Introduction
Neonatal hyperbilirubinemia or jaundice (yellow color of the mucous membranes) is defined as total serum bilirubin (TBS) concentration > 3-5 mg/dl1. This pathology is a benign and frequent condition in newborns2,3 that manifests itself due to the livers enzymatic immaturity4 and is more frequently observed in premature newborns5. The importance of early diagnosis rests on avoiding complications such as acute encephalopathy and kernicterus, the latter being a chronic complication associated with cerebral palsy, deafness, dental dysplasia, ophthalmoplegia, and personality disorders6.
Neonatal jaundice is a significant morbidity and mortality issue7, accounting for 1309.3 deaths per 100,000 live births worldwide in 2016, the seventh leading cause of neonatal death8. In 2012, the prevalence of neonatal jaundice was 29%, representing the leading cause of medical consultation and neonates hospitalization in Peru9.
The measurement of TSB is traditionally the most widely used diagnostic method worldwide, but it involves an invasive, costly, and time-consuming procedure. Another option is to measure transcutaneous bilirubin (TcB), a painless, cheap, fast, and non-invasive method that minimizes blood collection in newborns10,11. The American Academy of Pediatrics and the Canadian Academy of Pediatrics recommend TcB measurements on all newborns before discharge12,13. Moreover, it is used as a diagnostic method in some countries14. Due to these recommendations, studies have been conducted in different population groups to confirm the correlation of values obtained by transcutaneous and serum bilirubinometry and determine if the transcutaneous diagnostic method shows adequate sensitivity and specificity for diagnosing jaundice in newborns15.
The city of Cusco is located in the highlands of Peru at an altitude > 3400 m above sea level (masl). In this geographical area, it is necessary to verify if the transcutaneous method is as effective as the serum method since the physiological adaptation in healthy populations at high altitudes causes sustained hypoxia, increased cardiac output, and hemoglobin increase16,17 of 1.05 g/dl for every 1000 m of altitude18. This consideration is essential since bilirubin is the final product of the catabolism of hemoglobin1.
Methods
A prospective and cross-sectional study was conducted between January and March 2017 in the Neonatology Service of the Hospital Regional of the city of Cusco, located in the mountains of Peru, at an altitude of more than 3400 masl.
The study included full-term newborns with clinical jaundice, Apgar score > 7 (minute 1), and a normal physical exam. Newborns with the following characteristics were excluded: those born at home, needing resuscitation maneuvers, with congenital malformations, who received previous therapy, and those born to mothers with an obstetric pathology such as preeclampsia, diabetes mellitus, placental insufficiency, toxoplasmosis, rubella, cytomegalovirus, herpes simplex, or HIV.
Using convenience sampling, we collected data from newborns with clinical jaundice who met the selection criteria (the calculated sample size using the formula for diagnostic tests was 123 newborns) through a revised card evaluated by experts in the field. General data, such as gestational age by the Capurro method, life span in hours, sex, weight, height, birth route, Kramers scale, and measurement of total and transcutaneous serum bilirubin, were included in the study.
The Ethics Committee of the Hospital Regional of Cusco approved the research project. Data were collected under the authorization of the hospital director and the head of the Neonatology service, ensuring participants anonymity and confidentiality, and with the voluntary informed consent of the mothers of each newborn.
As soon as the selection criteria were met, venous blood samples were collected to measure TSB; after 30 min, transcutaneous bilirubin (forehead and sternum) was determined. We used the Konica Minolta/JM-103 transcutaneous bilirubinometer (Drager Medical Inc, Telford, Pennsylvania), which determines the intensity of yellow in the subcutaneous cellular tissue. The transcutaneous bilirubinometer use was standardized according to the manufacturers guidelines by placing it vertically to the skin and disinfecting it with alcohol (70%) before and after its use on each newborn.
The frequencies and percentages of the categorical variables were included in the descriptive analysis, as well as the best measure of central tendency and dispersion of the quantitative variables. Pearsons correlation was used to compare the measurement of transcutaneous bilirubin (in the forehead and sternum) with TSB values after the KolmogorovSmirnov normality test. A linear scatter plot was also calculated to determine the correlation curve trend. Bland-Altman graphs were used to determine if the difference between the methods was stable in all measured bilirubin ranges. TSB ≥ percentile 95 (high-risk hyperbilirubinemia) was used as the gold standard. According to the Buthani nomogram19, two cut-off points were used to determine transcutaneous bilirubin (in the forehead and sternum): percentile 95 and percentile 75 for postnatal age in hours, to compare the results in case the cut-off point was lowered. Sensitivity and specificity were calculated, and the receiver operating characteristic (ROC) curves were constructed to evaluate the predictive capacity of TcB (in the forehead and sternum). The results were considered statistically significant with p < 0.05. The SPSS software version 22.0 was used.
Results
A total of 195 patients were evaluated, of which 123 full-term newborns with clinical jaundice met the selection criteria. Seventy newborns (56.9%) were female, with a mean weight of 3251.79 ± 545 g, mean length of 49.29 ± 2.5 cm, mean age of 53 ± 16.6 h of life, and mean gestational age according to Capurro method of 38.8 ± 1.4 weeks. Of the total number of newborns, 115 (93%) showed an adequate birth weight. The most common delivery route was cesarean section in 72 (57.7%) births. Fifty-two (42.2%) were grade IV on the Kramer scale. The mean transcutaneous bilirubin in the forehead (TcBF) was 13.7 ± 3.5, the mean transcutaneous bilirubin in the sternum (TcBS) was 14.1 ± 3.1, and the mean TSB was 13.8 ± 3.9 (Table 1).
Table 1 General characteristics of newborns
| Characteristic | Mean ± SD (n = 123) |
|---|---|
| Gestational age (Capurro method) | 38.8 ± 1.4 |
| Weight (g) | 3251.7 ± 545 |
| Adequate birth weight | 115 (93%) |
| Low birth weight (LBW) | 8 (7%) |
| Length (cm) | 49,29 ± 2.5 |
| Hours of life | 53.9 ± 16.6 |
| Sex | |
| Male | 53 (43.1%) |
| Female | 70 (56.9%) |
| Birth route | |
| Cesarean section | 72 (57.7%) |
| Vaginal | 51 (42.4%) |
| Kramers scale | |
| II | 13 (9.7%) |
| III | 42 (34.9%) |
| IV | 52 (42.2%) |
| V | 16 (13.1%) |
| TcBF | 13.7 ± 3.5 |
| TcBS | 14.1 ± 3.1 |
| TSB | 13.8 ± 3.9 |
SD: standard deviation; TcBF: transcutaneous bilirubin in the forehead; TcBS: transcutaneous bilirubin in the sternum; TSB: total serum bilirubin.
Pearsons correlation between TSB/TcBF and TSB/TcBS was 0.90 and 0.91, respectively (p < 0.05), as shown in the linear scatter plots (Fig. 1).
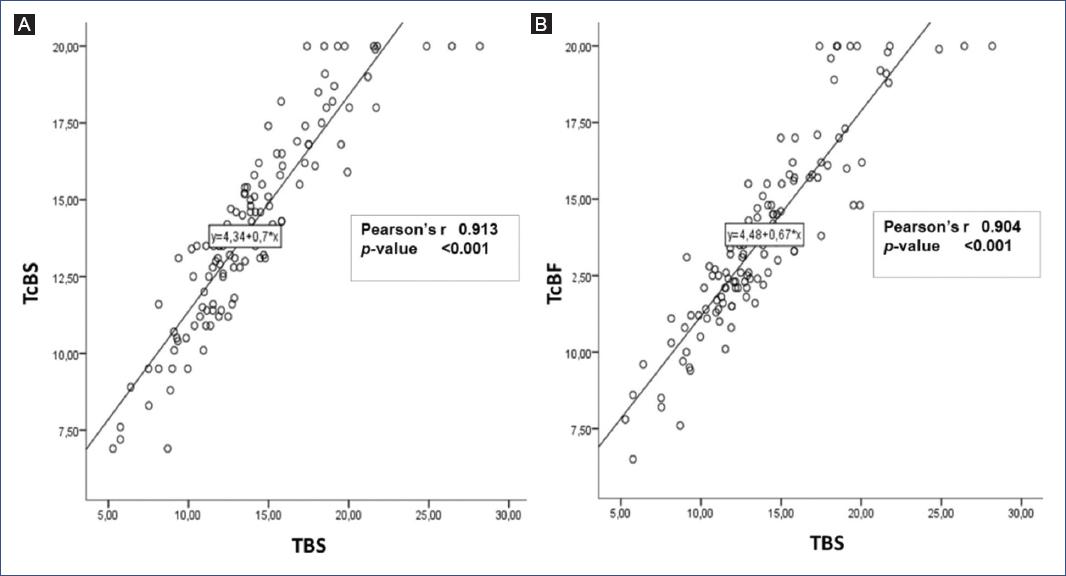
Figure 1 Linear regression plots for total serum bilirubin (TSB) versus transcutaneous bilirubin (TcB) at different measurement sites. A. Sternum (TcBS). B. Forehead (TcBF).
Table 2 presents the participants positive and negative results for TSB according to the cut-off points for transcutaneous bilirubin. The sensitivity, specificity, positive predictive value, negative predictive value, positive likelihood ratio, and negative likelihood ratio for TcB (in the forehead and sternum) at ≥ percentile 95 and ≥ percentile 75 cut-off points, according to the Bhutani nomogram, are represented.
Table 2 Predictive characteristics of the transcutaneous bilirubin indices in the forehead and sternum for the percentile 95 and 75 cut-off points according to total serum bilirubin
| Predictive TcB | Total | TSB ≥ p95 | Sen | Spe | PPV | NPV | LR+ | LR- | |
|---|---|---|---|---|---|---|---|---|---|
| (n = 123) | (+) | (-) | |||||||
| ≥ p95 TcBS | 70 | 57 | 13 | 100% | 80% | 81% | 100% | 5.08 | 0 |
| < p95 TcBS | 53 | 0 | 53 | ||||||
| ≥ p95 TcBF | 60 | 53 | 7 | 93% | 89% | 88% | 94% | 8.77 | 0.08 |
| < p95 TcBF | 63 | 4 | 59 | ||||||
| ≥ p75 TcBS | 106 | 57 | 49 | 100% | 26% | 54% | 100% | 1.35 | 0 |
| < p75 TcBS | 71 | 0 | 17 | ||||||
| ≥ p75 TcBF | 101 | 57 | 44 | 100% | 33% | 56% | 100% | 1.50 | 0 |
| < p75 TcBF | 22 | 0 | 22 | ||||||
Percentile cut-off points, according to the Bhutani nomogram. Total serum bilirubin determination is the diagnostic test of choice. F: forehead; LR: likelihood ratio; NPV: negative predictive value; p: percentile; PPV: positive predictive value; S: sternum; Sen: sensitivity; Spe: specificity; TcB: transcutaneous bilirubin; TSB: total serum bilirubin.
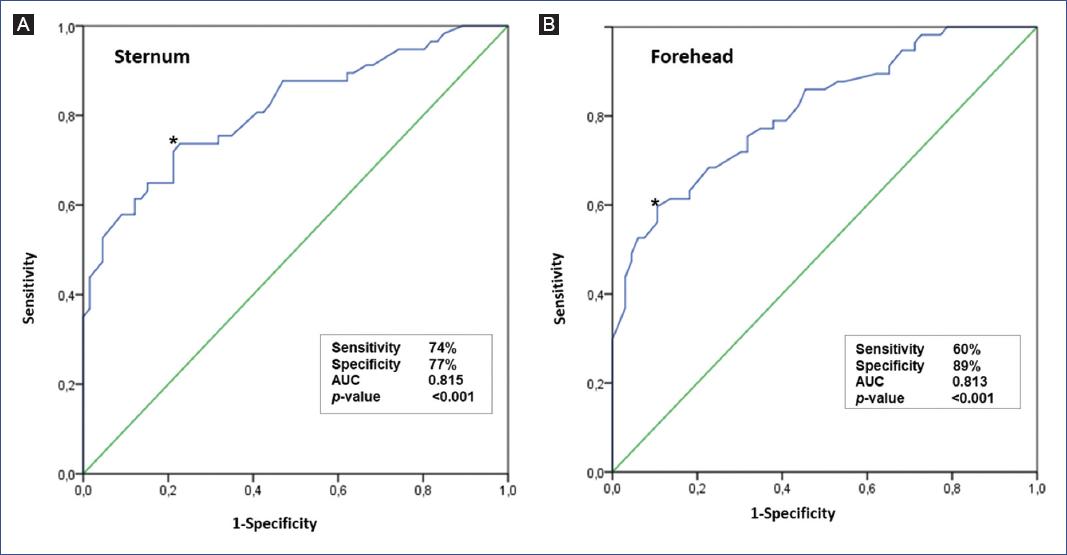
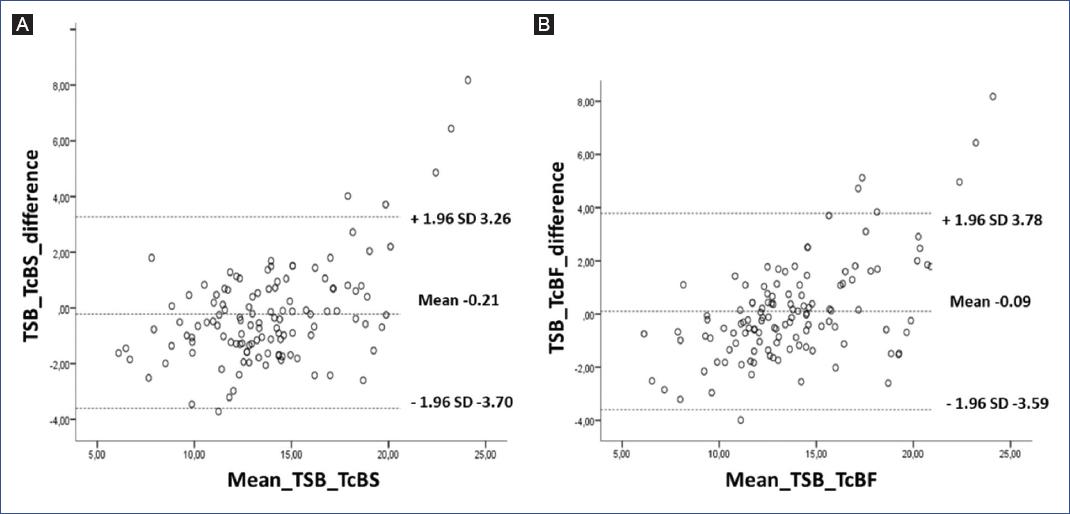
Figure 2 shows the sensitivity and specificity ROC curves, where the area under the curve for TcBS was 0.815 and for TcBF was 0.813 (p < 0.001). Figure 3 shows the Bland-Altman graph for the difference obtained between TSB and TcB (in the forehead and sternum) measurement methods.
Discussion
Our study showed a high correlation between the measurement of TcB (in the forehead and sternum) and serum bilirubin in newborns with clinical jaundice at more than 3400 masl. The correlations obtained were 0.91 and 0.90 for TcBS and TcBF, respectively, equal to or higher than other studies in populations at sea-level. In Mongolia, a Pearsons correlation coefficient of 0.78 was obtained20; in Bangladesh, 0.9321; in Iran, 0.823; in the southern United States, 0.6910; and in Israel, 0.7222. The correlation was also found in studies that included low birth weight newborns, neonates needing mechanical ventilation23, and newborns with different skin color11. Moreover, a meta-analysis showed that TcB is a reliable method in full-term and preterm infants24. Our study covered a population living at high altitude. Physiologically, higher concentrations of hemoglobin16,17 and, consequently, a greater amount of bilirubin are found in this population, which could explain the more significant correlation. No differences in mean serum or transcutaneous bilirubin levels were observed. These results could be used for reliable clinical follow-upas suggested by the Canadian Pediatric Society in its guidelines for detecting, managing, and preventing hyperbilirubinemia, in which correlation coefficients in the range of 0.77-0.93 are recommended for reliable clinical use (with some exceptions)25.
According to the Bhutani nomogram, our results showed an area under the curve of 0.815 for TcBS, slightly higher than that obtained for TcBF, which was 0.813 for the cut-off point ≥ percentile 95. Therefore, both methods are useful to measure transcutaneous bilirubin indicating a high predictive value. Similarly, for the cut-off point ≥ percentile 95 (high-risk zone), the sensitivity was 100% for TcBS and 93% for TcBF, with a specificity of 80% for TcBS and 89% for TcBF. Compared to other studies, the sensitivity found in this study was higher. Nahar et al.21 obtained a sensitivity of 77% and a specificity of 88%. The study conducted by Hemmanti and Kiyani26 showed a sensitivity and specificity of 96.6% and 99%, respectively, and Akahira-Azuma et al.20 found a sensitivity of 85% and a specificity of 84%.
In contrast, these studies used a 15 mg/dl of TSB cut-off point to measure sensitivity and specificity. This measure can be biased since bilirubin levels vary with the newborns age. However, the Bhutani nomogram covers this parameters measurement at all ages by percentiles. The sensitivity found for TcB is better in our study and exceeds sensitivity values found in the Kramer scale27, being more effective for screening a newborn with jaundice.
Our study demonstrated higher sensitivity than specificity in the TcB for a ≥ percentile 95 (high-risk zone) cut-off point. However, by decreasing the cut-off point to ≥ percentile 75 (high intermediate risk zone), we observed an increase in sensitivity and a decrease in specificity. These results indicate that both cut-off points could be recommended for the measurement of TcB as screening and, above all, for post-treatment follow-up of neonatal jaundice, but not for a definitive diagnosis to determine the use of phototherapy or exchange transfusion, as recommended by other studies11.
This study presented some limitations: newborns previously exposed to phototherapy were not considered; thus, it was not possible to verify whether TcB measurements were adequate for post-phototherapy follow-up. Furthermore, considering that the measuring instruments varied in each study, it was demonstrated that bilirubinometers of different brands, including the bilirubinometer used in our study, are reliable in estimating bilirubin levels28,29.
Peru is a developing country where multiple health problems and low resources affect 21.7% of the population30. In these areas, our findings might improve the clinical confidence of TcB measurement since it has been proven to correlate well with serum bilirubin, which is the diagnostic test of choice. Therefore, hospitalization time and costs could be reduced, and blood sampling from newborns with jaundice could be minimized. Furthermore, since this procedure can be performed with portable equipment, it would be useful in rural areas of Peru, where there is still a high prevalence of home births31.
Our study covered a high-altitude population, where a strong correlation between transcutaneous and serum bilirubin was confirmed. Therefore, measuring TcB instead of TSB could decrease hospital stay, costs, and blood sampling and could be used as a screening and follow-up tool for newborns with jaundice.











 nueva página del texto (beta)
nueva página del texto (beta)