. [ Links ]
[38] Mankin H.J., Dorfman H., Lippiello L., Zarins A., Biochemical and metabolic abnormalities in articular cartilage from osteoarthritis human hips. II. Correlation of morphology with biochemical and metabolic data, J Bone Joint Surg Am. 53(1971) 523-537. [ Links ]
[39] Buckleya M.R., Gleghorn J. P., Bonassar L. J., Cohen I., Mapping the depth dependence of shear properties in articular cartilage, J Biomech 41(2008) 2430–2437. [ Links ]
[40] Meachim G., Sheffield S.R., Surface ultrastructure of mature adult human articular cartilage, J Bone Joint Surg Br 51(1969) 529-539. [ Links ]
[41] Stockwell R.A., The interrelationship of cell density and cartilage thickness in mammalian articular cartilage, J Anat 109(1971) 411-421. [ Links ]














